Abstract
This study describes and evaluates the morphological and molecular relationship between canine mammary ductal hyperplasias with atypia and canine mammary neoplasias. Ductal hyperplasia was identified in association with malignant neoplasia in 56 of the 115 cases (48,8%), and although ductal hyperplasia without atypia was the type most frequently noted in the cases, most examples of hyperplasia with atypia were associated with mammary tumors. Estrogen receptor, E-cadherin, and cytokeratins 1, 5, 10 and 14 (CK34bE12) expression was quite lower than in normal mammary tissue, and HER2 overexpression was absent in all proliferative cells of ductal hyperplasia. The Ki-67 expression, epidermal growth factor receptor and progesterone receptor expression appeared higher in those hyperplastic lesions analyzed than in normal mammary glands. These findings suggest that canine mammary atypical hyperplasia may play an important role in the process of malignant neoplastic transformation, with molecular alterations that are similar to precursor lesions reported in humans.
The canine mammary gland is prone to a wide range of proliferative epithelial pathologies that are morphologically similar to those reported in the human breast. Epithelial lesions can be divided into 2 major groups: nonneoplastic and neoplastic.32,34,35 Among canine mammary neoplasms benign mixed carcinoma in benign mixed tumors and adenocarcinomas are considered to be the most prevalent and are noted for their negative prognosis.11,20
Epithelial nonneoplastic lesions, such as ductal hyperplasia (DH) and lobular hyperplasia (LH), are frequently diagnosed in canine mammary gland. 26 These lesions have been shown to be influenced by sex hormones as well as prolonged exposure to exogenous progestins administered for estrus prevention.8,19,25,45 There is no clinical evidence that canine mammary hyperplastic lesions are precancerous. However, in humans, mammary hyperplasia is recognized as a risk factor for invasive cancer.12,15,31
Several biological markers are expressed in different phases of mammary carcinogenesis; however, few attempts have been made to correlate biological markers in canine preneoplastic lesions with morphological criteria and associated malignant lesions.10,13,43
Although epidemiological studies in dogs are limited in the demonstration of premalignant potential of mammary hyperplasia, recent studies suggest that some hyperplastic lesions may have molecular features indicative of neoplastic transformation. Cell cycle–related markers (HER2, Ciclin, Ki67) are clearly increased with progression of carcinogenesis and are found in estrogen receptor (ER)–positive more often than in ER-negative specimens of ductal hyperplasia with atypia.14,22,37,2 Despite these reports, the precise anatomic definition of these lesions has only rarely been proposed, and their biological or prognostic significance has never been rigorously studied.
The present work aimed to stratify the pathologic spectrum of canine mammary hyperplasia, as has been done with the classification system used in human pathology. We evaluated molecular markers (ER, PR, p53, Ki-67, epidermal growth factor receptor (EGFR), HER-2, high-molecular-weight cytokeratins [34βE12], and E-cadherin) in epithelial hyperplasia of mammary specimens with and without concurrent neoplasia.
Methods
We evaluated histologic sections from 115 cases diagnosed as epithelial hyperplasia from canine mammary lesions selected from the archives of the Laboratory of Comparative Pathology of the Biological Science Institute of Federal University of Minas Gerais.
Hematoxylin and eosin (HE)–stained sections from a total of 115 biopsy specimens were examined and classified by 3 pathologists. These included a medical pathologist (HG) and 2 veterinary pathologists (EF and GDC), who individually reviewed and categorized the hyperplastic lesions using the medical 41 and veterinary classification systems. 26 Pathologists assessed the histopathology for epithelial hyperplasia and any associated epithelial tumors. The mammary neoplastic lesions were classified according to veterinary nomenclature. 26
According to the World Health Organizaton (WHO) veterinary classification, epithelial hyperplastic changes are divided into 2 categories, LH and DH, including subclassification based on the presence of cytological atypia (LHA and DHA). 26 When both LH and DH lesions were found in the same specimen, we grouped these lesions under the category of “mixed” hyperplasia without atypia, or mixed hyperplasia of usual type (MHU) and mixed hyperplasia with atypia (MHA) for purposes of analysis.
The criteria used to classify ductal hyperplasia in humans are those derived from the medical WHO classification. 41 The lesions were classified as ductal hyperplasia without atypia (hyperplasia usual type [HUT]) or atypical ductal hyperplasia (ADH).
Considering the small size of the certain lesions, immunohistochemical analysis was only performed on cases with enough material. Paraffin blocks were selected from 20 cases containing hyperplastic lesions classified by WHO human criteria (10 ADH and 10 HUT). ADHs were associated with 3 carcinomas, 4 ductal carcinomas in situ (DCIS), and 3 benign mixed tumors. HUTs were associated with 1 carcinoma, 3 DCIS, 4 benign mixed tumors, and 2 duct papillomas.
Consecutive 5-μm-thick sections were obtained and mounted on silanated slides for the immunohistochemical study. Sections were stained for rabbit polyclonal antibodies: HER2 (c-erbB-2; Dako; dilution: 1:40), EGFR (clone 31G7, Zymed, dilution: 1:100), P53 (clone CM1; Covance; dilution: 1:80); and mouse monoclonal antibodies: E-cadherin (clone 4A2C7; Zymed; dilution: 1:100), ER-LH2 (clone CC4-5; Novocastra; dilution: 1:25), PR (clone hPRa2; Neomarkers; dilution: 1:20), Ki-67 (clone Mib-1; Dako; dilution: 1:25), cytokeratins 1, 5, 10, and 14 (clone 34βE-12; Dako; dilution: 1:40). Heat-induced epitope retrieval (20 minutes) using Dako antigen retrieval solution (pH 6.0) was previously performed in a water bath. The slides were then cooled to room temperature for 20 minutes in the antigen retrieval buffer. The sections were incubated for 15 minutes at room temperature in 3% (vol/vol) H2O2, in primary antibodies for 16 hours, in reagent that contained anti-mouse and anti-rabbit secondary antibodies (Biotinylated Goat Anti-polyvalent, Laboratory Vision) for 15 minutes and streptavidin peroxidase (UltraVision Large Volume Detection System, HRP, Laboratory Vision) for 15 minutes. Between incubations, the slides were washed for 2 × 5 minutes in phosphate-buffered saline containing 1% (vol/vol) Tween 20. Immunoreactivity was visualized by incubating the slides for 10 minutes with diaminobenzidine (DAB Substrate System; Laboratory Vision). The slides were then counterstained with Harris hematoxylin. Positive and negative control slides were included in each batch. As a positive control we used human mammary neoplastic tissue known to express the antibodies. Negative controls were assessed using normal serum (Ultra V Block, Laboratory Vision) as the primary antibody.
Staining for cytokeratin 34βE12 and E-cadherin was evaluated semiquantitatively and scored according to 5 categories: negative (–), <5% of cells stained; positive (+), 5% to 25% of cells stained; positive (++), 25% to 50% of cells stained; positive (+++), 50% to 75% of cells stained; and diffusely positive (++++), >75% of epithelial cells stained. For both ER and PR, first a proportion score was assigned, representing the estimated proportion of positive-staining tumor cells (0, none; 1, <1/100; 2, 1/100–1/10; 3, 1/10–1/3; 4, 1/3–2/3; and 5, >2/3). Subsequently, an intensity score was assigned, representing the average intensity of positive tumor cells (0, none; 1, weak, 2, intermediate; and 3, strong). The proportion and intensity scores were then added to obtain a total score, which ranged from 0 to 8. 17 Cases were classified as ER and PR positive when the total score was greater than or equal to 3. The p53 staining was considered positive when more than 10% of cells exhibited positive nuclear staining, independent of staining intensity. The proliferative index was calculated by counting the nuclei positive for Ki-67 staining in a total of 500 epithelial cells from each lesion. HER2 and EGFR expressions were defined as epithelial cell membrane staining and scored according to the American Society of Clinical Oncology and College of American Pathologists. 46
Immunohistochemical data were analyzed with nonparametric methods on semiquantitative IHC scores using GraphPad InStat. The Kruskal-Wallis test was used to compare groups (HUT, ADH, mammary tumors, and normal mammary glands) for each antibody. Associations between categorical variables (ER, EGFR) in HUT and ADH were assessed by Fisher’s exact test. Statistical results were considered significant when P < .05. Morphologic lesions were expressed with descriptive statistics.
All procedures were performed under the guidelines and with the approval of the Ethics Committee for Animal Experimentation (CETEA/UFMG), protocol 192/2006.
Results
Histopathological analysis of the selected samples showed that ductal hyperplasia was associated with mammary tumors in the majority of the cases (98/115; 85.21%). It was possible to determine the age of animals and the affected mammary gland in 68 and 57 cases, respectively. The age of the animals at the time of surgery ranged from 3 to 16 years (mean, 9.5 years ± 2.5). In the 57 cases in which the affected mammary gland could be determined, the lesions were most commonly located in the abdominal mammary glands (21 of the 57 cases [36.8%] in caudal abdominal and 12 of the 57 cases [21%] in cranial abdominal) as opposed to the inguinal gland (20 of the 57 cases; 35%) or the thoracic glands (9 of the 57 cases [15.8%] in caudal thoracic and 5 of the 57 cases [8.7%] in cranial thoracic).
Ductal hyperplasia was diagnosed when mammary ducts showed increased cellularity and appeared to be composed predominantly of ductal epithelium and frequently of myoepithelium, both of which closely resembled normal structures within the adjacent unaffected mammary tissue in accordance with both the medical and veterinary WHO classification systems.26,41 In our study, hyperplastic changes were more commonly seen within small intralobular ductules and interlobular ducts, and occasionally hyperplasia was also noted in larger ducts.
Usual ductal hyperplasia was diagnosed in cases of increased ductal epithelial cellularity but without cellular atypia. Affected ducts were partially filled by irregularly shaped fenestrations lined by epithelial. Epithelial cells were arranged in cords that occasionally bridged the ductal lumina, and the hyperplastic epithelial cells often seemed to overlap because cytoplasmic boundaries were indistinct (Figure 1).26,41
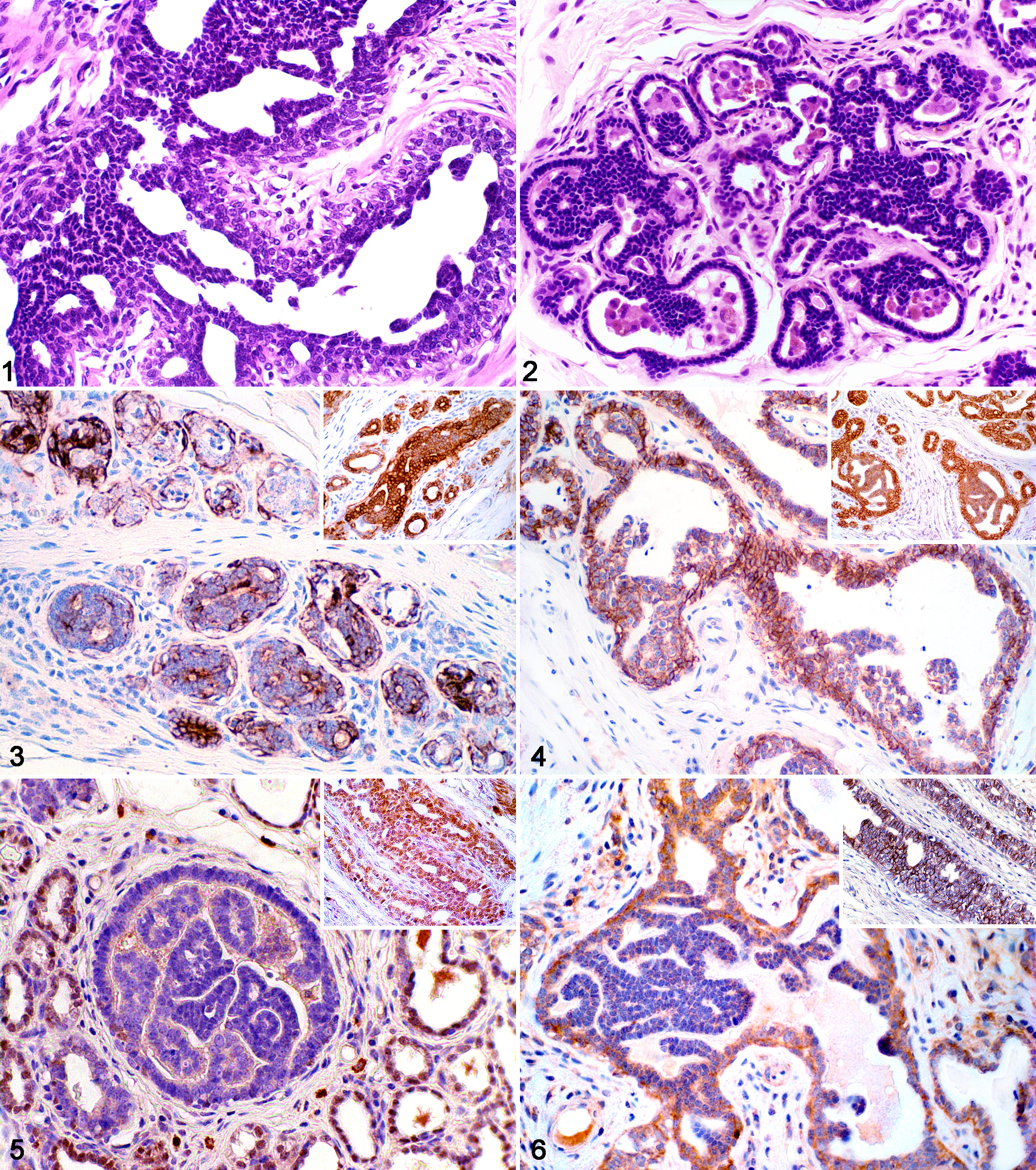
Mammary gland; Canis familiaris. Case 18. Ductal hyperplasia without atypia showing pleomorphic cells with micropapillary projections and irregular intraductal bridge formations (arrow). 40×. Hematoxylin and eosin (HE).
Atypical ductal hyperplasia was characterized by solid or papillary proliferations of small, dark, cuboidal to low columnar cells, often having a basaloid appearance and frequently palisading or forming parallel rows (Figure 2).26,41 Using the criteria of Schnitt et al, 36 ADH was distinguished from low- or intermediate-grade DCIS by the rudimentary and irregular shape of fenestrations and cytologic grade of atypia. In lesions with low-grade cellular atypia, the overall size of the lesion was also considered. Whereas high-grade cellular atypia always warranted the classification of high-grade DCIS, if features of low- or intermediate-grade DCIS were observed but did not involve more than 1 duct or space, the intraepithelial lesion was classified as ADH. 36
A total of 97 mammary epithelial neoplasms were diagnosed in female dogs, of which 41 were benign and 56 were malignant. The most prevalent neoplasms were DCIS (30 cases), carcinomas (26 cases), benign mixed tumors (19 cases), and other benign tumors (22 cases). Independent of the use of medical or veterinary classification, the mammary carcinomas were more often observed with the atypical hyperplasia. However, when the WHO veterinary classification was used, the prevalence of benign versus malignant neoplasia was similar in correlation with intralobular ductal hyperplasia, interlobular ductal hyperplasia, or mixed hyperplasia. Table 1 summarizes the occurrence of specific cases of ductal hyperplasia and epithelial mammary neoplasms included in this study, according to the criteria of the veterinary WHO classification system.
Distribution of Types of Ductal Mammary Hyperplasia and Associated Tumors According to Veterinary Pathology Classification
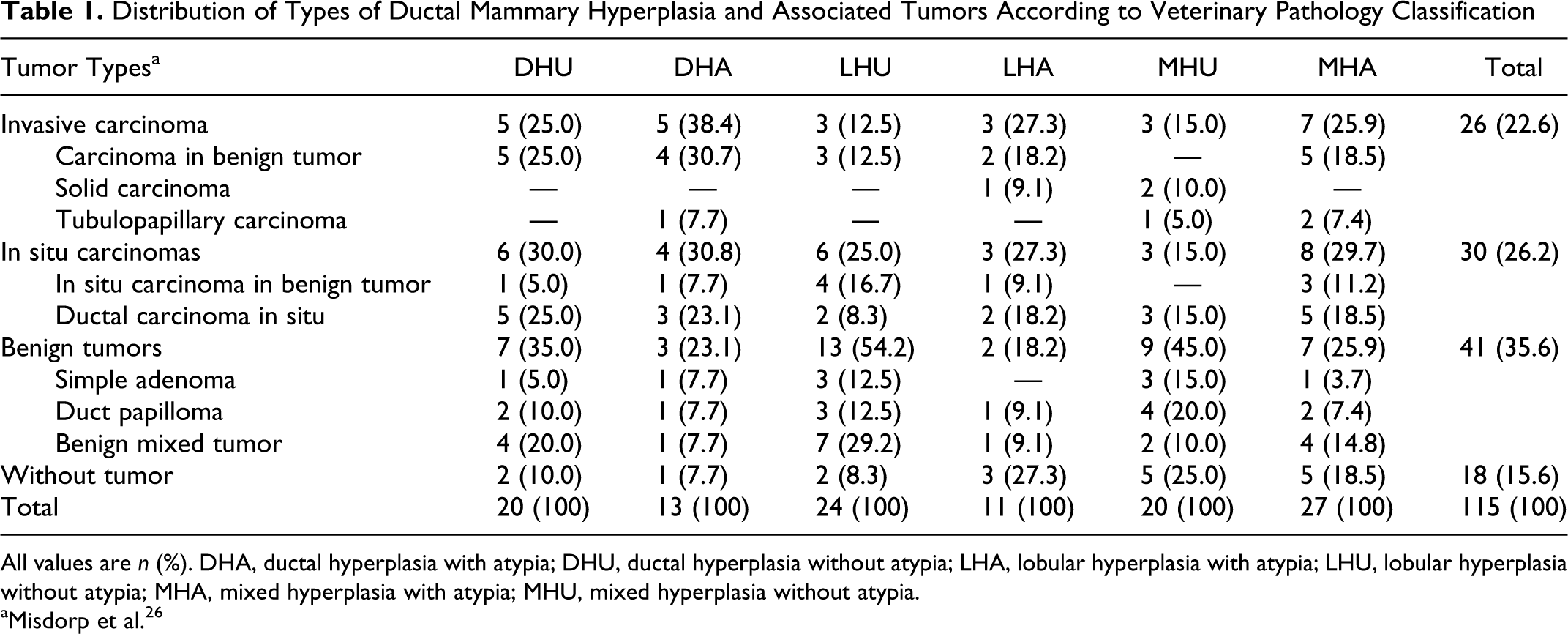
All values are n (%). DHA, ductal hyperplasia with atypia; DHU, ductal hyperplasia without atypia; LHA, lobular hyperplasia with atypia; LHU, lobular hyperplasia without atypia; MHA, mixed hyperplasia with atypia; MHU, mixed hyperplasia without atypia.
aMisdorp et al. 26
According to the medical WHO classification, of the 51 dogs with ADH, 30 (58.8%) had malignant mammary tumors and only 12 (23.5%) had benign neoplasms. Table 2 summarizes the occurrence of specific cases of ductal hyperplasia, which identified that mammary carcinomas are more often associated with ADH (P < .05).
Distribution of Types of Ductal Mammary Hyperplasia and Associated Tumors According to Human Classification
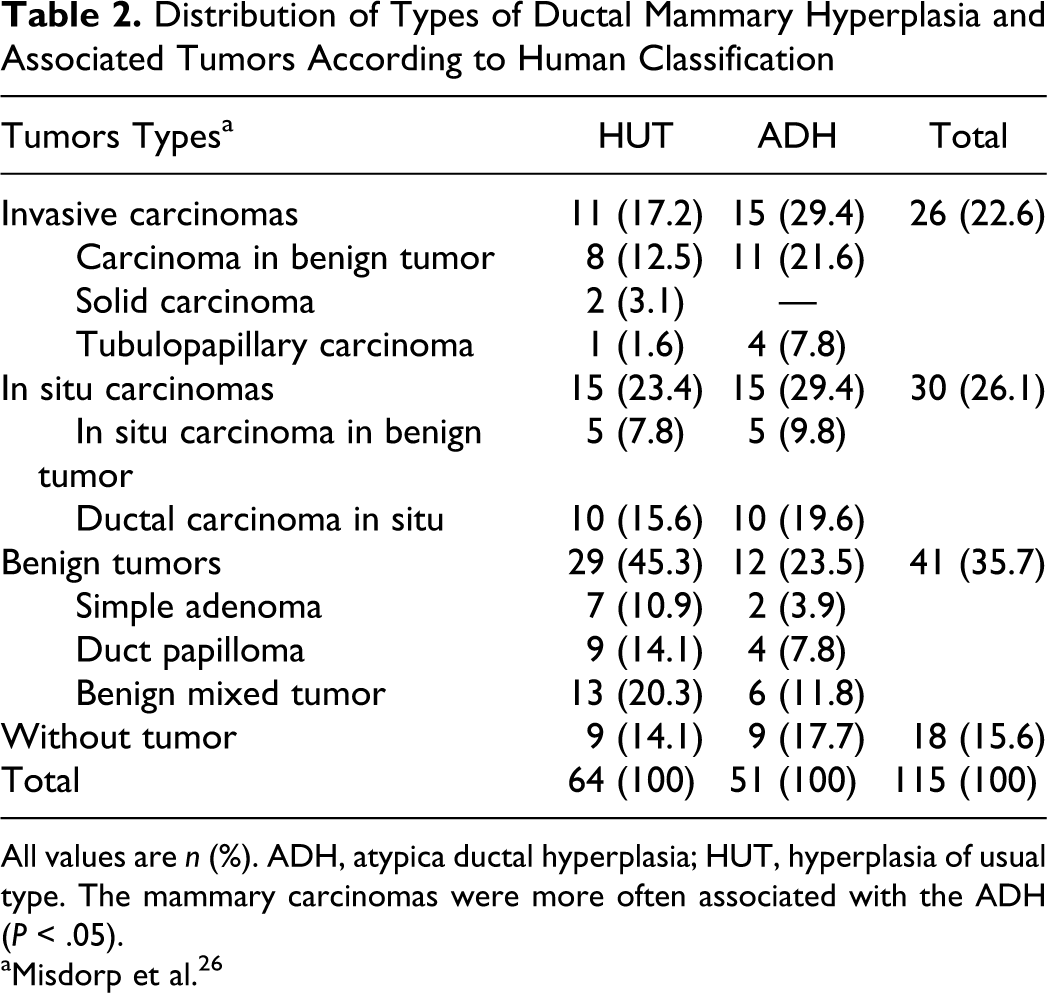
All values are n (%). ADH, atypica ductal hyperplasia; HUT, hyperplasia of usual type. The mammary carcinomas were more often associated with the ADH (P < .05).
aMisdorp et al. 26
Immunohistochemical staining showed positive cytoplasmic localization of cytokeratin 34βE12 in all hyperplastic cells adjacent to or distant from the mammary tumors, but with less intense staining than that noted in normal mammary epithelium (P < .05) (Figure 3). E-cadherin expression showed the same pattern of immunostaining in both mammary hyperplastic lesions and normal ducts. In ductal hyperplasia and various mammary tumors, E-cadherin staining was observed in both benign and malignant epithelial cells. However, luminal epithelial cells usually showed a reduced level of expression, with 4 cases being negative (2 HUT and 2 ADH) (P < .01) (Figure 4). No significant difference was observed between E-cadherin or cytokeratin 34βE-12 expression in cells of HUT, ADH, and DCIS.
The expression pattern of cell cycle–related proteins (ER, PR, p53, EGFR, HER2, and Ki-67) was variable in ductal hyperplasia and in associated tumors. The ER immunostaining was localized in the nuclei and showed some variability in intensity even in geographically distant individual lesions in the same case, with decreased expression in HUT and ADH in comparison with normal mammary gland (P < .01) (Figure 5). Only 3 cases of the HUT and 1 of ADH were positive for ER. The majority of the epithelial cells of HUT and ADH in all specimens showed positive staining for PR, and there was consistently high expression of Ki-67 and EGFR. Statistical analysis of the scoring of EGFR staining intensity revealed significant increased expression among HUT, ADH, benign tumors, and DCIS (P = .01). However, EGFR (Figure 6) overexpression was detected in only 2 cases of the HUT. The immunohistochemical results are summarized in Tables 3 and 4 .
Average Expression of KI-67, EGFR, ER, PR, E-Cadherin, and 34βE12 in Normal Mammary Epithelium, HUT, and Tumors From the Same Mammary Specimens
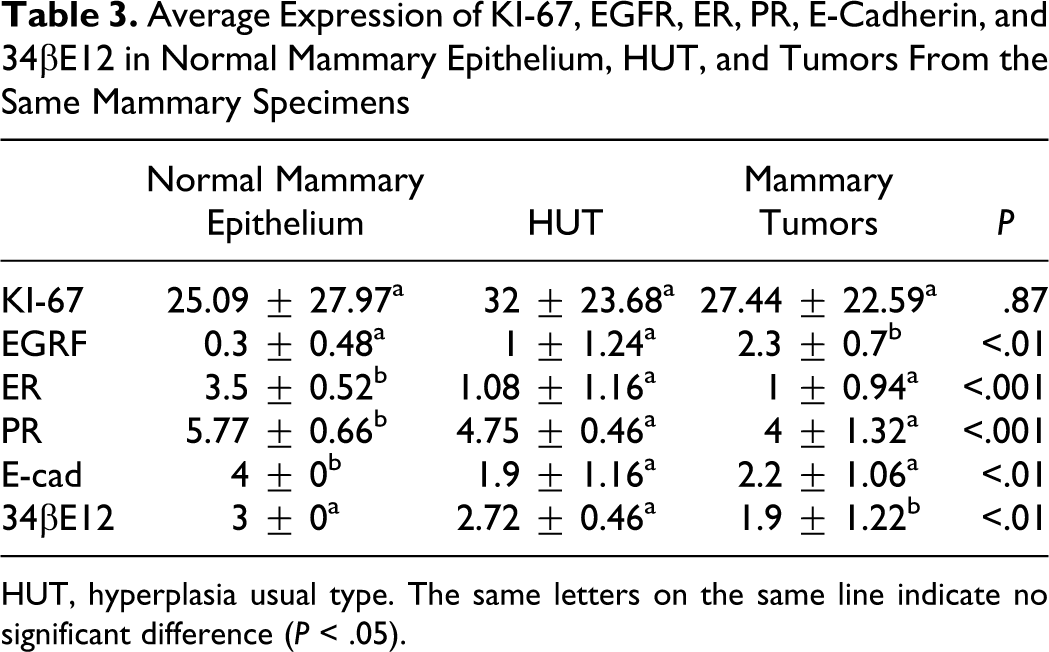
HUT, hyperplasia usual type. The same letters on the same line indicate no significant difference (P < .05).
Average Expression of KI-67, EGFR, ER, PR, E-Cadherin, and 34βE12 in Normal Mammary Epithelium, ADH, and Tumors From the Same Mammary Specimens
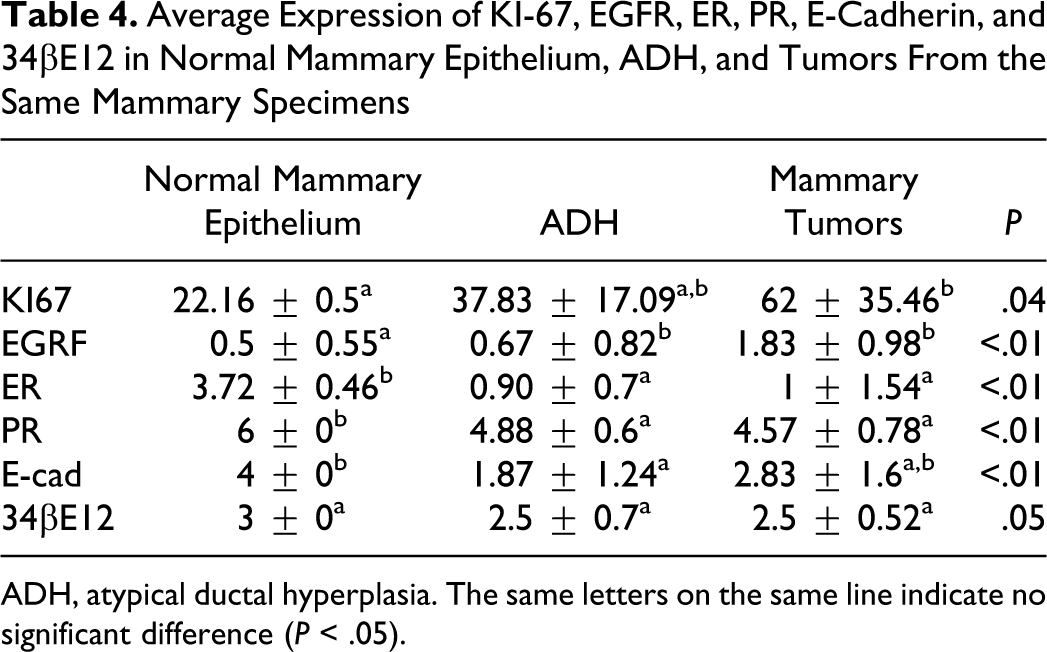
ADH, atypical ductal hyperplasia. The same letters on the same line indicate no significant difference (P < .05).
We noted p53 immunohistochemical positivity in 1 atypical ductal hyperplasia as well as in 4 tumors (2 DICS, 1 benign tumor, and 1 invasive carcinoma), but no evidence of the p53 expression was observed in adjacent cases of ductal hyperplasia. Two invasive carcinomas showed HER2 overexpression; however, all proliferative cells of ductal hyperplasia with or without atypia adjacent to tumors were negative for HER2.
Discussion
Hyperplastic lesions in dogs appear to follow a morphological pattern similar to that seen in human beings. However, the current veterinary classification criteria for epithelial hyperplasia in dogs do not correlate with possible prognostic features, as they are based only on the location and presence or absence of atypia. 26
We evaluated the presence of neoplastic and preneoplastic lesions, the sites they usually affect, and the mean age of occurrence, with results similar to the findings of previous published studies.3,32 About 56% of nonneoplastic mammary masses noted clinically are diagnosed as hyperplastic lesions, which occur in similar proportions in the inguinal, abdominal, and thoracic mammary glands. 5
According to the results of this study, ductal hyperplasia is mainly associated with mixed tumors whereas ADH is more commonly associated with invasive carcinomas. Our findings suggest that ADH is precursor lesion for invasive carcinomas of the canine mammary gland as has been shown in studies of human mammary neoplasia.24,30,31,33 Morphological and epidemiological studies of the human breast previously showed an increased risk of progression of atypical ductal hyperplasia toward ductal carcinoma in situ as well as invasive carcinomas.29,42
In our study, the high frequency of hyperplasia associated with neoplastic lesions may be attributable to the criteria used for case selection. Our samples were obtained from animals submitted for mammary biopsy as indicated by the finding of a mass during physical examination. However, within the scope of our selection criteria, our results show that atypical hyperplasia was more frequently associated with mammary carcinomas (in 30 of 51 cases) as opposed to those hyperplastic lesions associated with benign tumors.
The expression of different prognostic molecular markers has already been reported in studies examining both neoplastic and nonneoplastic lesions of canine mammary glands. A direct relationship has been shown between the expression of oncogenes and proteins involved in the process of neoplastic transformation and the malignant nature of the lesion.10,23,37
ADH and DCIS in human mammary lesions show decreasing levels of expression of high-molecular-weight cytokeratins CK5, CK14, and CK903. 28 In our study, we obtained similar results with the 34βE12 staining in both canine ADH and DCIS lesions. Our findings suggest that during malignant progression, the epithelial cells lose their 34βE12 expression. In addition to its possible role in cancer progression, high-molecular 34βE12 staining may be a useful tool to differentiate HUT and ADH/DCIS, as is currently used in immunohistochemical studies of human mammary biopsies. 21
Bratthauer et al 4 suggested that the combination of 34βE12 and E-cadherin antibodies could be useful in distinguishing lobular and ductal lesions that have overlapping morphological features. In addition to differentiating ductal lesions, a significant reduction of E-cadherin expression in atypical intraepithelial lesions may be a useful prognostic indicator in certain mammary pathologies (ADH and DCIS). 44 This decreased E-cadherin expression has a direct relationship with an increase of cell proliferation (Ki-67 index) and a reduction of ER expression, as seen in our results. These findings might be explained by transmembrane signaling mechanisms of growth factor receptors, hormone receptors, and molecules associated with epithelial-mesenchymal transition.1,16
The ER–estrogen complex is known for inducing genetic and epigenetic changes and for influencing different genes involved in the regulation of cell differentiation and proliferation. 39 From these mechanisms, an epithelial–mesenchymal transition (EMT) is suggested, and the progression to malignancy of mammary epithelial lesions is believed to be associated with a regulation of E-cadherin expression mediated by ER signaling. 47 There are few studies elucidating how this relationship occurs. However, a direct relationship between loss of E-cadherin expression and hormone receptors and increased expression of Ki-67 in mammary neoplasia has been reported. 18 There are no data to suggest a direct functional interaction between Ki67 and ER or E-cadherin proteins, so the strong association between these markers and poor prognosis is uncertain.
Previous results have led us to hypothesize that p53 may regulate ER expression. The negative immunohistochemical ER and positive p53 staining in mammary tumors seems to be related to poor prognosis. 40 In our series, those cases with neoplastic lesions in which there was positive p53 expression were found to be ER negative, and 1 case of ADH with associated DCIS was immunoreactive for p53. The simultaneous and coincidental occurrence of p53 mutants during neoplastic transformation cannot be excluded; however, our results should be interpreted with caution. The interpretation of these results is of course speculative, and a prospective study of dogs with ADH or DICS p53 positivity would be required to prove our hypothesis.
The ER activation process stimulates DNA synthesis, cell division, and the production of biologically active proteins, including transforming growth factor (TGF) and epidermal growth factor (EGF), both of which influence cell growth and differentiation.7,38 Atypical intraepithelial lesions in canine mammary glands apparently have a reduced ER expression and no overexpression of EGFR and HER2. These staining characteristics may be related to the activation of cell clones concurrent with loss of differentiation associated with the reduction of the existing regulation of the molecular pathway regulating ER and growth factor co-expression. Our results suggest that in ER-negative mammary lesions, cell proliferation is probably controlled by other paracrine growth factors (TGF-α and TGF-β) released by ER-positive cells and does not appear to be influenced by the autocrine ER activation of cyclins associated with the EGFR overexpression.6,9
The absence of ER expression in ductal hyperplasia and DCIS, as was noted in 75% of our cases, suggests that these lesions may be associated with tumor development secondary to decreased expression of HER2 and ER, as has been described in previous studies. On the basis of our data and those described in other publications, canine mammary intraepithelial lesions may be an interesting model to further study the correlation between decreased or absent HER2 and ER immunoreactivity and the development of mammary tumors.2,27
In conclusion, epithelial hyperplasias of canine mammary glands have the same ductal–lobular organization and similar molecular characteristics as those described in human lesions. We observed a direct relationship between the presence of in situ and invasive mammary carcinomas and the occurrence of atypical ductal hyperplasia. Additionally, gradual loss of expression of proteins related to proliferation control (ER, EGFR) and E-cadherin in intraepithelial lesions suggests that mammary hyperplasia in the dog may play an important role in the process of malignant neoplastic transformation. Prospective studies evaluating expression of molecular markers in epithelial proliferations may contribute to a better understanding of these changes in canine mammary tumor development.
Footnotes
Acknowledgements
We are thankful to Sandra J. Olson for the English review of the manuscript.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This work was financially supported by Fundação de Amparo a Pesquisa de Minas Gerais (FAPEMIG), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Brazil.
