Abstract
The clinicopathologic value of the immunohistochemical (IHC) expression of p53 protein was evaluated in 167 canine osseous tumors. p53 staining frequency and intensity in tumor cells was expressed as a p53 index. p53 index was significantly higher in osteosarcomas than in other sarcomas, chondrosarcoma, multilobular tumor of bone, and tumors initially misdiagnosed as osteosarcomas as well as in appendicular versus axial and in distal versus proximal osteosarcomas. A strong correlation is demonstrated between the p53 index and a range of clinicopathologic parameters in osteosarcoma, including the tumor site, histologic grade and score, mitotic index, degree of tumor necrosis, and pleomorphism. Chondroblastic osteosarcomas had significantly higher and telangiectatic osteosarcomas significantly lower p53 index than did osteosarcomas belonging to other histopathologic subtypes, a fact that tends to reinforce the perception of these osteosarcomas as distinct clinicopathologic entities. Entire males had higher p53 index than did neutered males. p53 index was higher in Rottweilers than in Great Danes and Terriers, confirming breed susceptibilities to osteosarcoma. p53 index showed no association with age, primary or secondary site status, or the presence of metastases or other tumor types. Biopsy samples had a higher p53 index than did postmortem samples, either because of differences in sample processing or the possibility that p53 overexpression is more evident at the earlier stages of osteosarcoma pathogenesis, presumably represented by the biopsy material. IHC examination for p53 and the derived index has the potential to be used as an additional diagnostic tool and prognostic indicator for osseous tumors.
The p53 gene encodes a nuclear phosphoprotein (p53) that normally acts as a tumor-suppressor gene. 10 p53 guards cells against replication when their genome is damaged or abnormal, by initially arresting the cell cycle and then by either activating and regulating DNA repair or by inducing programmed cell death (apoptosis) if genomic damage is excessive. 28 , 57 , 65
Absence of a functional p53 gene, p53 protein, or p53 pathways may remove mechanisms that normally arrest the proliferation of transformed cells, disrupting the apoptotic response that may ensue and therefore contributing to the development and progression of neoplastic clones. 93 Because of this central role in cell cycle and in carcinogenesis, 28 p53 is the most frequently altered gene in human tumors; 37 , 39 p53 gene mutations have been found in 40–45% of cases of the 10 most frequent cancers in humans. 99
Wild-type p53 is present in all cell types, usually in low quantities, 78 and has a short half-life that makes it difficult to detect by immunohistochemical (IHC) techniques. 53 However, abnormal, nonfunctional p53, either caused by mutations of p53 gene 40 or by the formation of p53 protein complexes with viral or endogenous proteins 87 such as a large T antigen of simian virus 40 or MDM2 (murine double minute-2), 58 may result in the synthesis of an abnormal protein that is metabolically stable 26 , 35 and has a 10- to 20-fold longer half-life than wild-type p53, 26 accumulating in the cell and reaching concentration levels detectable by immunohistochemistry. 44 IHC detection of p53 indicates, therefore, alterations in the p53 gene or p53 product and demonstrates a progression toward a tumor-associated phenotype. 60 , 105 p53 protein overexpression has been detected at high levels in a large number of sporadic tumors and in virally and chemically transformed cell lines from mice and humans. 3 , 41 , 44 , 78 Furthermore, p53 is a highly conserved gene in evolution, with regions of high homology in mammals, birds, fish, and amphibians; 77 most mutations occur within the highly conserved regions of p53. 40
Demonstration of the presence of p53 abnormalities is predictive of biological behavior in a wide spectrum of human malignancies, 18 , 86 , 117 including osteosarcoma, 64 synovial sarcoma, 94 breast cancer, 80 ovarian carcinoma, 20 , 96 prostate carcinoma, 11 , 12 , 115 renal adenocarcinoma, 62 gastric cancer, 43 , 95 adenoma and carcinoma of the ampulla of Vater, 85 colorectal adenoma 54 and carcinoma, 32 , 34 , 48 , 52 , 55 gliomas, 83 astrocytic tumors, 23 tumors of the urinary bladder, 24 , 25 , 97 cutaneous 68 and head and neck squamous cell carcinomas, 6 , 45 , 66 , 67 , 121 and advanced head and neck cancer treated with chemo/radiotherapy. 86 , 117
The canine p53 gene has been assigned to chromosome 5, 36 and the complete wild-type canine p53 cDNA has been isolated and sequenced 14 , 112 and the full length canine p53 protein characterized. 112 A high level of sequence homology was demonstrated with human sequences and other higher vertebrates, 112 with the conserved domains II, III, IV, and V being identical to those of humans. 82
Alterations in the p53 gene have been reported in a variety of canine cancers, including osteosarcoma. Mutations were found, depending on the study, in 7 of 15 appendicular osteosarcomas, 50 4 of 17, 110 and 8 of 21 75 primary osteosarcomas screened, as well as in osteosarcoma cell lines. 59 The locations and types of mutations were nearly identical to those reported in human cancer. Van Leeuwen et al. 111 also showed the presence of missense mutations accompanied by loss of the wild-type allele in three of nine canine mammary tumor cell lines and in two of the corresponding tumor tissues of origin analyzed. Again, all mutations were located in one of the four regions that are frequently affected in human cancers. Somatic mutations, accompanied in some cases by a spectrum of chromosomal aberrations, have also been reported in other canine benign 79 or malignant 14 , 69–71 , 79 , 114 mammary tumors, thyroid carcinoma, 19 skin tumors, 71 an adenoma of the circumanal gland, 73 lymphosarcoma, 81 and lymphoma, 113 as well as in keratoconjunctivitis sicca 30 but are not present in all cases and tumor types in dogs. 72 Germ-line mutations have been reported in one case each of canine lymphoma 113 and mammary cancer. 114
Tumors in bone are not infrequent in the dog, 8 , 17 the main type being osteosarcoma, followed by chondrosarcoma, 106 fibrosarcoma, haemangiosarcoma, and multilobular tumor of bone. In contrast with their counterparts in humans where metastasis to bone of carcinomas, in particular, is common, they are mainly primary tumors. 118 Osteosarcoma, in particular, accounts for the majority of these tumors 7–9 , 61 , 63 and is one of the most malignant and aggressive tumors in dogs, with a predilection in large and giant breeds. 109 The majority of cases that involve the appendicular skeleton often have lung metastases before the primary tumor is diagnosed. 84 Despite advances in its management, there are few established or proposed prognostic indicators for canine osteosarcoma, including age, 100 body weight, 38 , 90 , 109 breed, 15 , 90 tumor location, 38 , 76 serum alkaline phosphatase levels, 21 , 31 completeness of surgical excision, 38 tumor grade, 56 tumor diameter and volume, 27 , 76 tumor subtype, 38 , 76 tumor microvessel density, 16 and tumor necrosis after chemotherapy. 89
The aim of the present study was to characterize osseous tumors according to both their p53 protein IHC expression and their clinical and histopathologic characteristics as a basis for developing a prognostic evaluation protocol of these tumors in clinical patients.
Materials and Methods
One hundred sixty-seven archival cases of osseous tumors were retrieved and their clinical and pathologic features examined in detail. Histologic slides were examined by all investigators. The initial diagnosis of osteosarcoma was rejected in 32 cases (Table 1). The diagnoses offered are given in Table 1.
Tumor types examined and their p53 index and FPN values.
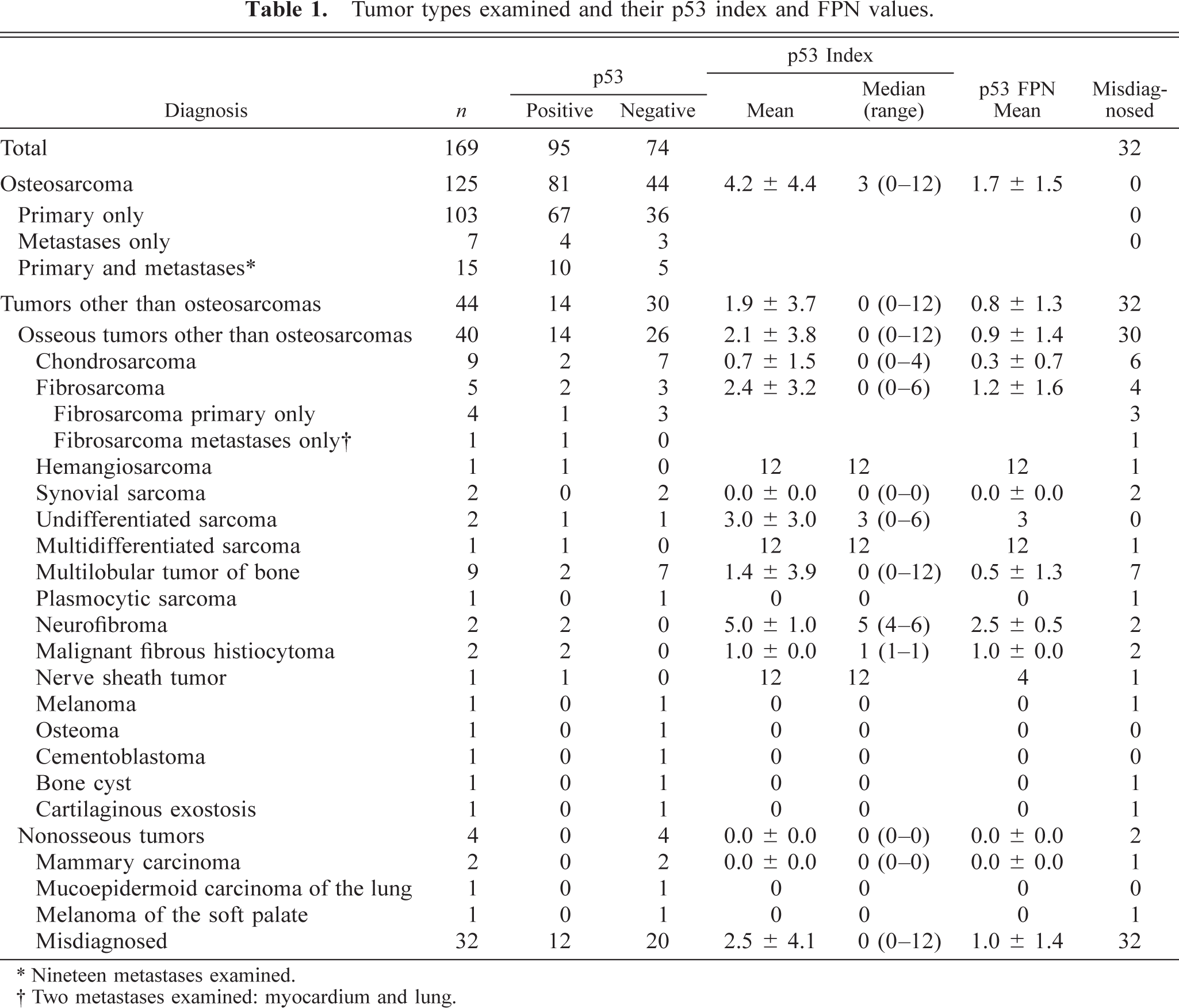
∗ Nineteen metastases examined.
† Two metastases examined: myocardium and lung.
Osteosarcomas were classified into histologic subtypes, as described previously, 98 on the basis of the quality and quantity of the extracellular matrix (ECM) produced (osteoblastic productive, minimally productive, chondroblastic and fibroblastic subtypes); the degree of differentiation (poorly differentiated subtype); the abundant presence of giant cells (giant cell subtype); and the dominant presence of blood-filled cystic lesions (telangiectatic subtype). Osteosarcomas were classified as simple if osteoid or bone was the only matrix or as compound if there were more than one type of ECM produced by the malignant cells.
The mitotic index, the degree of nuclear pleomorphism, and the degree of tumor necrosis of osteosarcomas were assessed to classify them into grades, as described previously. 102 The mitotic index (number of mitoses per 10 fields at 400×) was estimated and scored in the following way: 1–10 mitoses = score 1, 11–20 = score 2, 21–30 = score 3, 31 or more = score 4. The degree of tumor necrosis was scored in the following way: 0% = score 0, 1–15% = score 1, 16–50% = score 2, 51–100% = score 3. The degree of nuclear pleomorphism was estimated as mild, moderate, or marked (score 1, 2, or 3). The three scores obtained were added to calculate the histologic score (HS) of the tumors (2–10), which was then used to classify the tumors into grades in the following way: HS 1–5 = grade I, HS 6–8 = grade II, and HS 9 or 10 = grade III.
Each tissue sample had been fixed in 10% neutral buffered formalin for a varying period of time, routinely processed, and embedded in paraffin blocks. Five-micrometer-thick serial sections were prepared from each archival tissue block, mounted on silane-coated slides, and used for IHC staining. Tissue sections were similarly stained with hematoxylin and eosin for review of the initial morphologic diagnosis and for evaluation of nuclear pleomorphism, mitotic index, and the extent of necrosis.
Immunohistochemistry
An IHC protocol was developed and optimized for the demonstration of p53 protein in canine osseous tumors. In preliminary experiments, canine osteosarcomas and known p53-positive human ovarian carcinomas were examined with serially diluted primary antibodies to decide a suitable concentration of antibody (1:75). Microwaving was compared and preferred to boiling as a means of antigen retrieval. 88
Two human ovarian carcinomas known to be p53 positive from previous studies 20 , 119 were kindly provided by Mr. M. D. Walsh (Department of Pathology, University of Queensland) and used as positive controls (Fig. 1). For negative controls, the primary antibody was replaced with Tris-buffered saline (TBS), pH 7.6. Nontumorous areas of the positive control slides served as internal negative tissue controls.
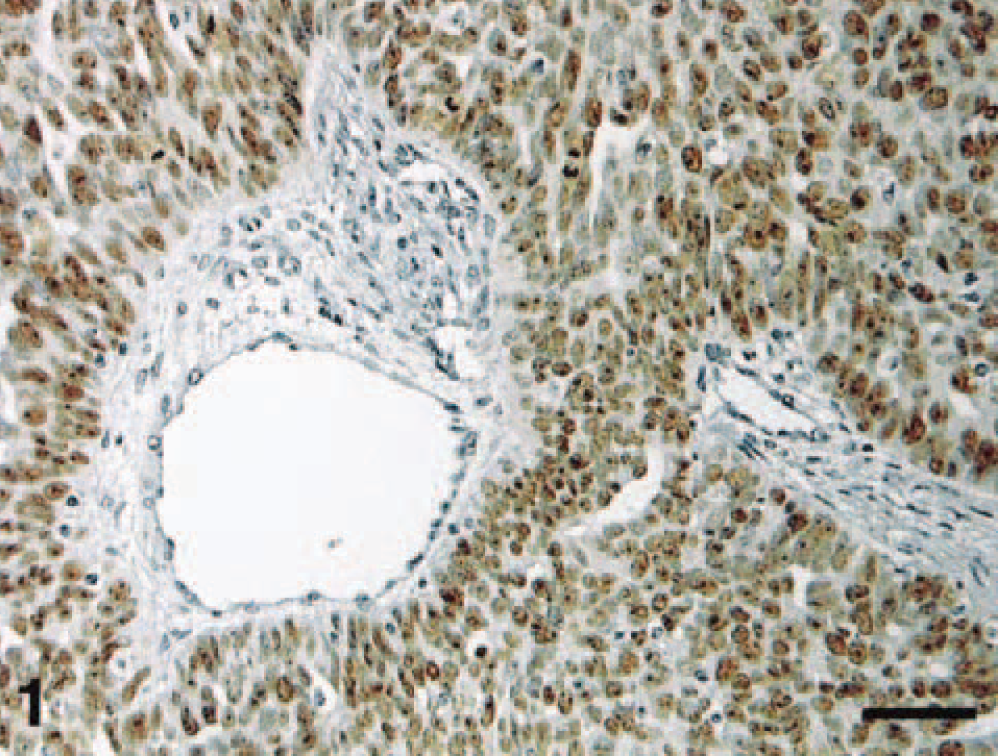
Human ovarian carcinoma. Positive control. IHC localization of p53 protein: intense nuclear staining of tumor cells. Streptavidin–biotin–peroxidase method, Mayer's hematoxylin counterstain. Bar = 60 μm.
Sections were deparaffinized in 100% xylene (3 × 2 minutes) and rehydrated through 100% ethanol (3 × 2 minutes). Sections were then subjected to heat for antigen retrieval by microwaving in preheated citrate buffer (pH 6.0) at 95 C for 10 minutes. Sections were allowed to cool for 20 minutes and rinsed with TBS. Sections were treated for 15 minutes with 3% (v/v) H2O2 in distilled water to inhibit endogenous peroxidase activity. Nonspecific antibody binding was reduced by incubating the sections with 4% commercial nonfat skim milk powder in TBS for 25 minutes. Sections were then incubated with primary antibody (diluted 1:75) overnight at 4 C. The antibody used was the polyclonal rabbit anti-human antibody CM-1 (Signet Laboratories, Dedham, MA), which recognizes both wild and mutant types of p53 protein. The sections were incubated with a rabbit biotin-labeled secondary antibody and then a streptavidin–biotin–peroxidase complex (both Dako, Carpinteria, CA) at room temperature.
After washing, 3,3′-diaminobenzidine tetrahydrochloride (Sigma Chemical Company, St. Louis, MO) was used as chromogen with H2O2 as substrate. The sections were counterstained lightly with Mayer's hematoxylin (Sigma), dehydrated, cleared in xylene, and mounted on cover slips with Permount (Fisher Scientific, Fairlawn, NJ).
All sections were evaluated independently by two observers (P. Loukopoulos and W. F. Robinson), and variations in the interpretation of results were resolved using a dual-headed microscope. The results of the IHC analysis were interpreted without knowledge of clinical data. Negative and positive control slides were compared for nonspecific and background staining or cross-reactivity. 2 Only those tumors containing cells with a distinct nuclear staining for p53 were considered to be positive. The fraction of positively stained nuclei (FPN) for p53 was scored in the following way: 0% = score 0 (negative tumors), 1–10% = score 1, 11–25% = score 2, 26–50% = score 3, 51–100% = score 4 (positive tumors).
Simultaneously, the mean nuclear staining intensity (NSI) was estimated and expressed as weak, moderate, or strong (score 1, 2, or 3). To express the staining semiquantitatively, a p53 index was calculated by multiplying the fraction of stained nuclei score (0, 1, 2, 3, or 4) by the nuclear staining intensity (1, 2, or 3). Cell staining patterns, regional variation, staining specificity, and agreement between multiple tumor sections were also noted.
Statistical analysis
The p53 index, NSI, and FPN were related to clinicopathologic parameters and compared between tumor types. To assess the statistical significance of intergroup differences in quantitative data, the Kruskal-Wallis one-way analysis of variance (ANOVA) on Ranks or the Mann-Whitney rank sum test were performed. Data grouped in contingency tables were analyzed statistically using the chi-square test or Fisher exact test for comparison of proportions.
Statistical significance was set at 0.05. The collected data were analyzed using the Jandel Sigmastat® Statistical Software (version 2.0, SPSS Science, Chicago, IL).
Results
A streptavidin–biotin IHC protocol for the detection of p53 protein in canine osseous tumors was developed and optimized. One hundred sixty-nine canine tumors were evaluated (Fig. 2). The number of positive cases for every tumor type is given in Table 1. FPN was significantly associated with NSI (P < 0.001).
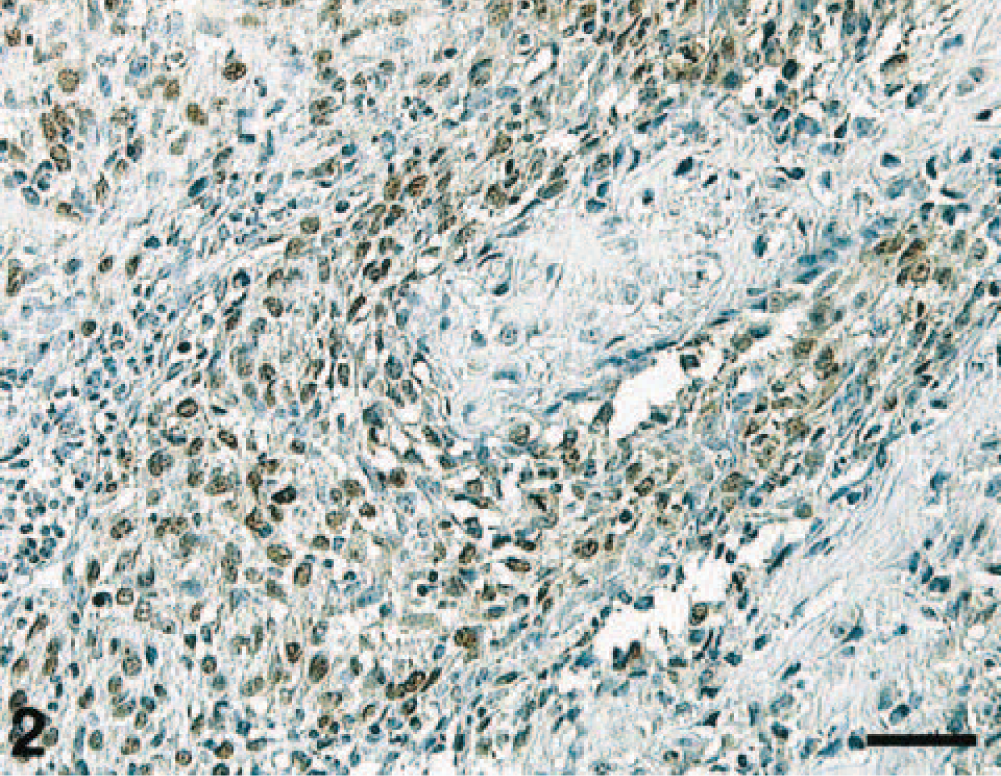
Canine osteosarcoma. IHC localization of p53 protein: intense nuclear staining of tumor cells. Streptavidin–biotin–peroxidase method, Mayer's hematoxylin counterstain. Bar = 60 μm.
Osteosarcomas showed significantly higher p53 index, FPN, NSI, and ratio of positive cases when compared with all other tumors examined as a group as well as when compared with the group of other osseous tumors, multilobular tumors of bone (MTB), and benign tumors as a group (Table 2). Osteosarcomas also had significantly higher p53 index and FPN than did osseous tumors that were initially diagnosed as osteosarcomas, the diagnosis of which was revised (misdiagnosed tumors), other sarcomas of bone as a group, and chondrosarcomas (Table 3).
P53 index and FPN values for selected osteosarcoma groups.
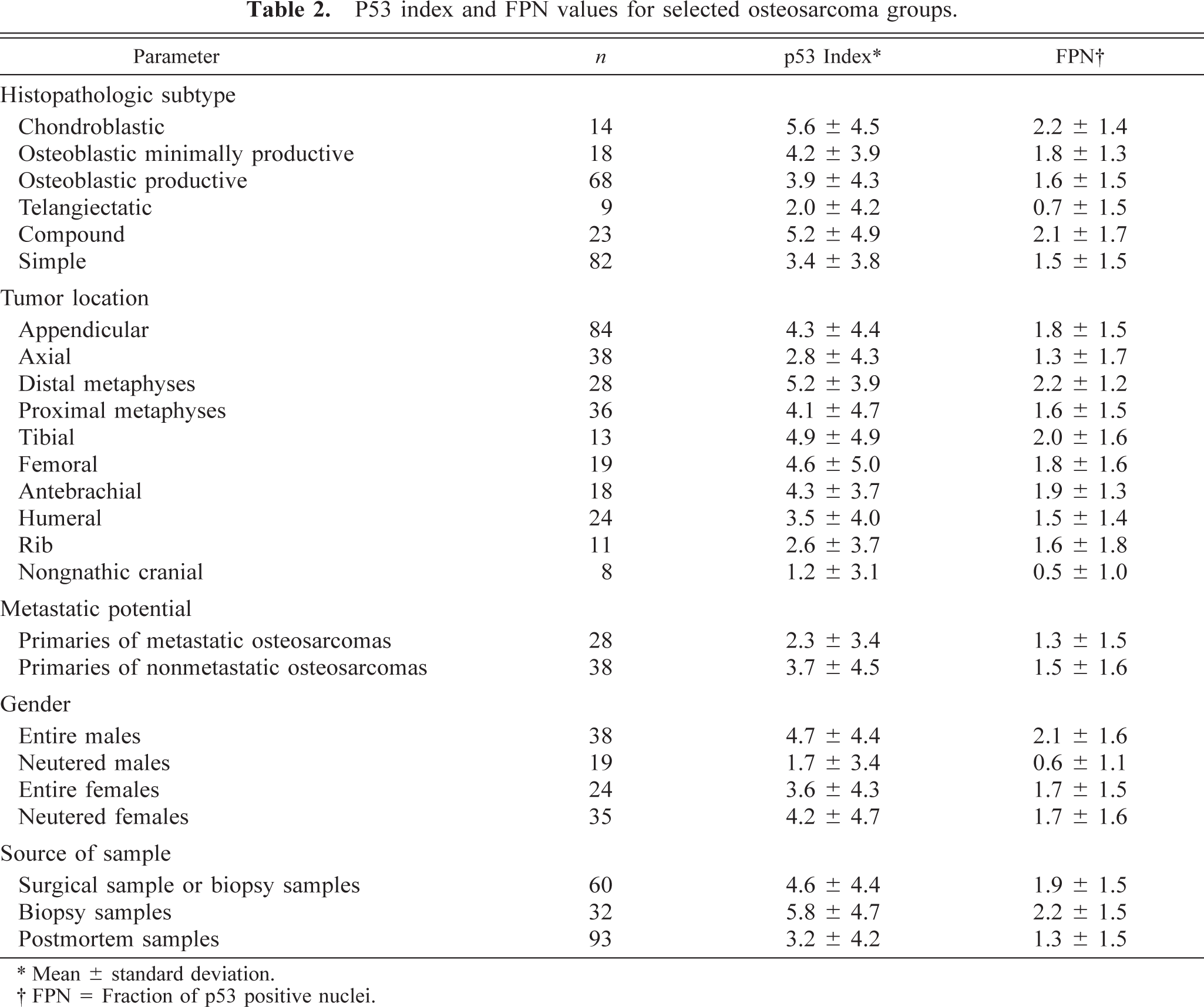
∗ Mean ± standard deviation.
† FPN = Fraction of p53 positive nuclei.
P values for p53 parameters resulting from the comparison of osteosarcoma with other tumor types.∗
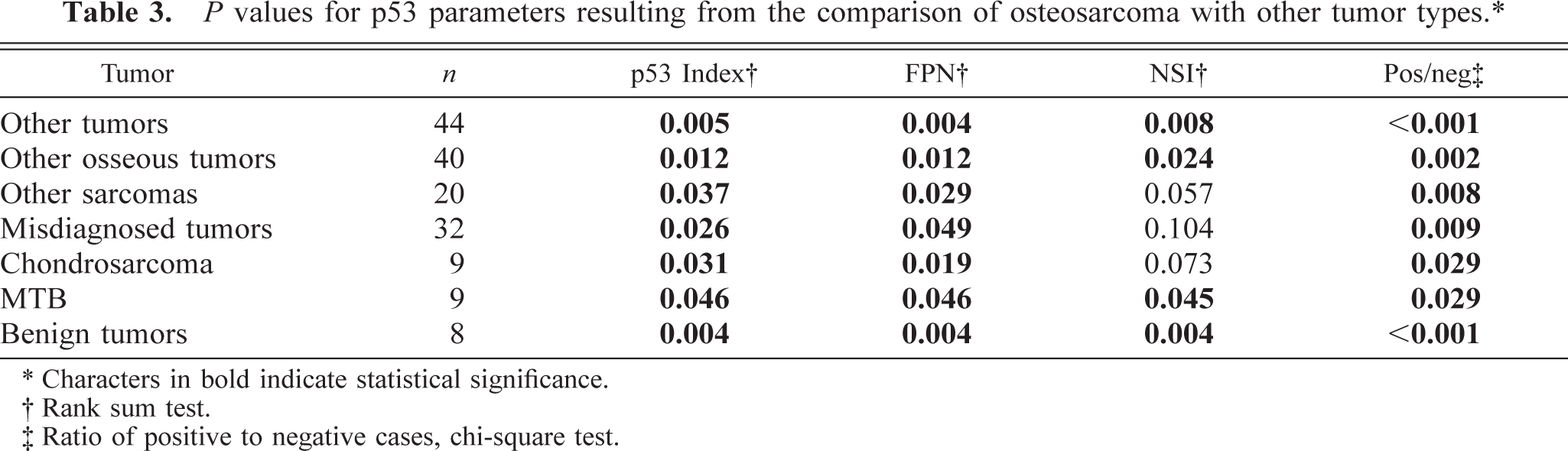
∗ Characters in bold indicate statistical significance.
† Rank sum test.
‡ Ratio of positive to negative cases, chi-square test.
There were no significant differences in p53 index, FPN, or NSI when tumors other than osteosarcomas were compared pairwise. All benign tumors (n = 8), including four cases of MTB and a case each of osteoma, cartilaginous exostosis, bone cyst, and cementoblastoma, were p53 negative.
p53 index, FPN, and NSI were significantly associated with the histopathologic grade of osteosarcomas (Kruskal-Wallis one-way ANOVA on ranks, P < 0.001 for all three parameters), the histologic score (one-way ANOVA on ranks, P < 0.001 for all parameters), the mitotic index (P = 0.004 for p53 index and FPN, P < 0.001 for NSI), the degree of tumor necrosis (P = 0.02 for p53 index, P = 0.028 for FPN, P = 0.032 for NSI), and the degree of nuclear pleomorphism (P < 0.001 for all parameters). Osteosarcomas with a high tumor grade, mitotic index, or degree of pleomorphism had significantly higher p53 index and FPN than did osteosarcomas where such parameters were low (Table 5). Tables 4 and 5 show the interrelationships between tumor grade, mitotic index, necrotic score, and degree of pleomorphism groups of osteosarcoma when compared pairwise for p53 index, FPN, and NSI values.
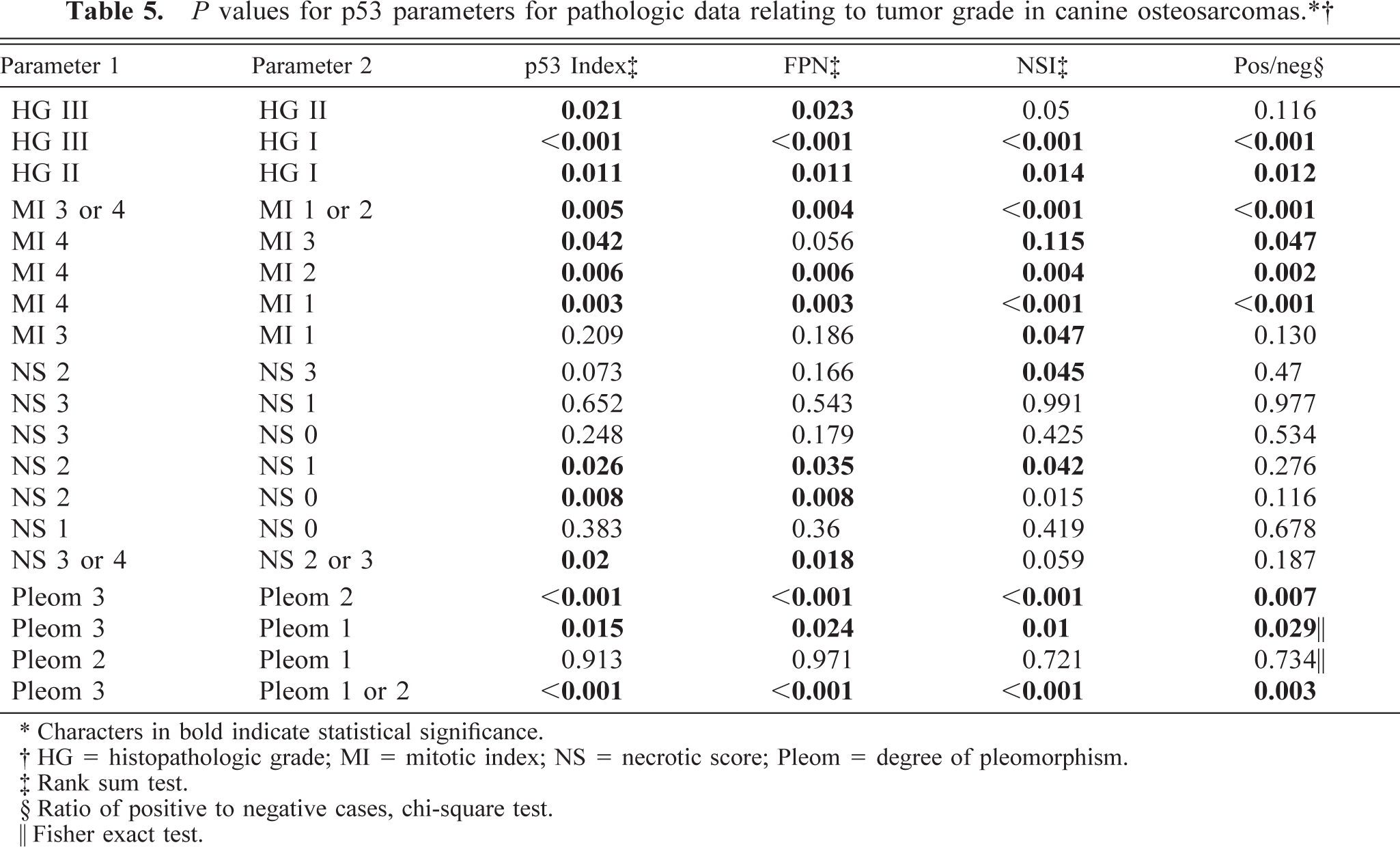
Histopathologic data relating to tumor grade for osteosarcoma cases.∗
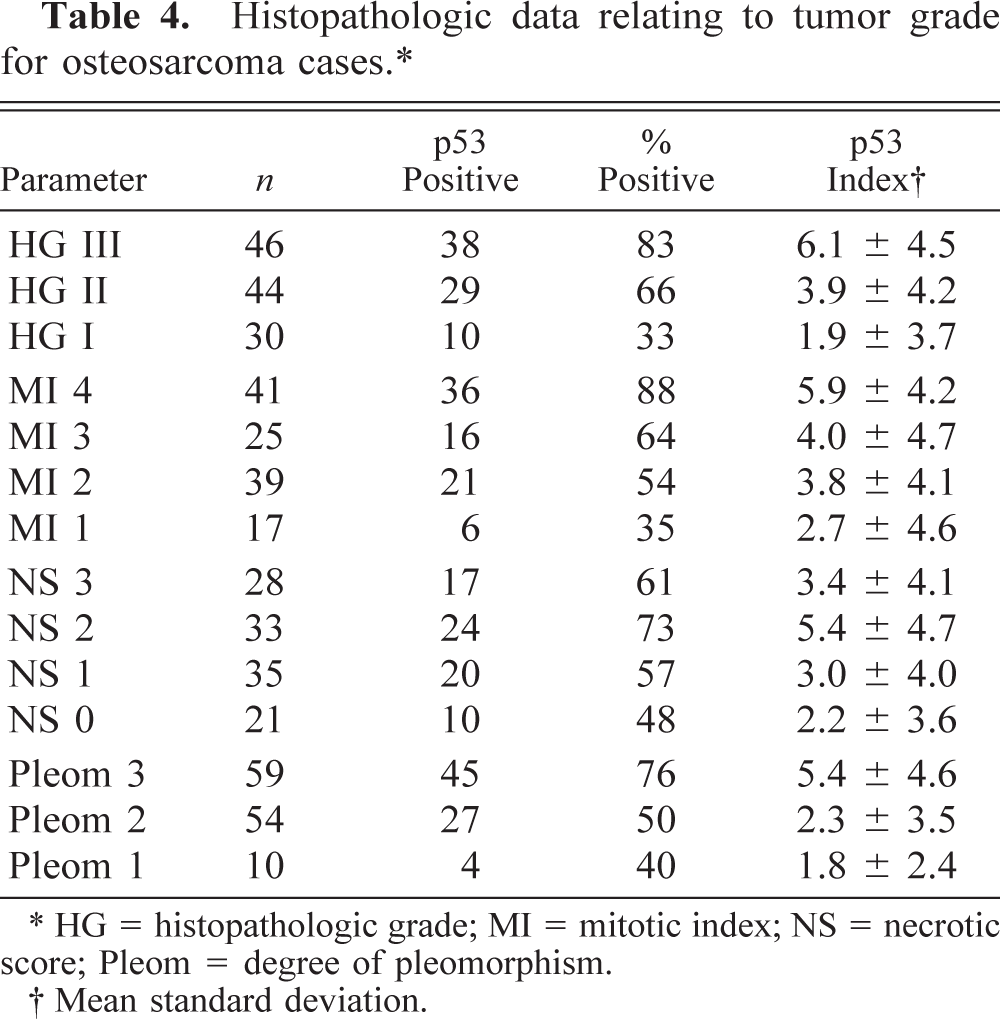
∗ HG = histopathologic grade; MI = mitotic index; NS = necrotic score; Pleom = degree of pleomorphism.
† Mean standard deviation.
∗ Characters in bold indicate statistical significance.
† HG = histopathologic grade; MI = mitotic index; NS = necrotic score; Pleom = degree of pleomorphism.
‡ Rank sum test.
§ Ratio of positive to negative cases, chi-square test.
‖ Fisher exact test.
Chondroblastic osteosarcomas had significantly higher p53 index (P = 0.043), FPN (P = 0.050), and NSI (P = 0.019) than telangiectatic osteosarcomas. Osteoblastic productive and osteoblastic minimally productive osteosarcomas had significantly higher NSI (P = 0.012 and 0.038, respectively) but not p53 index (P = 0.082 and 0.084, respectively) or FPN (P = 0.084 and 0.099, respectively), compared with telangiectatic osteosarcomas. Telangiectatic osteosarcomas also had significantly lower p53 index (P = 0.050) and NSI (P = 0.009) but not FPN (P = 0.058) than the group of all other osteosarcoma subtypes. Compound osteosarcomas did not have a significantly higher p53 index than did simple ones (P = 0.32).
Appendicular osteosarcomas showed significantly higher p53 index (P = 0.028), NSI (P = 0.006) and ratio of positive cases (58/84 versus 16/38, P = 0.009) but not FPN (P = 0.086), compared with axial osteosarcomas but not with extraskeletal ones (n = 4). There was no significant difference in p53 parameters between appendicular and axial osteosarcoma metastases.
The ratio of p53-positive cases was significantly higher in appendicular osteosarcomas that arose in distal (25/28 or 89%) compared with proximal metaphyses (23/36 or 63%) (chi-square test, P = 0.042).
In relation to tumor location, antebrachial osteosarcomas had the highest p53 index, whereas cranial ones had the lowest. p53 index was significantly lower in nongnathic cranial osteosarcomas compared with femoral (P = 0.049), tibial (P = 0.032), and antebrachial (P = 0.026) osteosarcomas but not with humeral (P = 0.09) or hind limb osteosarcomas (n = 38, P = 0.08).
There was no statistical difference in p53 index in the primaries of metastatic osteosarcomas (n = 28) compared with osteosarcomas without demonstrable metastases at the time of biopsy or necropsy (n = 38), in osteosarcoma metastasies (n = 24) compared with the primaries of metastatic or nonmetastatic osteosarcomas, in osteosarcoma cases with documented parallel presence of other tumors (n = 9) compared with osteosarcoma cases without it (n = 49), or in forelimb (n = 44) compared with hind limb osteosarcomas (n = 38).
p53 parameters were not associated with age or sex alone but were associated with the neutered or entire status of the animal alone or in combination with the sex of the animal. Entire animals, whether male or female, had significantly higher FPN than did neutered animals (P = 0.042). Entire males had higher p53 index, FPN, and NSI than did neutered females, entire females, and neutered males, in descending order. Entire males had significantly higher p53 index (P = 0.009) and FPN (P = 0.003) but not NSI (P = 0.058) than did neutered males. Entire females had significantly higher FPN (P = 0.025) but not p53 index (P = 0.065) or NSI (P = 0.145) than did neutered males. Neutered females tend to have higher p53 index than do neutered males but the difference was not significant (P = 0.066).
p53 expression was associated with the breed of the animal. Rottweilers (n = 23) had higher p53 index (4.8 ± 4.5) than did Great Danes (1.8 ± 2.9, n = 10)(P = 0.040) or Terriers as a group (1.0 ± 1.6, n = 6) (P = 0.038).
Samples taken as surgical specimens and biopsy samples, or biopsy samples alone, had a higher p53 index than did samples taken during postmortem examinations (P = 0.031 and 0.01, respectively).
Cytoplasmic staining was observed in 21 tumors (12%) including 15 osteosarcomas, 3 MTB, 2 chondrosarcomas, and 1 plasmocytic sarcoma. These tumors were registered as positive only if nuclear staining was present, as was the case for 17 cases, including all osteosarcomas. In all but three cases, cytoplasmic staining was moderate or intense and, where applicable, as intense as nuclear staining, a fact that sometimes created difficulties in the interpretation of the results, especially when complicated by background staining.
Discussion
In the present study, p53 overexpression is demonstrated in a number of canine osseous tumors, and evidence is provided for the clinical and pathologic relevance of p53 index in these tumors, with particular emphasis on osteosarcoma.
IHC overexpression of p53 in canine tumors has so far been reported in osteosarcomas and MTB, 92 benign and malignant mammary tumors, 46 , 91 epithelial colorectal tumors, 74 , 120 cutaneous mast cell tumors, 33 , 49 skin papillomas, 108 seminomas and Sertoli-cell tumors but not in Leydig-cell tumors, 47 squamous cell carcinomas, 107 nasal adenocarcinomas, 29 and a small number of other canine tumors. 29 , 51 In one study on mammary tumors, mammary osteosarcomas were the only tumor type that exhibited p53 immunoreactivity. 46
In the present study, p53 protein expression was detected immunohistochemically in 10 osseous tumor types, in most of them for the first time. Our results are in accordance with a previous study, 92 in that p53 protein is overexpressed in most of the canine osteosarcomas and that the prevalence of overall positivity is higher in osteosarcoma compared with MTB and in appendicular compared with axial osteosarcomas. Furthermore, in our study using the derived term, p53 index, osteosarcomas showed significantly higher p53 index compared with chondrosarcomas, benign tumors, other osseous tumors, and other osseous sarcomas, confirming the observation that p53 expression correlates with highly aggressive tumor behavior. Interestingly, osteosarcomas also had higher p53 index than did misdiagnosed tumors that were initially diagnosed as osteosarcomas. This fact, in conjunction with the above, leads us to suggest that the addition of IHC examination of canine osseous tumors for p53 may prove to be a valuable tool in their sometimes challenging differential diagnosis, particularly regarding cartilage-forming tumors, such as chondrosarcoma and MTB, which were misdiagnosed more frequently than other tumor types.
Previous studies suggested that p53 expression is not useful prognostically, in association with survival time, in canine epithelial colorectal tumors 120 or cutaneous mast cell tumors. 33 , 49 In one of the latter studies on cutaneous mast cell tumors, p53 expression was associated with tumor grade and site but not with breed predisposition, tumor size, grade, and differentiation. 33
In the present study, a direct correlation is demonstrated between the p53 index and a range of clinicopathologic parameters, including the histologic grade and score of osteosarcomas, the mitotic index, degree of tumor necrosis, degree of nuclear pleomorphism within the tumor, histopathologic subtype, appendicular or axial location of the tumor, tumor site and proximity to the body axis, breed, sex, and entire or neutered status.
The question of the association of survival time and the p53 index for osteosarcoma remains to be determined. However, recent studies have shown that the histologic grading of osteosarcomas is of prognostic significance 56 and is currently used routinely to make decisions on the clinical management of the tumors, 102 particularly regarding chemotherapy protocols. 4 , 103 , 104 p53 index may therefore be useful as an additional indicator to those traditionally used in the prognostic evaluation of osteosarcoma cases.
The relevance of classifying osteosarcomas into subtypes has been questioned in the past by some authors and the issue remains somewhat unresolved. 76 , 101 The significant difference in p53 index between the various subtypes in our study tends to support the view that subtypes represent distinct clinicopathologic entities. Whether certain subtypes have any association with increased or decreased survival times remains to be answered. We consider, however, that familiarity with the different histopathologic presentations of osteosarcoma may be helpful in establishing an accurate diagnosis and, consequently, a more suitable therapeutic approach, having an indirect positive effect on survival time. The higher p53 index observed in chondroblastic osteosarcomas may be caused by their, yet to be confirmed, more aggressive biological behavior and higher rate of unrestrained cell proliferation. Alternatively, our findings may suggest that p53 overexpression is not exclusively associated with malignancy but may in part be attributed to the increased rate of matrix turnover and tissue remodeling within chondroblastic osteosarcomas and the recently proposed role p53 may play in these processes in conjunction with matrix metalloproteinases. 13
Similarly, the lower p53 index observed in telangiectatic osteosarcomas may be attributed to their possibly less malignant phenotype or to the possibility that the formation of blood-filled cystic lesions, which characterizes telangiectatic osteosarcomas, 98 is a early event in their pathogenesis, unlike, perhaps, the inactivation of the p53 gene or p53 protein.
The presence of metastases or other tumors did not generally appear to influence p53 expression, either because p53 index alone truly does not reflect metastatic potential or because the retrospective nature of the study resulted in cases with possibly incomplete necropsies being included in the nonmetastatic group.
Body weight 90 and size 109 are established negative prognostic indicators in canine osteosarcoma. The higher p53 index observed in distal compared with proximal osteosarcomas may be attributed to the greater effect body weight has on the distal compared with the proximal metaphyses of long bones.
Interestingly, p53 index in osteosarcoma was higher in Rottweilers than in Great Danes and Terriers, a finding that may be related to selective breeding for desired traits in certain breeds of dogs. Knowledge of the relative frequency of the breeds within the canine population and the overall risk of osteosarcoma and other tumors for every breed would help create a more complete picture. In any case, further epidemiologic and genetic studies are needed to elucidate potential breed susceptibilities and we tend to agree with Veldhoen et al. 114 in that acquiring information about p53 inactivation in canine tumors could prove invaluable in the successful outbreeding of inherited predisposition to cancer in the dog.
An interesting finding of the study was the fact that entire males had higher p53 index than did neutered males. This suggests a role for androgenic hormones in the pathogenesis of canine osteosarcoma that requires further investigation.
The origin of the sample has a clear effect on p53 immunoreactivity. The reduced immunoreactivity of postmortem samples may be caused by their sometimes prolonged fixation time, the longer interval before fixation or the larger size of the samples compared with biopsy material, or it may be caused by the possibility that p53 overexpression is more evident at the earlier stages of osteosarcoma pathogenesis, presumably represented by the biopsy material. Cytoplasmic staining was detected in 21 (12%) of the tumors examined. Most previous reports have concentrated on nuclear immunolocalization of p53 protein, whereas only a few have reported or discussed its cytoplasmic localization, 42 , 116 including that in human osteosarcomas, 5 canine intestinal adenocarcinomas, 29 and testicular tumors. 42 , 116 Accumulation of p53 protein in the cytoplasm may be a result of an abnormal nuclear localization signal or may be caused by cytoplasmic factors, such as a 70-kDa heat shock protein (HSP70) that binds to the mutant p53 protein, causing its cytoplasmic localization. 42 , 26 Although p53 positivity is usually expressed as FPN, the nuclear staining intensity was associated with a number of clinicopathologic parameters in our study, in some cases in the absence of such association of these parameters with FPN, prompting us to include it in our analysis. p53 indices (positivity × staining intensity) have also been used in assessing p53 expression in other, human, malignancies. 1 , 22 , 95 The present study is, to our knowledge, the only one to describe a close relationship between the p53 index of a nonhuman tumor and its clinicopathologic parameters. It is also one of few studies to describe a relationship between the p53 index of any tumor and certain pathologic parameters such as the tumor subtype. It is evident that there is a fundamental abnormality in the function of the p53 tumor-supressor gene in canine osseous tumors and osteosarcomas in particular. This is clearly central not only to the development of the disease but, apparently, also to the differentiation of osteosarcomas into particular histopathologic subtypes. From this study, we consider that the addition of an IHC examination for p53 and the derived index has the potential to be used as an additional prognostic indicator for canine osseous tumors. A strategic approach incorporating other indicators will provide avenues to understanding the etiology of canine osteosarcoma and will help develop a broader-based, and therefore more accurate, diagnostic and prognostic index and therapeutic protocol for canine osteosarcoma patients.
Footnotes
Acknowledgements
This study was supported by a grant from the Australian Companion Animal Health Foundation. Mr. Loukopoulos was a University of Queensland Sister Janet Mylonas Memorial Scholarship holder. We thank Ms. J. Priest for statistical advice, Mr. C. Cazier for technical support, and the members of the Veterinary Teaching Hospital and the Departments of Companion Animal Sciences and Veterinary Pathology and Anatomy, University of Queensland, for providing the clinical and archival material. Preliminary results from this study were presented at the WSAVA/FECAVA World Congress 2000.