Abstract
The immunohistochemical expression of p63, ΔNp63, and p53 was studied in mixed tumors of canine mammary glands (13 benign mixed tumors and 19 carcinomas arising from benign mixed tumors) to determine the role of p63 and its isoform ΔNp63 in the development of mixed tumors, as well as to assess its relation with p53. P63 was expressed in myoepithelial cells of all benign mixed tumors and in 18 of 19 carcinomas in mixed tumors. The p63-negative carcinoma in mixed tumors was invasive, and a loss of p63 was detected in the other malignant tumors showing a discontinuous p63-stained myoepithelial layer. ΔNp63 was expressed in all benign mixed tumors but only in p63-positive carcinomas in mixed tumors. Despite its positive correlation with p63 expression in carcinomas in mixed tumors (r = 0.8323, P < .00001), ΔNp63 expression showed a decrease in benign tumors. Positivity for p53 was detected in 2 of 13 and 1 of 19 benign mixed tumors and carcinomas in mixed tumors, respectively. There was no correlation between p63 or ΔNp63 and p53 expression. Our data support the notion that the decrease of p63 expression, in particular of its isoform ΔNp63, seems to be an important factor in the development of carcinomas in mixed tumors.
Introduction
Mixed tumors are common neoplasias in canine mammary glands and human salivary glands and share many clinical and histomorphologic characteristics. 11 Benign mixed tumors are histologically characterized by a mixture of epithelial components (ductal and/or acinous cells and myoepithelial cells) within an apparently mesenchymal stroma, capable of producing different degrees of myxoid, chondroid, and bone tissues. 6, 25
Similarly to mixed tumors of human salivary glands, canine benign mixed tumors may develop carcinomatous foci resulting in carcinomas, 10, 18, 25 and an accurate distinction between these entities is critical in some cases. Difficulties arise, in particular, when distinguishing small and/or invasive carcinomatous foci.
In human breast tumors, the myoepithelial cells are considered the key to distinguish tumors in situ from tumors that are invading the stroma, because myoepithelial cells disappear in invasive tumors. Several studies have already investigated the applicability of myoepithelial cell markers 1, 42 The role of the myoepithelial cell markers has also been studied in the histopathology of mixed tumors of canine mammary glands. 4, 8, 20
The p63 gene, mapped in the chromosome 3q27-29, is a homologue of the p53 tumoral suppressor gene. The product encoded by p63 has been associated with an important role in cell proliferation, differentiation, senescence, and adhesion of stratified epithelial cells and can exert different biologic functions in several tumors. 3, 24, 36, 38
The p63 protein is essential in maintaining the precursor cell population in several epithelial tissues and may be considered a highly sensitive and specific immunohistochemical marker of human and canine myoepithelial and stem cells. 1, 8
The transcription of the p63 gene is based on 2 distinct promoters and alternative splicings responsible for 2 protein classes that, despite their structural homology, have apparently different functions and at least 6 isoforms.
Three isoforms contain an N-terminal transcription domain similar to p53 protein. It is known that this transactivation (TA) class is capable of activating p53 gene, that is, inducing apoptosis and regulating the cell cycle, and of acting as a suppressor gene of a nonclassical tumor. The other 3 isoforms do not have the NH2-terminal domain and are denominated ΔN. They act on the regulating cell cycling and apoptosis through the alternative mechanisms, as opposed to the way of TAp63/ p53. The distinct COOH domains (α, β, and γ) have functions that are not well known. 41 Only TAp63γ and TAp63α seem to be related to specific functions, such as transactivation of target genes of the p63 protein (p21, BAX and MDM2) 27 and differentiation of keratinocytes, respectively. 14
This study investigated the immunohistochemical expression of the p63 protein in benign mixed tumors and carcinomas arising from benign mixed tumors of canine mammary glands, as well as assessed possible biologic functions of its isoform ΔNp63 in the tumorigenesis of these lesions. Given that the interactions between the isoforms of p63 and p53 protein may be essential to understand the transition between normal cell cycling and the formation and development of tumors, we examined the correlation between these proteins.
Materials and Methods
Case selection
Thirty-two cases of canine mammary gland tumors were retrieved from the files of the Laboratory of Comparative Pathology, Department of General Pathology, Institute of Biological Science, Federal University of Minas Gerais, Brazil. The selection was carried out by 2 pathologists (G. D. Cassali, A. C. Bertagnolli) and based on the histopathologic diagnosis according to World Health Organization criteria. 25
After reviewing the cases, 13 benign mixed tumors and 19 carcinomas arising from benign mixed tumors were selected for immunohistochemical studies.
All dogs were females, ranging in age from 1 to 14 years. The tumor size (largest diameter) was determined by the pathologist on the gross specimen and classified as + = 3 cm, ++ = 3–5 cm, or +++ = <5 cm.
Immunohistochemical staining
All tissue samples were routinely fixed in 10% neutral formalin and embedded in paraffin. Sections of 3 µm were cut from one representative block of each case and collected onto glass slides. The slides were deparaffined, rehydrated in graded alcohols, and submitted to heat-induced antigen retrieval (water bath at 98°C) with antigen retrieval solution (DAKO, pH 6.0). Endogenous peroxidase activity block was performed with 3% hydrogen peroxidase in metanol.
Primary antibodies were diluted in phosphate-buffered saline and incubated for 60 minutes at room temperature. Primary antibodies used for immunohistochemical evaluation included a monoclonal anti-human p63 (1 : 100, clone 4A4, Neomarkers), which recognizes all known isotypes of p63, a rabbit anti-human polyclonal anti-p40 that recognizes only ΔNp63 isoforms (1 : 2,000, p40, Calbiochem), and an anti-human polyclonal p53 antibody (1 : 150, clone CM1, Novocastra). For confirmation of the myoepithelial phenotype, complementary immunohistochemical analysis using a monoclonal antibody anti-α-smooth muscle actin (α-SMA; 1 : 100, clone 1A4, DAKO) was performed on consecutive serial sections of 4 benign mixed tumors and 5 carcinoma arising from benign mixed tumors.
The reaction was amplified using the streptavidin-biotin-peroxidase complex (Lab Vision) method. Diaminobenzidine was used as a chromogen. Slides were counterstained with Mayer's hematoxilin, washed in running water for 5 minutes, dehydrated, and mounted in a synthetic medium. Adjacent normal canine mammary tissue was used as positive control for p63, ΔNp63, and α-SMA.
Canine mammary tumors previously known to express high levels of p53 were used as positive controls for p53. Negative controls were obtained by omission of primary antibodies.
Evaluation of immunohistochemical data
The immunohistochemical results for p63 and α-SMA were evaluated for each of the mammary cell types, which were classified in accordance with their histologic morphology: 1) alveolar and ductal epithelial cells; 2) myoepithelial cells located above the basal lamina, which constitutes the periductal and periacinar basal layer (resting and proliferating); 3) spindle- and stellate-shaped cells proliferating in the interstitial areas; and 4) chondrocytes and osteoblasts of the ectopic cartilage and bone.
The morphology of resting myoepithelial cells is similar to that of normal cells. The proliferative myoepithelial cells form a small mass adjacent to the alveolar ducts. Spindle-shaped myoepithelial cells have distinct eosinophilic to clear cytoplasm and form solitary foci. Star-shaped cells have abundant mucinous stroma, occasionally accompanied by hyaline or chondroid changes. 4
The number of p53, p63, ΔNp63, and α-SMA reactive cells were assessed semiquantitatively using a scoring system: − = no staining, + = focal stained or <5% cells stained, ++ = between 5 and 50% cells stained, and +++ = tumors with >50% cells stained.
The cases were interpreted as positive if >5% of the neoplastic cells displayed distinct brown nuclear (p63 and ΔNp63) or cytoplasmic (α-SMA) staining. 31
The cases were considered positive for p53 if more than 10% of the neoplastic cells displayed distinct brown nuclei. 12
Statistical analysis
Statistical analysis was performed using the Graph Pad Prism v.3 software. The comparisons between different tumors and p63 and p53 expression were analyzed by Mann-Whitney test. To determine the correlation between p63, p53, and ΔNp63, data were analyzed by Spearman correlation test.
Results
The clinical, histopathologic, and immunohistochemical features of the tumors are summarized in Tables 1, 2, and 3. In the normal glandular parenchyma adjacent to the tumors, the myoepithelial cells were highly reactive to p63, ΔNp63, and α-SMA (+++), forming a continuous layer surrounding ducts and acinus (Figs. 1, 2). Immunoreactivity for p63 and ΔNp63 was detected in the cell nuclei, whereas for α-SMA it was observed along the cell cytoplasm of the myoepithelial cells. No immunoreactivity was observed for p53.
Characterization of cell types and immunoreactivity for p63 and ΔNp63 in mixed tumors of canine mammary gland. ∗
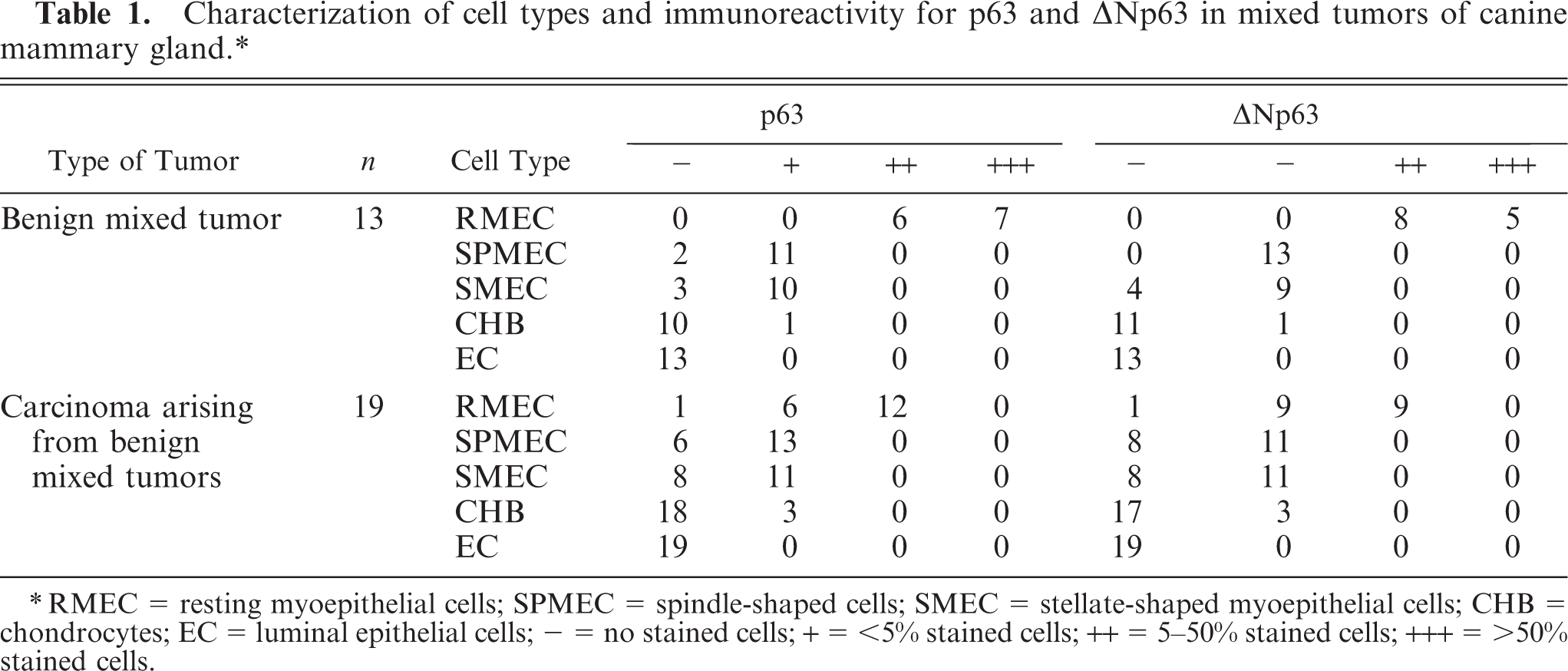
∗RMEC = resting myoepithelial cells; SPMEC = spindle-shaped cells; SMEC = stellate-shaped myoepithelial cells; CHB = chondrocytes; EC = luminal epithelial cells; - = no stained cells; + = <5% stained cells; ++ = 5-50% stained cells; +++ = >50% stained cells.
Characterization of tumor size and p63, p40, p53, and α-smooth muscle actin (SMA) expression in benign mixed tumors of canine mammary gland.

∗+ = 3 cm; ++ = 3-5 cm; +++ = <5 cm.
† - = no stained cells; + = <5% stained cells; ++ = 5-50% stained cells; +++ = >50% stained cells.
Characterization of tumor size and p63, p40, p53, and α-smooth muscle actin (SMA) expression in benign mixed tumors of canine mammary gland.
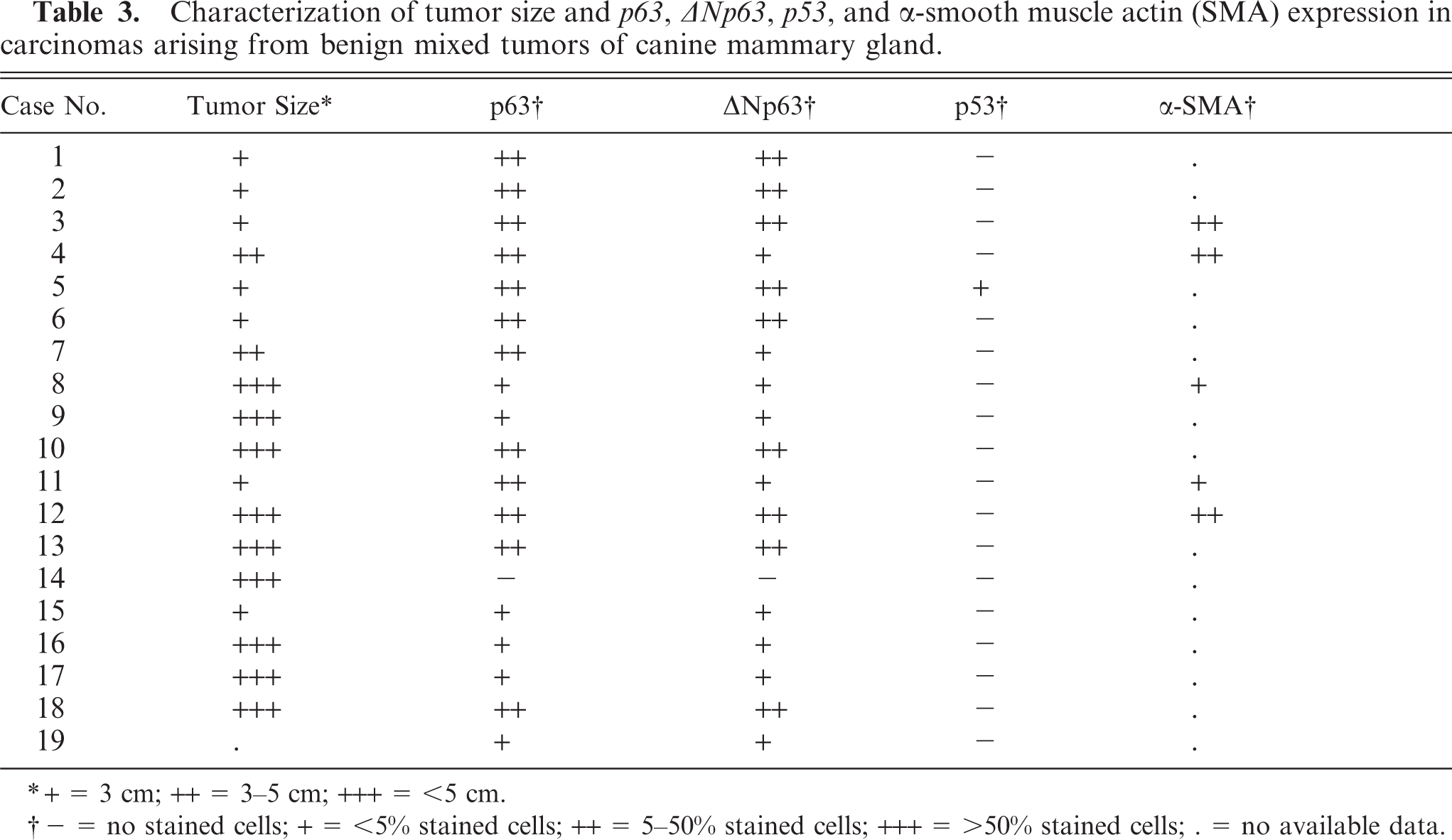
∗+ = 3 cm; ++ = 3-5 cm; +++ = <5 cm.
† - = no stained cells; + = <5% stained cells; ++ = 5-50% stained cells; +++ = >50% stained cells; . = no available data.
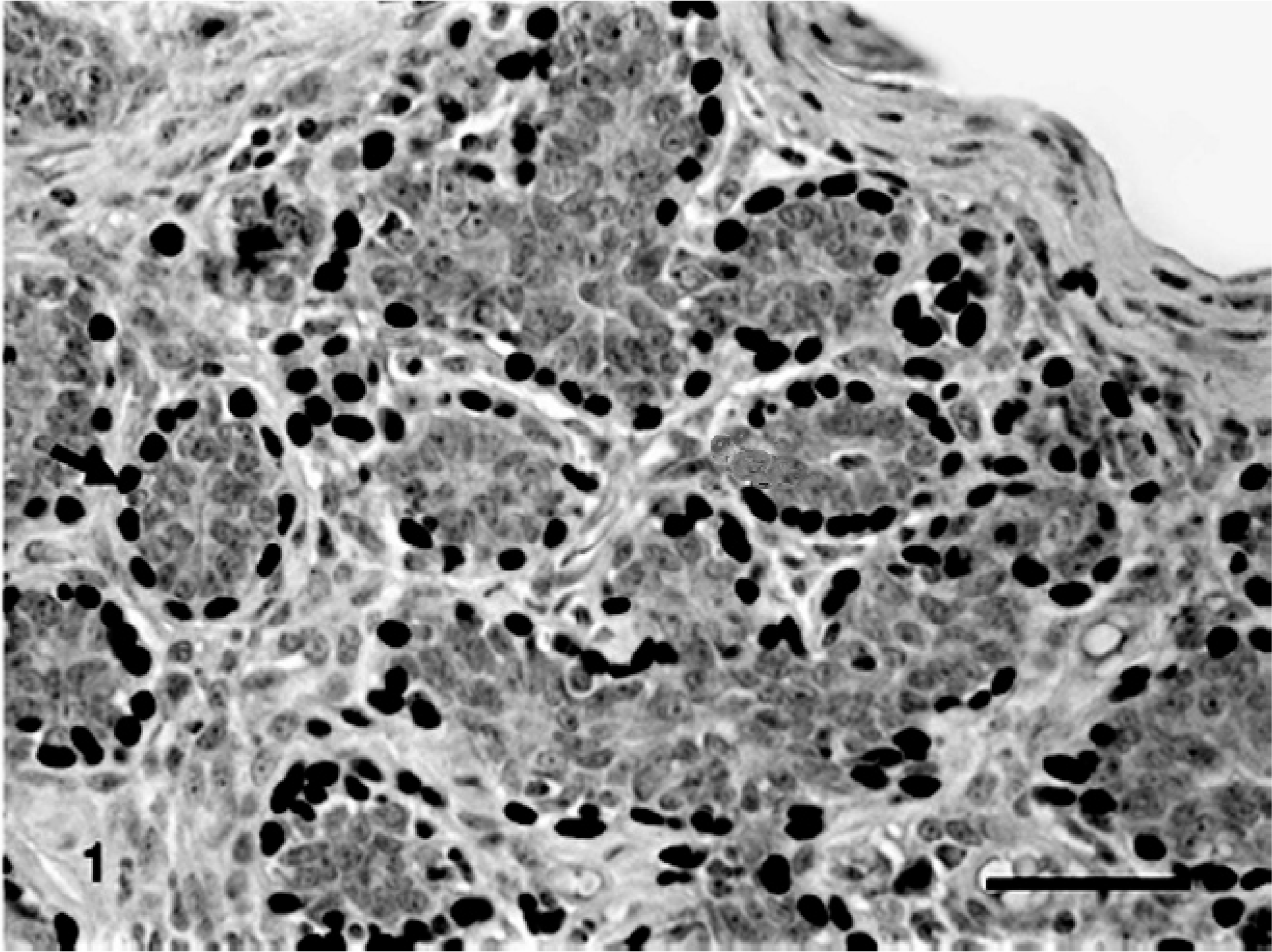
Benign mixed tumor of the mammary gland of dog No. 13. Periductal and periacinar myoepithelial cells p63-immunoreactives stained in brown (nuclei, arrow) (+++). Bar = 40 μ
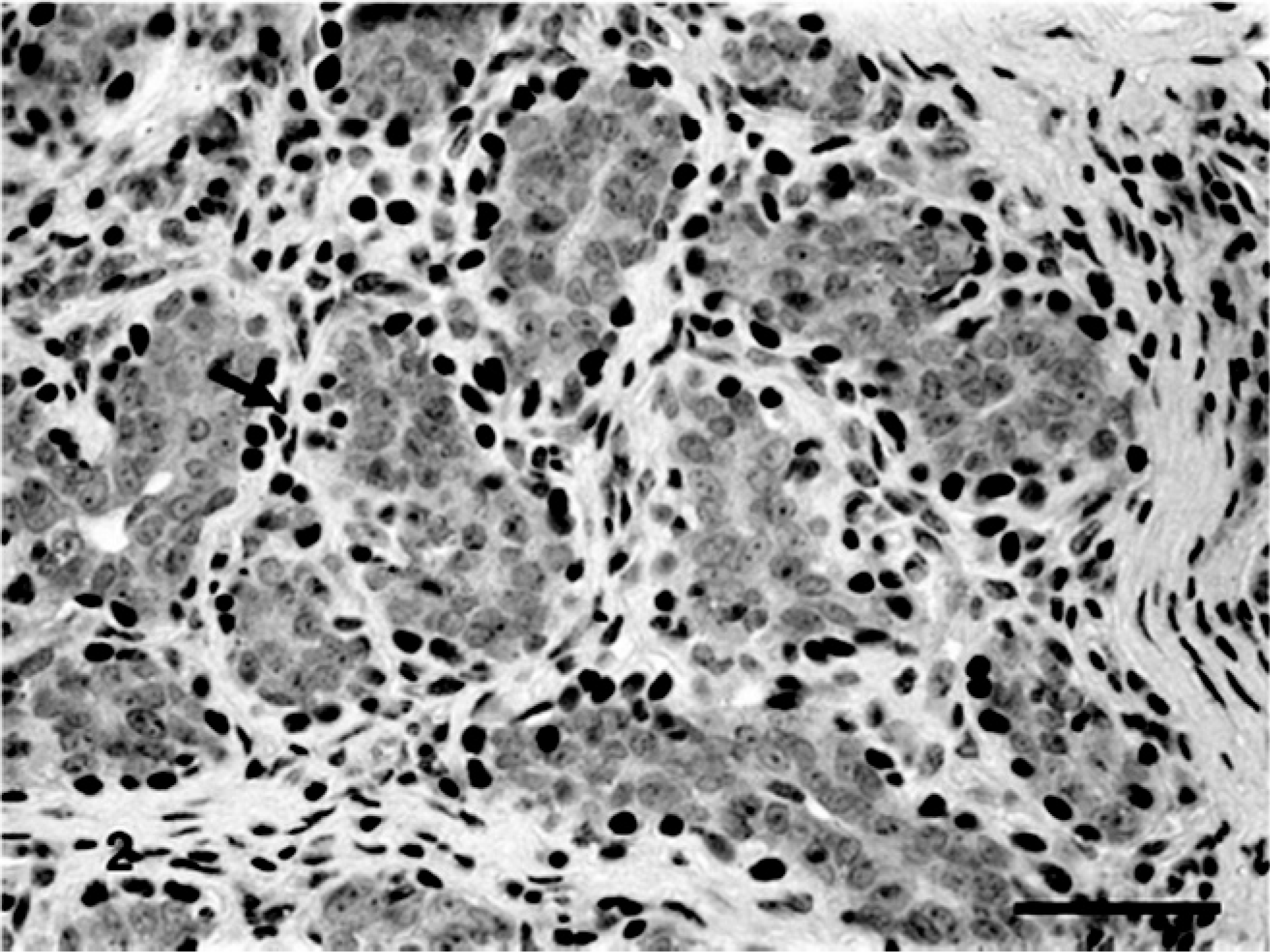
Benign mixed tumor of the mammary gland of dog No. 13. Periductal and periacinar myoepithelial cells ΔNp63-immunoreactives stained in brown (nuclei, arrow) (+++). Bar = 40 μ.
The benign mixed tumors submitted to further immunohistochemical study using α-SMA showed moderate cytoplasmic reactivity. All benign mixed tumors showed nuclear reactivity for p63 and ΔNp63 in the myoepithelial cells around ducts and acini. Seven cases showed high immunoreactivity (+++, Table 1), forming a continuous layer of p63-stained myoepithelial cells surrounding epithelial neoplastic cells, and 6 cases showed moderate reactivity (++), forming layers of separated p63-stained cells. For ΔNp63, 5 cases showed high immunoreactivity (+++) and 8 showed moderate reactivity (++). In myxoid and chondro-myxoid components, spindle and stellate cells showed focal reactivity (+) for p63 in 11 and 10 cases, respectively. Spindle cells and stellate cells were also immunoreactive for ΔNp63 (+) in 13 and 9 cases, respectively. Chondrocytes were immunoreactive for p63 (Fig. 3) and ΔNp63 in 1 case. Osteoblasts were p63 negative.
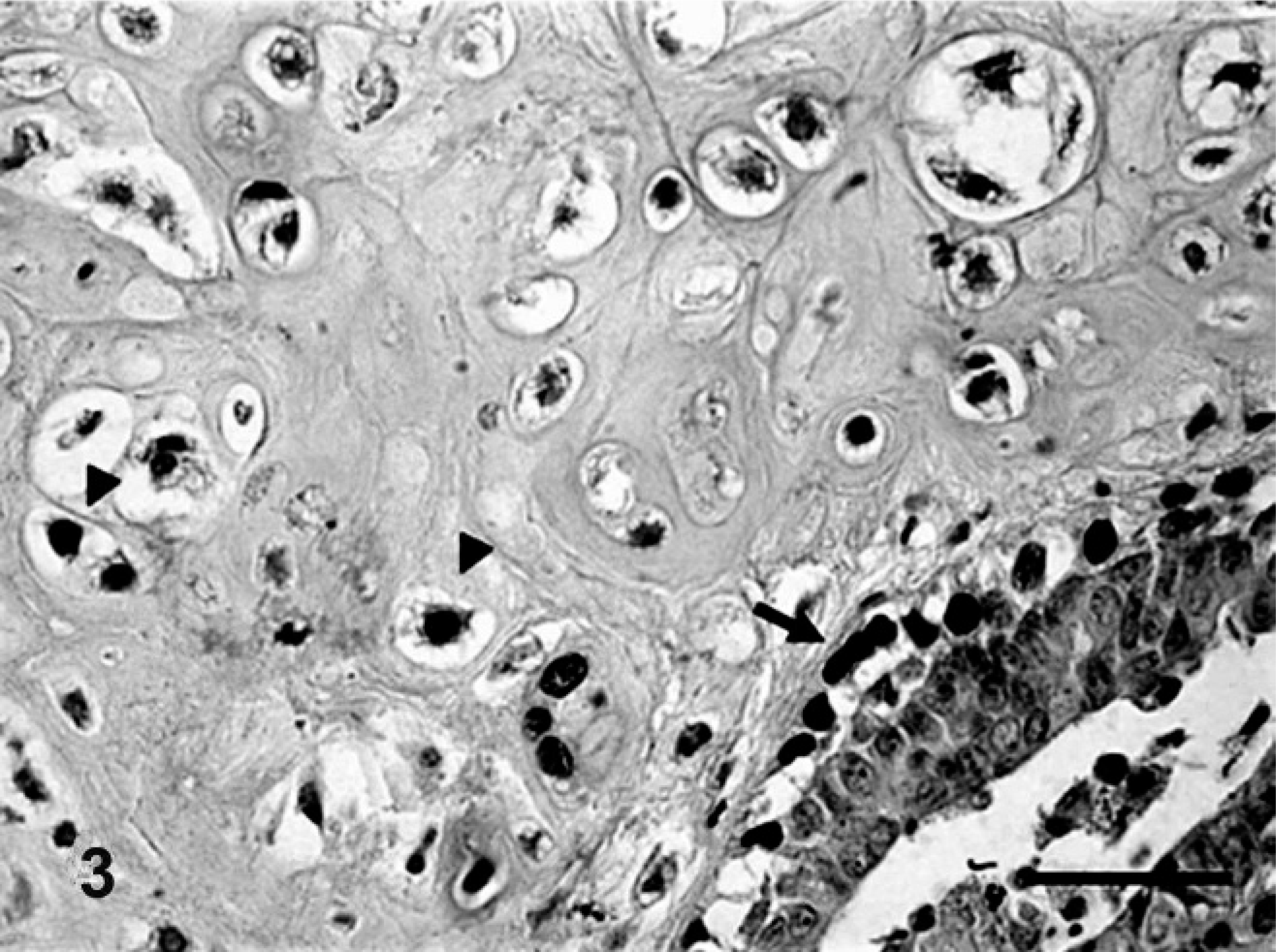
Benign mixed tumor of the mammary gland of dog No. 3. Periductal and periacinar myoepithelial cells immunoreactive for p63 (arrow) (+++). Note myoepithelial cells immunoreactive for p63 in areas undergoing chondroid differentiation (arrowhead). Bar = 20 μ.
Carcinomas in benign mixed tumors showed immunoreactivity for p63 in myoepithelial cells surrounding carcinomatous areas in situ. Reactivity varied from focal to moderate for p63 (Fig. 4) and ΔNp63 (Fig. 5), forming discontinuous stained layers. However, an invasive carcinoma arising from benign mixed tumor was totally devoid of p63 or ΔNp63 expression.
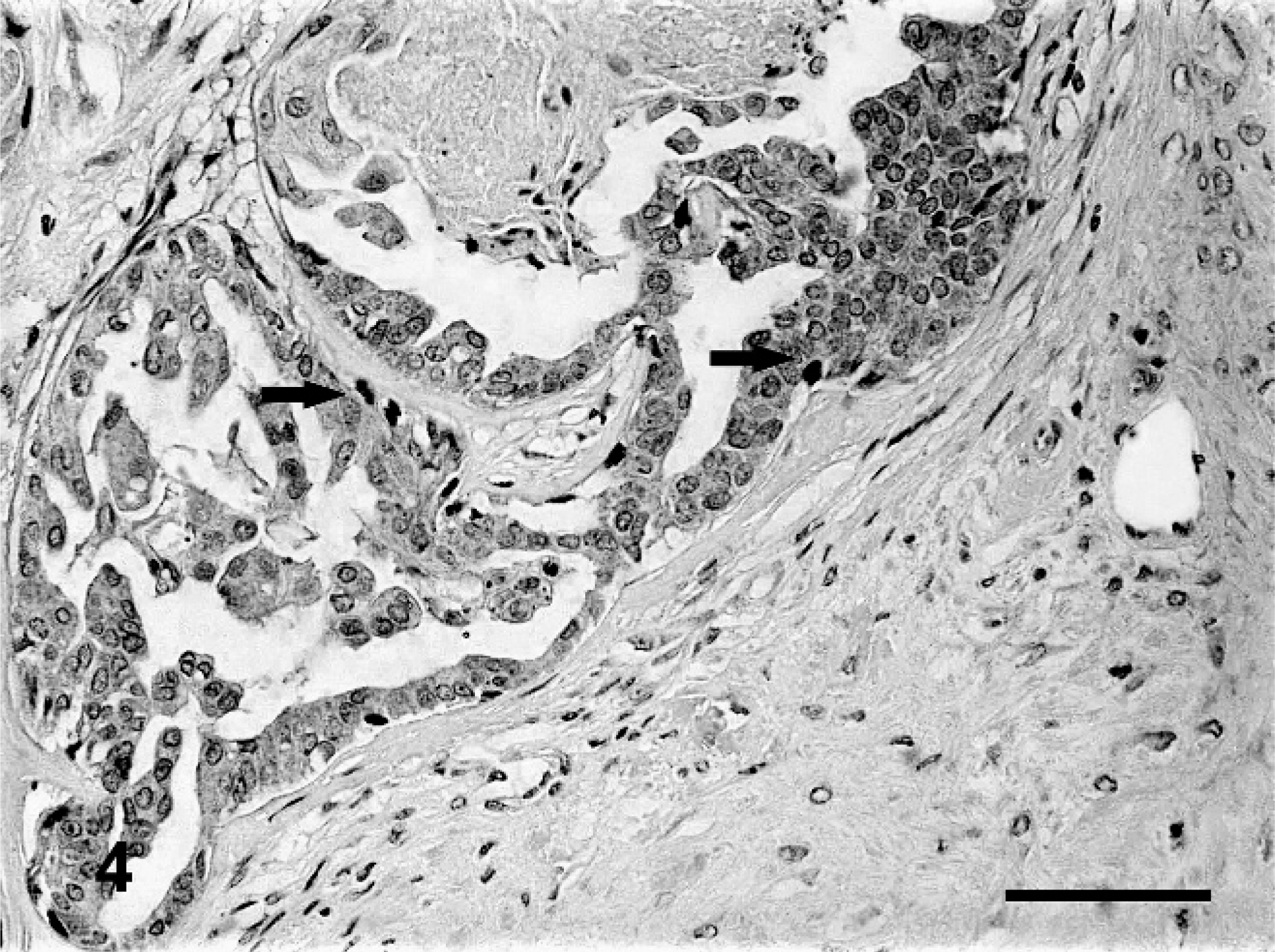
Carcinoma arising from benign mixed tumor of dog No. 19. Myoepithelial layer surrounding the acinar epithelium with reduced immunoreactivity for p63, forming a discontinuous layer (arrows) (+). Bar = 40 μ.
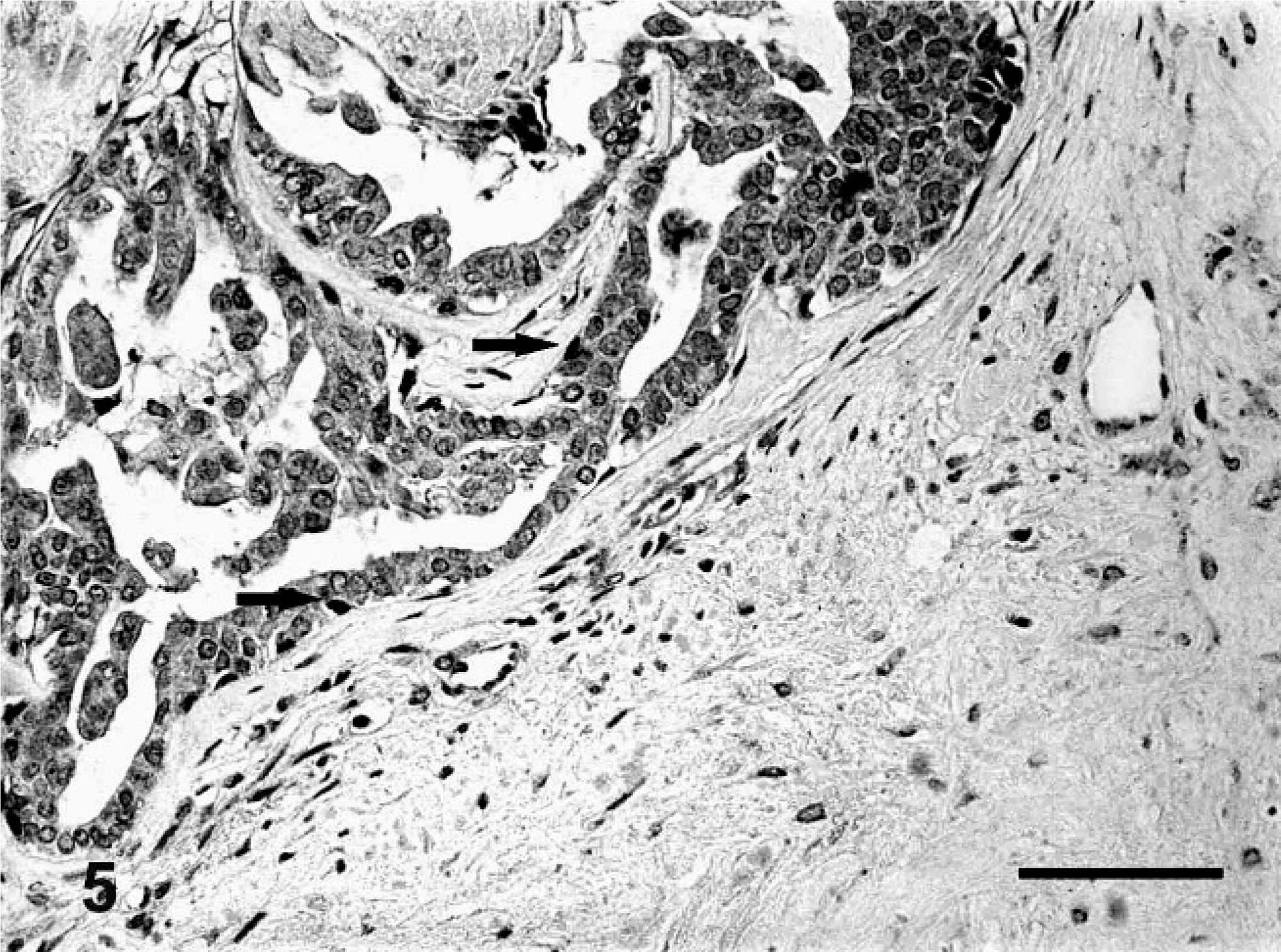
Carcinoma arising from benign mixed tumor of dog No. 19. Myoepithelial layer surrounding the acinar epithelium with reduced immunoreactivity for ΔNp63 (arrow) (+). Bar = 40 μ.
Spindle and stellate myoepithelial cells showed focal reactivity (+) for p63 in cases 13 and 11, respectively. Spindle cells and stellate cells were also immunoreactive for ΔNp63 in 11 cases. Chondrocytes were immunoreactive for p63 and ΔNp63 in 3 cases. Osteoblasts were p63 negative.
Positivity for p53 was detected in only 1 of the 19 cases of carcinomas in benign mixed tumors and in 2 cases of benign mixed tumors.
Immunoreactivity for p63 and ΔNp63 in benign mixed tumors was significantly higher than in carcinomas in mixed tumors (P < .05). No statistical difference was found for p53 positivity between benign mixed tumors and carcinomas in benign mixed tumors.
There was no correlation between p63, ΔNp63, or p53 expression with clinical characteristics, such as animal age and benign mixed tumor size. In carcinomas in benign mixed tumors, p63 was negatively correlated with tumor size (r = −0.4361; P < .0001) and positively correlated with Δ Np63 expression (r = 0.8323; P < .0001).
Discussion
The myoepithelial cells play an important role in the distinction of tumors in situ from invasive tumors, which show loss of myoepithelial cells and contain discontinuous myoepithelial cell layers. 21, 22 In the present study, myoepithelial cells showed a progressive loss of immunoreactivity for p63 as tumors developed from benign to malignant, in situ and invasive. Similar results were observed in human mammary 1, 32 and salivary glands tumors, 10 where the loss of p63 expression occurred simultaneously with malignancy progression.
A parallel decrease of α-SMA expression was detected in benign as well as malignant tumors, although to a lesser degree than in the cases of p63 and ΔNp63. This may be explained by the fact that p63 is a highly specific and sensitive myoepithelial cell marker in human 1 as well as canine 8, 29 mammary glands. Other myoepithelial markers, such as α-SMA, also react with cytoskeleton proteins from both myoepithelial and basal cells. Therefore, the advantage of using p63 is that it does not cross-react with the stromal myofibroblasts and is expressed in the nuclei of myoepithelial cells. 37
Because p63 encodes multiple products with transactivating, cell death–promoting, or dominant negative activities, 41 we analyzed the expression of ΔNp63 isoforms. Several studies in human complex epithelia have shown that ΔNp63, mainly ΔNp63α, is the predominant isoform of p63 expressed in the proliferative, basal compartment and is necessary for the regenerative proliferation of epithelial progenitor cells. 1, 5, 26, 28 It is consistent with the role in opposing p53-mediated growth arrest and apoptosis. 24, 40
The definitive role of the p63 gene in tumor formation and progression is controversial. Studies using the p63 deficient model verified that mice were not susceptible to chemically induced tumors, suggesting an oncogenic function for p63. 15 This is supported by the fact that ΔNp63α is overexpressed in several human epithelial cancers and has been correlated with the survival and maintenance of the proliferative capacity of cancer cells. 13, 23
In contrast to a role in promoting tumorigenesis, p63 expression is associated with favorable prognosis in other cancers, being the loss of expression associated with progression to more invasive and metastatic tumors. 38 This evidence indicated that p63 can act as a metastasis suppressor.
This study demonstrates that all benign tumors were positive for ΔNp63 at different reactivity degrees. The same was observed in case of malignant tumors, except for one invasive tubular carcinoma that was not immunoreactive for p63 and ΔNp63.
The ΔNp63 and pan-p63 expressions were identical in the vast majority of samples. This indicates that ΔNp63 might be the predominant isoform in mixed tumors. However, both p63 and ΔNp63 in malignant tumors showed loss when compared with benign tumors. The results may seem contradicting, because a loss of ΔNp63 expression would decrease the evolution capacity of the tumors. It is possible that the 4A4 antibody might not have recognized the TAp63 isoforms, because there is evidence that TAp63 isoforms can be transcriptionally active at levels below the detection limit by immunohistochemistry in some tumors. 5, 37
We cannot discard the possibility that TAp63 isoforms may also be decreased in carcinomas in mixed tumors. In this case, the biologic relations between TAp63 and ΔNp63 could be unbalanced in carcinomas in mixed tumors, leading to predominance of the oncogenic role of ΔNp63 isoforms.
It is also possible that the decrease of ΔNp63 in carcinomas in benign mixed tumors might be important for the development of benign mixed tumors to carcinomas by the 2 mechanisms, depending on the development stage. In this case, ΔNp63 acts initially as an oncogene and simultaneously as a tumor suppressor.
The predominance of ΔNp63 may suggest its role in early stages of mixed mammary tumors through maintaining the proliferative potential of the cells, as well as protecting the apoptosis. There is additional evidence in vivo from human tumors that loss of p63 seems to coincide with tumor progression. In addition to the transitional carcinomas of the bladder, human mammary and lung tumors also show decreased p63 expression. 1, 38
A possible mechanism of how the loss of ΔNp63 can lead to tumoral progression was supported by in vitro data, which revealed that loss of ΔNp63 expression in squamous cell lines leads to upregulation of invasion and metastasis-related genes, acquisition of mesenchymal characteristics, and downregulation of epidermal differentiation markers. 2 Furthermore, it was demonstrated that ΔNp63 is essential for normal mammary epithelial cell-matrix adhesion. The downregulation of p63, mainly ΔNp63 in primary mammary epithelial mouse cells, induced cell detachment and apoptosis. 3 The same study verified that several genes involved in cellular adhesion, such as integrins and laminin, displayed reduced expression levels when p63 was downregulated.
In the mammary gland, p63 is expressed in myoepithelial cells, which can act as tumor suppressors and express high levels of cell adhesion proteins. Therefore, it is suggested that downregulation of ΔNp63 may also influence the expression of these proteins and contribute to higher motility. 3 However, further investigation is needed.
In this study, p53 immunoreactivity was verified in both benign mixed tumors (15%) and carcinomas in mixed tumors (10%), and there was no significant difference between the 2 groups. These results differ from data derived from similar immunohistochemical analyses of canine mammary tumors, which showed that the incidence of the p53 positive staining in carcinomas was significantly higher than that in benign tumors. 34 The high proportion of canine benign mammary tumors positive to p53 may be due to the detection of the wild-type p53 protein by the antibody used, which recognized mutant and wild-type p53. 35 However, it may also mean a precocious molecular event in the tumorigenesis of benign mixed tumors. Because canine benign mixed tumors may develop carcinomatous foci resulting in carcinomas, the overexpression of p53 may indicate a higher malignant potential of these tumors. Nevertheless, because few carcinomas arising in mixed tumors showed p53 overexpression, the significance of this observation requires further analysis of the mutation profiles.
Based on evidence that in vitro studies suggest that mutant forms of p53 may interact with p63 and, thus, change its stability, 7, 30 we investigated a possible association between the p63 and p53 expression; no correlation was observed. In human mammary carcinomas, Ribeiro-Silva, Ramalho, Garcia, and Zucoloto also verified that p53 was rarely coexpressed with p63. 33
Although mutations in p53 generally lead to the formation of a defective protein that accumulates in the cellular nucleus and can be detected by immunohistochemistry, 16 the lack of concordance between the immunohistochemical overexpression and mutations was demonstrated. 39 It is possible that the p53 expression alterations observed in our cases do not correlate with p53 gene mutations. The direct molecular analysis of the p53 gene may offer more consistent information about the possible interactions.
The most well-known function of ΔNp63 is the downregulation of the p53 activity inhibiting the apoptosis. ΔNp63 can exert the dominant negative effects either by direct protein-protein interaction or by competition for binding sites at the DNA level. 30, 40 The present results suggest that ΔNp63 promotes an apoptosis inhibition in the tumors studied independently of protein interactions. It is consistent with a recent study that verified that ΔNp63a plays an antiapoptotic role regardless of the p53 status. 17
The presence of some interstitial myoepithelial cells of the mesenchymal component expressing p63 have been previously described 8, 29 and favor the participation of the myoepithelial cells in the histogenesis of the mesenchymal component of mixed tumors. This hypotesis is supported by previous immunohistochemical studies describing that some spindle- and stellate-shaped myoepithelial cells proliferating in the interstitial areas lost the expression of cytokeratins and gained mesenchymal protein expression. 4, 9, 19, 20
In conclusion, the expression of p63 and ΔNp63 is reduced in carcinomas in benign mixed tumors and may be related to the development of these tumors. The higher specificity and sensitivity of p63 to detect myoepithelial cells indicate that p63 may be a useful tool in diagnosing carcinomas in mixed tumors and may help indentify small carcinomatous foci. Despite the already well-established relation between p53 and p63, no there was no evidence of protein correlation in this study, which suggests that other interaction mechanisms may be involved.
Footnotes
Acknowledgements
This research was supported by Coordenação de Apefeiçoamento de Pessoal de Nível Superior (CAPES), Conselho Nacional de Desenvolvimento e Pesquisa (CNPq) and Fundação de Amparo a Pesquisa do Estado de Minas Gerais (FAPEMIG).
