Abstract
Twenty-seven feline cutaneous mast cell tumors (MCTs) were selected for this retrospective study. Samples were routinely processed and stained with hematoxylin and eosin (HE) and toluidine blue, and tumors were classified as well-differentiated (19/27), atypical or poorly granulate (7/27), and pleomorphic (1/27). Immunohistochemistry to detect KIT protein was performed on all samples. The immunoreactivity was recorded by distribution within the tumor, cellular location, and intensity. Well-differentiated MCTs were predominantly characterized by diffuse cytoplasmic (8/19) and membranous stain (7/19); a diffuse distribution of KIT positive cells was displayed in most of these tumors as well (15/19). Atypical MCTs showed diffuse distribution of labeled cells (4/7), and diffuse cytoplasm immunostaining was seen most (5/7). The pleomorphic MCT showed diffuse cytoplasmic KIT stain, with moderate labeling intensity, typically displaying focal distribution in deeper areas of the neoplasm. According to the results, there was no correlation between the type of MCTs and KIT expression, although the use of feline KIT immunohistochemistry could be useful to assess the mast cell origin.
Introduction
Mast cell tumors (MCTs) are frequently diagnosed in dogs and less commonly in cats. They have cutaneous or systemic presentations, and MCTs represent the second most commonly encountered cutaneous neoplasm in cats, with an estimated prevalence up to 20% of all skin tumors. 5,6 Feline cutaneous MCTs usually appear as single or multiple masses, most frequently in the head and neck. Clinically, cutaneous MCTs are nodular, discrete, often ulcerated lesions reported in animals older than 4 years of age, without sex or breed preference. 6,7
Microscopically, cutaneous MCTs of cats are classified into 3 categories: 1) well-differentiated, 2) atypical or poorly granulated, also called histiocytic-like, and 3) pleomorphic or poorly differentiated. 6,7 Well-differentiated and pleomorphic MCTs usually behave benignly, but atypical MCTs are considered malignant. Some authors have suggested that the Siamese breed is at higher risk to develop atypical or poorly granulated MCTs, 1,6,7 although another study indicates no clear breed predilection. 11
The KIT protein is the product of the c-kit proto-oncogene, and it is a type-III receptor tyrosine kinase expressed by mast cells and mast cell progenitors. It consists of an extracellular ligand-binding domain, made up of 5 immunoglobulin-like repeats, a transmembrane hydrophobic region, and a cytoplasmic domain with tyrosine kinase (TK) activity. 3,22,27 Stem cell factor (SCF), the ligand of the KIT protein, is a cytokine that stimulates mast cell growth and differentiation. In the skin, SCF is produced by fibroblasts, keratinocytes, and endothelial cells. 4,14,15,24
In spontaneous canine cutaneous MCTs, mutations have been detected in the juxtamembrane region of KIT, suggesting that they could be implicated in the pathogenesis of MCTs. 13,15,25 Alterations in the process of ligand binding and constitutive TK activation, caused by c-kit mutations, have been described as triggering effects for MCTs development. 13,15
KIT expression has been demonstrated by immunohistochemistry (IHC) in normal cells from dogs and cats such as mast cells, mammary gland epithelial cells, interstitial cells of Cajal, oocytes, endometrium and Purkinje cells, as well as neoplastic cells in malignant melanoma, mammary gland adenoma and carcinoma, gastrointestinal stromal tumors, seminoma, Sertoli cell tumor, interstitial cell tumor, ovarian papillary carcinoma, endometrial carcinoma, pheocromocytoma, and MCTs. 9,16,19,23
Previous studies in our department demonstrated that canine cutaneous MCTs showed KIT protein expression; 2 patterns of protein immunolocation were described, and an inverse correlation between the degree of differentiation and expression of KIT was reported. 23 Researchers have proposed the use of the KIT-staining pattern as a prognostic tool in routine evaluation of canine MCTs. These patterns are defined by membranous (pattern I), focal or stippled cytoplasmic or paranuclear (pattern II), and diffuse cytoplasmic (pattern III) protein location. 10,21
There are no reports on the relationship between the histologic type of feline cutaneous MCTs and KIT expression patterns, as described for canine cutaneous MCTs. Therefore, the aim of the present study was to investigate the expression of the KIT protein in feline cutaneous MCTs and to elucidate whether there is any relationship between histologic types of feline MCTs and expression of KIT.
Materials and Methods
Samples
Twenty-seven feline cutaneous MCTs were selected for the present retrospective study. All tumors were diagnosed at the Department of Veterinary Pathology at the Universitat Autònoma de Barcelona between 1997 and 2005. Tissues were received at the laboratory already fixed by immersion in 10% neutral buffered formalin; they were routinely processed and stained with HE and toluidine blue. The tumors were reevaluated by 2 veterinary pathologists and classified according to Hendrick et al. 7 and Gross et al. 8 Clinical data, histologic diagnosis, and classification are described in Table 1.
Feline mast cell tumors: clinical data, tumor anatomic location, histologic classification and immunohistochemical detection of KIT protein.∗
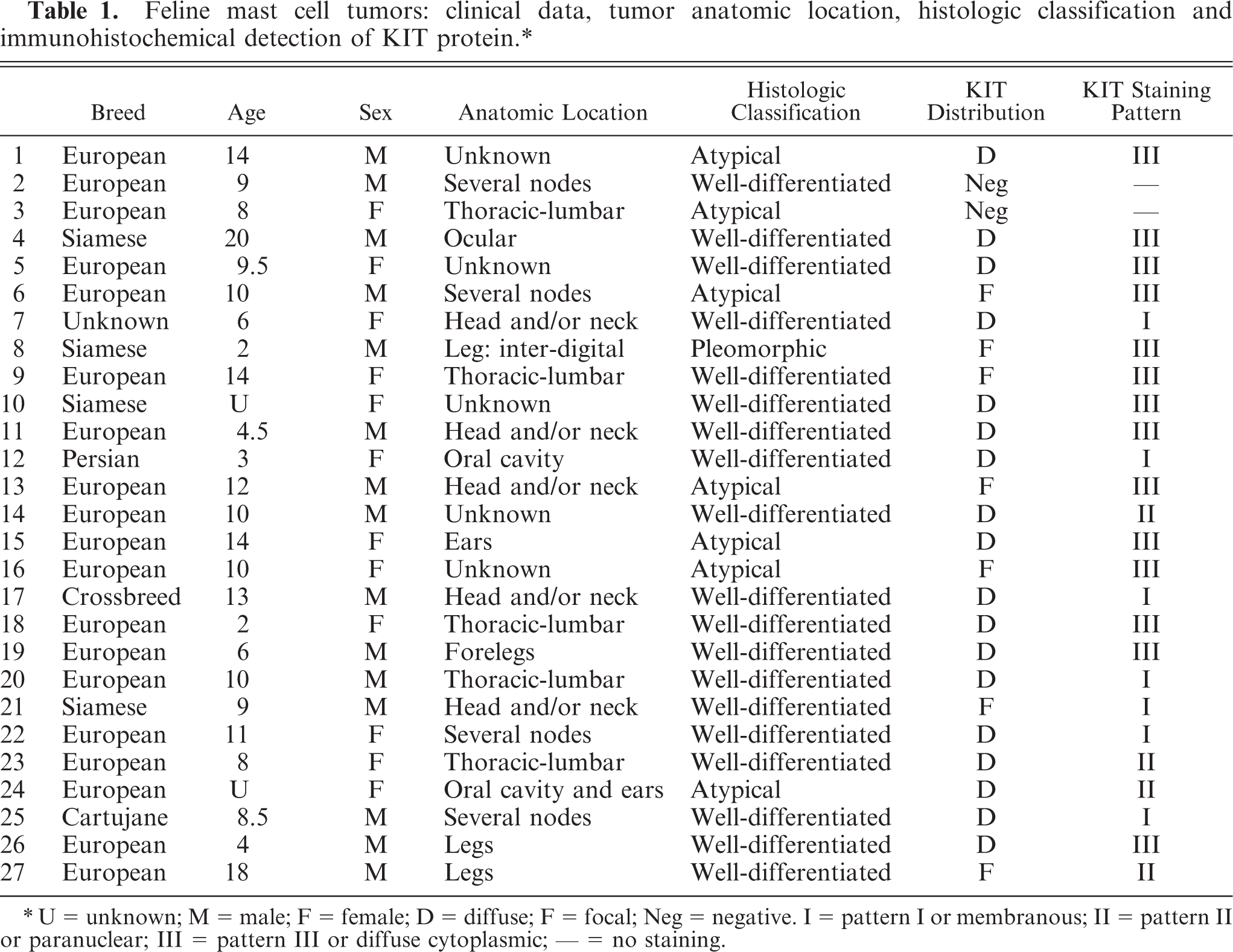
∗U = unknown; M = male; F = female; D = diffuse; F = focal; Neg = negative. I = pattern I or membranous; II = pattern II or paranuclear; III = pattern III or diffuse cytoplasmic; – = no staining.
Immunohistochemistry (IHC)
Studied tissues were cut to a thickness of 5 µm, placed on glass slides coated with 3-triethoxysilyl-propylamin, deparaffinized in xylene, rehydrated in graded alcohols, and rinsed in distilled water. Sections were then treated with hydrogen peroxide in methanol (3%) for 30 minutes for inhibition of the endogenous peroxidase. As antigen retrieval treatment, citrate buffer pH 6.0 was used for 15 minutes at 95°C in a water bath. Before application of the primary antibody, blocking of nonspecific binding was done for 1 hour with 10% normal goat serum in Tris-buffered saline (TBS). Primary antibody (CD117/c-kit polyclonal rabbit anti-human, Dako-Cytomation, Glostrup, Denmark) was incubated overnight at 4°C, at dilution 1 : 300 in TBS. The peroxidase-base detection system used was Envision system (Dako-Cytomation). Normal feline cerebellum and 2 canine MCTs (tested previously as a membrane and a diffuse cytoplasm immunostaining) were used as KIT-IHC positive control tissue. As a negative immunohistochemical control, the primary antibody was replaced by TBS. Immunoreactions were visualized using diaminobenzidine (Sigma-Aldrich, St. Louis, MO) plus 0.05% H2O2 in imidazole buffer. Samples were counterstained with Mayer's haematoxylin.
Results
Details of the 27 cats included in this study are given in Table 1. Selected cutaneous MCTs included 8 intact and 4 spayed females, as well as 6 intact and 9 castrated males. Five different breeds were represented, including 19 European, 4 Siamese, 1 Persian, 1 Cartujane, and 1 crossbreed. Breed information was unavailable for 1 female cat. The average age of affected cats was 9.36 years within a range of 2 to 20 years.
Twenty MCTs were solitary nodules (74.07%). The most common anatomic locations of cutaneous MCTs were head (including nose, mouth, eyes, and ear locations) and neck (n = 9, 33.33%), followed by lumbar and costal areas (n = 5, 18.51%), legs and forelegs (n = 4, 14.81%); 4 cats had multiple tumors without indication of specific locations (14.81%). The tumor location was not described in 5 of the studied cats.
The immunoreactivity for each tumor was recorded by distribution of positive cells within the tumor (diffuse or focal) and cellular location (membranous, or pattern I; paranuclear or focal cytoplasmic, or pattern II; and diffuse cytoplasmic staining, or pattern III) (Table 1). Tumors that had a diffuse cytoplasmic stain or pattern III showed different intensities of labeling; therefore, the stain was described as strong, moderate, or weak.
Most of feline cutaneous MCTs in this study were diagnosed as well differentiated (n = 19, 70.37%), and 18 out of 19 were positive to KIT. The distribution of the positive neoplastic cells was variable. Fifteen MCTs (83.33%) displayed a diffuse labeling distribution, while only 3 MCTs (16.67%) showed a focal staining in deeper areas of the neoplasms. Related to the cellular location, 7 MCTs (38.89%) had a membranous and weak cytoplasmic staining, or pattern I (Fig. 1); 3 MCTs (16.67%) showed a strong labeling in paranuclear location and weak cytoplasmic staining, or pattern II (Fig. 2); and 8 MCTs (44.45%) displayed a diffuse cytoplasmic reaction, or pattern III. Among the 8 neoplasms with pattern III, diffuse cytoplasmic labeling was strong in 2 cats (25%) (Fig. 3), moderate in 1 cat (12.5%), and weak in 5 cats (62.5%).

Well-differentiated mast cell tumor; cat No. 7. Labeled for KIT protein. Membrane-associated with weak cytoplasmic staining, pattern I. Immunoperoxidase method, counterstained with Mayer's hematoxylin.
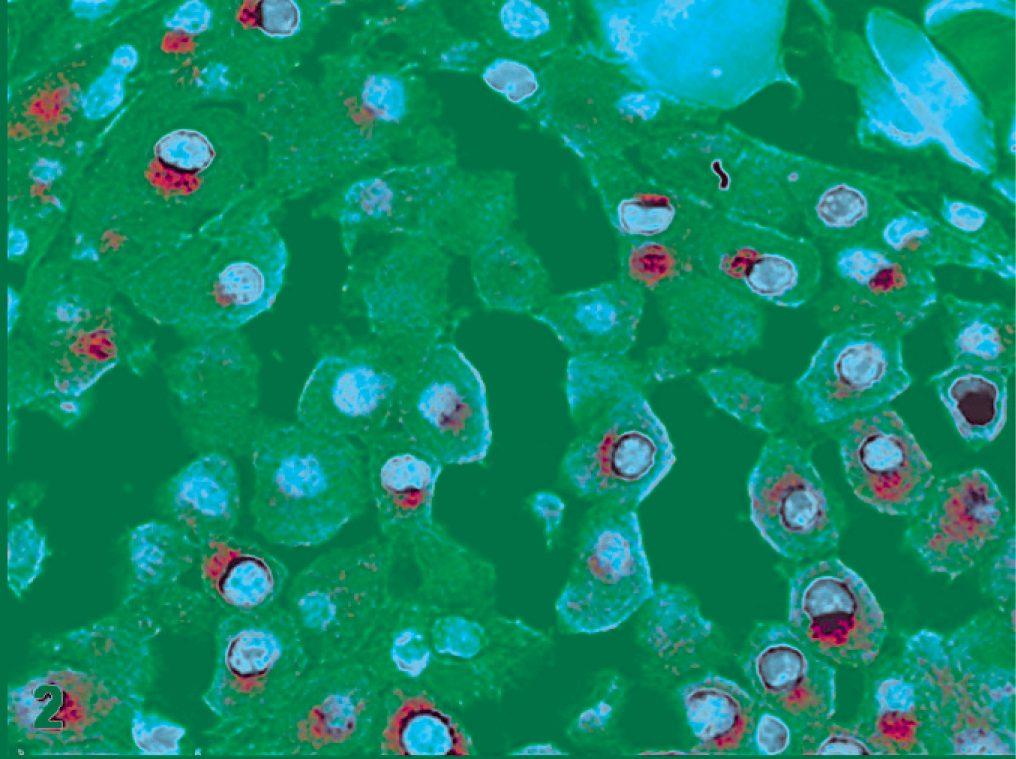
Well-differentiated mast cell tumor; cat No. 2. Labeled for KIT protein. Paranuclear with weak cytoplasmic staining, pattern II. Immunoperoxidase method, counterstained with Mayer's hematoxylin.
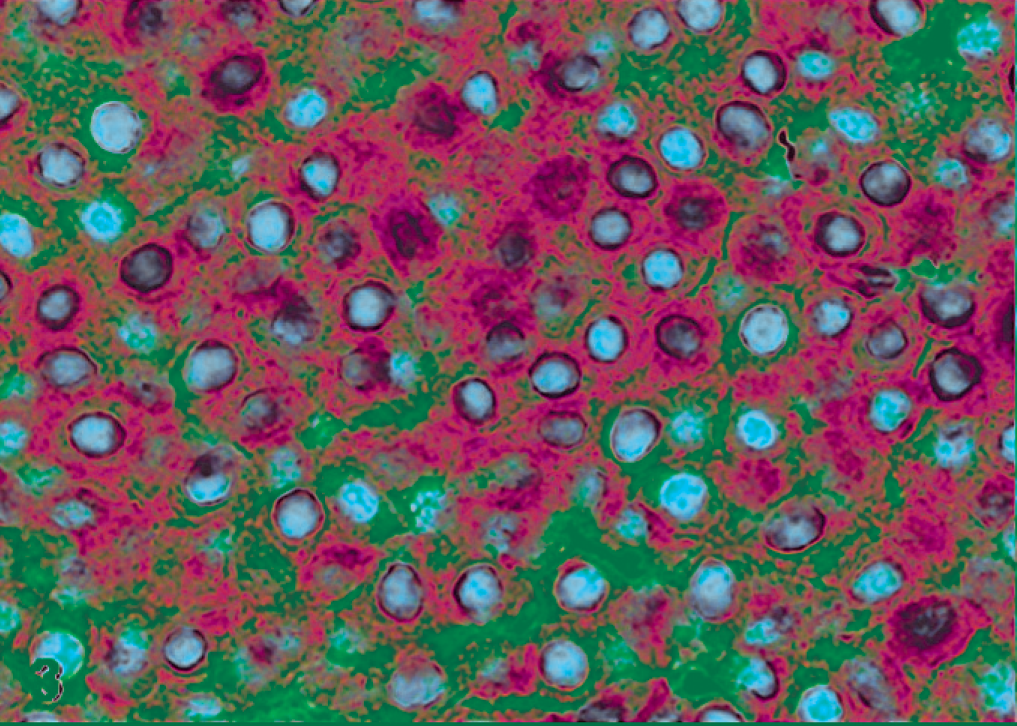
Well-differentiated mast cell tumor; cat No. 19. Labeled for KIT protein. Diffuse and strong cytoplasm labeling, pattern III. Immunoperoxidase method, counterstained with Mayer's hematoxylin.
Seven out of 27 (25.93%) tumors were classified as atypical or poorly granulated MCTs, and 6 of them (85.71%) showed positive KIT immunoreaction. The distribution of KIT staining was diffuse in 4 cats (66.67%) and focal in 2 cats (33.33%). In these latter cats, the immunoreaction was located in deeper areas of the neoplasms. One out of 6 IHC-positive neoplasms (16.67%) showed positive staining in paranuclear location, pattern II (Fig. 4), and 5 (83.33%) displayed strong (n = 1) (Fig. 5) or moderate (n = 4) diffuse cytoplasmic reaction, or pattern III. None of the atypical MCTs studied were classified as having pattern I or membranous staining.
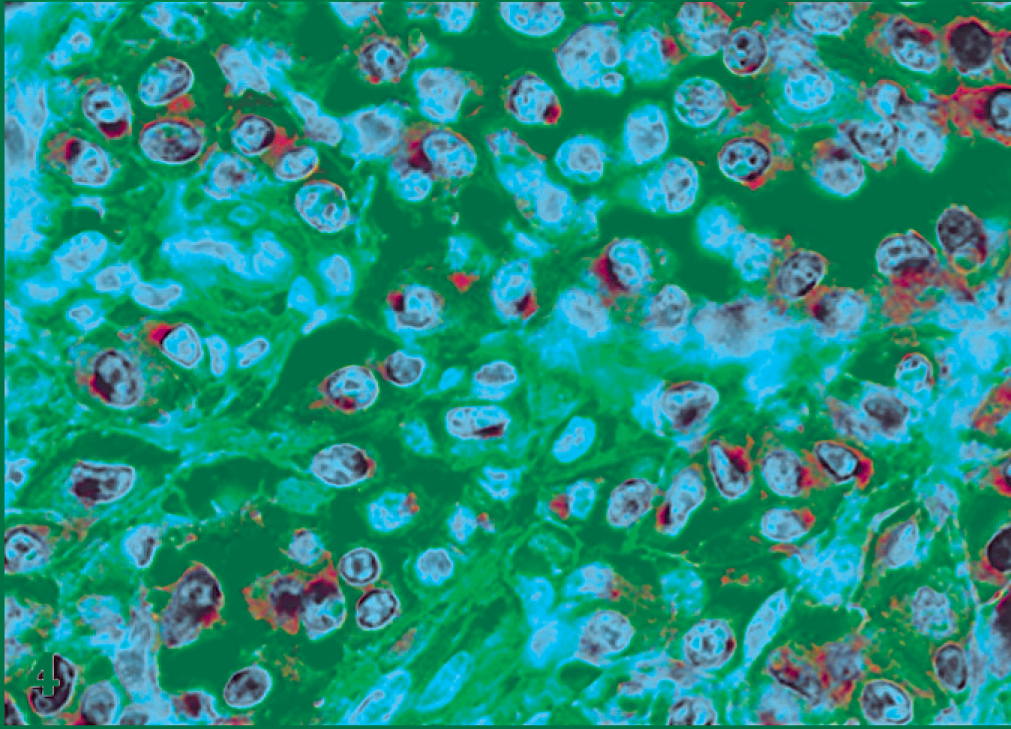
Atypical mast cell tumor; cat No. 1. Labeled for KIT protein. Paranuclear with weak cytoplasm staining, pattern II. Immunoperoxidase method, counterstained with Mayer's hematoxylin.
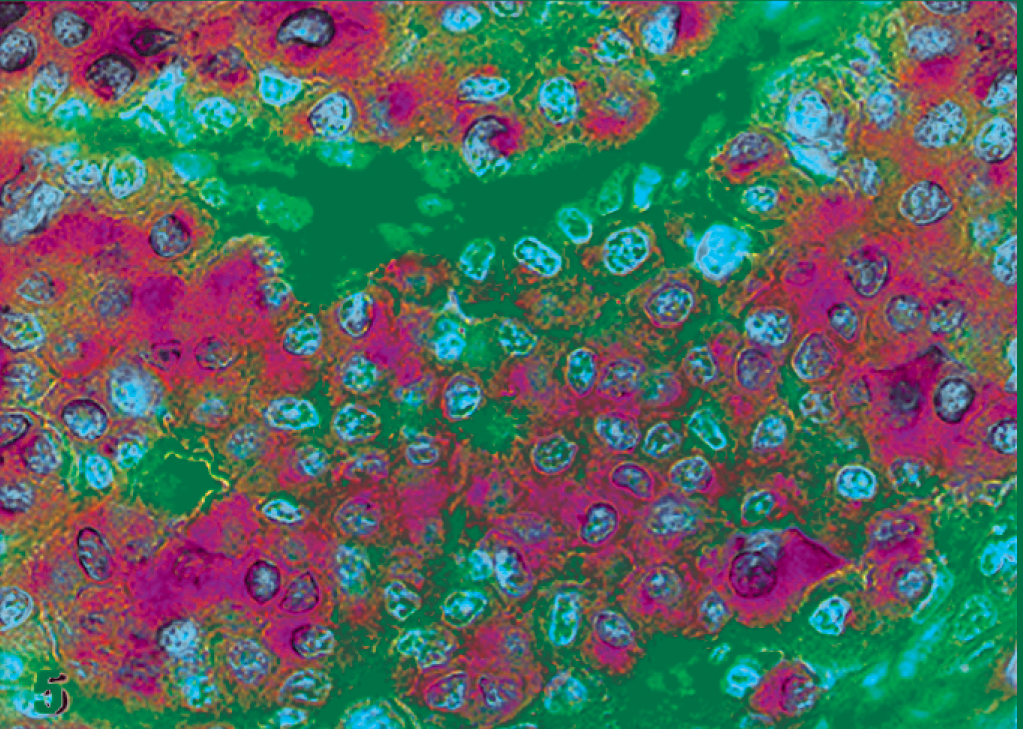
Atypical mast cell tumor; cat No. 16. Labeled for KIT protein. Diffuse and strong cytoplasm labeling, pattern III. Immunoperoxidase method, counterstained with Mayer's hematoxylin.
One out of 27 tumors was classified as a pleomorphic or poorly differentiated MCT. This neoplasia displayed focal distribution of KIT-IHC labeling, with immunoreaction located in deeper areas of the neoplasm. Positive cells showed a moderate diffuse cytoplasmic staining, and the tumor was classified as pattern III (Fig. 6).
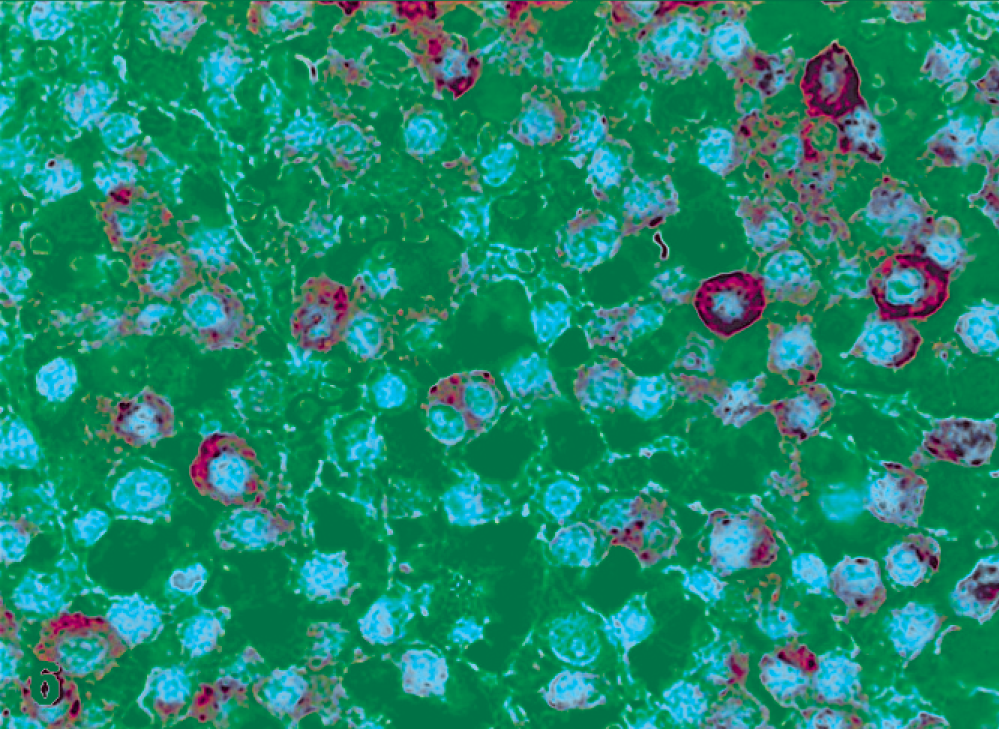
Pleomorphic mast cell tumor; cat No. 8. Labeled for KIT protein. Diffuse and moderate cytoplasm labeling, pattern III. Immunoperoxidase method, counterstained with Mayer's hematoxylin.
Discussion
MCT is a common neoplasm in cats and may represent up to 20% of all cutaneous tumors. 5 The grading system for canine cutaneous MCTs proposed by Patnaik et al., 20 which is in current use, establishes a correlation between MCT histopathologic grade and survival time in dogs. However, this grading system does not seem to be useful for cats, since most feline MCTs are benign and the microscopic features of canine MCTs are not evident in cats. Therefore, several authors have proposed 3 different feline tumor types that include well-differentiated, atypical, and pleomorphic MCTs. 6,9,18 Unfortunately, there is no epidemiologic data concerning overall prevalence or incidence of this feline neoplasm related to its microscopic classification.
In this study, the average age (9.36 years) and anatomic locations were in accordance with most published data. 2,6,7,9,11,12,18 The distribution of cat breeds, mostly European, must be understood as a representation of the local pet population sampled, and a higher number of cases would be needed to conclude true breed susceptibility for MCT development. The Siamese breed, considered to be more prone to suffer from atypical MCTs by several authors, 6,17,26 did not appear to be overrepresented in this study (n = 4, 10.3%). On the other hand, in this breed, MCT is usually mentioned in younger cats; 6,26 however, in this study, Siamese cats ranged from 2 to 20 years of age, with an average of 10.3 years.
A slightly higher percentage of cutaneous MCTs corresponded to male cats. 2 However, most published studies indicate no sex predisposition for MCTs in cats. 11,12,17,18
As general features, and according to some researchers, 19 feline cutaneous MCTs express KIT in a similar proportion in membranous and diffuse cytoplasmic locations, and diffuse cytoplasmic immunoreaction is usually moderate. As compared with other studies, a significant number of MCTs in this study corresponded to atypical MCTs (n = 7, 25.95%). 11,26 Taking into account that atypical MCTs are considered a challenge for the histologic diagnosis by conventional methods (HE stain), the use of KIT-IHC may be important to characterize these tumors. Most of these atypical MCTs (6/7) were positive for this antibody.
Although 2 feline cutaneous MCTs were negative to KIT-IHC, it cannot be ruled out that inadequate antigen preservation may have taken place. Further studies should be conducted on feline MCTs in order to elucidate whether, as in canine MCTs, KIT-IHC should become a definitional feature for these neoplasms. A prognostic indication might be obtained from simultaneous evaluation of clinical, histologic, and KIT-IHC features.
KIT immunoreaction from well-differentiated feline MCTs was detected mainly in membrane (pattern I) and diffuse cytoplasmic (pattern III) locations, while atypical ones showed mostly diffuse cytoplasmic labeling. Taking into account that KIT, a transmembrane protein, is located in the cell membrane of normal canine mast cells and neoplastic cells from well-differentiated MCTs of dogs, 10,23 it seems that KIT protein in cats can be also present in cytoplasm in some feline MCTs. In fact, it is known that numerous mutations in c-kit have been implicated in the progression of MCTs in humans and dogs with aberrant KIT location. One possible explanation for this phenomenon is that the cells that present KIT in their cytoplasm are actively synthesizing this receptor, which later migrates to the cell membrane. Alternatively, it is also possible that KIT is mostly in an activated form in these cells. 23
In summary, the aim of the present study was to investigate the expression of the KIT protein in feline cutaneous MCTs and to establish a potential relationship between histologic types and expression of KIT similar to what is described in cutaneous canine MCTs. 10,21,23,25 However, even though a greater number of tumors were investigated than in previous studies, 19 it was not possible to establish any correlation between the type of MCTs and KIT expression.
Footnotes
Acknowledgements
We are grateful to CReSA personnel and Veterinary Pathology Diagnostic Service from the Universitat Autònoma de Barcelona, especially to Mrs. Blanca Pérez and Ms. Aida Neira for their excellent technical help. This work was funded by the Project No. 163253 from UAB. PhD studies of Carolina Rodriguez Cariño are funded by a bursary of Universidad Central de Venezuela.
