Abstract
The BH3-only protein Bad is a proapoptotic Bcl-2 family member that acts as a sensitizer in intrinsic apoptosis by inactivating antiapoptotic members through heterodimer formation. Bad has been shown to contribute to tumorigenesis, including lymphoma formation in humans and mice, through alteration in expression or functional status. Here, its immunohistochemical expression was analyzed in canine nonneoplastic and lymphoma tissues using tissue microarrays. Bad was expressed in the cytoplasm of a wide range of nonneoplastic tissues, especially epithelial cells. Nonneoplastic lymph nodes displayed weak immunostaining in the follicular germinal centers only. Immunoblotting supported these observations but also revealed presence of nonspecific labeling in some organs. Of 81 lymphomas, 29 (35.8%) displayed moderate to strong immunohistochemical Bad labeling, and a significant expression increase was found in lymphomas (especially B cell and double negative) compared to nonneoplastic lymph nodes. These findings warrant further investigations of the functional status, the involvement of partner proteins, and a possible impact of Bad on prognosis in canine lymphoma.
Keywords
Apoptosis is essential for normal development and tissue homeostasis and protects against diseases, including cancer. 3 Several members of the Bcl-2 family proteins, which are critical regulators of intrinsic apoptosis, have been assigned either oncogenic or tumor suppressor properties. 2 The proapoptotic BH3-only subgroup member Bad (Bcl-XL/Bcl-2-associated death promoter) acts as a sensitizer early in the course of intrinsic apoptosis by binding and neutralizing its antiapoptotic partners Bcl-2, Bcl-XL, and Bcl-w. 1,16 This binding is inhibited by phosphorylation of Bad at conserved serine residues, which leads to the inactivation of Bad through sequestration by cytosolic 14-3-3 proteins. 4,11,18
Ablation of Bad in mice induced diffuse large B-cell lymphomas, 10 and loss of Bad expression was observed in spontaneous epidermal neoplasms in humans. 13 Increased levels of phosphorylated Bad were found in follicular lymphomas and colorectal carcinomas. 7,17 Publicly available immunohistochemical data indicate that Bad is widely expressed in normal human organs. 8,12 In some tissues, including lymphatic organs and a limited number of lymphoma cell lines and samples, expression levels of Bad appeared to be low or even absent. 8,12
Due to the apparent lack of canine data, the aim of the present study was to assess the expression of Bad in nonneoplastic tissues and lymphomas in this species by means of immunohistochemistry.
Material and Methods
Canine Tissues and Cultured Cells
Tissues
Tissue microarrays (TMAs) containing archival canine nonneoplastic organs (Table 1) were available from a previous study. 14 In addition, normal nonneoplastic mandibular lymph node samples were available; they comprised a TMA containing six to ten 1.2-mm cores from each of 18 dogs and a multiblock containing larger lymph node probes from 7 dogs. Selected normal tissues were freshly collected and snap-frozen for Western blot analysis. TMAs containing 4 cores of 0.6-mm diameter each from 90 cases of canine lymphoma immunophenotyped and classified were available from previous studies. 5,14 Representativeness of cores was assessed on consecutive sections labeled with hematoxylin and eosin and with immunohistochemical CD3 and CD79αcy markers as described. 14
Immunohistochemical Detection of Bad in Nonneoplastic Canine Tissues.
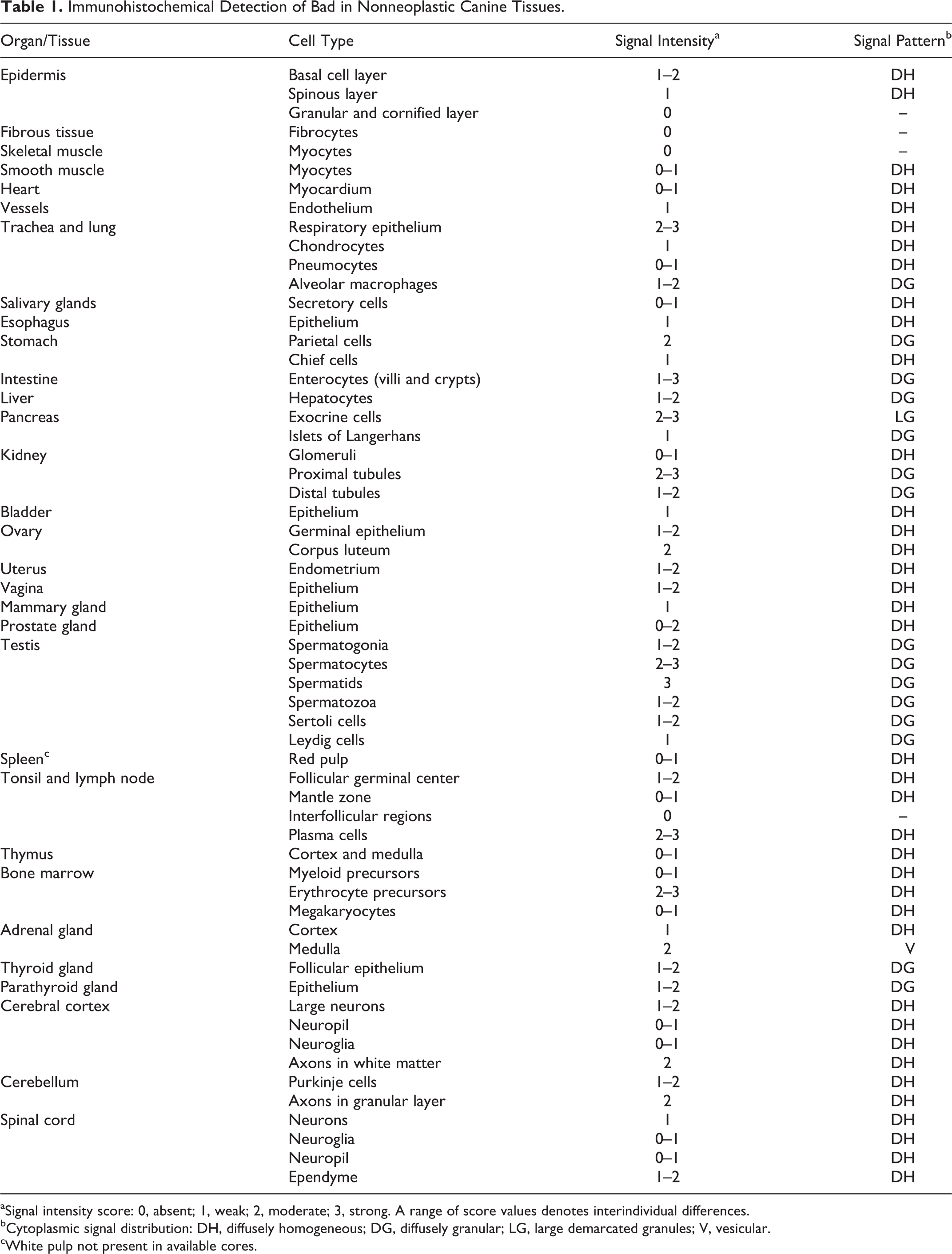
aSignal intensity score: 0, absent; 1, weak; 2, moderate; 3, strong. A range of score values denotes interindividual differences.
bCytoplasmic signal distribution: DH, diffusely homogeneous; DG, diffusely granular; LG, large demarcated granules; V, vesicular.
cWhite pulp not present in available cores.
Canine T-Cell Lymphoma CL-1 Cells
The cells were cultured in RPMI 1640 medium with stable glutamine (E15-885, PAA Laboratories, Pasching, Austria) supplemented with fetal calf serum 10% (BioConcept Ltd., Allschwil, Switzerland) and penicillin/streptomycin 1% (Gibco BRL Life Sciences, Basel, Switzerland) under standard conditions. Pellets were constructed for immunohistochemistry as described using cells fixed for 24 hours in 4% formaldehyde. 6 For Western blot analysis, the cells were washed with ice cold phosphate buffered saline (PBS) and pelleted in 15-ml tubes at 6000 g for 2 minutes at 4°C. The supernatant was discarded and the pellet resuspended in PBS and centrifuged at 16 000 g for 1 minute at 4°C in a 1.5-ml Eppendorf tube. The pelleted cells were lysed in ice-cold lysis buffer (20 mM Tris, 100 mM NaCl, 1 mM EDTA, 0.5% Triton X-100) supplemented with protease inhibitors (1 mM PMSF, 0.3 µM Aprotinin, 0.5% Leupeptin, 0.01% Pepstatin-A), centrifuged at 16 000 g for 15 minutes at 4°C, and the supernatant was stored at 4°C until further processing.
Immunohistochemistry
Sections (2 µm) were mounted on positively charged glass slides and immunostained in a Discovery XT autostainer (Ventana Medical Systems, Inc., Tucson, AZ) using the CC1 antigen retrieval program and the monoclonal antibody 1541-1 against human Bad (Epitomics, Burlingame CA, USA) in a 1:1500 dilution. The immunohistochemical procedure—as well as the antibody selection and validation process, which relied on the use of canine recombinant Bad protein and ultraviolet-irradiated canine keratinocytes—is extensively described in the supplementary material (http://vet.sagepub.com/supplemental). Negative controls using 1% BSA in PBS instead of the primary antibody were carried out for each section. All tissues were scored according to the cytoplasmic immunohistochemical signal intensity as follows: 0 = signal absent, 1 = weak, 2 = moderate, 3 = strong signal. The score assigned to a given cell type corresponded to the signal intensity displayed by more than 50% of the cells. The reported final scores for each cell type were calculated as mean values of 2 independent readings by one author (MD). For a lymphoma case to be scored, presence of at least 2 properly labeled representative cores with more than 50% of the core well preserved was required. A mean score for all cores of each tumor was calculated. The immunohistochemical signal intensity scores of B-cell, T-cell, and double-negative (DN) lymphoma immunophenotypes and the nonneoplastic lymph nodes (n = 26) were compared statistically using the nonparametric Kruskal-Wallis rank-sum test and Dunn post hoc z value test. P values lower than .05 (z values > 2.638) were considered to indicate statistical significance. Analyses were performed using SPSS 20.
Western Blots
Tissue pieces (approximately 200 mg) were thawed, lysed in 1.5-ml lysis buffer (15 mM HEPES pH 7.5, 15 mM NaCl, 60 mM KCl, 0.27 M sucrose, 2 mM EDTA pH 8.0, 1% Triton X-100, protease inhibitors as above)using a pestle, and centrifuged 3 times at 16 000 g for 15 minutes at 4°C. Lysates of tissues (containing 100 µg of protein as determined with the Bradford method) and CL-1 cells (18 µg, measured using a NanoDrop-1000 (Thermo Fisher Scientific, Wilmington, DE)) were loaded with 4x SDS loading buffer (0.25 M Tris/HCl pH 6.8, 8% SDS, 10% glycerol, 0.02% bromphenol blue, 10% β-mercaptoethanol) on a 12% polyacrylamide gel and blotted (Fastblot B43, Biometra GmbH, Göttingen, Germany) onto a PVDF membrane (Immobilon-P Transfer Membrane, Millipore Corporation, Billerica, MA). After blocking with 3% BSA in TBST (Dako Wash Buffer 10×, Dako Schweiz AG, Baar, Switzerland), membranes were incubated with antibody 1541-1 (Epitomics) diluted 1:5000, washed with TBST, incubated with a horseradish peroxidase–conjugated polyclonal goat-anti-rabbit antibody (Jackson ImmunoResearch, Soham, UK) diluted 1:10 000, washed, incubated with LumiGLO (Kirkegaard & Perry Laboratories Inc., Gaithersburg, MD), and developed on an enhanced chemiluminiscence film (Amersham Hyperfilm ECL, GE Healthcare Limited, Buckinghamshire, UK). For negative control, 1% BSA in TBST was used instead of the primary antibody.
Results
This study analyzed immunohistochemically the expression of Bad in canine nonneoplastic tissues and lymphomas assembled in TMAs using an extensively validated monoclonal antibody (http://vet.sagepub.com/supplemental). The results were partially corroborated by Western blot analyses.
Nonneoplastic Tissues
The immunohistochemical signal (Table 1, Figs. 1–11) was limited to the cell cytoplasm. It was either diffusely homogeneous or coarsely granular, except for cells of the exocrine pancreas and adrenal medulla, where it appeared as well demarcated, large, intensely labeled granules or as large, empty vesicles, respectively. Several organs and cell types displayed weak to moderate labeling intensities. A strong labeling was generally present in exocrine pancreas cells (Fig. 4), renal proximal tubular epithelium (Fig. 5), spermatids (Fig. 7), and the tracheal epithelium. Labeling was completely absent from superficial epidermal cells (granular and cornified layer), fibrous tissues, and striated muscles (Fig. 9) and from all negative controls. Cores from different dogs often displayed small differences in signal intensities (Table 1). Rare cell types with interindividual differences greater than 1 score point included small intestinal enterocytes and prostatic epithelial cells. In nonneoplastic lymph nodes, immunohistochemical labeling of lymphocytes was in general mainly restricted to germinal center lymphocytes (score 1), while mantle and interfollicular zones were negative (Fig. 10).
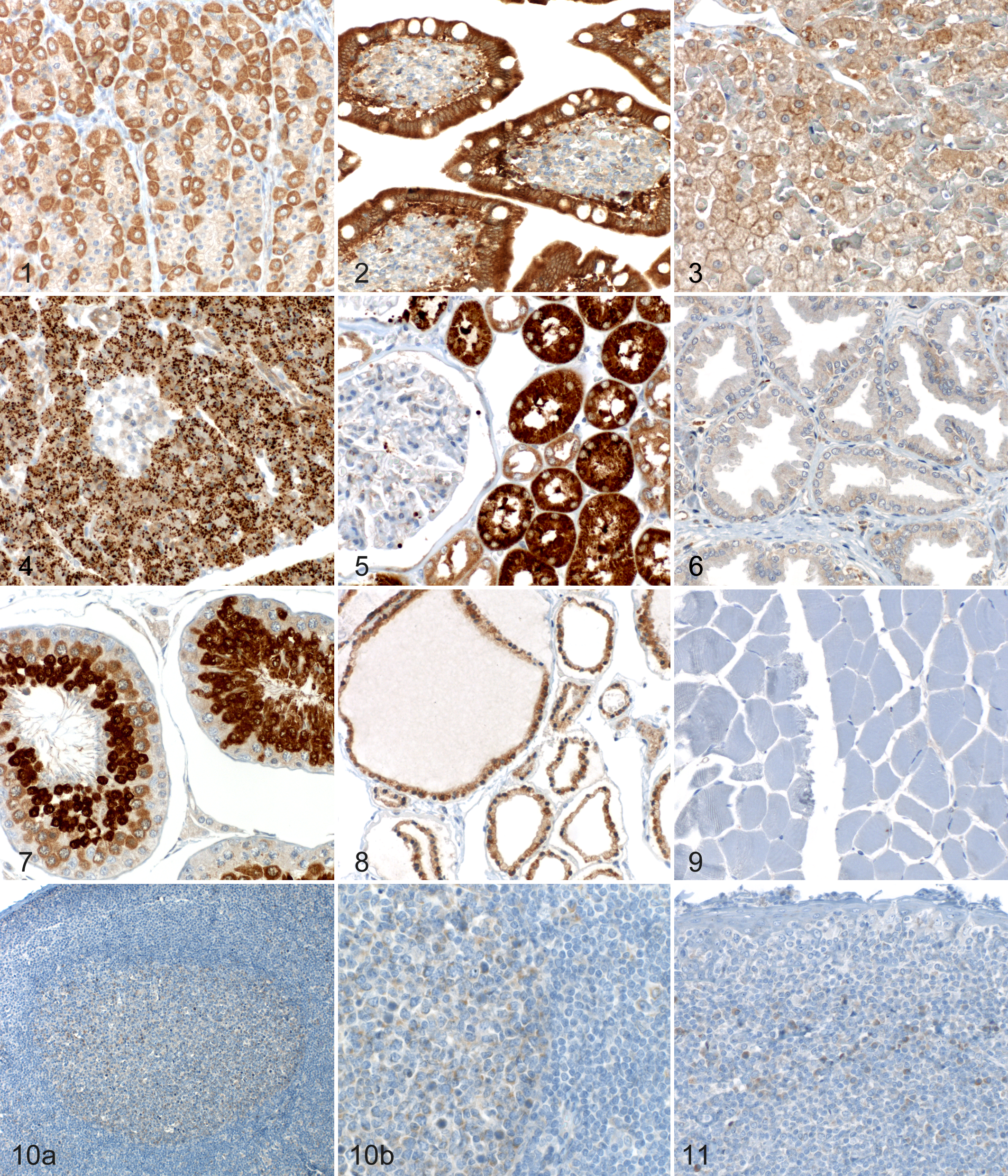
Immunohistochemical Bad labeling of nonneoplastic canine tissues (each from a different dog). The immunohistochemical signal was limited to the cytoplasm. Anti-Bad antibody 1541-1, detection with Omni-UltraMap, hematoxylin counterstain.
Western blots run for Bad with lysates of selected nonneoplastic tissues to test the reliability of immunohistochemistry (Fig. 12) showed, in general, overall intensities matching well with the immunohistochemical signals. However, kidney, pancreas, and liver lysates showed, besides the expected single band at 21 kDa or double band at 21 and 18 kDa, 1 or 2 additional, prominent, likely nonspecific bands at 40 and 50 kDa. In contrast, the major product labeled by the antibody in extracts of normal lymphatic tissues and testis had the expected size.
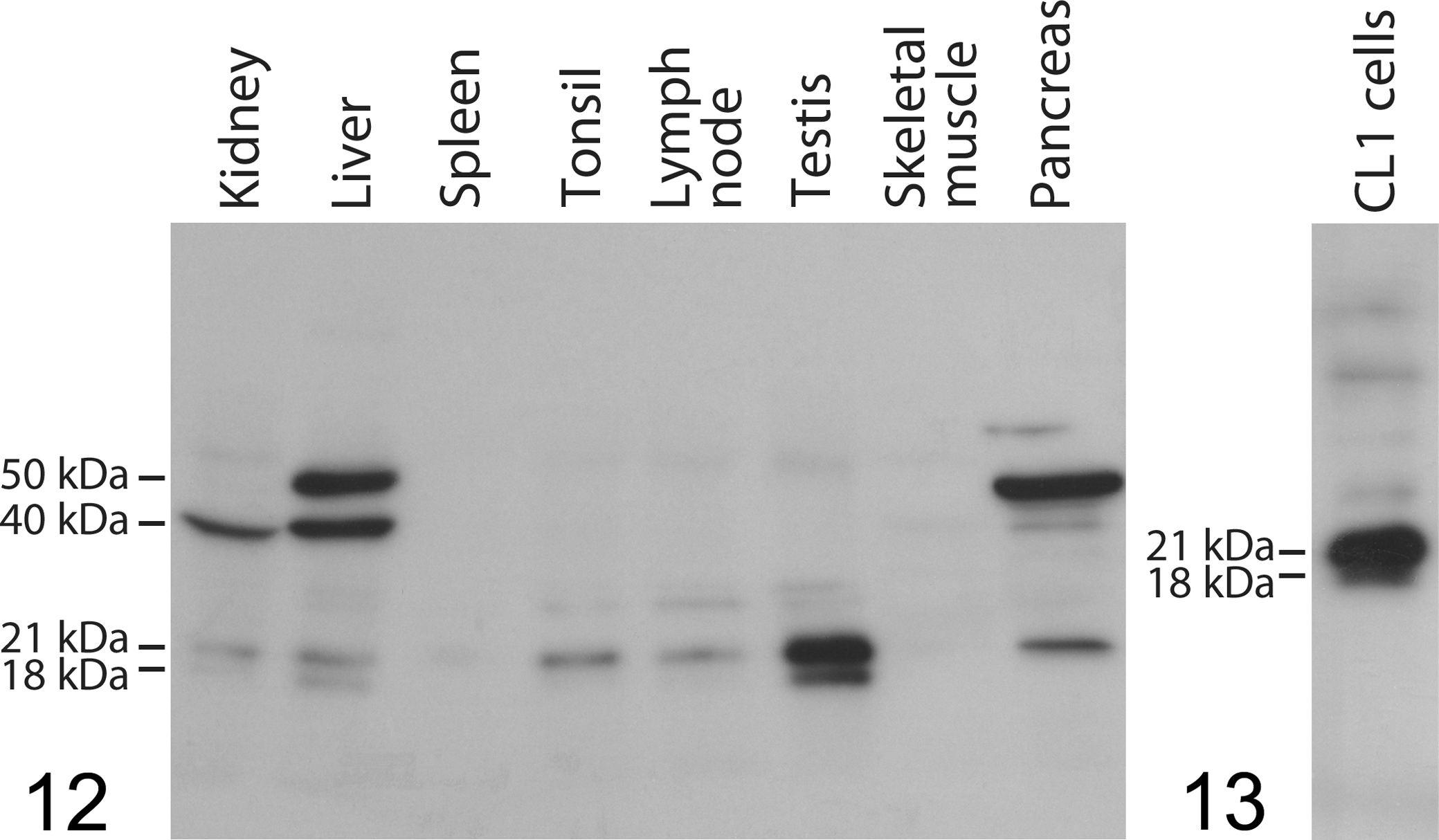
Western blot for Bad with antibody 1541-1 in nonneoplastic tissue lysates of indicated organs. Distinct bands compatible with Bad at 21 kDa and partly also at 18 kDa are present in liver, kidney, tonsil, lymph node, testis, and pancreas lysates; liver, kidney, and pancreas lysates show additional prominent bands at 40 and/or 50 kDa.
Lymphomas
Bad immunohistochemistry of CL-1 lymphoma cells resulted in a generalized, marked cytoplasmic signal (Fig. 14), while a Western blot showed exclusive labeling of a double band of 21 kDa (major product) and 18 kDa (Fig. 13) supporting specificity of the antibody. Neoplastic cells of the 81 cases fulfilling the inclusion criteria predominantly displayed a diffusely homogeneous cytoplasmic signal, with a superimposed granulated pattern observed in a few cases. Overall, the lymphoma cases showed scores ranging from 0 to 3 (Figs. 15–18) with a mean value of 1.08, a median value of 1.00, and a standard deviation of 0.73. The labeling intensity varied very little within cores, while in about 50% of the lymphoma cases, a slight intercore variability of up to maximally 1 scoring point (in a single case, 2 points) was noted. Table 2 lists the scoring results of the lymphoma cases grouped according to immunophenotype and most frequent subtypes, as well as all normal lymph node tissues available on TMAs. While most nonneoplastic lymph nodes (n = 25 of 26, 96.2%) displayed an overall signal intensity score ≤ 1, this applied to only 52 of 81 (64.2%) lymphoma cases. The remaining 29 cases (comprising 17 B-cell, 4 T-cell, and 8 DN lymphomas) showed scores > 1. Among B-cell lymphomas, the heterogeneous group with less common subtypes and nonclassifiable lymphomas displayed higher signal intensities than the other groups. One-way analysis of variance with the Kruskal-Wallis rank-sum test showed a statistically significant difference in the labeling intensity between nonneoplastic lymph nodes and the whole group of lymphomas (P < .001). A Dunn post hoc z value test revealed a statistically significant difference between nonneoplastic lymph nodes and B-cell lymphomas (z = 3.602, P = .002), between nonneoplastic lymph nodes and DN lymphomas (z = 4.557, P = .001), and between T-cell lymphomas and DN lymphomas (z = 3.739, P = .001).
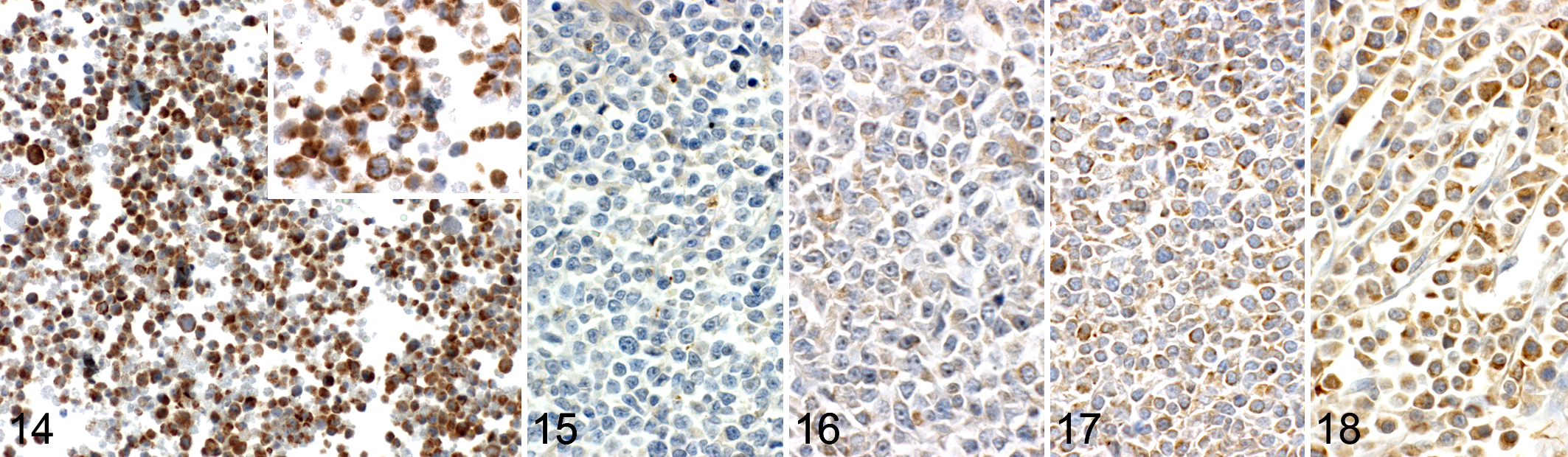
Immunohistochemical Bad labeling in canine lymphoma. Anti-Bad antibody 1541-1, detection with Omni-UltraMap, hematoxylin counterstain.
Immunohistochemical Detection of Bad in Canine Lymphomas and Nonneoplastic Lymph Nodes.
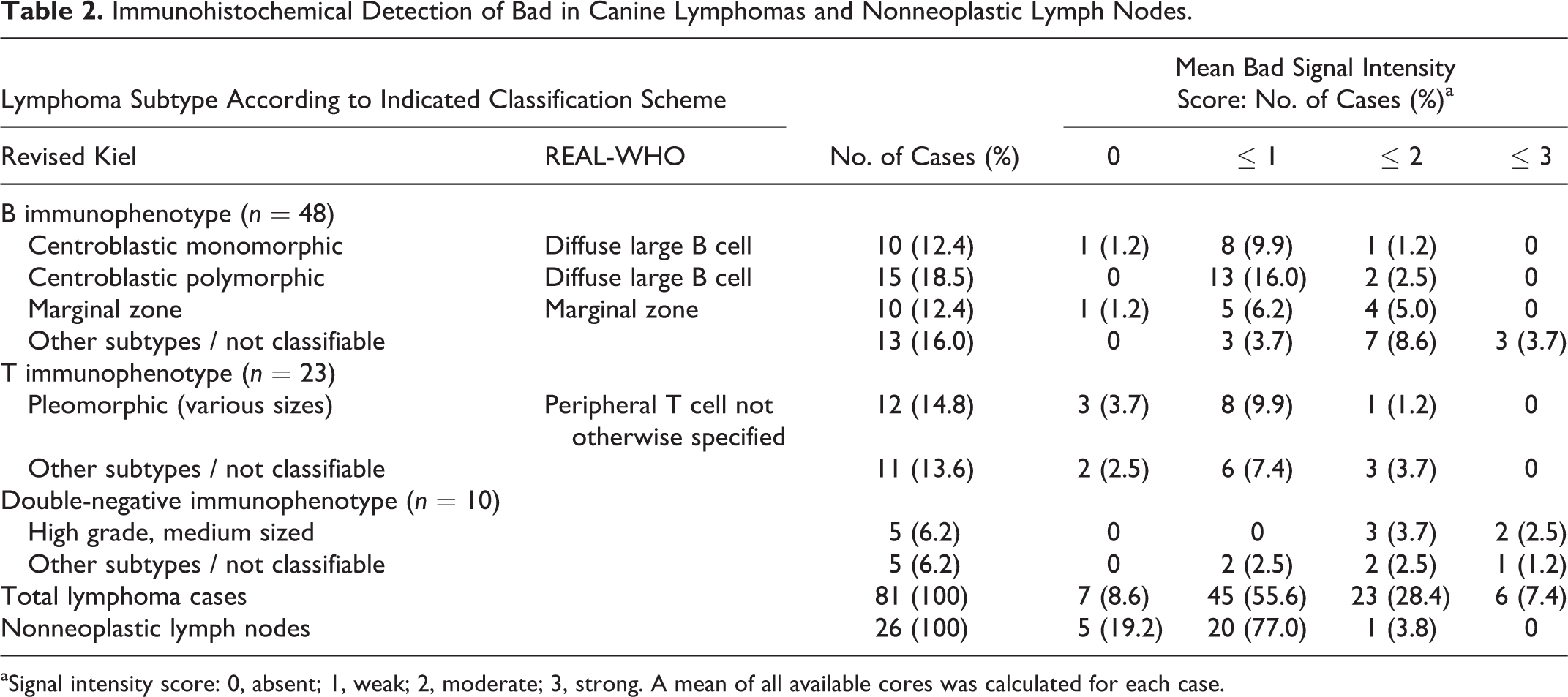
aSignal intensity score: 0, absent; 1, weak; 2, moderate; 3, strong. A mean of all available cores was calculated for each case.
Discussion
The present study analyzed the expression of Bad protein in canine lymphomas. Since ablation of Bad was purported to promote lymphoma development in mice, 10 a decreased Bad expression in canine lymphoma compared to nonneoplastic lymph nodes was considered as a possible outcome. However, no such finding was observed here. Instead, a considerable fraction of the lymphomas (35.8%) showed higher Bad expression than nonneoplastic lymphocytes, a difference that was statistically significant for B-cell and DN lymphomas, possibly indicating enhanced apoptotic signaling in the affected tumors. These findings contrast with the results of previous human studies showing little or no Bad expression in seven lymphoma and leukemia cell lines 8 and in 20 of 22 malignant lymphomas. 12 However, another report indicated higher Bad expression levels in diffuse large B-cell lymphomas compared to follicular and mantle cell lymphomas. 15 Unfortunately, the number of cases in the present canine study was too small to assess statistical differences between subtypes. Moreover, there was no relationship between Bad expression and markers of apoptotic activity, such as cleaved caspase-3 or cleaved lamin A, in our material (unpublished data). This can be considered as an additional argument against a prominent role of Bad in the genesis of canine lymphoma. Since execution of intrinsic apoptosis is thought to depend on stoichiometric interactions of Bcl-2 family members, the role of this protein might be more precisely assessed with a comprehensive approach probing the whole family.
Two reports indicate the presence of phosphorylated Bad in human follicular and cutaneous T-cell lymphomas. 17,19 Thus, it might be of interest to determine the functional status of Bad in future studies, which was omitted here due to failure to find a suitable phosphorylation-specific antibody (data not shown). However, the diffusely homogeneous signal observed in the canine tumors suggests that the enhanced signal may be due to an increase of inactive hyperphosphorylated Bad, which is sequestered in the cytoplasm by the scaffold protein 14-3-3. An involvement of Bad has been hypothesized in the development of chemotherapy resistance in human diffuse large B-cell lymphomas found to overexpress the isoform 14-3-3ζ. 9
The other main part of this investigation was the analysis of Bad expression in nonneoplastic tissues. Western blot and immunohistochemical data indicate that Bad is expressed in the cytoplasm of a wide range of normal canine cell types. This is consistent with investigations of human tissues. 8,12 However, Western blot results indicate that the immunohistochemical findings in this broad tissue collection should be interpreted with caution, since in some organs, such as liver, kidney, and pancreas, additional nonspecific bands were present. There is no reported evidence for homodimerization of Bad, 18 and the reducing conditions of the SDS-PAGE used here should have separated heterodimers as well. Thus, the marked immunohistochemical signal observed in these 3 organs has a major nonspecific component. Interestingly, the immunohistochemical labeling pattern in the exocrine pancreas cells clearly differed from the either diffusely homogeneous or diffusely granular pattern observed in most other cell types. In summary, our data show the importance of validating antibodies with respect to their reactivity with the specifically targeted tissue or cell type. Specific immunohistochemical labeling of lymphomas in this study is supported by the lack of nonspecific bands in lysates of nonneoplastic lymphatic tissues and, notably, CL-1 lymphoma cells. However, validation by Western blot is strongly advised for future prospective studies.
In general, similar to previous human studies, normal epithelial cells displayed a stronger signal than mesenchymal cells. 8,12 Findings in several mesenchymal tissues, including all types of musculature, differed to some extent among human studies, thus hampering the interpretation of the canine data for these cell types. In canine nonneoplastic lymph nodes, a weak to moderate immunohistochemical signal was mainly observed in the cytoplasm of germinal center lymphocytes, while mantle and interfollicular zone lymphocytes were negative, as previously observed by Kitada and colleagues. 8 The Protein Atlas 12 indicates a weak to moderate signal in lymphatic tissue depending on the antibody used. Common explanations for discrepancies among studies include use of different antibodies targeting different epitopes and even different protocols, the occurrence of different isoforms, and differences in tissue fixation and processing. Further reasons, possibly applicable here, include interspecies differences, interindividual variability, the small amounts of tissue provided by TMAs, and nonspecific antibody binding in certain tissues.
In conclusion, the present study showed that the proapoptotic Bcl-2 family member Bad is expressed in a wide range of normal canine tissues. Our findings indicate that the results of immunohistochemical studies should be interpreted with great caution and that accurate validation (eg, Western blotting, as in this study) is crucial to rule out nonspecific cross-reactions. Expression levels of Bad were generally low in nonneoplastic canine lymphocytes, but they were increased in a portion of the lymphomas examined. The significance of this finding is unknown, and further investigation of the functional state of Bad, involved partner proteins, and a possible impact on prognosis is required.
Footnotes
Acknowledgements
We are very grateful to Sabina Wunderlin for excellent technical help. We thank Dr Sonja Hartnack for support with the statistical analysis. We are indebted to Profs E. J. Müller and M. M. Suter, University of Berne, Switzerland, for providing the Bskin cell line and Dr Y. Goto-Koshino, University of Tokyo, Japan, for providing the CL-1 cell line.
Declaration of Conflicting Interests
The authors declared that they had no potential conflicts of interest with respect to their authorship or the publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
