Abstract
Mammary tumors are among the most common neoplastic processes in female dogs. Prostaglandin E2, the catalytic product of Cox-2, may promote tumor development and angiogenesis. It has been investigated in several human cancers and also correlated with the evolution of the disease. However, the clinical implications of tumor pathology require more investigation in veterinary medicine. Angiogenesis is essential for the growth and metastasis of major solid tumors and has been correlated with prognosis in human and canine breast cancer. The aim of this study was to evaluate Cox-2 expression and microvessel density in canine mammary carcinomas and to correlate them with overall survival of the animal. Cox-2 and angiogenesis were assessed by immunohistochemistry in 46 mammary carcinomas (19 ductal and 27 metaplastic) and in healthy mammary glands. To assess tumor angiogenesis, microvessel density (MVD) was determined by CD31 staining. Immunostaining revealed that 46/46 (100%) of the tumors were positive for Cox-2 and CD31, and there was no statistical difference among tumor types. Cox-2 protein expression correlated positively with CD31 staining (r = 0.3742, P = .0104) but did not correlate significantly with tumor type. Longer overall survival was observed in metaplastic carcinomas (P = .028), in tumors with low microvessel density (P = .0002) and with low Cox-2 score (P = .01). Our results demonstrate that increased microvessel density and increased Cox-2 expression were linearly related in the canine mammary tumors studied and were also related to worse prognosis and shorter overall survival. This suggests that Cox-2 inhibitors could be an alternative for the treatment and control of advanced neoplastic mammary disease in female dogs.
Introduction
Mammary tumors are among the most common neoplastic processes in female dogs, and 50% of them are malignant.16, 25 Canine mammary tumors are of interest for both veterinary and human oncologists because they are similar to human breast cancers. During the last 3 or 4 decades, they have been proposed as animal models for human breast cancer research.1, 6, 17
Prognosis is directly related to factors such as tumor size, lymph node involvement, presence of distant metastasis, histologic type, histologic grade, nuclear differentiation level, invasion level, intravascular growth and presence of estrogen receptors. Large tumors (>3.0 cm) with lymph node metastases indicate a worse prognosis; approximately 80% of patients show recurrence 6 months after treatment, leading to short overall and disease-free survival.2, 25
Cox-2 is an inducible enzyme that interferes with tumor development and angiogenesis. During cancer progression, it participates in arachidonic acid metabolism, generating prostaglandins that can mediate various mechanisms, including cellular proliferation, apoptosis, immune system modulation, and angiogenesis.10
In human medicine, Cox-2 expression in colon and breast cancer is increased under preneoplastic conditions and in more aggressive neoplasias.4 Carcinomas with increased Cox-2 expression have been correlated with worse prognosis for women with breast cancer.4 Malignant canine mammary tumors express Cox-2 more strongly than benign tumors, irrespective of histologic type. However, the precise implication of this for animal survival has not yet been studied.5
Cellular conditions such as hypoxia and expression of cytokines (interleukin 6), oncogenes (ras), and vascular endothelial growth factor (VEGF) lead to increased Cox-2 expression. This helps to promote angiogenesis and tumor invasiveness, which is mediated through increased activity of matrix metalloproteinases 2 and 9 (MMP-9, MMP-2).12 Cox-2 has also been related to the inhibition of apoptosis through inhibition of the pro-apoptotic Bax protein and overexpression of the anti-apoptotic bcl-2 protein.3
Angiogenesis is essential for the growth and metastasis of major solid tumors. High levels of angiogenic factors and histologic evidence of increased tumor neovascularization through microvessel density measurement are considered to have important prognostic value in human medicine for various solid tumors.7, 22 Many authors correlate the capillary microvessel index with prognosis in human breast cancer,8, 9, 11, 13 and recent studies have reported similar results in veterinary medicine.18
Other studies have demonstrated that VEGF expression is increased in malignant canine mammary tumors, with increased risk of disease dissemination and tumor mass development, suggesting that microvessel density may be an indicator of malignancy and represent the angiogenic potential of the neoplasm.19, 20
The aim of the present study was to evaluate Cox-2 expression and microvessel density in canine mammary carcinomas and to correlate them with overall survival of the animal.
Materials and Methods
Case selection
Mammary tumor samples from female dogs were obtained after surgical removal in the Veterinary Teaching Hospital of the Federal University of Minas Gerais, Brazil. The samples were fixed in 10% neutral formalin and embedded in paraffin. Sections of 4 μm were cut from 1 representative block for each case, collected on glass slides, and stained with HE.
Histologic diagnosis was conducted on HE-stained sections according to the World Health Organization's diagnostic criteria for dog mammary gland tumors.15 All tumors were revised by 2 pathologists using the same guidelines to ensure consistency of diagnosis. We used human diagnostic criteria and human classification in order to compare lesions in the 2 species.6
After revision by 2 pathologists, 46 malignant tumor samples (27 carcinomas in mixed tumors, 11 tubular carcinomas, and 8 solid carcinomas) were selected for immunohistochemical and survival analysis. The tubular and solid carcinomas were combined to form the ductal carcinoma group, and the carcinomas in mixed tumors formed the metaplastic group.
Immunohistochemical analysis
Sections of 4 μm were cut from one representative block for each case and collected on gelatin-coated slides. The slides were deparaffinized and rehydrated in an alcohol series. Endogenous peroxidase was blocked by immersion in 3% hydrogen peroxide.
Deparaffinized tissue sections were subjected to heat-induced antigen retrieval (water bath at 98°C) with antigen retrieval solution (DAKO, SA, Denmark, pH 6.0). Next, the slides were incubated at 4°C for 16 hours with a primary rabbit monoclonal anti-human Cox-2 antibody (SP21, 1 : 20, Lab Vision, Freemont, CA) and an anti-human Cd31 mouse monoclonal (JC70A, 1 : 50, DAKO),14 followed by the EnVision polymer HRP and Envision+ (DAKO) for 1 hour at 37°C. The sections were stained with the chromogen 3,3–diaminobenzidine tetrahydrochloride (DAB Substrate System, Lab Vision), incubated for 10 minutes and counterstained with Mayer's hematoxylin.
Sections from a human colon carcinoma known to express Cox-2 were used as positive controls for Cox-2, and adjacent normal mammary tissues were used as internal negative controls. The antibody had previously been tested on normal canine kidney to demonstrate its specificity for canine tissues. For CD31, sections from canine mammary carcinomas known to express CD31 were used as positive controls. Histologically recognizable blood vessels within tissue sections served as internal controls for CD31 immunostaining. Negative controls were obtained by substituting normal serum for the primary antibody.
Cox-2 expression analysis in tumoral cells
Positivity for Cox-2 was indicated by cytoplasmic staining. The number of Cox-2 positive cells was evaluated semiquantitatively, with the distribution score defined by the estimated percentage of positive cells in 5 fields at 400× magnification: 0 = absence, 1 = fewer than 10% stained cells, 2 = between 10% and 30%, 3 = between 31% and 60%, 4 = more than 61% stained cells. For staining intensity, values from 0 to 3 were attributed: 0 = absence (−), 1 = weak staining (+), 2 = moderate staining (++), and 3 = strong staining (+++). Distribution score and intensity were multiplied to obtain a total score, which ranged from 0 to 12.5, 12
Analysis of microvessel density
CD31 positivity was indicated by membrane staining (Fig. 1). To analyze microvessel density (immunohistochemistry with CD31), structures with or without lumens that were positive for CD31 were considered as individual vessels. Areas of fibrosis, necrosis and inflammation, and vessels with muscular walls were not counted. Microvessels were counted in the 3 most vascularized areas, known as hot spots, in 200× magnification fields (corresponding to approximately 0.76 mm2), from which the median was obtained.24
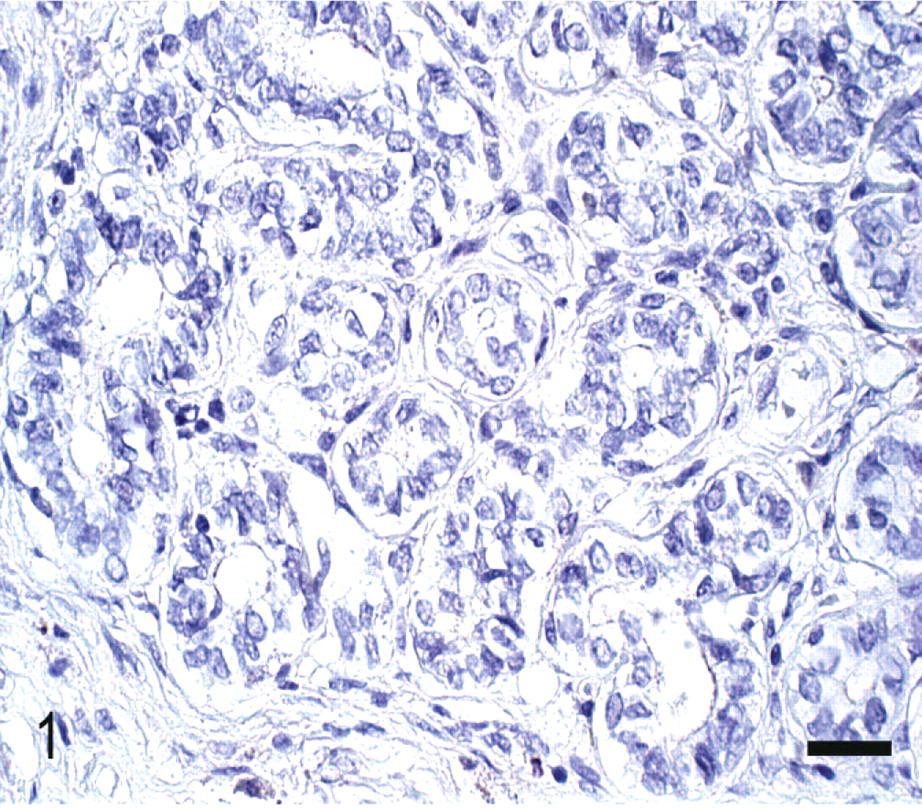
Normal mammary gland; Canis familiaris. Epithelial cells from normal acini showing absence of Cox-2 staining. Streptavidin–biotin complex method. Mayer's hematoxylin counterstain. Bar = 30 μm.
Survival analysis
Follow-up was from 2003 to 2007 through telephone communication and periodic returns to the Veterinary Teaching Hospital of the Federal University of Minas Gerais. The owner of the canine patient reported the cause of death (evolution of the neoplastic process or other). Overall survival was defined as the period (days) between surgical removal and death caused by the neoplastic process. The animals whose deaths occurred from any other cause were not taken into account.
The animals were divided in groups for correlating survival with microvessel density and Cox-2 expression. For microvessel density, the animals were classified according to the median values. Four groups were obtained: 1 = median 30 or below, 2 = median from 31 to 60, 3 = median from 61 to 90, and 4 = median from 91 to 120. For Cox-2 expression, there was a range from 0 to 12 as described above. The animals were classified into 2 groups: 1 = score from 0 to 5 and 2 = score from 6 to 12.
Statistical analysis
To determine whether Cox-2 staining intensity, distribution score, total score, staining indices, and microvessel density differed by histologic subtype of tumor (metaplastic carcinoma vs. ductal carcinoma), a Mann-Whitney test was performed. An unpaired Student's t-test was performed to detect statistically significant differences in microvessel density between metaplastic and ductal carcinomas. The tests were 2-sided, and a P value of less than .05 was considered statistically significant.21 The correlation between Cox-2 staining and microvessel density or tumor type was assessed using a Spearman's rank correlation coefficient test.21 The analyses were performed using Graph Pad Prism V.3 software (San Diego, CA). Survival curves were generated using the Kaplan-Meier method, and differences in survival times were compared using log-rank analysis. Values were considered statistically significant when P < .05.
Results
Cox-2 and CD31 immunohistochemistry
Of the 46 cases analyzed, 27 were metaplastic carcinomas and 19 ductal carcinomas. The results showed that Cox-2 was not expressed in normal canine mammary gland (Fig. 1). Immunohistochemical analysis revealed some degree of positivity in 100% (46/46) of the tumors examined. In both metaplastic carcinomas and ductal carcinomas, Cox-2 expression was located in the tumor cell cytoplasm.
Immunohistochemistry for Cox-2 revealed that 74% (20/27) of the metaplastic carcinomas had a final score of 0–5 and 26% (7/27) of 6–12. Of the 19 ductal carcinomas, 53% (10/19) showed a Cox-2 final score of 0–5 and 47% (9/19) of 6–12. Ductal carcinomas stained more intensely for Cox-2 than metaplastic carcinoma (P = .0221) (Figs. 2, 3). However, analysis of the distributions and total scores showed no statistical significance among tumor types (P = .0914).
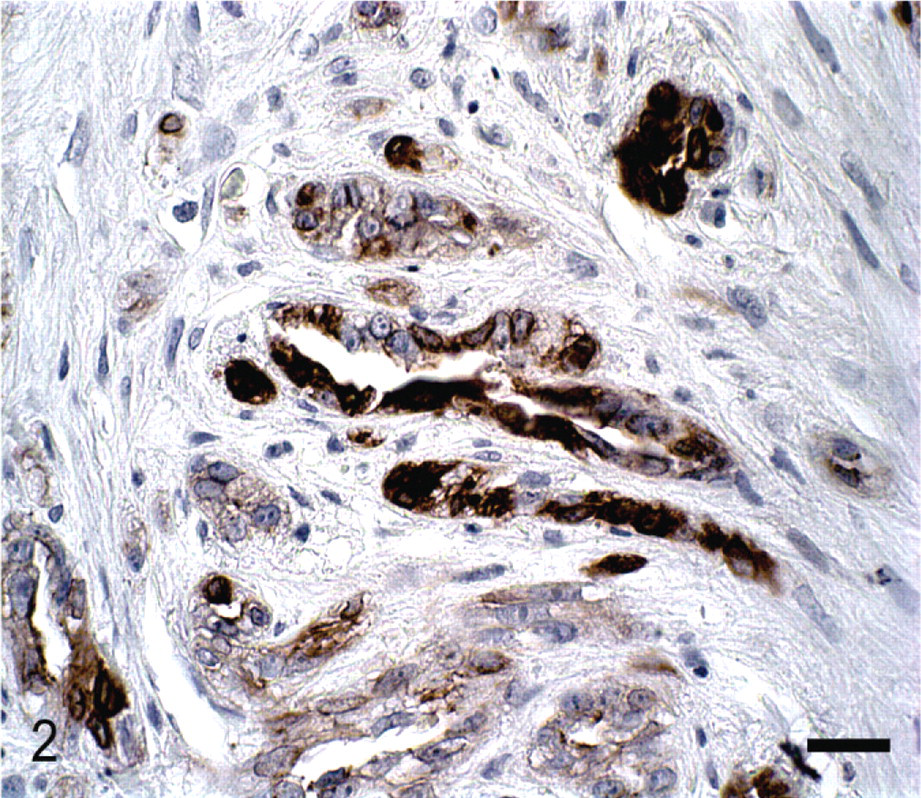
Mammary gland; Canis familiaris. Ductal carcinoma neoplastic epithelial cells showing immunoreactivity for Cox-2 (scoring 6). Streptavidin–biotin complex method. Mayer's hematoxylin counterstain. Bar = 30 μm.
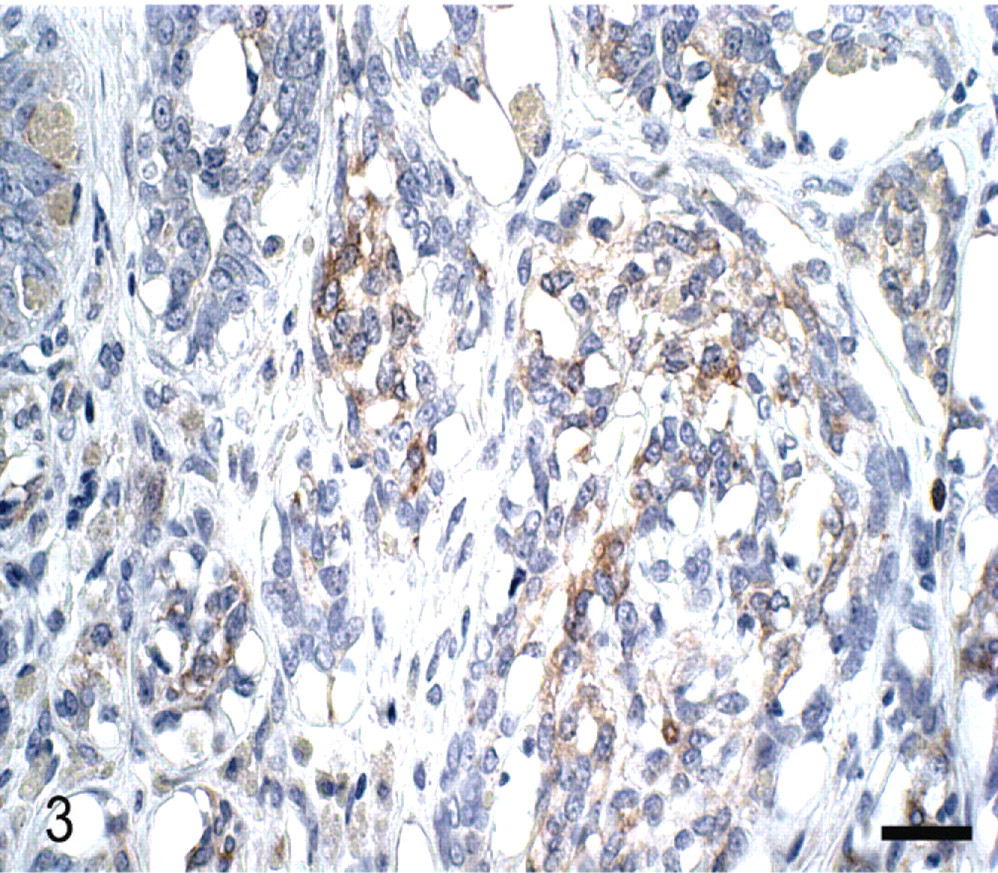
Mammary gland; Canis familiaris. Metaplastic carcinoma. Neoplastic epithelial cells showing weak Cox-2 staining (scoring 4). Streptavidin–biotin complex method. Mayer's hematoxylin counterstain. Bar = 30 μm.
CD31 immunohistochemistry revealed positive staining in 46/46 (100%) of the tumors (Fig. 4). CD31 expression did not differ significantly among tumor types (P = .747). Cox-2 protein expression correlated positively with CD31 staining (r = 0.3742, P = .0104) but did not correlate significantly with tumor type.
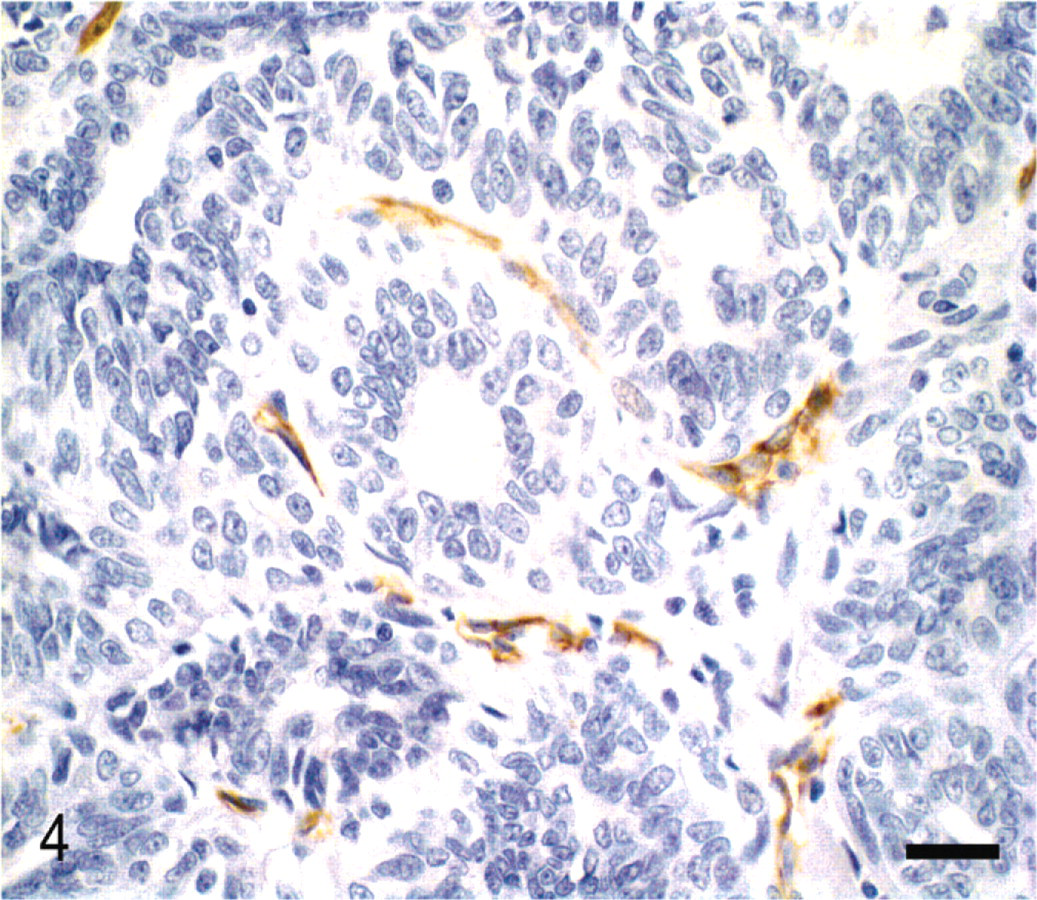
Mammary gland; Canis familiaris. Ductal carcinoma. Immunostaining for CD31 shows small vessels without lumen and some isolated positive endothelial cells. Streptavidin–biotin complex method. Mayer's hematoxylin counterstain. Bar = 30 μm.
Survival analysis
Considering only the histologic subtype, survival was found to be longer in female dogs with metaplastic carcinomas than in those with ductal carcinoma (P = .028) (Fig. 5). When overall survival and microvessel density were analyzed, animals with increased vascularization presented shorter overall survival (P = .0002) (Fig. 6). Patients with Cox-2 scores ranging from 0 to 5 showed longer overall survival than those with Cox-2 scores ranging from 6 to 12 (P = .01) (Fig. 7).
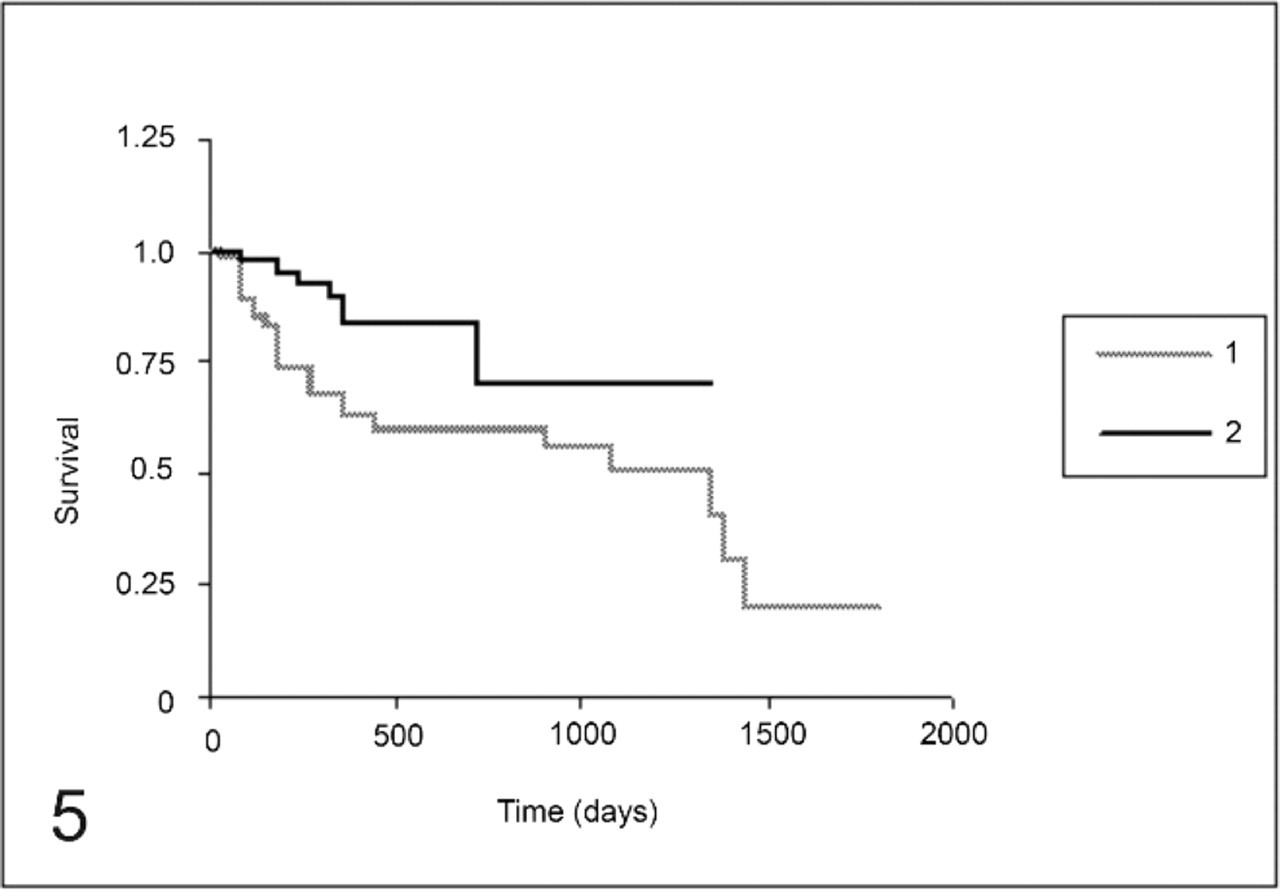
Overall survival curves for the group of 46 female dogs. Metaplastic carcinoma, 27 cases (curve 1) and ductal carcinoma, 19 cases (curve 2) groups (P = .028).
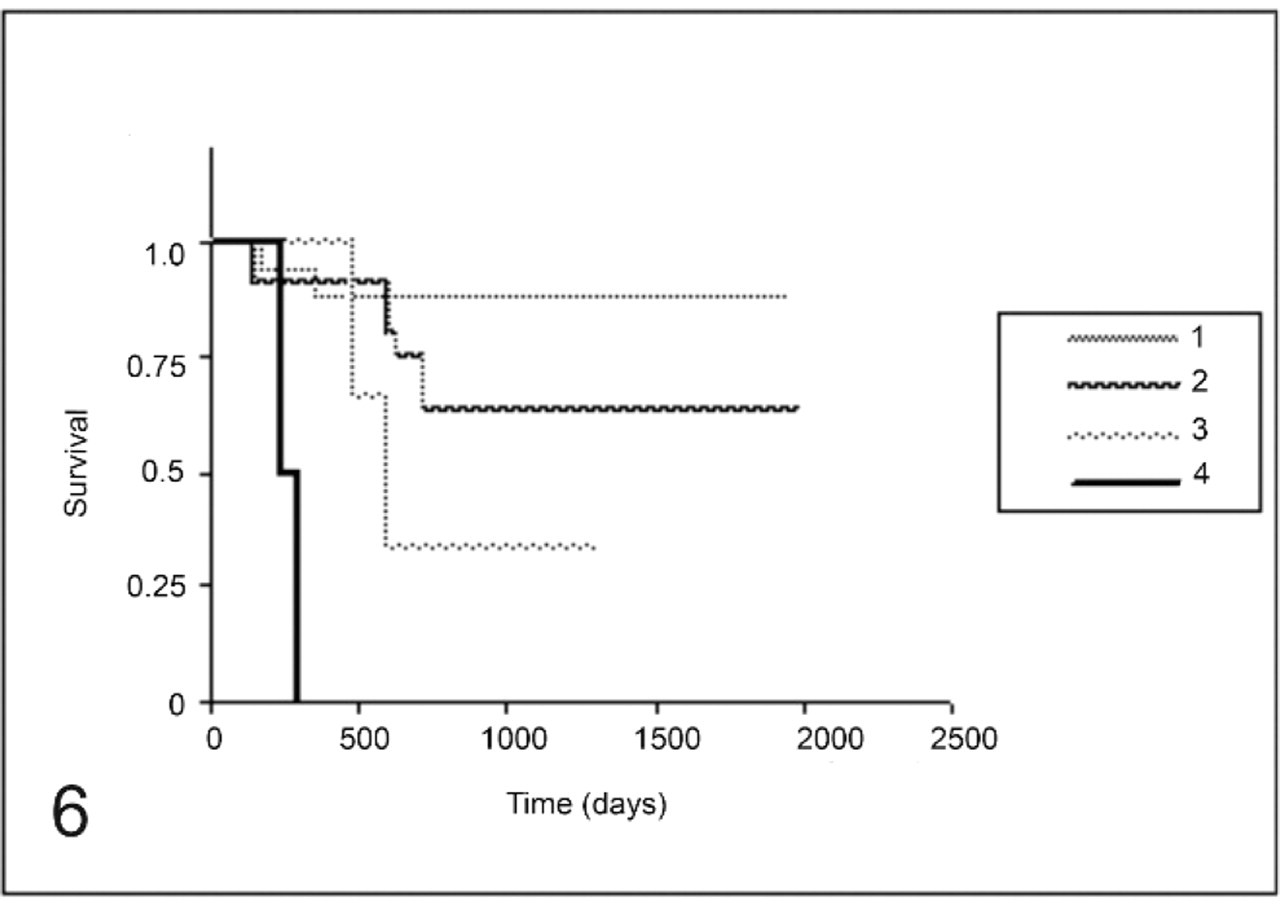
Overall survival curves for the group of 46 female dogs with mammary carcinomas. MVD until 30 (curve 1), MVD from 31 to 60 (curve 2), MVD from 61 to 90 (curve 3), and MVD from 91 to 120 (curve 4) (P = .0002).
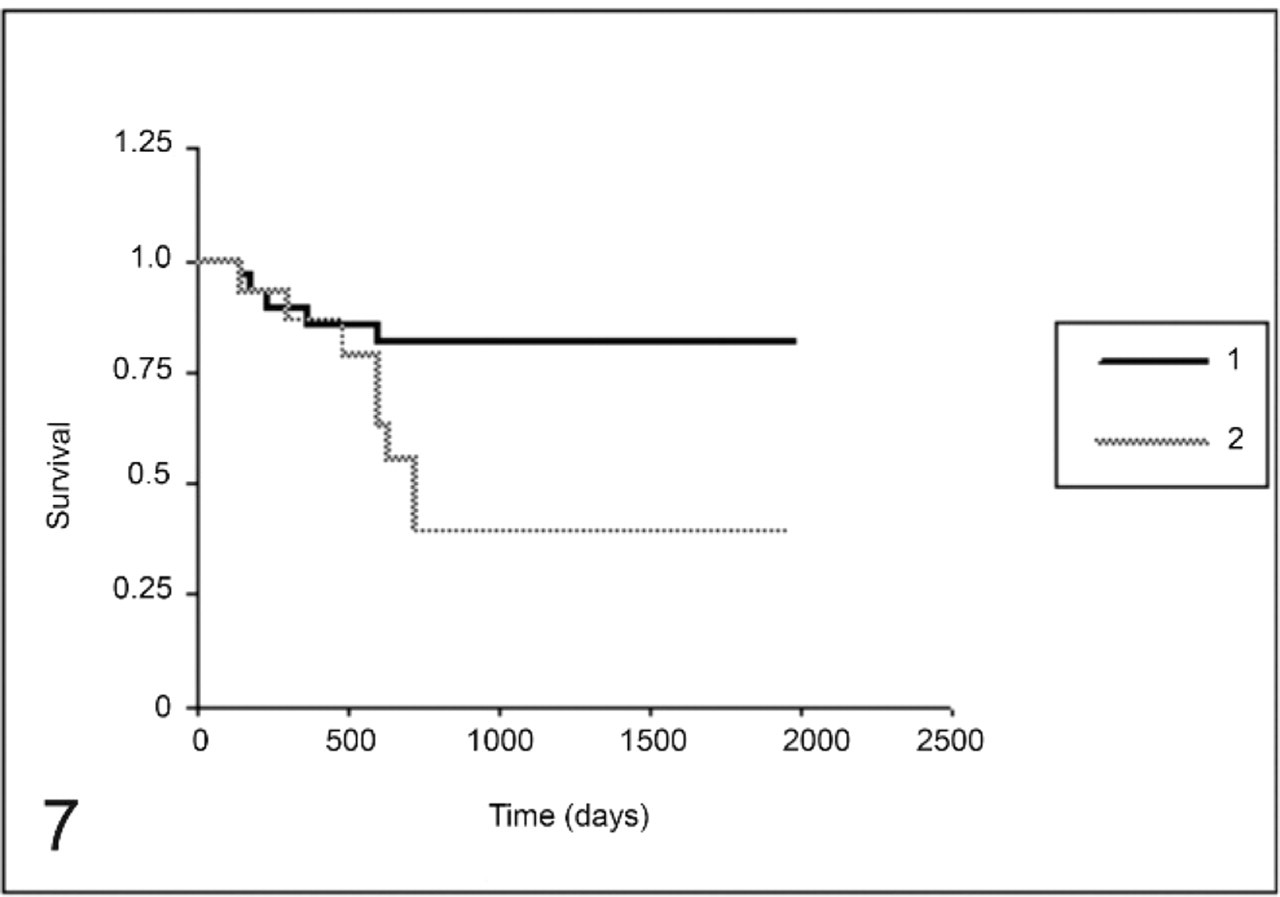
Overall survival curves for the group of 46 female dogs with mammary carcinomas. Low Cox-2 score 0–5 (curve 1) and high Cox-2 score 6–12 (curve 2) (P = .01).
There were 10 censored animals that died for reasons unrelated to the tumor. The most common cause was visceral (Calazar) leishmaniasis (L. chagasi), an endemic disease where the research was conducted. The overall median survival time of the entire population was 907 days. Follow-up of the studied population lasted at least 2 years, equivalent to 730 days, except for the ones that died of the tumor before this deadline.
Discussion
The results of this study demonstrate that patients with increased Cox-2 expression have shorter survival times. Millanta et al.14 reported a strong correlation between Cox-2 expression and prognostic factors such as histologic grade and HER-2 expression in canine mammary tumors, also suggesting that increased Cox-2 expression was related to worse prognosis, as observed in the survival analysis in our study.
When we assessed overall survival and angiogenesis, our results corroborated the literature,8, 9, 11, 13, 18, 19 suggesting that angiogenesis is essential for the development and evolution of neoplastic disease, resulting in shorter overall survival and worse prognosis.
We suggest that microvessel density and angiogenesis could be a sensitive indicator of malignancy in veterinary medicine. The increased expression of VEGF in canine mammary tumors indicates a higher risk of development and dissemination of the disease. Those tumors could be used as therapeutic models of the inhibition of angiogenesis,19, 20 as confirmed in our study by the evaluation of survival and angiogenesis.
Our results showed no association between microvessel density and Cox-2 expression, as suggested for women by Costa et al.4 It is possible that increased Cox-2 expression could be related to other factors besides neovascularization, resulting in the progression of neoplastic disease. Such factors could include the inhibition of apoptosis, induction of cellular proliferation and destabilization of the immune system.3, 12
In this study, we chose to compare the 3 histologic types of canine mammary tumors. Carcinomas in mixed tumors are characterized by carcinomatous transformation of epithelial components in benign mixed tumors, corresponding morphologically to the matrix-producing human female breast carcinoma. Tubular and solid carcinomas, simple or complex, correspond morphologically to ductal carcinomas in women.2, 15, 23
Cavalcanti and Cassali2 verified that female dogs with metaplastic carcinomas have a longer overall survival than those with ductal carcinomas. Wargotz and Norris23 showed that women with metaplastic carcinoma had better prognoses than those with ductal carcinoma. This study confirms the results obtained by Cavalcanti and Cassali,2 demonstrating a longer overall survival for metaplastic carcinoma bearers.
We suggest that the small difference between Cox-2 scores in the ductal carcinoma group may be related to the differentiated behavior of these tumors, confirming the association between the Cox-2 expression and disease aggressiveness. Besides showing shorter overall survival, it was verified that Cox-2 expression correlated positively with ductal carcinoma, suggesting a worse prognosis as described for women by Costa et al.4 In female dogs, Doré et al.5 observed higher Cox-2 expression in malignant than in benign tumors, but they did not evaluate patient survival. In our study, we observed shorter survival in patients whose tumors expressed more Cox-2. Therefore, we emphasize the possibility of using Cox-2 inhibitor anti-inflammatory drugs to treat canine mammary tumors in accordance with Costa et al.4 and Doré et al.5
Our results demonstrate that increased microvessel density and increased Cox-2 expression were correlated in the canine mammary tumors studied and were also related to worse prognosis and shorter overall survival, suggesting that Cox-2 inhibitors could be an alternative for treating and controlling advanced mammary neoplastic disease in female dogs.
Footnotes
Acknowledgements
This research was supported by CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior), CNPq (Conselho Nacional de Desenvolvimento e Pesquisa), and FAPEMIG (Fundação de amparo à pesquisa do Estado de Minas Gerais).
