Abstract
Hepatocyte growth factor (HGF) and the proto-oncogenic receptor c-Met are implicated in growth, invasion, and metastasis in human cancer. Little information is available on the expression and role of both gene products in canine osteosarcoma. We hypothesized that the expression of c-Met is associated with malignant histologic characteristics, a short survival time, and a reduced disease-free interval in canine osteosarcoma. Quantitative real-time polymerase chain reaction was used to analyze the messenger RNA (mRNA) expression of both HGF and c-Met in 59 canine osteosarcoma samples. The relationship between HGF and c-Met expression, patient outcome, and histologic characteristics of the tumor were studied. Western blot analysis was performed to investigate the presence of active HGF protein. The expression pattern of c-Met in 16 slides of canine osteosarcoma was identified by immunohistochemistry. Coexpression of HGF and c-Met mRNA in all canine osteosarcoma samples suggested autocrine or paracrine receptor activation. A significant, moderately positive correlation was found between c-Met and HGF mRNA expression. c-Met mRNA expression was not associated with survival time or disease-free interval. Expression of c-Met was significantly associated with metastasis via the lymphogenic route. Immunolabeling with c-Met revealed a cytoplasmic staining pattern in all osteosarcoma cell types. In this study, c-Met mRNA expression in canine osteosarcoma was found to be of no influence on survival time and disease-free interval. Further studies are necessary to confirm the involvement of the c-Met pathway in the lymphogenic route of metastasis.
Osteosarcoma, the most common primary bone tumor in dogs, accounts for 85% of all skeletal malignancies. 6 Osteosarcoma primarily affects large- and giant-breed dogs of middle to older age and is a locally aggressive tumor, with the appendicular skeleton most frequently affected. Metastatic disease is very common and occurs mainly to the lungs via the hematogenous route. 6 In most cases, the dogs will die or euthanasia will be indicated because of malaise from metastatic disease.
Canine osteosarcoma resembles osteosarcoma in humans. On histologic examination, canine and human osteosarcomas are alike, i.e., both are of high histologic grade. The tumor rapidly metastasizes to the lungs, and survival is improved with adjuvant chemotherapy in both species. Other similarities include the relative size of the patient (with a larger individual being affected more often) and presentation at metaphyseal locations of major weight-bearing bones. 27 Canine osteosarcoma differ in some respects from the human counterpart: dogs are affected at a later stage in life and the disease has a higher prevalence and a more rapid progression in the dog. 27
The etiology of osteosarcoma in both species is largely unknown. The ligand hepatocyte growth factor (HGF) and the proto-oncogenic receptor c-Met have been implicated in the genesis and malignant progression of several human malignancies, including osteosarcoma. 2 A causative role for HGF/c-Met signaling in the development and/or progression of canine osteosarcoma is still under debate.
HGF, first identified as a potent mitogen of primary cultured hepatocytes, 18 is produced by several mesenchymal cells and is secreted as the biologically inactive precursor pro-HGF. Pro-HGF (92 kDa) is proteolytically activated by HGF-activator. The activated product, a disulfide-linked heterodimer, consists of a 69-kDa α-subunit and a 34 kDa β-subunit. The latter is involved in c-Met binding and subsequent transmembrane signaling. 10 Research on human and canine osteosarcoma cell lines has shown a possible role for HGF in enhanced proliferation, invasion, and scattering of tumor cells after stimulation of c-Met. 3,5,8 More recently, Patanè et al. 20 found that overexpression of c-Met, obtained by lentiviral vector-mediated gene transfer, resulted in the conversion of human osteoblasts into osteosarcoma cells.
Coexpression of HGF and c-Met has been detected in a variety of human tumors, including glioma, myeloma, breast, lung, pancreatic, and thyroid cancers; glioma; and myeloma. 13 Autocrine stimulation of the c-Met receptor by HGF may lead to a more malignant phenotype: autocrine stimulation of c-Met by HGF implicates an advantage for tumor cells in colony forming in anchorage independent conditions. 16 In addition, the nontumorigenic mouse cell line C127 (expressing very low levels of HGF and c-Met proteins) was engineered to overexpress both HGF and c-Met proteins. This cell line became phenotypically transformed, highly tumorigenic, and metastatic in vivo. Increased levels of either HGF or c-Met alone did not result in a tumorigenic switch. 12
A body of publications discuss expression of HGF and c-Met in human osteosarcoma samples, 1,8,9,17,19,21,22,26 but only one study reports on canine osteosarcoma, noting c-Met expression in 5 of 7 clinical samples. 7 HGF expression and histologic malignancy characteristics and patient outcome in relationship to c-Met expression, however, were not addressed in that study.
In the study reported here, messenger RNA (mRNA) expression levels of both HGF and c-Met were measured in 59 canine osteosarcoma samples with quantitative real-time polymerase chain reaction (Q-PCR), and the data were related to histologic malignancy characteristics and patient outcome. The amount of proteolytically activated HGF in tumor tissue was determined with Western blot analysis. The cell-specific expression pattern of c-Met in histologic slides of canine osteosarcoma was identified by immunohistochemistry.
Materials and Methods
Dogs
A total of 59 osteosarcoma samples were obtained from dogs that were referred to the Department of Clinical Sciences of Companion Animals, Utrecht University, The Netherlands, between 1993 and 2004. The age of the dogs at presentation ranged from 1.0 to 13.0 years (median 7.4 years). The most common breeds from which osteosarcoma were obtained were Rottweiler (n = 17), cross breed (n = 6), Labrador Retriever (n = 5), Great Dane (n = 4), and Doberman (n = 4). The remaining 23 dogs comprised several breeds. The weights of the dogs ranged from 16.0 to 87.0 kg (median 41.0 kg). The majority of the dogs had appendicular osteosarcoma (n = 51), 7 dogs had axial osteosarcoma, and 1 dog was diagnosed with an extraskeletal osteosarcoma. Clinical data were collected from the medical records. Follow-up information was obtained from the medical records or via direct communication with owners or referring veterinarians. None of the dogs received chemotherapy before harvesting of tumor material.
Sample handling
Osteosarcomas were harvested under sterile conditions during surgery or during postmortem within 30 minutes after euthanasia of the dog. Samples were immediately snap-frozen in liquid nitrogen and stored at −70°C. An adjacent sample was fixed in paraformaldehyde (4%) for at least 24 hours, decalcified in ethylenediaminetetraacetic acid (EDTA) (10%) and embedded in paraffin. Sections (4 µm) were stained with hematoxylin and eosin for confirmation of the diagnosis and to determine the histologic classification. Before mRNA and protein extraction, samples were pulverized, under cooling in liquid nitrogen, by using a 5-mm ball bearing in a Braun Mikro-dismembrator U (Braun Biotech International, Melsungen, Germany) at 2,000 rounds per minute for 45 seconds in ribonuclease free plastic containers.
Histologic classification
Osteosarcoma were classified by one certified veterinary pathologist (MK) by using the osteosarcoma grading system previously described. 14 Osteosarcomas were sorted into osteoblastic and mixed-type histologic subtypes. Three grades with increasing histologic malignancy characteristics were distinguished; these characteristics included the following: nuclear pleomorphism, amount of matrix, percentage of tumor cells, number of mitotic cells, extent of necrosis, number of multinucleated giant cells, number of whirls, and extent of tumor cell invasion in vessels or lymph nodes. The association between c-Met mRNA expression and these histologic characteristics was studied.
RNA isolation and reverse-transcription PCR
Total cellular RNA was isolated from each tumor powder specimen by using RNeasy Mini Kit (Qiagen, Venlo, The Netherlands), according to the manufacturer's instructions. An additional pretreatment with >600 mU/ml Proteinase K (Qiagen) was used to improve the RNA isolation procedure. The RNA samples were treated with Dnase-I (Qiagen) to exclude contamination with traces of genomic DNA. The amount of isolated RNA was quantified with a Nanodrop-1000 spectrophotometer (Thermo Scientific, Wilmington, DE, USA). In total, 3 µg of RNA was incubated with oligo(dT) primers at 42°C for 45 minutes in a 60-µl reaction volume by using the Reverse Transcription System (Promega Benelux, Leiden, The Netherlands) according to the manufacturer's instructions.
Q-PCR
Q-PCR was performed on HGF and c-Met and on reference genes glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and hypoxanthine phosphoribosyltransferase (HPRT). Real-time quantitative PCR was based on the high affinity double-stranded DNA-binding dye SYBR green I (SYBR green I, BMA, Rockland, ME, USA) and was performed in triplicate in a spectrofluorometric thermal cycler (iCycler, BioRad, Veenendaal, The Netherlands). For each PCR reaction, 2 µl (of the 2× diluted stock) of copy DNA (cDNA) was used in a reaction volume of 50 µl that contained 1× manufacturer's buffer, 2 mM MgCl2, 0.5 × SYBR green I, 200 µM deoxyribonucleotide triphosphates, 20 pmol of both primers, 1.25 units of AmpliTaq Gold (Applied Biosystems, Nieuwerkerk a/d IJssel, The Netherlands), on 96-well iCycler iQ plates (BioRad). Primer pairs, depicted in Table 1, were designed by using PrimerSelect software (DNASTAR Inc., Madison, WI, USA). All PCR protocols included a 5-minute polymerase activation step and continued for 40 cycles (denaturation) at 95°C for 20 seconds, annealing for 30 seconds, and elongation at 72°C for 30 seconds, with a final extension for 5 minutes at 72°C. Annealing temperatures were optimized at various levels that ranged from 56°C to 58°C (Table 1). Melt curves (iCycler, BioRad), agarose gel electrophoresis, and standard sequencing procedures were used to examine each sample for purity and specificity (ABI PRISM 3100 Genetic Analyser, Applied Biosystems). Standard curves constructed by plotting the relative starting amount versus threshold cycles were generated by using serial fourfold dilutions of pooled cDNA fractions from 44 samples. The amplification efficiency, E (%) = (10(1/−s) − 1) × 100 (s = slope), of each standard curve was determined and appeared to be >95%, and <105% over a wide dynamic range. For each experimental sample, the amount of the gene of interest, and of the endogenous references GAPDH and HPRT were determined from the appropriate standard curve in autonomous experiments. If relative amounts of GAPDH and HPRT were constant for a sample, then data were considered valid and the average amount was included in the study (data not shown). Results were normalized according to the average amount of the endogenous references.
Nucleotide sequences of dog-specific primers for real-time quantitative PCR.

Western blot analysis for HGF protein
Extracted protein from 6 osteosarcoma samples was used in the Western blot assay. Two samples with the highest HGF mRNA expression were selected as well as two samples with the lowest HGF mRNA expression. In addition, two samples with medium expression were analyzed. Normal canine liver tissue was taken as a positive control. Extracted protein from a canine bile-duct epithelial cell line, which has no endogenous HGF production, was taken as a negative control. The cell line was cultured in-house as previously described. 23
For the extraction of soluble protein, 50 mg of frozen, pulverized tumor tissue (and frozen liver tissue as a positive control) was used. The extraction of protein, measurement of protein concentration, electrophoresis, Western blotting procedure, immunodetection, and exposure were carried out according to our standard laboratory protocols, as previously described. 24 The primary HGF antibody (Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA) was diluted 1 : 1,000 with Tris buffered saline and Tween 20 (TBS-T) that contained 4% bovine serum albumin (BSA). Beta-Actin (Neomarkers, Lab Vision Corporation, Fremont, CA, USA) was used as a loading control in a dilution of 1 : 2,000 in TBS-T that contained 4% BSA. After washing with TBS-T, the membranes were incubated with their respective secondary antibody; chicken anti-goat IgG conjugated with horseradish peroxidase (HRP) (dilution 1 : 20,000) (Santa Cruz Biotechnology) or anti-mouse IgG HRP–conjugated (dilution 1 : 20,000) (R&D Systems, Minneapolis, MN, USA) for 1 hour at room temperature.
Immunohistochemistry for c-Met
Sixteen large intact histologic osteosarcoma samples were selected for immunohistochemical staining for c-Met. Excluded were samples of core biopsies or samples with mainly necrotic areas. Seven samples were from an appendicular location, and two were from axial osteosarcoma. Seven samples were of the osteoblastic subtype, and 9 samples were of the mixed subtype. Two of these samples were classified low malignant (grade 1), 4 showed a medium malignant grade (grade 2), and 10 samples were of a high malignant grade (grade 3). Freshly cut paraffin sections (4 µm) were mounted on 3-aminopropyltriethoxysilan coated slides, dried for 48 hours at 40°C, and stored at 4°C until use. Deparaffinized sections were treated with 0.3% H2O2 in methanol for 20 minutes at room temperature to block endogenous peroxidase activity. After rehydration, antigen retrieval was performed by boiling the sections in 0.01 M citrate, pH = 6.0, in a microwave oven for 10 minutes. The sections were quickly cooled down by rinsing in demineralized water, followed by washing in Tris buffered saline (TBS) solution. Background staining was blocked by incubating sections with normal rabbit serum 1 : 50 diluted for 15 minutes at room temperature. The sections were incubated with primary antibody (human c-Met, clone 8F11, cat. no. MONX10170 Monosan, Uden, The Netherlands), diluted 1 : 1,000 in TBS-BSA 1%, overnight at room temperature. After washing in TBS, the slides were incubated with rabbit–anti-mouse biotinylated secondary antibody (1 : 200 in TBS-BSA 1%) (Dako Cytomation, Glostrup, Denmark) at room temperature, followed by washing in TBS and 30 minutes incubation with horseradish peroxidase–streptavidin complex (Dako Cytomation). Color was developed in 3-3′-diaminobenzidine (0.5 mg/ml) and 0.02% H2O2 in Tris buffer (pH 7.6). Sections were counterstained in Mayer's hematoxylin counterstain. Positive control tissue consisted of human prostate (n = 1), canine prostate (n = 1), and canine liver (n = 1), all without EDTA treatment, as well as human osteosarcoma (n = 3) and normal canine humerus (n = 2), decalcified in EDTA as described above. In negative controls, the first antibody was omitted.
Statistical analysis
Statistical analyses were carried out with SPSS 15.0 statistical package (SPSS Benelux BV, Gorinchem, The Netherlands). A multivariate stepwise linear regression was performed to evaluate the association of c-Met mRNA expression with histologic characteristics. Predictors included histologic subtype, nuclear pleomorphism, amount of matrix, percentage of tumor cells, number of mitotic cells, extent of necrosis, number of multinucleated giant cells, and number of whirls. The level of significance for inclusion was set at P < .05, and the level of significance for exclusion was set at P > .10. Association of c-Met mRNA expression and tumor invasion in lymph nodes and vessels was analyzed in a separate analysis by using analysis of variance (ANOVA), because values for these characteristics were missing (in 3 samples, no vessels were present, and, in 37 cases, no draining lymph nodes were available for analysis). c-Met mRNA expression in relation to tumor grade was analyzed separately by using ANOVA, because grade is a derivate of the histologic characteristics (save the histologic subtype).
Log values for HGF and c-Met mRNA expression, corrected for the values of the reference genes, were distributed normally, and the relationship between expression levels of HGF and c-Met mRNA were evaluated by using the Pearson correlation coefficient. A univariate Cox regression model was used to compare c-Met mRNA expression with survival data. Survival time and disease-free interval were studied. Survival time in days was defined as the time between presentation in the clinic and death of the dog. Disease-free interval in days was defined as the time between the day of surgery and recurrence or the appearance of metastases. Dogs that had died from nonosteosarcoma related causes were counted as censored cases. None of the dogs were alive at the time of analysis.
Results
Detection of c-Met and HGF on mRNA level with Q-PCR
Samples of 59 canine osteosarcoma were analyzed with Q-PCR for c-Met and HGF mRNA expression levels. In all 59 samples, both c-Met and HGF mRNA were detected. The specimens showed a great variation in expression levels of both c-Met and HGF (Figs. 1, 2). Expression levels were related to expression of the reference genes. Expression of c-Met and of HGF exceeded mean expression in 29% and 47% of cases, respectively. Expression of both c-Met and HGF exceeded mean expression in 24% of cases. A significant, moderately positive correlation was found between c-Met and HGF mRNA expression (Fig. 3).
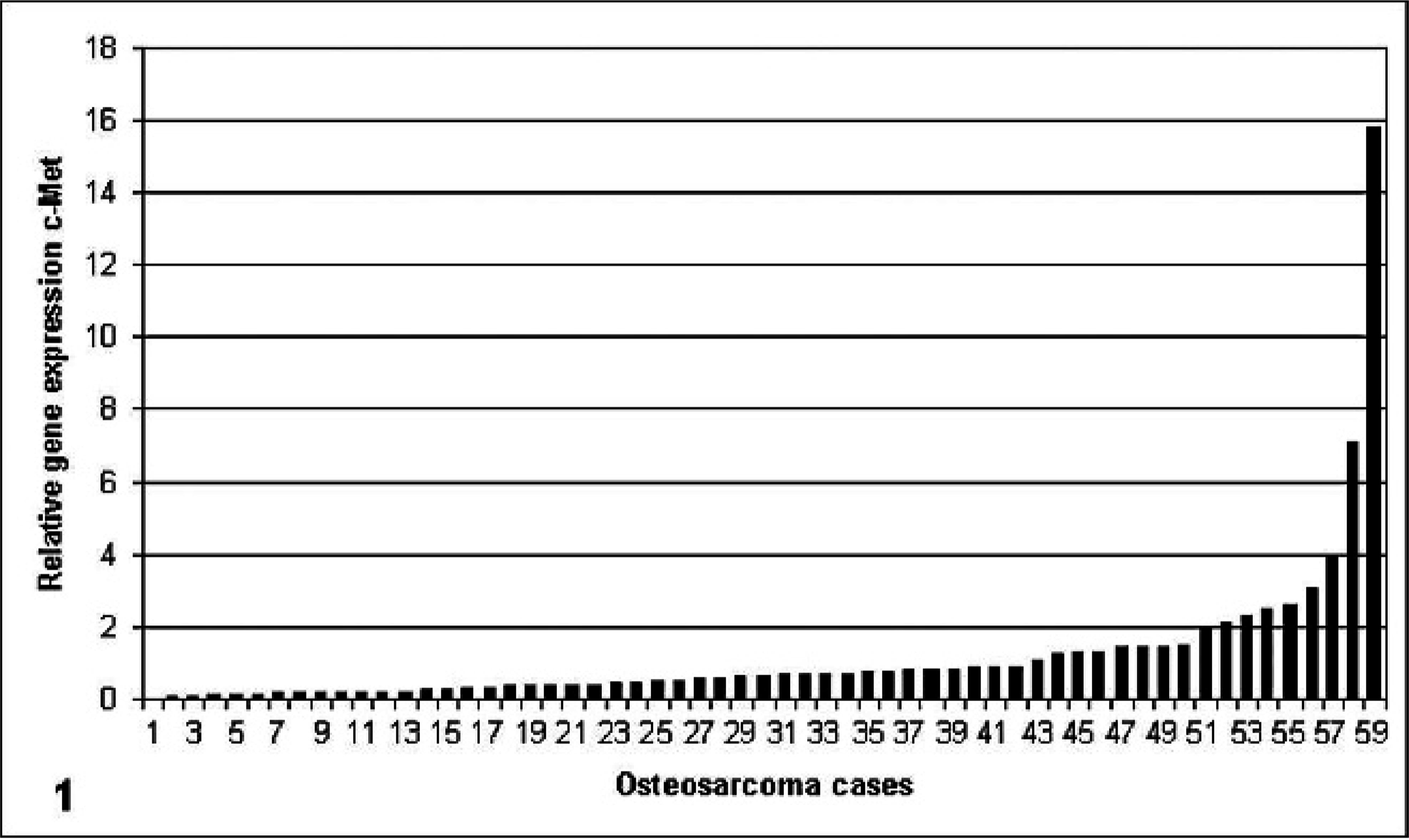
Relative gene-expression c-Met. Data depicted as individual relative gene expression after normalization against endogenous reference genes.
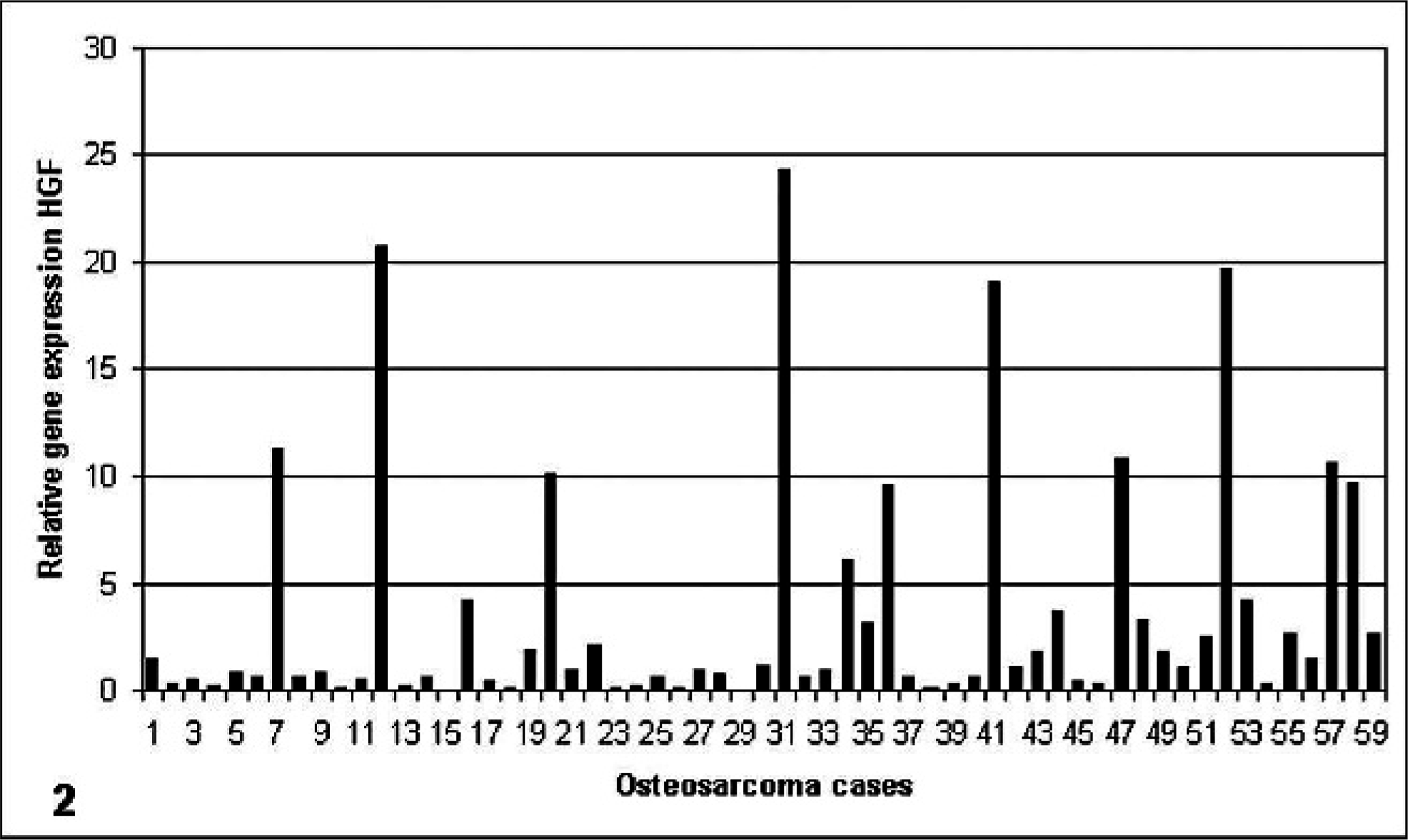
Relative gene-expression HGF. Data depicted as individual relative gene expression after normalization against endogenous reference genes.
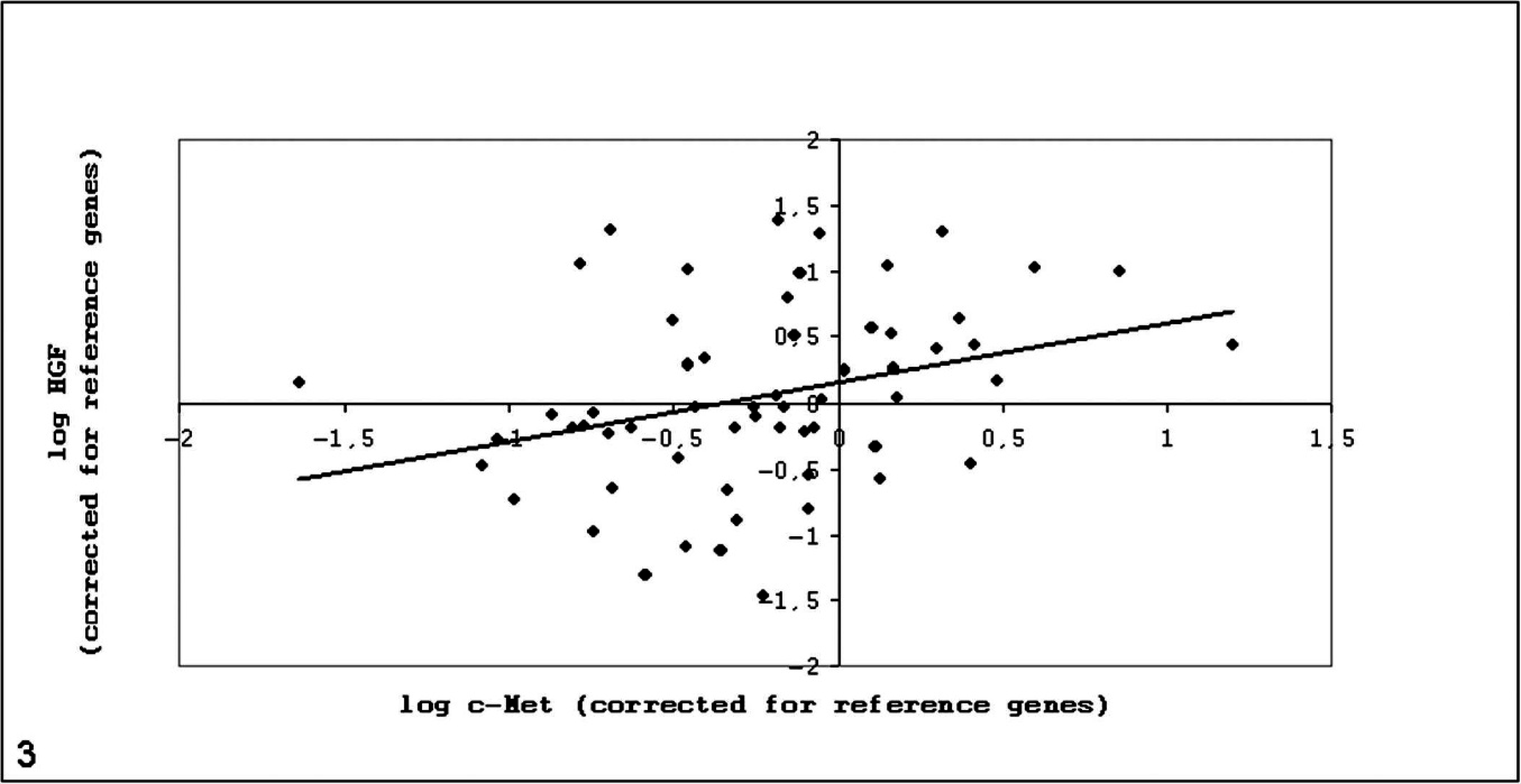
Correlation between HGF and c-Met mRNA expression. Pearson correlation coefficient: 0.323 (P = .013). Samples are depicted in order of increasing c-Met expression (nos. 1–59).
Western blot analysis for HGF protein
Proteolytically activated HGF-α protein was detected in the liver sample and in all 6 osteosarcoma samples (Fig. 4). The canine bile-duct epithelial cell line remained negative for HGF-α, which verified the specificity of the antibody. The osteosarcoma samples showed no large differences in protein expression for HGF, although there was a wide variation in the mRNA expression levels as measured with Q-PCR. Therefore, HGF mRNA expression levels were not used for outcome analysis and association with histologic characteristics.
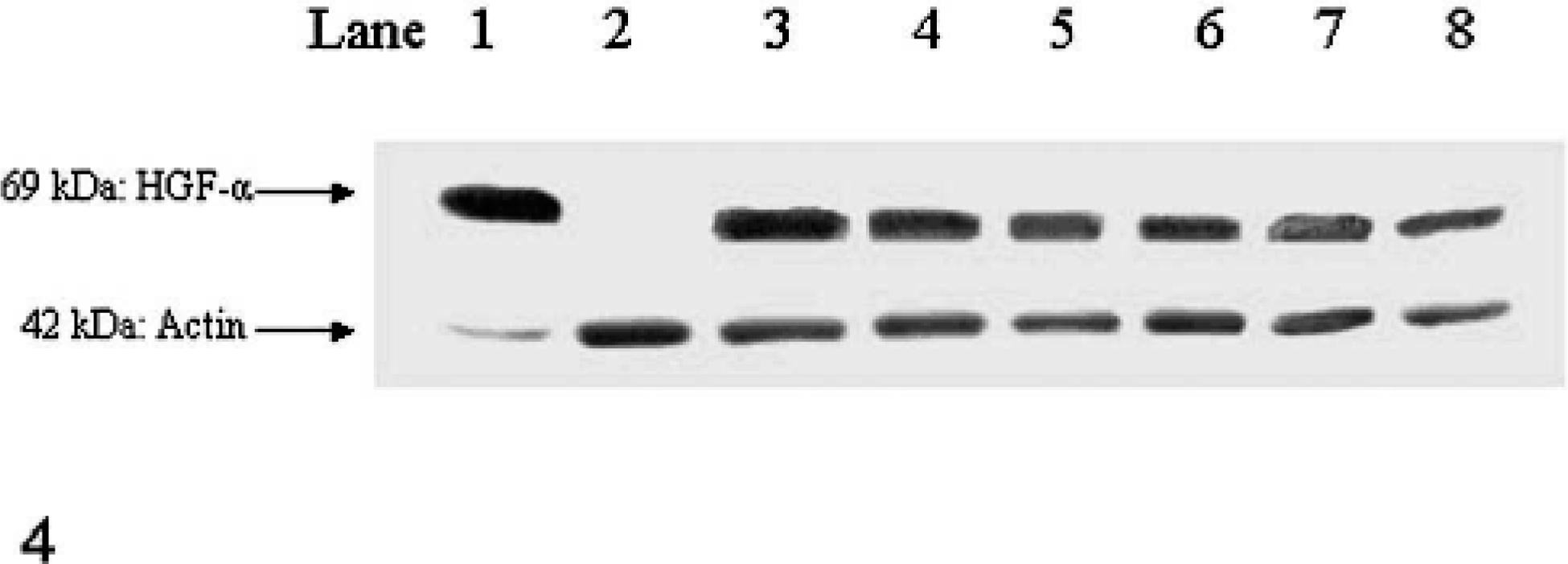
Western blot analysis for HGF alpha. In parentheses: relative HGF mRNA expression levels of canine osteosarcoma (OS) 1: Liver, 2: Bile duct epithelium, 3: OS (0.1), 4: OS (0.34), 5: OS (3.33), 6: OS (9.71), 7: OS (20.67), 8: OS (24.27).
Association between c-Met mRNA expression level and histologic characteristics in canine osteosarcoma
The majority of osteosarcoma was of high histologic grade: 47 samples were classified as high malignant, 10 samples as medium malignant, and 2 samples as low malignant. Twenty-six samples were of the osteoblastic subtype, the remaining 33 samples were mixed-type tumors. A negative association between the number of mitosis and c-Met mRNA expression level (P = .036) was revealed by multivariate stepwise linear regression analysis for histologic characteristics. As shown through ANOVA, c-Met mRNA expression was not associated with grade and metastasis in blood vessels. Draining lymph nodes of 23 dogs were evaluated. Of these lymph nodes, 3 contained metastatic osteosarcoma cells. A significant positive association of c-Met mRNA expression and metastasis in lymph nodes was discovered with ANOVA F(1, 22) = 7.042, P = .015.
Effects of c-Met mRNA expression on survival and disease-free interval
Dogs that were euthanatized on the day of diagnosis or were treated conservatively were excluded from the analysis (n = 21). Of the remaining 38 dogs, 32 dogs had appendicular osteosarcoma, 5 had an axial osteosarcoma, and one presented with an extraskeletal osteosarcoma. The majority of the osteosarcomas were graded as highly malignant (n = 29), followed by medium malignant (n = 8), and low malignant (n = 1). From this group of patients, 8 dogs underwent surgery without additional chemotherapy. Two dogs underwent limb amputation for appendicular osteosarcoma, a mandibulectomy was performed in 2 dogs, 2 dogs underwent ulnectomy, and an osteosarcoma of the rib was resected in 2 dogs. The remaining 30 dogs received chemotherapy in addition to surgical resection of the primary tumor.
Twenty-one dogs of the chemotherapy group underwent total limb amputation for appendicular osteosarcoma. Four dogs underwent scapulectomy, and the arcus zygomaticus was removed in 1 dog. In 1 dog, an extraskeletal osteosarcoma in the pectoral region was removed. In 3 dogs, limb sparing surgery for osteosarcoma of the distal ulna (n = 1) and distal radius (n = 2) was performed. Fifteen of the 30 dogs that received chemotherapy as additional therapy received lobaplatin at an intravenous dose of 35 mg/m2, once every 3 weeks, for a maximum of 4 doses as previously described, 15 5 dogs received carboplatin 300 mg/m2 every 3 weeks, and 8 dogs received alternating carboplatin 300 mg/m2 and doxorubicin 30 mg/m2 every 3 weeks. One dog received carboplatin, and, after metastatic disease was discovered, lobaplatin was given. One dog received doxorubicin monotherapy (30 mg/m2 every 3 weeks).
For statistical analysis of survival time, 38 patient records could be used, and 35 records were used for the analysis of disease-free interval. Three dogs were excluded from the disease-free interval analysis: one was excluded because the primary tumor in the vertebrae could not be completely resected, another was excluded because lung metastasis were present at the time of diagnosis, and a third developed lung metastasis, but the dog suffered from a histiocytic sarcoma at the same time, so the origin of the metastases was inconclusive.
Four cases were censored in both analyses: one dog was euthanatized because of a thoracic fibrosarcoma, one dog died after surgery for a herniated disc, one dog developed an anaplastic carcinoma at the amputation site, which had metastasized to the lungs, and the last dog was euthanatized because of an adenocarcinoma in the nasal sinus. In this analysis, no effect of c-Met mRNA expression levels on survival time (P = .795) or disease-free interval (P = .939) was detected.
Immunohistochemistry for c-Met
In normal canine bone samples, osteoblasts, osteoclasts, and the vast majority of all bone marrow cells showed strong positively staining cytoplasm for c-Met antibody. Some background reactivity was present in arterial smooth muscle cells, whereas endothelial cells, osteocytes, and striated muscle cells remained negative. Negative controls of bone tissues (n = 2) remained negative. Both human and canine prostate positive controls showed a strong cytoplasmic staining pattern in epithelial cells. The human osteosarcoma samples exhibited a strong cytoplasmic staining pattern in tumor cells. All tested canine osteosarcoma stained positive for c-Met antibody, with minor inter- and intratumor variability for the specific cell types. Background staining corresponded to the decalcified controls. Generally, neoplastic osteoblasts, fibroblasts, chondroblasts, and multinucleated giant cells exhibited strong cytoplasmatic positivity (Fig. 5).
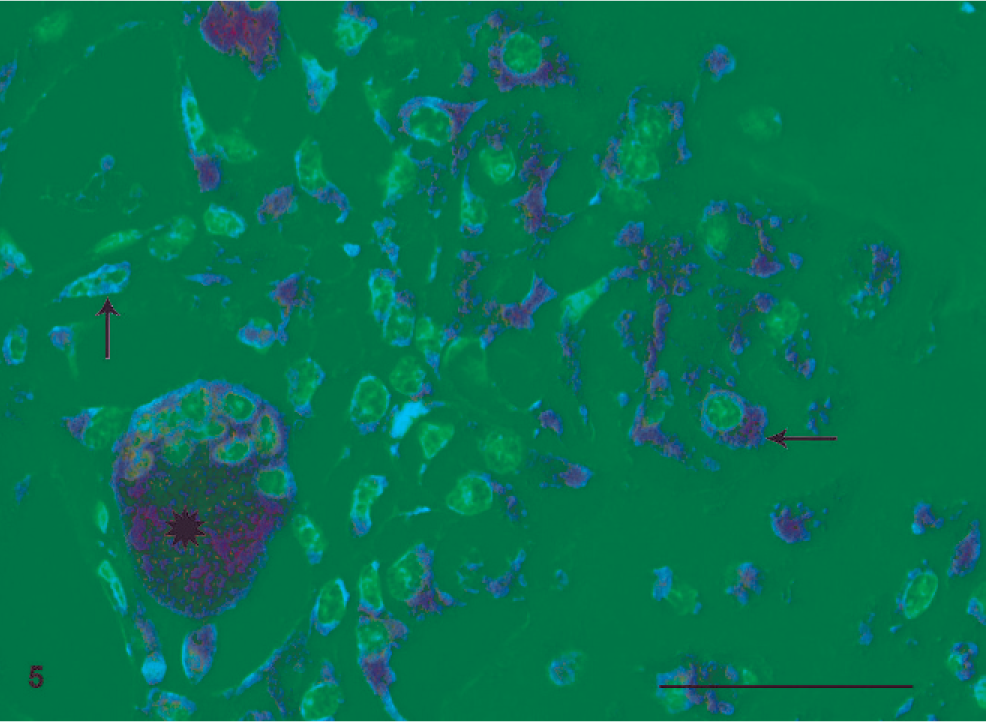
Scapula: mixed type osteosarcoma grade III; dog. Strong cytoplasmic staining pattern (brown) is present in multinucleated giant cells (asterisk), neoplastic osteoblasts (horizontal arrow), and neoplastic fibroblasts (vertical arrow). c-Met immunolabeling, Mayer's hematoxylin counterstain. Bar = 50 µm.
Discussion
In human carcinogenesis, the HGF and c-Met signaling systems were confirmed to play an important role in stimulating cell division and motility, resulting in the promotion of progression and metastasis in various epithelial and mesenchymal malignancies. 2 Coexpression of HGF and c-Met is reported in human osteosarcoma and indicates an autocrine- or paracrine-mediated signal transduction pathway. 8 The coexpression of HGF and c-Met in canine cancers had not previously been investigated in a large cohort. In the current study, we found expression of HGF and c-Met in all canine osteosarcoma samples. The difference between previously reported data on c-Met expression in canine osteosarcoma, 7 and the current report is most likely caused by the higher sensitivity of the Q-PCR detection method compared with Northern blotting.
Other researchers suggested that autocrine or paracrine stimulation, or overexpression of c-Met is responsible for activation of the receptor. 8 In the current study, HGF and c-Met mRNA expression were detectable in all osteosarcoma samples. This means that HGF was produced locally, which suggests an autocrine activation loop in our samples. Thus, the receptor was probably being activated through autocrine or paracrine stimulation.
For both HGF and c-Met, mRNA expression levels showed a wide variation. In the majority of cases, the expression of HGF and c-Met decreased compared with the reference genes. Variation in expression levels of HGF and c-Met might be caused by the great intra- and intertumor variability in morphologic appearance in canine osteosarcoma.
Because HGF is post-translationally modified, mRNA expression levels do not necessarily indicate the amount of active protein. Furthermore, the HGF protein can be produced by various tissues throughout the body. 28 Together, these two facts indicate that HGF mRNA levels in the tumor do not necessarily reflect the amount of active protein present locally. Therefore, Western blot analysis was added to measure the availability of active HGF protein. HGF protein levels were more or less comparable between the samples with highest and lowest mRNA expression levels (Fig. 4). This may indicate that, besides locally produced HGF, HGF supplied from surrounding tissues is present in the osteosarcoma samples. In conclusion, based on our findings, both an autocrine and paracrine mode of activation might play a role in the activation of the c-Met receptor in canine osteosarcoma.
This study showed no significant correlation between patient outcome and c-Met mRNA expression. A limitation in this study is the wide variability of ascertained cases and different treatment strategies. However, there was no significant difference in survival time between different treatment strategies applied. The findings reported here confirm results from a previous study that investigated the correlation of c-Met expression and outcome in dogs. 7 Although an eightfold larger number of cases was analyzed and a more sensitive technique was used (Q-PCR in our study versus Northern blotting in the previous study), the lack of correlation of c-Met expression and patient outcome is concluded in both the study by Ferracini et al. 7 and in the current study. Similar conclusions were reported in human osteosarcoma. 17,19 Based on our findings, c-Met expression in tumor samples cannot be used as a predictor for prognosis in canine osteosarcoma. A crucial causative role for the c-Met pathway in progression and hematogenous metastasis of canine osteosarcoma is not confirmed based on our results.
A positive relationship was found between lymph-node metastasis and c-Met mRNA expression. Canine osteosarcoma metastasize mainly through the hematogenous route, and lymph node metastases are rare. 6 The 3 dogs in our study with lymph-node metastasis all had high-grade osteosarcoma of the appendicular skeleton and were treated with amputation and chemotherapy: one dog died from lung metastasis, the other two dogs (with both increased c-Met and HGF expression) did not develop lung metastasis. This correlation between c-Met expression and the lymphogenic metastatic pathway has not been described previously in either human or canine osteosarcoma, however, c-Met and stromal HGF were found to significantly correlate with regional lymph-node metastasis in human head and neck squamous cell carcinoma, salivary gland carcinoma, and mammary carcinoma. 4,11,25 Determining the significance of the role of c-Met in lymphogenic metastasis in canine osteosarcoma requires studies of a larger number of dogs with lymph-node metastases.
A negative association was detected between number of mitoses, counted histologically, and c-Met expression. This was an unexpected finding, because the c-Met pathway has been previously reported to be associated with a stimulation in cell division. 2 To confirm the importance of this finding and to quantify expression levels of HGF and c-Met in a cell-specific way, laser microdissection of tumor cells and subsequent RNA isolation and Q-PCR are recommended to exclude noise because of expression of HGF and c-Met in reactive bone and stromal component present in whole tumor samples.
In the immunohistochemical study with c-Met antibody, a cytoplasmatic staining pattern was detected in the majority of tumor cells. The histology is similar to the staining pattern in the human osteosarcoma samples, which were used as a positive control. Several immunohistochemical studies for c-Met are performed on human osteosarcoma. A diffuse, cytoplasmatic staining pattern with or without a linear membranous staining pattern is described in human osteosarcoma, which is concordant with what we described in canine osteosarcoma. 1,9,17,19,22,26 The proportion of c-Met–positive tumors in human studies varies between studies. Some studies report c-Met positivity in the majority of tumors, 1,22,26 whereas other studies detect c-Met positivity only in several samples. 9,17,19 c-Met positivity in osteoblastic cells, multinucleated giant cells, and fibroblastic tumor cells between canine and human osteosarcoma is comparable; however, one study describes a negative staining in fibroblastic tumor cells. In this study, however, the overall positivity of c-Met in tumor samples, as well as in cellular subtypes, except for the chondroblastic subtype, was low. 17 Differences in c-Met detection with immunohistochemistry could be because of differences in c-Met expression. In the cited human studies herein, no expression profiling were performed to study c-Met expression on the mRNA level. Important factors that will account for differences in the number of positive tumors are the following: differences in tissue preparation, fixation (frozen versus formalin sections), formalin fixation time, decalcification (time and method), and used antibodies. For future immunohistochemical studies, we propose a standardized protocol for decalcification and formalin fixation for osteosarcoma samples to obtain comparable staining patterns.
This study is the first to describe mRNA expression levels of HGF and c-Met in a large number of canine osteosarcoma samples. This coexpression of HGF and c-Met indicates an autocrine activation loop. In addition, Western blot analysis of active HGF protein indicated that a paracrine loop of activation might occur as well. Immunohistochemistry detected c-Met protein of all different cell types within the osteosarcoma cells. No major effect of c-Met expression on patient outcome is detected; therefore, c-Met expression in tumor tissue is not recommended as a prognostic indicator. c-Met seems involved in the lymphogenic route of metastasis, but a larger number of samples is necessary to prove this effect. We conclude that the c-Met pathway is present in canine osteosarcoma. The effects described here are very similar to effects previously described in human osteosarcoma, but the specific effects of the c-Met pathway in canine osteosarcoma on the biological level require further investigation. For further studies, we would propose tissue collection and array analysis in a matched fashion. To elucidate c-Met–related and other pathways in canine osteosarcoma, we would propose microarray analysis for a hypothesis free approach.
Footnotes
Acknowledgements
We thank Prof. P. C. W. Hogendoorn, Dr. A. M. Cleton-Jansen, and I. H. Briaire-de Bruijn from the Leiden Universitair Medisch Centrum, Leiden, The Netherlands, for assisting in immunohistochemical staining and kindly providing the antibody for c-Met. We also thank Dr. E. Teske, Dr. S. M. van den Bergh, and Dr. J. van den Broek for statistical advice. We thank Daniel J. Compton and Linda McPhee for critically reviewing the manuscript.
