Abstract
Bovine spongiform encephalopathy (BSE) is a transmissible spongiform encephalopathy of cattle, first detected in 1986 in the United Kingdom and subsequently in other countries. It is the most likely cause of variant Creutzfeldt-Jakob disease (vCJD) in humans, but the origin of BSE has not been elucidated so far. This report describes the identification and characterization of two cases of BSE diagnosed in the United States. Case 1 (December 2003) exhibited spongiform changes in the obex area of the brainstem and the presence of the abnormal form of the prion protein, PrPSc, in the same brain area, by immunohistochemistry (IHC) and Western blot analysis. Initial suspect diagnosis of BSE for case 2 (November 2004) was made by a rapid ELISA-based BSE test. Case 2 did not exhibit unambiguous spongiform changes in the obex area, but PrPSc was detected by IHC and enrichment Western blot analysis in the obex. Using Western blot analysis, PrPSc from case 1 showed molecular features similar to typical BSE isolates, whereas PrPSc from case 2 revealed an unusual molecular PrPSc pattern: molecular mass of the unglycosylated and monoglycosylated isoform was higher than that of typical BSE isolates and case 2 was strongly labeled with antibody P4, which is consistent with a higher molecular mass. Sequencing of the prion protein gene of both BSE-positive animals revealed that the sequences of both animals were within the range of the prion protein gene sequence diversity previously reported for cattle.
Introduction
Transmissible spongiform encephalopathy (TSE) agents or prions induce fatal neurodegenerative diseases in humans and in other mammals. They are transmissible among their species of origin, but they can also cross some species barriers and induce infection with or without disease in other species. Human TSEs include Creutzfeldt-Jakob disease (CJD), Gerstmann-Sträussler-Scheinker syndrome, Kuru, and fatal familial insomnia. 38 In animals, 4 distinct TSE diseases are recognized: scrapie in sheep and goats, transmissible mink encephalopathy (TME) in mink, chronic wasting disease (CWD) in cervids, and bovine spongiform encephalopathy (BSE) in cattle. BSE is transmissible via BSE-contaminated feed to felines (feline spongiform encephalopathy, FSE) and exotic ungulates (exotic ungulate encephalopathy, EUE). 50,51
Prions are proteinaceous infectious particles and are the causative agents of TSEs. They are host-coded proteins that have undergone conformational changes and have biological and physicochemical characteristics that differ significantly from those of other infectious agents. For example, they are resistant to inactivation processes that are effective against conventional viruses including those that alter nucleic acid structure or function. These include ionizing and UV radiation, 1 and inactivation by formalin. 21 In contrast, infectivity is highly susceptible to procedures that modify protein conformation. 41 In TSE disease, the normal cellular protein, PrP c , is converted to abnormal prion protein, PrPSc. PrPSc exhibits increased beta sheet content, a change that may drive the additional changes in solubility and protease resistance. 40 Unlike normal cellular protein, PrPSc is relatively insoluble in detergents, is relatively resistant to proteases, 39 and is capable of causing a conformational change in additional molecules of PrP c . The precise function of the normal PrP c in healthy animals remains unknown. There is some evidence to show that PrP c might play a role in sleep physiology, in resistance to oxidative stress, in signal transduction, and in self-renewal of hematopoietic stem cells. 17,31,33,55
TSE disease involves the accumulation of PrPSc in the central nervous system (CNS) of the host, eventually leading to neurodegeneration and disease. In TSE-affected animals, PrP c has a determinant role in the incubation time and species barrier. 8 Mice lacking prion protein gene (Prnp) expression are not susceptible to TSE agents or prion infection, demonstrating the key role of PrP c in TSEs. 8 Susceptibility to prions thus depends upon the presence of PrP c on the cell membrane of the host; prions do not propagate in brains that lack PrP c . 6
BSE first emerged in 1986 in the United Kingdom, where more than 180,000 cases have been diagnosed to date (August 2006). Widely referred to as “mad cow disease”, BSE subsequently spread to many countries, predominantly in Western Europe. These outbreaks, caused by the consumption of infected meat and bone meal containing the malformed prion protein, have resulted in the destruction of thousands of cattle and have caused significant economic losses. BSE is a chronic degenerative disease affecting the central nervous system of cattle. Affected animals display changes in temperament, abnormal posture, incoordination and difficulty in rising, decreased milk production, and/or loss of body weight despite continued appetite. 42 The average incubation period is about 4–6 years and all affected animals succumb to the disease. 30 Following the onset of clinical signs, the animal's condition deteriorates until it either dies or is destroyed. This process usually takes from 2 weeks to 6 months. Most cases in the United Kingdom occurred in dairy cows between 3 and 6 years of age, with the highest susceptibility to infection being in the first 6 months of life; adult cattle appear to be at relatively low risk of infection. 3
Epidemiological surveillance programs carried out in many European and non-European countries have discovered BSE-positive animals within the last decade. 18,36 In May 2003, Canada reported its first indigenous case of BSE, detected as part of the Canadian BSE surveillance program on a commercial cow-calf operation in Alberta, Canada 13,45 . Since then, Canada has reported 7 additional cases of BSE (two in 2005, five in 2006 — as of August 2006), all of them detected as part of the ongoing Canadian BSE surveillance and the majority of cases stemming from a distinct area in Alberta.
All currently validated diagnostic tests for BSE require brain tissue. 35,49 There is currently no validated ante mortem test for BSE. The original diagnostic test method was histopathology in which brain sections exhibiting the classical vacuoles and spongiform changes in specific areas are used for diagnosis. 35 In the mid-1990s, immunohistochemistry (IHC) and Western blotting were developed for the detection of PrPSc in tissues. 35 Both IHC and Western blot are considered confirmatory tests for BSE by the World Organization for Animal Health (OIE). 35 In the past decade, “rapid tests” have been introduced commercially for BSE surveillance. 35
This report describes the identification and characterization of 2 BSE cases diagnosed in the United States. The first US BSE case (December 2003) was identified by using the BSE surveillance system that was in place in the United States from May 1990 until May 2004. Using this system, brainstem samples from field cases of cattle exhibiting signs of neurologic disease, cattle condemned at slaughter for neurologic reasons, rabies-negative cattle submitted to public health laboratories, neurologic cases submitted to veterinary diagnostic laboratories and veterinary teaching hospitals, and cattle that were nonambulatory (downer cattle/fallen stock) were sent to the National Veterinary Service Laboratory (NVSL) for confirmatory IHC testing. Before 1995, BSE testing at the NVSL was done by histological analysis of brain sections for the presence of BSE-typical spongiform lesions. After 1995, testing included the use of the newly established IHC procedures for the detection of PrPSc in brain sections of TSE-infected animals. 30 After the first case of BSE in the United States was diagnosed, an enhanced BSE surveillance program was established on June 1, 2004, based on the use of a rapid screening test a , followed by confirmatory testing (IHC and/or Western blot, since June 2005 both methods are used in parallel) for any reactive sample, designated “inconclusive.” As of August 6, 2006, more than 775,000 samples have been tested and 2 samples were found to be BSE positive. Currently, an “inconclusive” sample is defined as being positive by the initial one-well ELISA and again positive in at least 1 out of 2 wells in a repeat test of the original homogenate. USDA has designed its enhanced testing program to collect the majority of samples from high-risk animals in the following categories: nonambulatory cattle, cattle exhibiting signs of a central nervous system disorder, cattle exhibiting other signs that may be associated with BSE, such as emaciation or injury, and dead cattle. In addition, all cattle condemned on ante mortem inspection are sampled. In this report, detection of PrPSc by immunohistochemistry and Western blot analyses are presented. Brain material from both cases is compared with each other and with well-defined BSE isolates from other countries. In addition, sequences of the prion protein gene of both BSE-positive animals are discussed.
Material and methods
Animals and tissues
Case 1 was an approximately 6.5-year-old, nonambulatory cow, slaughtered in December 2003 in Moses Lake, Washington State. The animal was imported from Canada in 2001. Case 2 was a downer cow, approximately 12 years old, and sent to a pet food plant in November 2004. The animal was born and raised in Texas. Brain samples were taken from both animals according to the USDA BSE surveillance plan and shipped to the National Veterinary Service Laboratories (NVSL) in Ames, Iowa. The obex sample from Case 2 was found to be reactive twice in a rapid BSE test performed by the Texas Veterinary Medical Diagnostic Laboratory at Texas A&M University, College Station, TX, before being shipped to NVSL. The rapid BSE test a used in the United States has been validated by the European Union, Canada, and the United States and has demonstrated both high sensitivity and specificity for detecting BSE prion protein in cattle. Frozen and formalin fixed samples from the medulla oblongata and the cerebellum were available for analysis. No portion of either cow's carcass entered the animal or human food chain.
Histopathology
Brain tissue was fixed in 10% buffered formalin, embedded in paraffin wax, sectioned at 5 micron thickness, and stained with hematoxylin and eosin (HE) for light microscopic examination.
Immunohistochemistry (IHC)
Brain tissue was placed in 10% buffered formalin and after a minimum of 4 days of fixation appropriate sections of obex were put in cassettes and kept in fresh formalin until they were processed for routine paraffin embedding. Using automated tissue processing, tissues were cut at 5 micron thickness and mounted onto charged glass slides b . Slides were set upright, drained and air-dried for a minimum of 3 hr, followed by 15 min at 80°C. The slides were then deparaffinized (xylene, ethyl alcohol gradient, distilled H2O). Additionally, 5-micron sections of US BSE case 2 were treated with 95% formic acid for 5, 15, and 30 min at room temperature. Slides were incubated with a target retrieval solution c , placed in a medical decloaking chamber d , autoclaved for 30 (case 1) or 30 and 45 (case 2) min at 121°C, and then cooled for 25 min. Slides were soaked for a minimum of 5 min in Ventana APK Wash Solution e . The Ventana NexES carousel e , filled with APK Wash Solution e and Liquid Coverslip bottles e , was used with the PrP-specific antibody F99/97.6.1 f at a concentration of 10 μg/ ml. Slides were processed to completion using alkaline phosphatase red paraffin protocols as suggested by the manufacturer e . Then, slides were removed, placed in a rack and dipped thirty times in 250 ml of warm soapy tap water containing 2–3 drops liquid dishwashing detergent g . Afterwards, slides were rinsed in running tap water for 2 min and dehydrated in ethyl alcohol and xylene. Coverslips were added to slides and IHC results were interpreted as follows: 1) positive for PrPSc: pink to red and 2) background and negative for PrPSc: only blue background. It should be noted that the IHC procedure did not incorporate a proteinase K digestion step. For the purpose of simplicity of nomenclature, the term “PrPSc” is used to describe the abnormal prion protein. As positive controls, slides from the brainstem of a BSE-positive cow, obtained from the United Kingdom, and from the brainstem of a scrapie-positive sheep were used for Case 1; slides from brainstem of a BSE-positive cow obtained from the United Kingdom and from the US BSE Case 1 were used for Case 2. As negative controls, slides from brainstem material of BSE-negative cattle and scrapie-negative sheep were used.
Western blot analyses
Brain homogenates from US BSE case 1 were prepared from approximately 0.5 gram brainstem material and analyzed using the Prionics®-Check Western Kit h as suggested by the manufacturer with minor modifications regarding the detection system. It should be noted that the Prionics®-Check Western Kit h method is not a confirmatory assay for BSE. Samples were homogenized at room temperature with homogenization buffer h (10% w/v) using a PowerGen125 homogenizer i with a disposable probe i (5 times, 30 sec), and digested with proteinase K (PK) h for 40 min at 48°C. PK-digestion was stopped according to the manufacturer's protocol and 10–15 μl volume of sample was loaded onto pre-cast sodium dodecyl sulfate (SDS)-12% polyacrylamide gel electrophoresis (PAGE) gels j . SDS-PAGE was performed as described by the manufacturer and the proteins transferred from the gel to a polyvinylidene difluoride (PVDF) k membrane with transfer buffer h . The membranes were blocked with PVDF blocking buffer j and either incubated with antibody 6H4 h (1:10,000 dilution or 0.1 μg/ml of a mouse IgG1 monoclonal antibody antiserum raised against human residues 144–152 [DYEDRYYRE] of the PrP peptide) or antibody P4 l (1:10,000 dilution or 0.1 μg/ml of a mouse monoclonal antibody raised against synthetic ovine PrP residues 89–104 [GGGGWGQGGSHSQWNK] of the PrP peptide) for 1 hr at room temperature or overnight at 4°C. After three washes in Tris-Buffered Saline Tween®-20 (TBST) j , the membranes were incubated for 60 min at room temperature with a biotinylated sheep anti-mouse antibody m (1:10,000 dilution or 0.05 μg/ml in TBST). After 3 washes in TBST j , a streptavidin-horseradish peroxidase conjugate m was added for 60 min. After another round of washes with TBST, bound antibodies were detected by using the ECL Plus m chemiluminescent substrate.
For US BSE case 2, the Prionics®-Check Western Kit h and the OIE-recommended Scrapie Associated Fibril (SAF)-Immunoblot method (http://www.oie.int/eng/normes/mmanual/A_summry.htm) were used 35 with minor modifications. In contrast to the Prionics®-Check Western h method, the SAF Immunoblot method enriches brain samples for PrPSc by ultracentrifugation prior to loading them onto a SDS-PAGE gel. The SDS-PAGE electrophoresis conditions, subsequent transfer and immunodetection of PrPSc were carried out as described above. Therefore, only the enrichment method will be described in more detail. Material for analysis of case 2 was taken from the brainstem (2 g) and cerebellum (2 g) area and minced with a new blade after removal of dura mater. A 10% (w/v) tissue homogenate in 10 mM Tris, pH 7.5, containing 5 mM MgCl2 was prepared using a homogenizer g with a disposable probe g (5 times, 30 sec). The homogenate was mixed well and then again sonicated for 30 sec on ice bath (5-10 times). Benzonase®n was added to the mixture for a final concentration of 100 U/ml and incubated for 1 hr at 37°C while shaking. An equal volume of 20% (w/v) N-lauroylsarcosine o in 10 mM Tris, pH 7.5 and 1 mM DTT o was added to each homogenate, vortexed for 1 min every 10 min for a total of 30 min at room temperature. Homogenates were transferred to polyallomer tubes p and centrifuged at 20,000 × g for 25 min at 10°C. Supernatant was centrifuged again using polyallomer tubes p at 200,000 × g for 55 min at 10°C. The resultant supernatant was discarded, the pellet was resuspended in sterile, distilled H2O (1 μl/mg tissue equivalent) and sonicated until suspended. Sample was split into 2 aliquots into microcentrifuge tubes and one sample was treated with PK q (concentration 0.4 U/ml) by incubation at 37°C for 60 min with agitation while the control sample was not treated with PK. Phenylmethylsulphonyl fluoride r (PMSF) was added to a final concentration of 5 mM, incubated on ice for 15 min and transferred to a new 1.5-ml ultracentrifuge tube. Volume was brought up to 500 μl with H2O and centrifuged at 200,000 × g for 1 hr at 10°C. Pellet was resuspended in SDS-PAGE sample buffer to at least 10 mg tissue equivalent per μl. Samples were sonicated on wet ice before loading on SDS-PAGE gel. For both Western blot techniques, detection was performed either on Biomax films s or scanned images were obtained with a Typhoon m imaging system. As positive control samples, BSE-positive brain material from Canadian and Swiss cattle, cattle-and sheep-passaged scrapie and CWD-positive elk and mule deer material were used for Case 1, brain material from Case 1 and sheep scrapie were used for Case 2. As negative controls, brain material from a BSE-negative cow was used.
DNA isolation and PCR amplification
Genomic DNA was extracted from 200 μl of a 10% brain homogenate (cerebellum) using the DNeasyTM tissue kit t according to the manufacturer's instructions. PCR was performed in a 100 μl final reaction volume containing 0.2 pmole of forward primer (5′-CAT ATG ATG CTG ACA CCC TC −3′), 0.2 pmol of reverse primer (5′-AGA AGA TAA TGA AAA CAG GAA G-3′)1X Easy-A PCR buffer, 2.5 mM MgCl2, 0.8 mM each deoxyribonucleotide triphosphate (dNTP Master Mix) u , 2.5 U of Easy-A™ high-fidelity cloning Taq DNA polymerase v , and 0.4 ug of total DNA. Amplification was performed with the following conditions: 94°C for 5 min, followed by 30 cycles of 94°C for 30 sec (denaturation), 59°C for 30 sec (annealing), and 72°C for 1 min. A final extension step at 72°C was performed for 10 min. Amplified DNA product was purified using a GENECLEAN® spin kit w and sequencing was performed using the ABI 3700 DNA sequencer with the cycle sequencing kit x . The fragment was sequenced in duplicate using the original 2 primers and 2 internal primers 4142 and 9612 4 for a total of 8 reactions.
Databases were searched using standard nucleotide-nucleotide BLAST at the National Center for Biotechnology Information Web Site (http://www.ncbi.nlm.nih.gov). The database is a collection of sequences from several sources, including GenBank, the Reference Sequence and the Protein Data Bank. The nucleotide sequences of cases 1 and 2 were aligned using both CLUSTAL V 26,27 and CLUSTAL W 48 with the following GENBANK accession numbers: AY335912 (bovine), AY367641 (bovine), AF016227 (elk), AY275712 (white-tailed deer), AF166334 (sheep), AJ567986 (sheep), and the Canadian BSE case using Lasergene version 5.07 software (DNASTAR-Madison WI).
Results
ELISA results
When brainstem samples from case 1 and case 2 were repeatedly tested for the presence of PrPSc using a validated ELISA test system, the mean optical density (OD) values were 1.86 for case 1 (n = 2) and 2.49 for case 2 (n = 6).
Histological and immunohistochemical examination
Brainstem samples at the level of obex were available from both cases and analyzed for the presence of spongiform changes and deposition of PrPSc. Lesions of spongiform encephalopathy diagnostic for BSE were detected in the obex region of case 1 (Fig. 1A), and were not present in the obex of case 2 (Fig. 1C). The tissue of case 2 had been frozen and therefore artifactual changes were present, but definitive lesions of BSE were not observed (Fig. 1C). In both cases the medulla at the level of the obex was examined. In case 1 there was neuropil vacuolation present in several areas and occasional scattered neuronal vacuolation. The neuropil vacuolation was most pronounced in the solitary nucleus and tract, the spinal nucleus and tract of the trigeminal nerve, the olivary nuclei, and less pronounced in the motor nucleus of the vagus. In case 2 there was a freezing artifact that precluded definitive histological interpretation of vacuolar changes, however overt TSE related vacuolar lesions were not observed. When IHC was performed, both cases were positive for the presence of PrPSc. Whereas significant amounts of PrPSc were detected in the obex area of case 1 (Fig. 1B), only weak staining for PrPSc was observed in the obex area of case 2 (Fig. 1D). Initially, the IHC of case 2 was negative, however, after formic acid treatment and extended antigen retrieval the case was positive by IHC (Fig. 1D). Extended antigen retrieval (up to 45 minutes) and the treatment of the slides with formic acid prior to immunostaining were necessary to obtain unambiguous PrPSc signals. The distribution of PrPSc in brainstem of case 2 was not as uniform nor as intense as seen with case 1 (Fig. 1B). Case 1 had intense widespread immunostaining throughout the obex. This included most nuclei and gray mater neuropil. Both intracellular and extracellular staining was present. Case 2 also had widespread neuropil and intracellular immunostaining however the staining was markedly less intense than case 1 and more prominent in the lateral and ventral portions of the section as compared to case 1. The staining was most pronounced in the nucleus of the solitary tract and the nucleus of the spinal tract of the trigeminal nerve as well as the olivary nuclei.
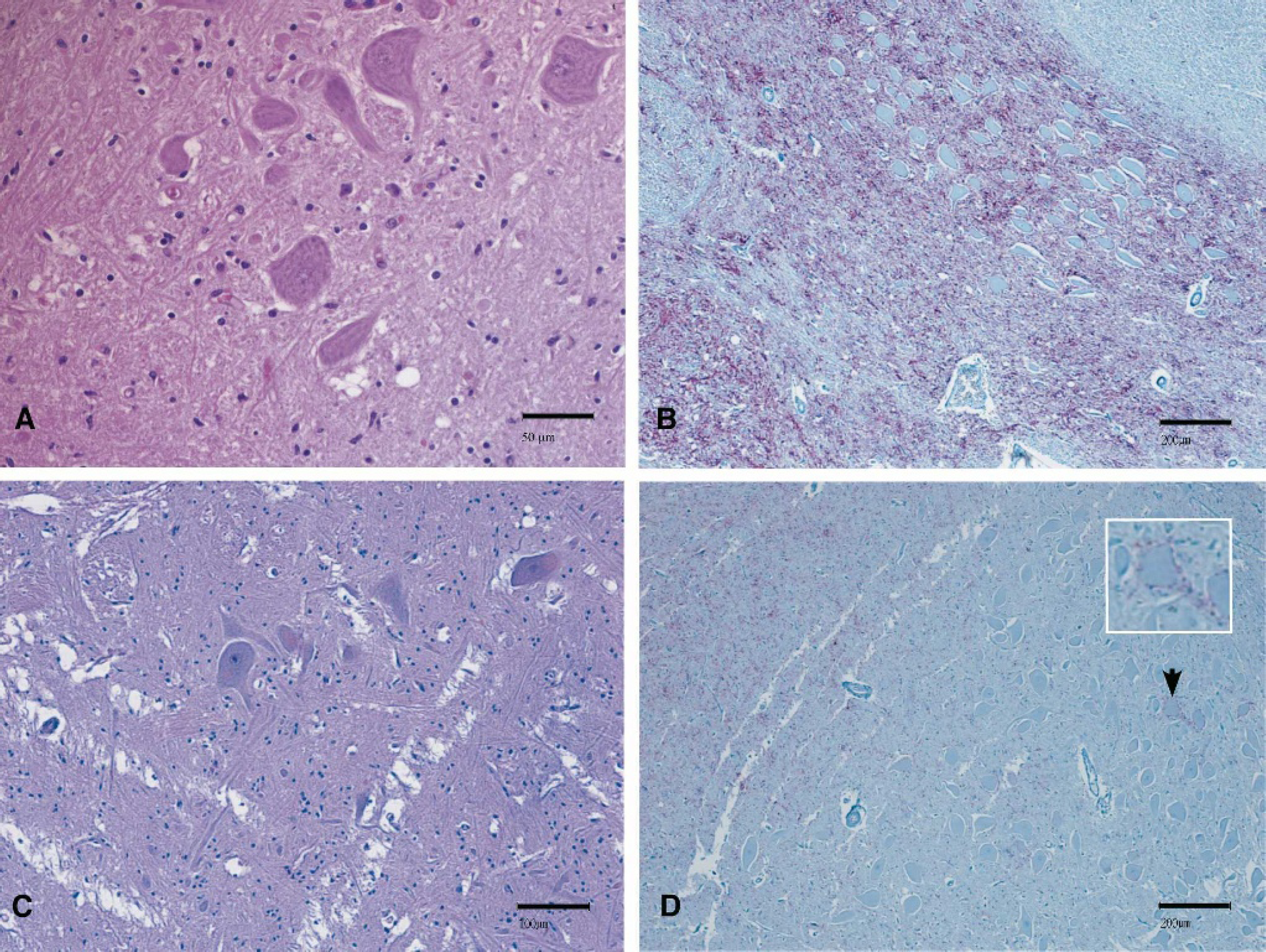
Histopathology and IHC on brain samples from US BSE cases. In both cases the medulla at the level of the obex was examined. US BSE case 1 is depicted in
Western blot analysis
Western blot analysis (employing the Prionics®-Check Western Kit f ) of brainstem homogenates of case 1 revealed a strong positive reaction at 1 mg tissue equivalent (Fig. 2A-C). All 3 isoforms of PrPSc were easily detected using the monoclonal antibody 6H4 and showed similar molecular masses when compared to the May 2003 Canadian and a Swiss BSE isolate (Fig. 2B). The glycoform profile of the 3 PrPSc-isoforms of case 1 was determined by analyzing 5 independent Western blots using the Typhoon m imaging system employing software ImageQuant 5.2 m . The diglycosylated isoform was the most prominent isoform (72.0% ± 4.7), followed by the monoglycosylated (23.3% ± 3.6) and the unglycosylated (4.0% ± 1.2) isoforms. The unglycosylated isoform of case 1 showed a lower molecular weight when compared with mule deer CWD, sheep-passaged mule deer CWD, elk CWD and sheep scrapie samples (Fig. 2A, B). Using a hybrid Western blot employing monoclonal antibodies P4 and 6H4, 44 all TSE-infected samples reacted well with antibody 6H4, however, US BSE case 1 and a cattle-passaged scrapie isolate did not react with antibody P4 (Fig. 2C, D). Sheep scrapie and mule deer CWD reacted with both antibodies. As already discussed above (see Fig. 2A), the relative molecular mass of the unglycosylated isoform of the mule deer CWD isolate and the two sheep scrapie isolates was higher than the respective isoform of case 1 (Fig. 2C).
Brain material (brainstem and cerebellum) from case 2 was analyzed by Western blot with and without enrichment of PrPSc. It should be noted that it was not possible to determine the precise anatomical location where the samples were taken, since the material was frozen and thawed several times before Western blot analysis was performed. Nonenriched samples (Prionics®-Check Western Kit f ) were analyzed at a concentration of 1–1.5 mg brain tissue equivalent (mg eq) and enriched samples (SAF Immunoblot) at a concentration of 10–150 mg eq. As shown in Fig. 3A, brainstem from case 2 was definitely positive after enrichment (≥20 mg eq), whereas the nonenriched sample (1 mg eq) was inconclusive. All 3 isoforms of PrPSc were definitely present at 20 mg eq with a strong reaction at 40 and 80 mg eq (Fig. 3A). In addition, brainstem and cerebellum samples were taken from different regions of the respective tissues and analyzed by enrichment Western blot. However, only 2 out of 4 cerebellum and 3 out of 6 brainstem samples were positive at 20 mg eq (data not shown). Glycoform profile analysis (analyzing 4 independent Western blots) revealed a significant proportion of the diglycosylated isoform (61.2% ± 2.9), and lesser amounts of the monoglycosylated (29.8% ± 2.3) and unglycosylated (9.0% ± 2.5) isoforms. Furthermore, cerebellum and brainstem samples from case 1 and case 2 were compared side by side employing monoclonal antibody 6H4. All of these samples, except for the sheep scrapie control sample, were enriched for PrPSc. The unglycosylated and monoglycosylated isoforms of PrPSc from case 2 migrated with an apparent molecular mass higher than the respective isoforms of case 1 (Fig. 3B, 3C), indicating a relative difference in molecular mass between the two BSE cases. The reaction pattern was very similar, independent of the region of the brain (brainstem or cerebellum) used for analysis (Fig. 3B, 3C). In hybrid Western blots, enriched cerebellum samples from case 1 reacted strongly with antibody 6H4 (Fig. 3C) and either weakly or not at all with antibody P4 (Fig. 3D), a similar reaction pattern as described for non-enriched brainstem samples of case 1 (Fig. 2C, 2D). Cerebellum samples from case 2 reacted strongly with both antibodies, 6H4 and P4 (Fig. 3C, 3D). The intensity of the reaction of case 2 brain material with antibody P4 (Fig. 3D) was even stronger than with 6H4 (Fig. 3C) at similar milligram equivalent amounts. The sheep scrapie control sample showed a similar antibody reaction pattern as seen for case 2 (Fig. 3C, 3D).
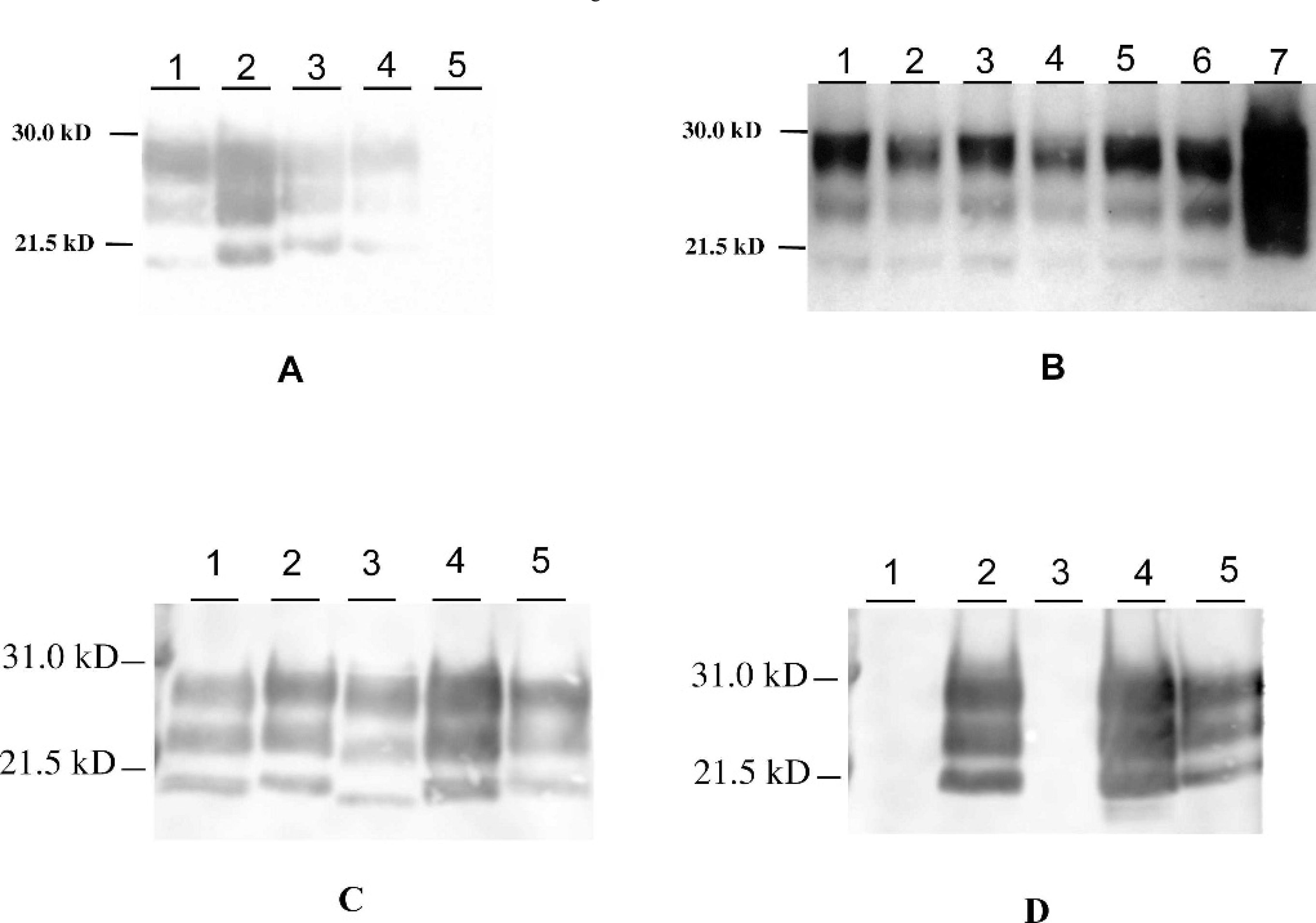
Western blot analysis of US BSE case 1.
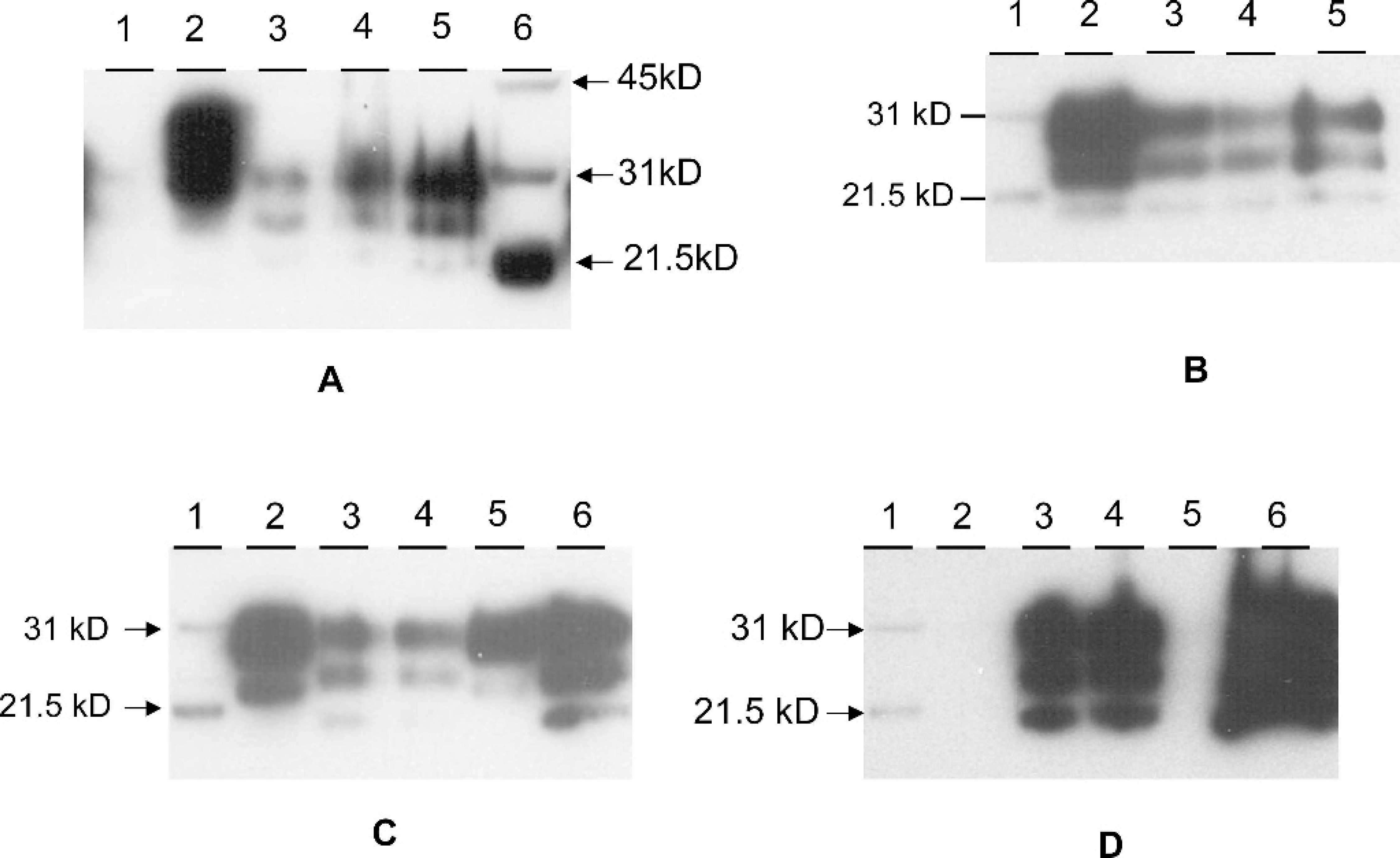
Western blot analysis of US BSE case 2.
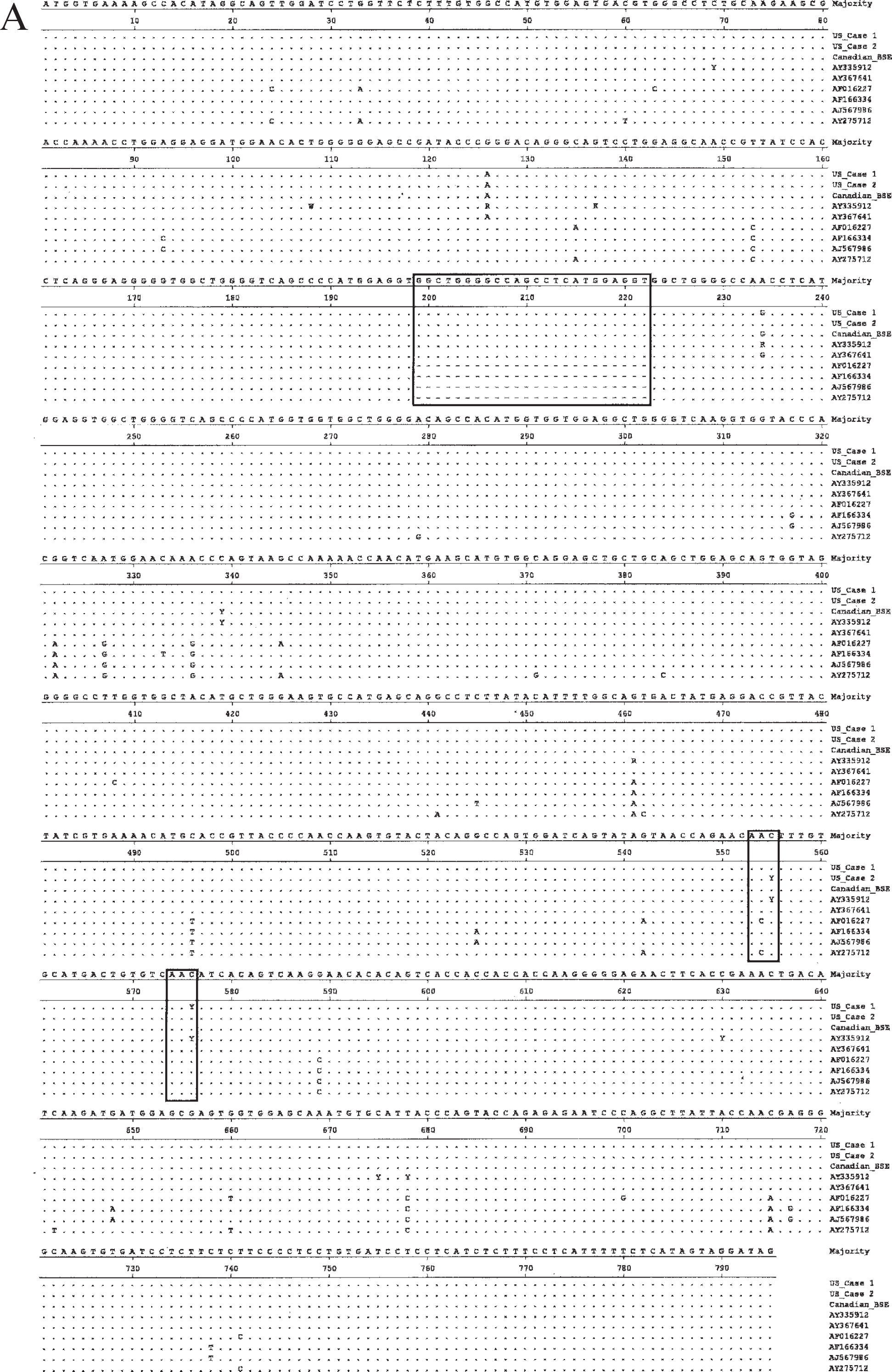
Alignment of bovine, ovine and cervid Prnp sequences including US BSE cases 1 and 2.
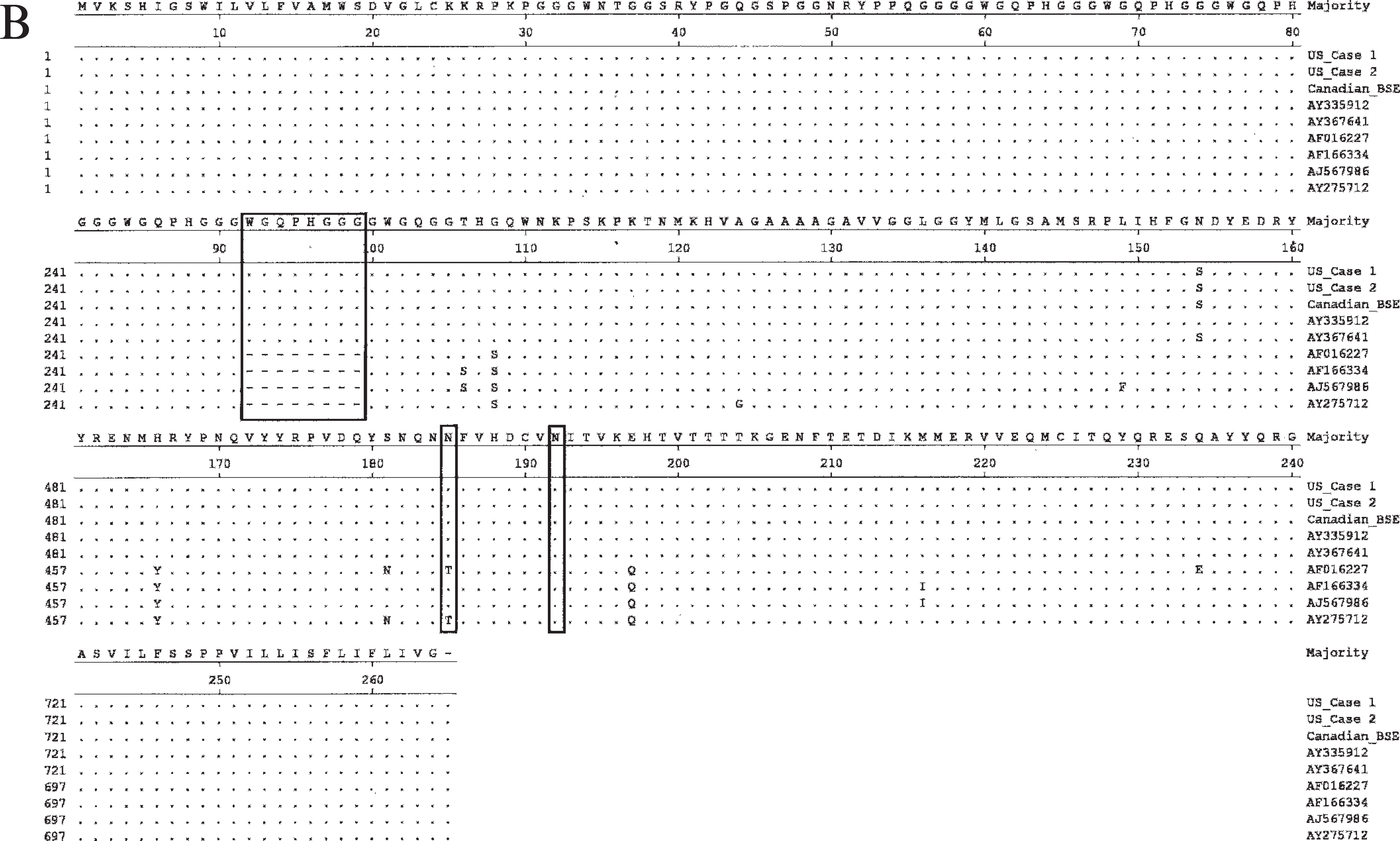
Continued.
Analysis of the Prnp
In order to confirm that the specimens from both cases were of cattle origin and to determine whether they might be associated with a spontaneous germline mutation, the full coding sequence from exon 3 of the Prnp was amplified and aligned with known PrP sequences from cattle, sheep, and cervids. DNA was isolated from brainstem material of both cases. As shown in Fig. 4A, the DNA sequence of both cases was identical to representative bovine Prnp sequences, but different from sheep and cervid sequences. The Prnp sequence of case 1 was found to be normal with a synonymous polymorphic position at codon 192 (T or C; no amino acid substitution). The animal had 6 copies of the octapeptide repeat region on both of their Prnp alleles. The Prnp gene of case 2 was also found to be normal with a synonymous polymorphism at codon 185 (T or C; no amino acid substitution), and both alleles contained the 6-copy octapeptide repeat region. The amino acid sequences of the 2 cases did not differ from each other and other bovine PrP protein sequences, however, there were differences when compared with the sheep and cervid sequences (Fig. 4B).
Discussion
The BSE epidemic in the United Kingdom was a food-borne disease in cattle, associated with feeding of meat and bone meal (MBM) that contained BSE infected central nervous system tissue. 52 The cause of the original case or cases of bovine spongiform encephalopathy (BSE), however, remains an enigma. Sheep- or goat-derived scrapie-infected tissues included in the MBM rations fed to cattle or a previously undetected sporadic bovine TSE (maybe due to a germline mutation in the Prnp of affected cattle) have been considered as possible origins. Recently, another theory namely that BSE originated from a human TSE through animal feed containing imported mammalian raw materials contaminated with human remains from the Indian subcontinent has been brought forward. 12 Unlike CWD and scrapie, there is little evidence for either direct horizontal or vertical transmission of BSE among cattle. 2,53 Since the introduction of a ban on the use of ruminant proteins for ruminant feed in affected countries, the incidence rate of BSE in these countries has been steadily declining. 47
This report presents the prion protein polypeptide profile and genotype from 2 cases of BSE diagnosed in the United States in 2003 and 2004. The obex area of the brainstem of case 1 was positive by a rapid BSE test, contained spongiform changes and had extensive deposition of the abnormal form of the prion protein, PrPSc, by IHC (Fig. 1A, 1B). Western blot analyses using brain material revealed a positive reaction using a 1-mg brain tissue equivalent (mg eq). The PrPSc polypeptide profile from BSE case 1 was characterized by 1) a lower molecular mass of the unglycosylated PrPSc polypeptide fragment compared to samples from sheep with scrapie and deer or elk with CWD, 2) good immunoreactivity with monoclonal antibody 6H4, and a lack of or weak staining with monoclonal antibody P4, and 3) a glycoform profile with a predominant proportion of the diglycosylated PrPSc isoform (see Fig. 2). Western blot comparison of the US BSE case 1 to the May 2003 Canadian and European BSE isolates revealed similar sized PrPSc polypeptide fragments (Fig. 2B). The 2003 Canadian BSE isolate from Alberta was reported to be a typical BSE isolate with similar molecular properties (including a lack of or weak staining with monoclonal antibody P4) to the PrPSc isolated from BSE cases in Switzerland (Fig. 2B) and the United Kingdom. 45 PrPSc isolated from elk with CWD had a higher molecular mass profile than did the corresponding PrPSc for the Canadian BSE case. 45 Similarly, PrPSc from mule deer and elk with CWD had a higher molecular mass profile than did the corresponding PrPSc for the US BSE case 1 (Fig. 2A, 2B).
United States BSE case 2 was born and raised in Texas, and represents the first native case of BSE in the United States. The brainstem of this animal reacted positive in the rapid BSE test used for BSE surveillance in the United States. The tissue of US BSE case 2 had been frozen before formalin fixation. Therefore, artifactual histopathological changes were present in the brainstem of this animal (Fig. 1C), however, unambiguous vacuolar changes diagnostic for BSE were not detected. PrPSc was detected in the brainstem by IHC (Fig. 1D). However, the staining intensity was less intense when compared with the signal found in the brainstem of case 1 (Fig. 1B). Moreover, the IHC staining pattern of case 2 was rather localized and not as diffuse as found with case 1. Interestingly, case 2 was only positive after formic acid treatment and extended antigen retrieval. Initially, case 2 was negative by IHC, even though the IHC positive control brainstem sections (from case 1) were strongly positive under the initial fixation and antigen retrieval conditions used. A positive Western blot reaction using antibody 6H4 was only seen after sample enrichment using the OIE SAF Immunoblot method and only 50% (5 out of 10) of brainstem or cerebellum samples were positive for the presence of PrPSc when tested at 20-mg brain tissue equivalent. This indicates that PrPSc was not uniformly distributed in the brainstem or cerebellum of case 2 and the PrPSc content per mg tissue equivalent was significantly lower (at least 20 times less) than found in brain material of case 1. Surprisingly, brain material from case 2 reacted strongly with antibody P4 (Fig. 3D). The intensity of the reaction of cerebellum and brainstem from case 2 with antibody P4 was even stronger than with 6H4 at similar mg tissue amounts (Fig. 3 C-D). A similar pattern was also seen with the sheep scrapie control sample (Fig. 3C-D). The unglycosylated and monoglycosylated isoforms of PrPSc from case 2 migrated higher than the respective isoforms of case 1 (Fig. 3B-C), indicating a difference in molecular mass between the 2 US BSE cases. The migration pattern of case 1 is typical for BSE (Fig. 2B), whereas the migration pattern of case 2 was reported as being unusual for BSE. 5 In addition, glycoform profile analysis of case 2 revealed a significant proportion of the diglycosylated isoform (approximately 61%), however, the percentage was lower than observed for the diglycosylated isoform of case 1 (approximately 72%). The monoglycosylated and unglycosylated isoforms of case 2 were more prominent (approximately 30% and 9%, respectively) than observed with the respective isoforms of case 1 (approximately 23% and 4%, respectively). The glycoform profile for case 1 is similar to that described for typical cases of BSE 5,28 , whereas the profile found for case 2 is rather unusual. 5 The differences observed between case 1 and 2 were not caused by sampling of different regions of brain, since both, brainstem and cerebellum samples of case 1 and 2 showed the above described reaction patterns.
The Prnp gene of Bos taurus contains multiple polymorphic sites: binary single nucleotide polymorphisms and an insertion-deletion polymorphism with 5, 6, or 7 repeats in the octapeptide-repeat region have been reported for exon 3. 13,25 The Prnp sequences of case 1 and 2 clearly fall within the range of cattle sequence diversity previously reported for cattle (Fig. 4A, 4B). Therefore, one etiological possibility for the disease condition of these animals, namely a germline mutation similar to one of the genetic forms of human prion disease can most likely be ruled out. 37 Prnp sequences from both US BSE cases contained 6 octapeptide-repeat regions on both alleles, a number found in cattle but not in either sheep or cervids. United States BSE case 1 had a polymorphic site at codon 192, a position previously reported to be variable in the bovine Prnp gene with a genotype frequency of about 18% in the US beef cattle population. 25 Both, DNA amplified from fresh as well as paraffin-embedded tissues, showed the codon 192 polymorphism, indicating that these samples were derived from the same animal. United States BSE case 2 had a synonymous polymorphism at codon 185, reported to be variable with a genotype frequency of about 6% in the US beef cattle. 25 We conclude from these data that samples from both US BSE cases had normal, unremarkable cattle-like Prnp gene sequences. Polymorphisms in the sheep prion protein have been correlated with both variable incubation periods and degrees of resistance to scrapie. 19,20 This has not been the case for BSE in cattle. 29,34 However, noncoding regions of the Prnp gene locus of cattle (e.g., prion protein gene promoter polymorphisms) might have an influence on BSE susceptibility as reported recently. 43,44 These regions have not been analyzed in this study.
Unusual cases of BSE have been reported in the past 2 years by investigators from several countries. There have been 2 molecular types of unusual BSE isolates described in the literature: 1) a type with a lower molecular mass of the unglycosylated isoform (L-type) and 2) a type with higher molecular mass of the unglycosylated isoform (H-type). The L-type has been found in cattle in Italy, 11 Japan, 54 and Belgium. 15 In Italy, 2 cattle, older (11 and 15 years) than other bovines affected with BSE, showed an unusual molecular phenotype. Western blot analysis showed a PrPSc type with a predominance of the monoglycosylated isoform and the unglycosylated isoform fragment of lower molecular mass than usually seen with BSE. 11 The disorder was pathologically characterized by the presence of PrPSc-immunopositive amyloid plaques, as opposed to the lack of amyloid deposition in typical BSE cases, and by a different pattern of regional distribution and topology of brain PrPSc accumulation. 11 On the basis of the above features, the authors proposed to name the disease bovine amyloidotic spongiform encephalopathy, BASE. 11 A Japanese case was identified in an ELISA-positive specimen from a 23-month-old Holstein steer slaughtered in September 2003. The animal was reportedly healthy before slaughter and histology showed no spongiform changes and IHC revealed no signal of PrPSc accumulation typical for BSE. Western blot analysis of brainstem homogenate revealed a small amount of PrPSc with an electrophoretic profile different from that of typical BSE-associated PrPSc with 1) a lower content of the diglycosylated molecular isoform of PrPSc, 2) a faster migration of the unglycosylated isoform of PrPSc, and 3) less resistance against PK digestion when compared with typical BSE. 54 Another case involved a 64-month-old Belgian cow, whose obex sample was positive by ELISA, and the cow was reported healthy before slaughter. The histopathology of the obex, pons, and midbrain showed no spongiform changes and IHC of the brainstem revealed no signal of PrPSc accumulation. Western blot analysis of the obex region revealed a small amount of PrPSc with an electrophoretic profile of the unglycosylated isoform of PrPSc showing a lower migration pattern compared with that of a typical BSE case. 15
So far, the H-type has been only described in cattle from France and Germany. 5,9 A distinct molecular phenotype was found in 1 German and 3 French BSE cattle following active surveillance of the disease at slaughterhouses or in rendering plants using rapid BSE tests. The 3 French cases did not have clinical signs suggestive of BSE during their life and the German and French animals were older cattle (8-15 years old). The unusual molecular phenotype of the German 9 and French 5 cases was characterized by 1) a higher molecular mass of the unglycosylated PrPSc isoform, 2) a strong labeling of all 3 PrPSc polypeptides with antibody P4, and 3) the French cases by a glycoform profile with a less prominent diglycosylated PrPSc isoform. 5 All of the H-type features were also observed with US BSE case 2 (Fig. 3).
Unusual cases of BSE are an unexpected finding since it was previously believed that BSE disease in cattle is caused by a single strain of infectious agent, which has been shown to be very consistent and uniform in appearance, even after transmission to other species. 7,28,46 The reports of unusual phenotypes of BSE in cattle suggest that different PrPSc phenotypes exist in cattle with BSE. There are several hypotheses which can explain these findings: 5 1) there are manifestations of the BSE agent with different molecular features in cattle. It is known that sequence differences in the Prnp gene give rise to variants in electrophoretic profiles of PrPSc, as shown in cases of human CJD, 10 however, comparison of the Prnp alleles available from cattle with unusual BSE phenotypes (including US BSE case 2) and the general cattle population have shown normal cattle Prnp sequences; 2) cattle may have been infected by another source of infectious agent (e.g., scrapie or CWD). However, a survey of brain material derived from 262 high-risk, adult cattle in CWD-endemic areas in Colorado for the presence of changes indicative of a TSE infection was negative. 22 Interestingly, experimental infection of cattle with either US sheep scrapie isolates 14 or CWD sources 23,24 led to a cattle disease with clinicopathological features different from typical BSE in cattle; 3) a rare sporadic form of TSE disease could exist in cattle as described for human TSEs. 16 Further studies are needed to determine the frequency and origin of such novel BSE phenotypes. Critical studies would elucidate the transmissibility of unusual BSE cases to cattle or other mammalian hosts; transmission has not been reported in the literature so far.
It is concluded from the studies reported here that 1) the PrPSc profile from the first US BSE case showed similar molecular properties to the typical PrPSc pattern described for the May 2003 Canadian and European BSE isolates, 45 and 2) the PrPSc profile from the second US BSE case showed unusual molecular properties similar to atypical high molecular weight BSE cases reported in France and Germany. 5,9 Both cases were identified by the USDA surveillance program in place and the carcasses did not enter the human or animal food chain. IHC staining in brainstem sections for an unusual high molecular BSE case is described here for the first time. A germline mutation as an etiological possibility for the disease conditions of both cases can be most likely ruled out. Future work will address the question whether brain material from both US BSE cases are infectious in cattle after intracerebral and oral inoculation.
Acknowledgements
The authors would like to thank S. Lebepe-Mazur, D. Clouser, K. Hassall, L. Manning, Shelley Ganske, Sharon Lund, Nadine Beckwith, and D. Orcutt for excellent technical support and Drs. M. Kehrli and R. Levings for their encouragement and support during the BSE diagnostic work. The authors also wish to thank the 7 State/University Veterinary Diagnostic Laboratories for their hard work screening US cattle for the presence of BSE.
Footnotes
a.
Platelia/TeSeE™ ELISA BSE test, Bio-Rad, Hercules, CA.
b.
Fisher Superfrost Plus, Fisher Scientific, Hampton, NH.
c.
Dako, Carpinteria, CA.
d.
Biocare, Walnut Creek, CA.
e.
Ventana Medical Systems Inc., Tucson, AZ.
f.
VMRD, Pullman, WA.
g.
Non Ultra Dish Liquid, Original Scent, Dawn. Procter & Gamble, Cincinnati, OH.
h.
Prionics, Schlieren, Switzerland.
i.
Fisher Scientific, Pittsburgh, PA.
j.
Invitrogen, Carlsbad, CA.
k.
Immobilon-P, Amersham Biosciences, Piscataway, NJ.
l.
R-Biopharm Inc, Marshall, MI.
m.
Amersham Biosciences, Piscataway, NJ.
n.
Novagen, Madison, WI.
o.
Sigma, St. Louis, MO.
p.
Beckman Coulter Inc, Fullerton, CA.
q.
USB, Cleveland, OH.
r.
Roche, Indianapolis, IN.
s.
Kodak, St. Louis, MO.
t.
Qiagen, Valencia, CA.
u.
Amresco, Solon, OH.
v.
Strategene, La Jolla, CA.
w.
Q-BIOgene, Irvine, CA.
x.
Big Dye Teminator, Applied Biosystems, Foster City, CA.
