Abstract
Feline cutaneous mast cell tumors (FeCMCTs) are characterized by variable biological behavior. Development of multiple nodules and potential visceral involvement, along with inconsistency of conventional prognostic aids, justify uncertainty in differentiating benign from malignant forms. c-Kit proto-oncogene activating mutations have been reported in feline mast cell tumors (MCTs), but their prognostic relevance was not investigated. This study was performed on FeCMCTs with variable clinical outcome to assess whether Kit cytoplasmic immunohistochemical labeling can be regarded as indicative of c-Kit mutations and to evaluate the relationship between Kit dysregulation and survival. Twenty-four cats diagnosed with a primary cutaneous MCT were enrolled. Kit immunohistochemical pattern and c-Kit (exons 8, 9, 11) mutational status were assessed in 34 tumor samples. Risk factors affecting survival were a number of mitoses greater than 5 per 10 HPFs (P = .017) and cytoplasmic Kit labeling (P = .045). Increased mitotic activity was associated with Kit cytoplasmic expression (P = .01). c-Kit encoding mutations were present in 19 (56%) tumors (exon 8, 19%; exon 9, 71%; exon 11, 10%), however, they were not significantly related to protein expression and they had no influence on prognosis. Additionally, in 6 of 9 (67%) cats, multiple nodules from the same cat had different mutational statuses. Mutations in the fifth immunoglobulin-like domain of Kit occur frequently in FeCMCT, but they are variably associated with aberrant protein expression and do not appear to be strictly correlated with biological behavior. These findings need to be confirmed in larger series, and exploration of further genomic regions of c-Kit is warranted.
Mast cell tumors (MCTs) globally account for 15%–21% of all feline skin neoplasms. 26,29 Although clinical and histological features are well described, the biological behavior of these tumors is still controversial.
Several authors consider the majority of feline cutaneous mast cell tumors (FeCMCTs) to be benign, despite their tendency to recur locally or to develop at additional skin sites, but in almost all series there is a variable proportion of tumors that behave aggressively and are ultimately fatal.9,22,31,37,38
The veterinary literature has conflicting reports on the coexistence of cutaneous and visceral MCTs and on which primary form of the tumor is predominant. 10 Cats usually have the cutaneous form diagnosed first and then later succumb to the visceral form. 4,10,18,33 Systemic spread of cutaneous tumors has been reported in 0%–22% of cases, 20 and disseminated cutaneous nodules seem to be more frequently associated with systemic disease. 29 However, cats with solitary tumors may later develop multiple tumors without evidence of visceral involvement, and this condition also appears to be associated with a guarded prognosis. 1,6,22,29
Additionally, most studies indicate that the histological features usually associated with malignancy, such as pleomorphism or infiltrative growth, are not constantly related to clinical behavior in FeCMCTs, with the exception of mitotic activity. 2,16,28
For these reasons, the detection of FeCMCTs with potential aggressive behavior is still a challenge, and there is a need to find methods for identifying this subset of tumors at the time of diagnosis.
The c-Kit proto-oncogene encodes the receptor tyrosine kinase Kit, which has been shown to regulate normal mast cell survival, proliferation, differentiation, and migration. 5,25,35
Kit protein includes an extracellular domain composed of 5 immunoglobulin-like domains (encoded by exons 1–9), a transmembrane domain (exon 10), and an intracellular domain (exons 11–21). The intracellular domain is further divided into a negative regulatory juxtamembrane domain (exons 11 and 12) and a cytoplasmic tyrosine kinase domain that is split by an insert into ATP-binding (exon 13) and phosphotransferase lobes (exon 17). Ligand binding induces receptor dimerization, which activates the tyrosine kinase, resulting in autophosphorylation and in the phosphorylation of exogenous substrates. These phosphorylations then lead to downstream signal transduction. 21
In canine MCTs, juxtamembrane domain c-Kit mutations and aberrant cytoplasmic Kit protein localization are significantly associated with increased cellular proliferation and with both reduced progression-free and overall survival rates. 36,37 As a consequence, Kit receptor tyrosine kinase inhibitors (TKIs) are an active part of MCT treatment in dogs. 23
In feline MCTs, aberrant cytoplasmic Kit immunohistochemical expression has been reported in 29%–67% of cases, and a higher frequency was observed in tumors with unfavorable outcome. 24,30,31 c-Kit proto-oncogene mutations in exons 6, 8, 9, and 11 have been detected, and in a pilot clinical study, beneficial response to molecular targeted therapy was obtained in 7 cats. 15 However, the prognostic relevance of c-Kit mutations has never been investigated in this species. A thorough understanding of the role that c-Kit plays in the development of feline MCTs may aid the clinical approach and successful treatment of this disease.
This study was aimed at characterizing Kit receptor dysregulations (ie, cytoplasmic immunohistochemical expression and/or c-Kit mutations) in a series of FeCMCTs with variable disease progression. Specifically, our goals were (1) to assess whether aberrant cytoplasmic expression of Kit could be regarded as indicative of c-Kit mutations and (2) to evaluate the relationship between Kit dysregulation and tumor proliferation, disease progression, and overall survival.
Materials and Methods
Study Overview and Case Inclusion Criteria
Medical records of cats diagnosed with MCTs between January 2005 and December 2010 were retrieved from the archives of the Department of Veterinary Medical Sciences (University of Bologna, Italy) and reviewed. Selection criteria were a histological diagnosis of MCT (1 or more nodules) of reported primary cutaneous origin and availability of follow-up information regarding disease progression and outcome. A minimum follow-up time of 12 months was required for inclusion.
Medical Data
Data collected in all cases included signalment, tumor location (head and neck, trunk, limbs), and number of nodules at presentation.
Additional information included the results of staging procedures, when available. Treatment details were also recorded, including use of surgical resection—complete (removal of all the cutaneous MCTs with histologically clean margins) or incomplete—and administration of adjuvant therapies.
Follow-up information was gathered from online forms or telephone interviews with the referring veterinarian or the cat’s owner. Information and dates were recorded about disease progression and outcome. Progression-free survival (PFS) was defined as the time (days) from the date of diagnosis to the earliest evidence of local tumor recurrence, new cutaneous MCT development, or metastasis to visceral organs. Overall survival (OS) was defined as the time (days) from the date of diagnosis to the last reported date that the patient was seen alive. At the end of the study, the patient status was recorded as alive, dead because of non-tumor-related causes, or dead because of tumor-related causes.
Histological Examination
For each cat, formalin-fixed, paraffin-embedded (FFPE) tumor samples were retrieved from the archives of the Pathology Unit of the Department of Veterinary Medical Sciences. In the case of multiple concurrent MCTs or the development of new cutaneous MCTs after the initial diagnosis, all the nodules with available histological material were included in order to evaluate the differences in Kit expression pattern and c-Kit mutations.
Histological sections were cut at 4 μm, stained with hematoxylin and eosin (HE) and toluidine blue, and reviewed to confirm the original diagnosis. Tumor proliferation was assessed by calculating the mitotic count (MC). MC was defined as the number of mitotic figures per 10 high-power fields (400×). The areas with the highest mitotic activity were chosen for evaluation.
Immunohistochemical Analysis
Immunohistochemical staining for CD117 (rabbit polyclonal antibody, 1:100, Dako, Glostrup, Denmark) was applied to assess Kit protein expression.
Immunohistochemistry (IHC) was performed with a strep-tavidin-biotin-peroxidase technique. Four-micrometer-thick FFPE tissue sections were dewaxed and rehydrated. Endogenous peroxidase activity was blocked by incubation for 30 minutes with 3% hydrogen peroxide in distilled water. Slides were then microwaved in citrate buffer (2.1 g of citric acid monohydrate per liter of distilled water; pH 6.0) for 2 cycles of 5 minutes at 750 W for antigen retrieval. Sections were incubated overnight at 4°C in a humid chamber with the primary antibody diluted in phosphate-buffered saline (PBS; pH 7.4, 0.01 M) with 1% bovine serum albumin to prevent nonspecific labeling. Following washing in PBS, sites of primary antibody binding were identified using a commercial streptavidin-biotin-peroxidase kit (LSAB, Dako), and diaminobenzidine was used as chromogen (0.04% for 10 minutes at room temperature).
Sections were rinsed in PBS and in running tap water, counterstained with Papanicolaou’s hematoxylin, dehydrated, and mounted under DPX (Sigma-Aldrich, St. Louis, MO). A Kit-positive canine cutaneous MCT and feline normal cerebellum were used as positive controls to assess the specificity of the reaction. Negative controls were obtained substituting the primary antibody with an unrelated antibody of the same isotype.
Kit protein expression was classified using the patterns described for canine MCTs. 17 For data analysis, Kit patterns II and III (focal to stippled and diffuse cytoplasmic staining) were grouped and challenged versus Kit pattern I (membranous staining).
Mutation Analysis
Genomic DNA was purified from 6- to 8-μm sections of FFPE tumor samples by using a commercial kit (Nucleospin Tissue DNA, Macherey-Nagel, Düren, Germany). Sections were stained with HE to identify the most appropriate areas to sample (ie, containing a high proportion of neoplastic cells). The selected areas were collected using a cotton swab soaked with phosphate buffer saline by gently rolling the cotton tip on the glass surface. DNA was extracted per the manufacturer’s instructions with a slight modification represented by an heat incubation step at 90°C for 1 hour aimed at removing DNA crosslinks immediately after the prelysis phase of the purification protocol. A negative extraction control was also included for each batch.
Exons 8, 9, and 11 of feline c-Kit were PCR amplified using the oligonucleotides already described. 15 Furthermore, for exons 8 and 9, whenever the full-length amplicon could not be obtained using those primers, the target was amplified using different combinations of the same outer and novel inner primers, as schematically indicated in Fig. 1. Detailed primer sequence is reported in Table 1. PCR products were then purified using Nucleospin PCR and Gel Clean Up kit (Macherey-Nagel), forward and reverse cycle-sequenced using Big Dye Terminator v. 1.1 kit (Applied Biosystems, Foster City, CA), and purified again with Centri-Sep columns (Applied Biosystems). After denaturation with formamide (HiDi, Applied Biosystems), the amplified DNA segments were electrophoresed on ABI Prism 310 DNA sequencer (Applied Biosystems). Obtained electropherograms were analyzed by Sequencing Analysis v. 5.2 software (Applied Biosystems). If DNA amplification was not successful, the case was excluded from the study.
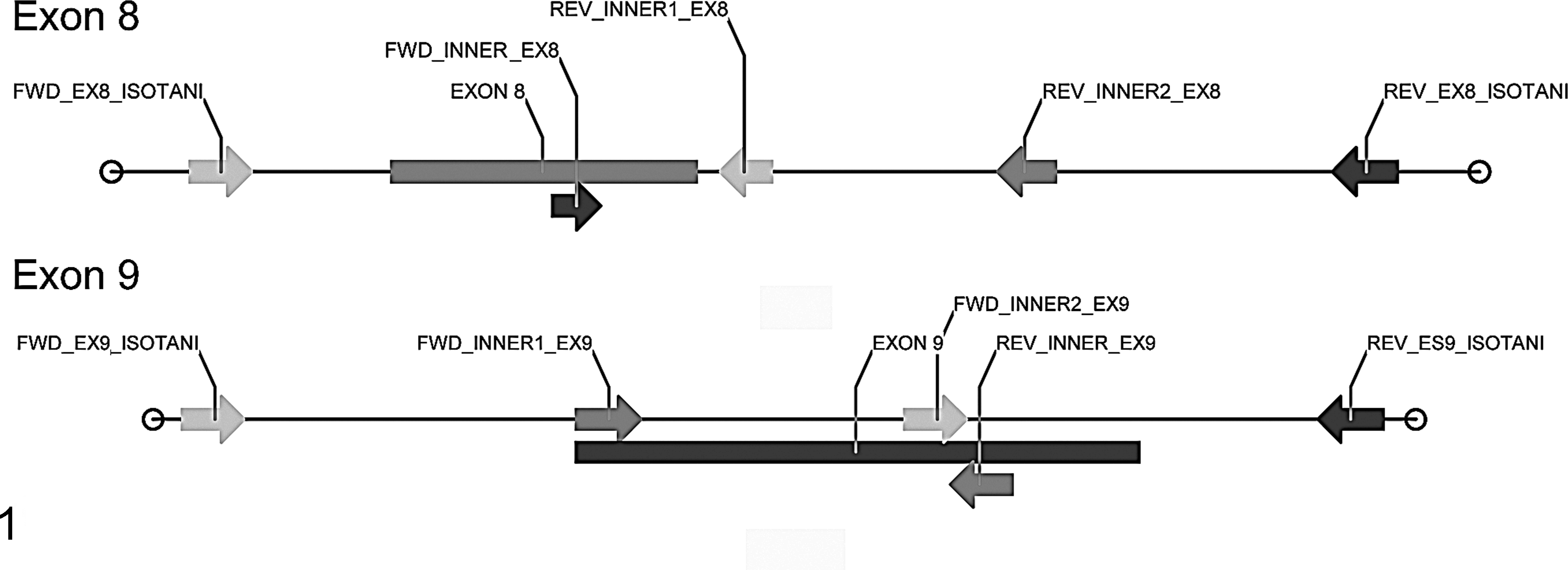
Schematic diagram of the position of the outer and inner primers used for the amplification of exons 8 and 9 of feline c-Kit.
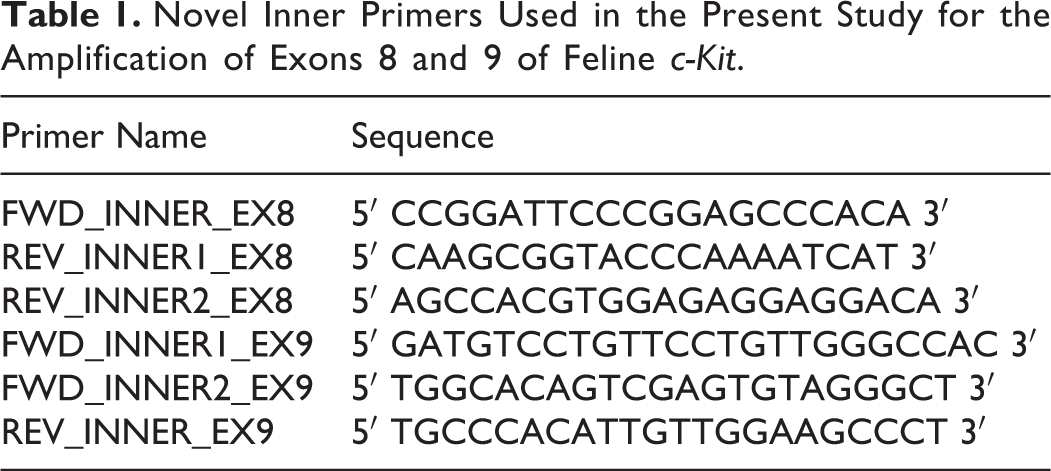
Novel Inner Primers Used in the Present Study for the Amplification of Exons 8 and 9 of Feline c-Kit
Tumors were classified as mutated when presenting point mutations, duplications, or deletion/insertions (delins) in 1 or more of the amplified exons of feline c-Kit, resulting in amino acid changes in the protein.
Statistical Analysis
Data were analyzed by use of commercial software programs (SPSS Statistics v. 19, IBM, Somers, NY, and Prism v. 5.0, GraphPad, San Diego, CA); P values ≤.05 were considered significant. When appropriate, data sets were tested for normality by use of the D’Agostino and Pearson omnibus normality test. Values were expressed as mean ± standard deviation in case of normal distribution or as median with a range in case of nonnormal distribution. Median values were compared by use of the Mann-Whitney U-test for unpaired data.
The relationship between Kit immunohistochemical pattern (membranous or cytoplasmic) and c-Kit mutations (present or absent) was assessed by use of Fisher’s exact test. Kit-negative tumors were not considered in this analysis.
Risk analyses for PFS and OS were performed with Cox proportional hazards model. Cats developing local tumor recurrence, new cutaneous MCT, or metastasis to visceral organs were recorded as events for PFS analysis; cats that were reported dead because of MCT were recorded as events for OS analysis. Variables analyzed for prognostic significance were age, tumor location, number of nodules at presentation, surgical resection, MC, Kit immunohistochemical pattern, and c-Kit mutations. The sensitivity and specificity of MC as a predictor of death due to MCT were analyzed using the receiver operating characteristics (ROC) curves. Individual values with the respective sensitivity and specificity were scrutinized to individuate the optimal cutoff for risk analysis. In the case of multiple concomitant tumors from the same cat, the highest mitotic count and the presence of Kit cytoplasmic pattern and c-Kit mutations in at least 1 tumor were considered. The new MCTs arisen after the initial diagnosis were not included in survival analysis.
Those risk factors having a P value <.05 in univariable analysis were included in multivariable model building. Because of the small number of cats and the heterogeneity of adjuvant therapies, multivariable analysis could not be performed to account for the effect of those treatments.
Results
Medical Data
Twenty-four cats were included in the study. A table with all the information available about signalment, history, and clinical course is available online as supplementary material. There were 21 domestic shorthairs, 1 domestic longhair, 1 Siamese crossbreed, and 1 Chartreux. Mean age was 8.4 ± 3.31 years. There were 13 females (54%; 2 intact, 11 spayed) and 11 castrated males (46%).
At diagnosis, 15 cats (63%) had a single cutaneous nodule, 5 (21%) had 2 nodules, 1 (4%) had 6 nodules, and 3 (12%) had disseminated nodules all over the body. Excluding disseminated MCTs, 16 tumors (52%) were located on the head and neck, 11 (35%) on the trunk, and 4 on the limbs (13%).
Staging workup (consisting of physical examination, complete blood count, buffy coat examination, biochemistry panel, chest radiography, and abdominal ultrasound) was performed in 17 cats (71%) and did not indicate any systemic involvement, although ultrasound-guided fine-needle aspirates of spleen and liver were only obtained in 1 cat. In the remaining 7 cats (29%), physical examination of the abdomen was unremarkable and abdominal ultrasound was not performed.
All cats underwent surgery. Excision was complete in 16 cats (67%) and incomplete in 5 (21%, including the cats with disseminated lesions), whereas in 3 cats (12%) the completeness of excision was not assessed. Adjuvant treatment with corticosteroids was administered in 6 cats (25%). Lomustine, vincristine, and the tyrosine-kinase inhibitor masitinib were administered in 1 cat each (17%).
Median follow-up time was 740 days (range, 372–1922 days). According to the observed clinical course, there were 12 (50%) progressions of disease. Median PFS was 113 days in those 12 cats (range, 5–555 days). Disease progression consisted of new cutaneous occurrence in all cats, whereas none of the cats had local MCT recurrence. One of the 12 cats developed histologically confirmed splenic and hepatic metastases; in the other 5 cats, the occurrence of visceral metastases was suspected by the referring veterinarian. Thirteen cats (54%) were alive at the end of the study, 3 (13%) died because of non-tumor-related causes, and 8 (33%) were reported dead because of tumor-related causes. All of these 8 cats were euthanatized due to disseminated cutaneous lesions, anorexia, and severe depression, although no end-of-life staging tests or necropsies were performed. Median OS in those cats was 284 days (range, 48–616 days).
Samples
Thirty-four nodules were included for subsequent evaluations. In 15 cats (62%) a single nodule was available, 2 concurrent nodules were obtained in 5 cats (21%), and 3 concurrent nodules were obtained in 1 cat (4%), whereas in the remaining 3 cats (13%), 2 nonconcurrent nodules were obtained (excised 7, 10, and 11 months apart, respectively).
Histological Examination
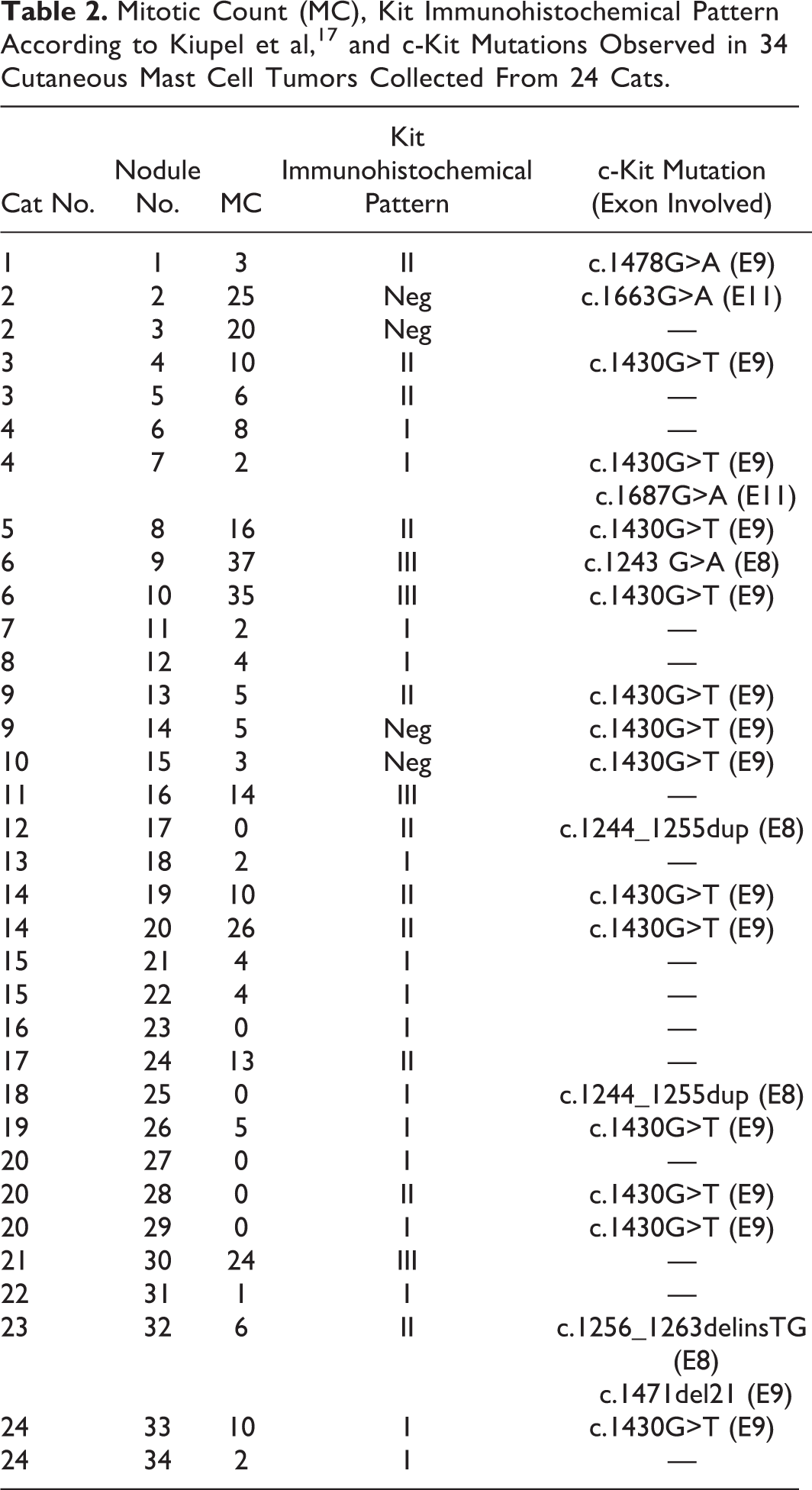
There were 31 of 34 mastocytic well-differentiated MCTs and 3 atypical/poorly granulated MCTs. Median mitotic count estimated in the 34 nodules was 5 (range, 0–37; Table 2).
Mitotic Count (MC), Kit Immunohistochemical Pattern According to Kiupel et al, 17 and c-Kit Mutations Observed in 34 Cutaneous Mast Cell Tumors Collected From 24 Cats
Immunohistochemical Analysis
Kit immunohistochemical expression was evaluated in all 34 nodules, and a positive signal was obtained in 30 (88%). Fifteen tumors (44%) were Kit pattern I, 11 (32%) were Kit pattern II, and 4 (12%) were Kit pattern III (Figs. 2 –4; Table 2).
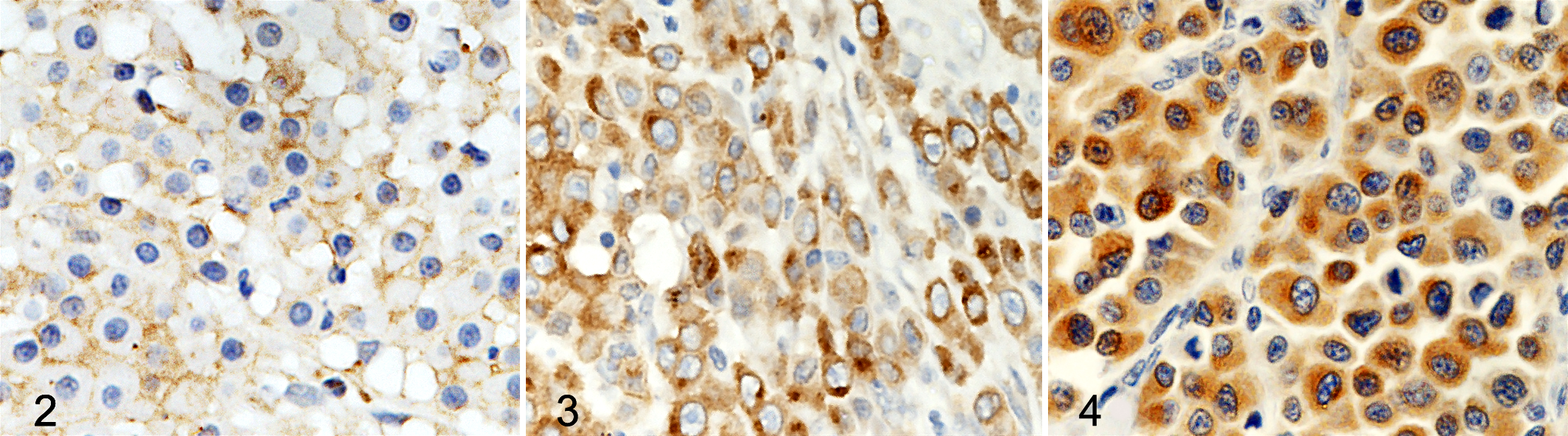
Skin; feline; cutaneous mast cell tumor. Immunolabeling for Kit protein (CD117); hematoxylin counterstain. Kit staining pattern I, consisting of perimembrane protein localization.
Mutation Analysis
Mutations were detected in 19 of 34 tumors (56%) (Table 2). Overall, there were 21 mutations: 4 (19%) were located in exon 8, 15 (71%) in exon 9, and 2 (10%) in exon 11. Two tumors had concomitant mutations, located in exons 9 and 11 and in exons 8 and 9, respectively.
Mutations were of 8 different types, including 5 point mutations, 1 duplication, and 2 delins. A comprehensive scheme of the observed mutations is provided in Fig. 5.
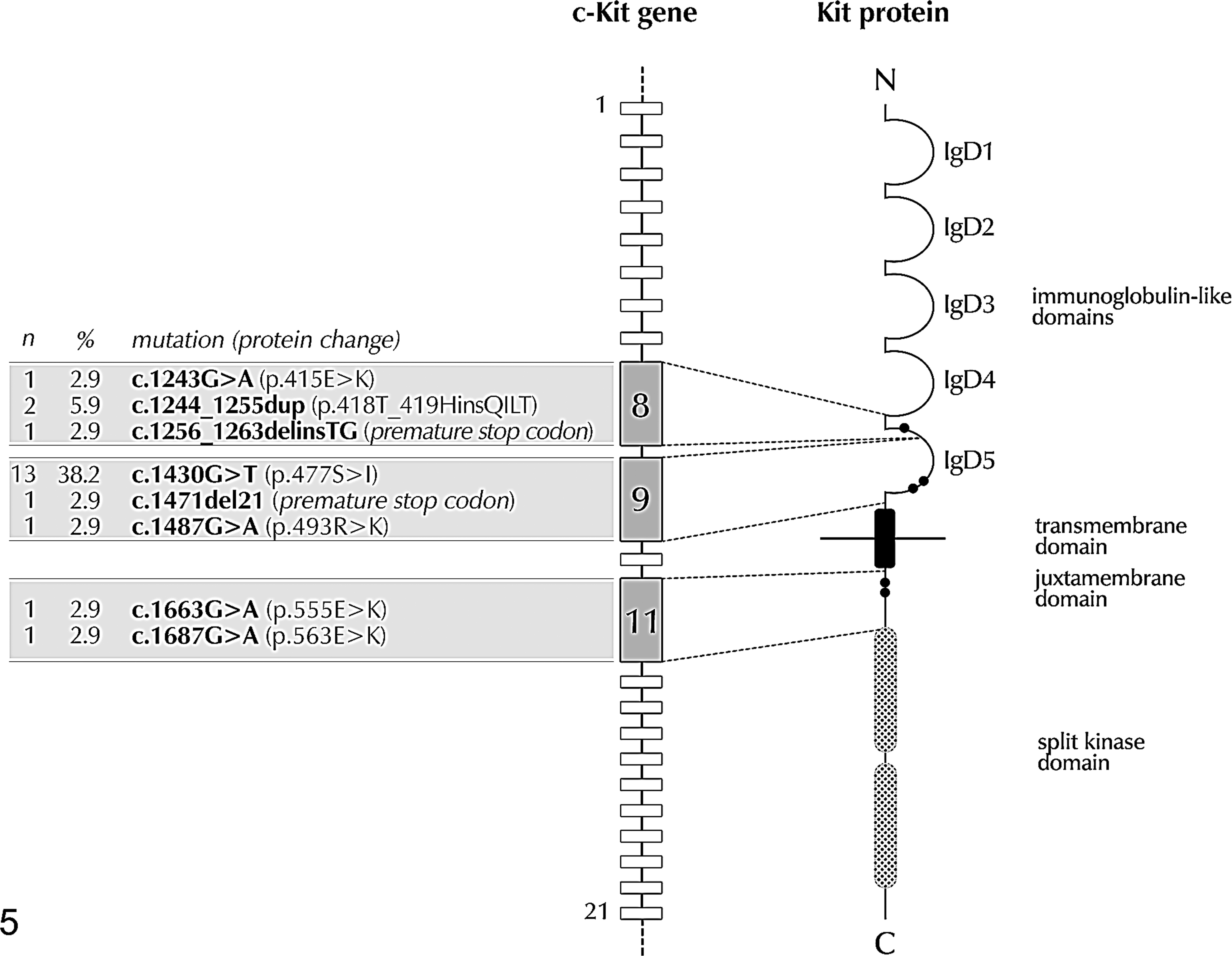
Location and frequency of c-Kit mutations identified in 34 FeCMCTs.
In 6 of the 9 cats (67%) with multiple nodules from the same cat, there were different mutational statuses, including 2 cats with disseminated cutaneous MCT (Table 2).
Statistical Analysis
Mitotic count was significantly higher in the tumors with a cytoplasmic expression of Kit (median, 10; range, 0–37) compared with those with a membranous expression or negative to Kit (median, 3; range, 0–25; Mann Whitney U-test, P = .01).
Eleven of 15 tumors (73%) with cytoplasmic labeling had c-Kit mutations, compared with 5 of 15 (33%) with a membranous expression (Fig. 6); however, this difference did not reach statistical significance (Fisher’s exact test, P = .066), not even considering single types of mutation. Three of 4 (75%) Kit-negative tumors had c-Kit mutations (2 in exon 9 and 1 in exon 11). There were no correlations between tumor histotype and mitotic index, IHC, or mutational status.
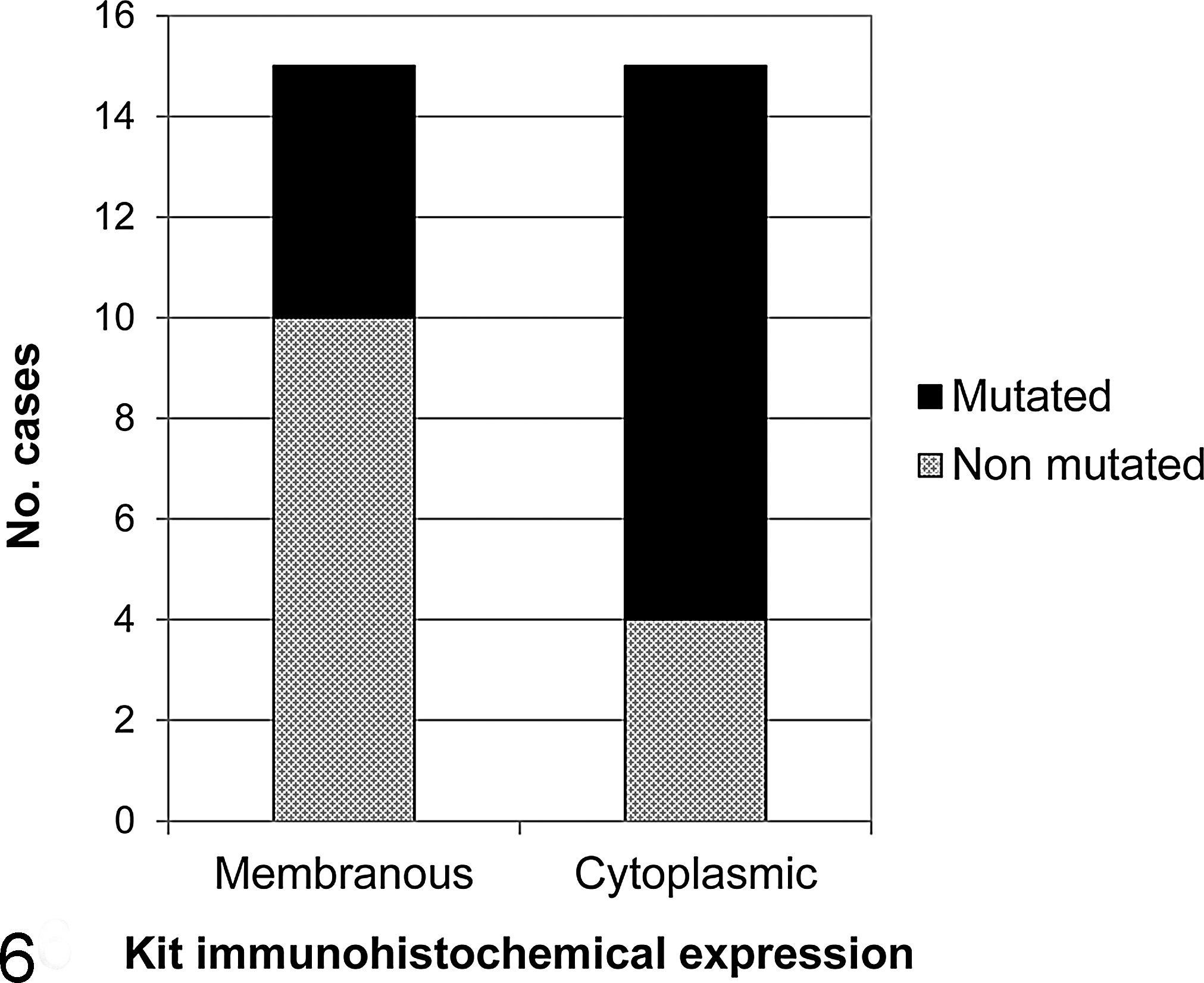
Relationship between c-Kit mutations and Kit protein localization in 34 feline cutaneous mast cell tumors. Eleven of 15 (73%) MCTs with cytoplasmic Kit immunohistochemical expression had c-Kit mutations versus 5 of 15 (33%) with membranous expression. This different distribution was not statistically significant (P = .066).
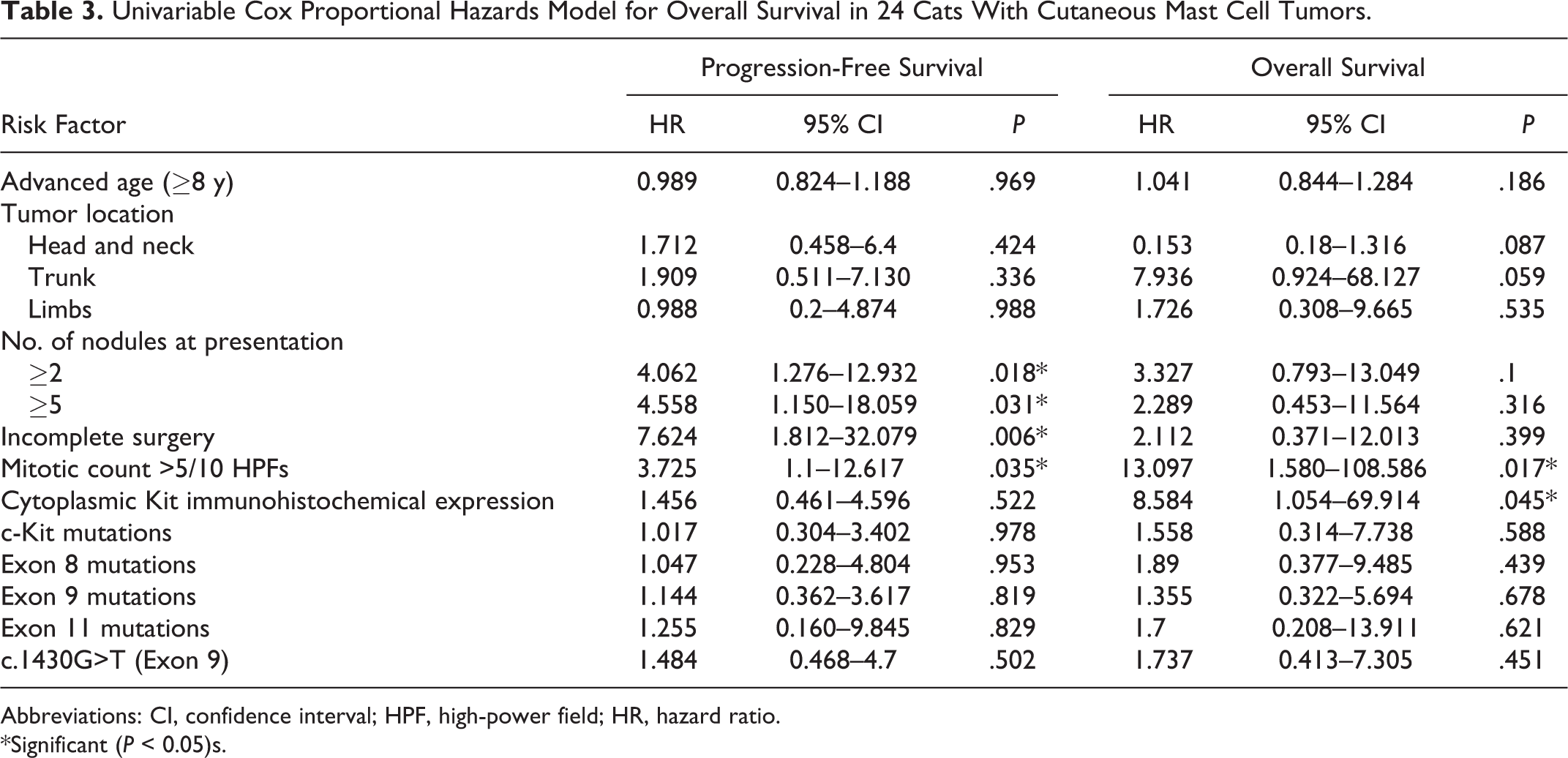
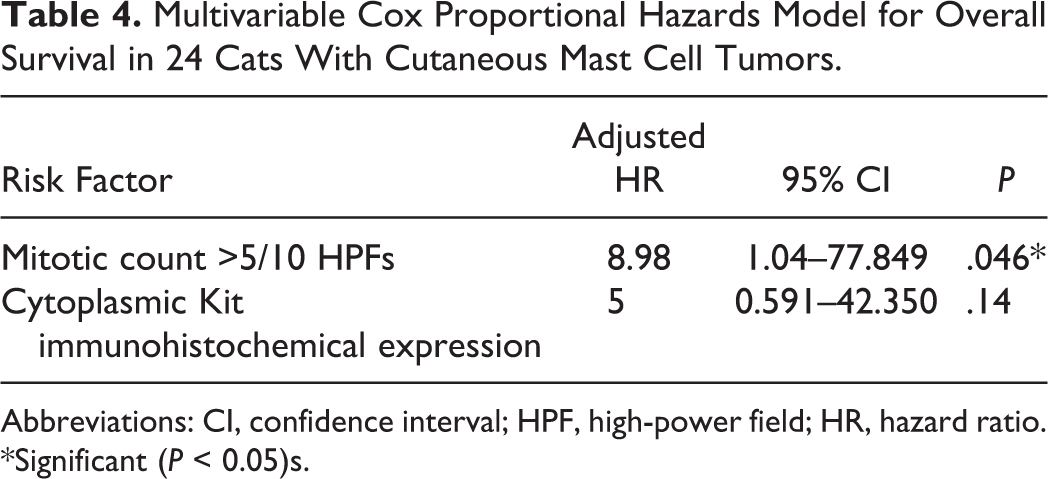
Risk factors significantly affecting PFS were incomplete surgical resection and high MC, whereas risk factors significantly affecting OS were high MC and cytoplasmic Kit immunohistochemical pattern (Table 3). c-Kit mutations were not related to PFS or OS when challenging cats without mutations versus cats with mutated tumors considered altogether or grouped according to exons 8, 9, and 11. The relation between specific mutation and survival outcome was statistically evaluated only for the most frequent mutation c.1430 G>T (exon 9), which resulted in nonsignificance (Table 3). On multivariable analysis, MC turned out to be the best predictor of MCT-related death (hazard ratio [HR], 8.98; confidence interval [CI], 1.04–77.85; P = .046) (Table 4).
Univariable Cox Proportional Hazards Model for Overall Survival in 24 Cats With Cutaneous Mast Cell Tumors
Abbreviations: CI, confidence interval; HPF, high-power field; HR, hazard ratio. *Significant (P < 0.05)s.
Multivariable Cox Proportional Hazards Model for Overall Survival in 24 Cats With Cutaneous Mast Cell Tumors
Abbreviations: CI, confidence interval; HPF, high-power field; HR, hazard ratio. *Significant (P < 0.05)s.
Discussion
The goal of this study was to investigate the nature and incidence of Kit dysregulations at both the gene and protein levels to better define the role played by this receptor in the development and progression of FeCMCTs.
This preliminary analysis was performed on a relatively small number of cases, which may have resulted in loss of statistical power. The retrospective nature of this study is a further limitation, as cases often lacked uniformity in staging and therapy protocols; in particular, the incidence of visceral involvement may have been underestimated due to lack of fine needle aspirates.
Similar to previous surveys, we observed a predominance of middle-aged to older cats, with a minority of young cats (<4 years old) and no sex predilection. This report also confirms the tendency of these tumors to arise more frequently in the head and neck region. However, neither age nor tumor location proved to be prognostically relevant.
Mitotic activity is reportedly the only reliable histological prognostic indicator in FeCMCT. 16,20 Indeed, in this study, MC emerged as the most powerful predictor for OS, and it also significantly affected PFS. Furthermore, a higher mitotic activity was observed in tumors with a cytoplasmic Kit pattern, suggesting that a downstream consequence of aberrant Kit protein localization in feline MCTs may be an increase in cellular proliferation, similarly to canine MCT. 37 The cats with cytoplasmic Kit labeling had an increased risk of MCT-related death (HR, 8.58; CI, 1.0–69.91; P = .045). This is consistent with our previous report in which we observed a tendency for a higher frequency of Kit cytoplasmic expression among the cases of FeCMCT with an unfavorable outcome. 31 The higher number of MCT-related deaths and the use of a proportional-hazards regression model in this study may have helped reach statistical significance.
In the present study, c-Kit mutations occurred in FeCMCTs in 56% of cats, although these mutations did not appear to influence the survival outcome. The vast majority of these mutations were located in exons 8 and 9, which correspond to the fifth immunoglobulin-like domain (IgD5) of the receptor, reaching a predominance of this region of 90%, whereas exon 11 was involved with a frequency of 10%. In people, c-Kit IgD5 mutations are reported in acute myeloid leukemia (26%) and gastrointestinal stromal tumors (18%). 3,12 In comparison with human mast cell neoplasms, the mutational status observed in this study is different from that in adult mastocytosis, in which the prevailing mutation is c.7176A>T (p.816D>V), in exon 17, whereas it is similar to that reported in pediatric mastocytosis, although the prevalence in cats appears to be higher. 8,19 Known Kit mutations occurring in canine MCTs include more frequently internal tandem duplications and small insertions and deletions in the extracellular juxtamembrane region (exons 11 and 12); less commonly, mutations involve the IgD5, including exon 8 (4.7%) and exon 9 (4.2%). 21 In feline MCTs, few studies have been carried out investigating c-Kit mutations. 7,11,14,15,34 Of these, only 1 was performed on a relevant number of cases and nucleotide sequences. 15 In that study, the incidence of mutations was also very high (68%), and IgD5 was similarly involved with a prevalence of 95%, which led us to focus on exons 8, 9, and 11. According to the same study, spontaneous feline MCTs with Kit mutation responded to the TKI imatinib mesylate in 7 of 8 cats. 15
The 3 types of mutations found in exon 8 (c.1243G>A, c.1244_1255dup, c.1256_1263delinsTG), occurring overall with a frequency of 19%, were located in a neighboring residue of mutations that have been reported in patients with pediatric mastocytosis. 13,19 Mutations at this level were considered to affect the affinity of the hemophilic dimerization of Kit, resulting in the constitutive activation of the kinase domain without ligand binding. In particular, the duplication (c.1244_1255dup), consisting of the insertion of 4 amino acids (glutamine, isoleucine, leucine, and threonine) between residues 418 and 419, was the most frequent (45%) mutation observed by Isotani et al 15 in their series of cutaneous and visceral MCTs and had been previously reported in 4 cats with systemic mastocytosis. 11,14 Because this study, which is specifically focused on cutaneous MCT, has demonstrated exon 8 duplication in only 2 cats, we can hypothesize that this mutation is more common in visceral tumors.
The most frequent mutation encountered in this study (c.1430G>T, exon 9) was also previously reported 15,34 and was demonstrated to cause ligand-independent activation of Kit by substituting Ser477 to Ile477. Mutations around this residue are not observed in human tumors; however, identical substitutions were reported on the corresponding codon of Ser479 in canine MCT as an activating mutation. 21 Despite its frequency, this mutation was not associated with any specific behavior.
Exon 11 encodes the intracellular juxtamembrane region, which, in the inactive state, maintains the 2 lobes of the kinase domain in a closed conformation. Thus, mutations in exon 11 probably activate Kit tyrosine phosphorylation by disrupting the structure of the juxtamembrane region, releasing it from the kinase domain. 21,27 The 2 mutations observed in exon 11 (c.1663G>A and c.1687G>A) are located neighboring a hotspot, between residues 555 and 590, which is frequently involved in canine MCTs.
Webster et al 36 identified a significant (P = .046) relationship between the presence of c-Kit mutations and aberrant Kit protein localization in 60 canine MCT, demonstrating the importance of c-Kit mutations in the tumorigenesis of these neoplasms and clearly identifying the c-Kit proto-oncogene as a potential target for therapeutic intervention.
In the present study, performed on 34 samples of FeCMCT, a statistically significant association between c-Kit mutations and Kit pattern was not demonstrated, although a tendency was observed (P = .066). This tendency could be likely confirmed in larger series. Alternatively, the lack of significant correlation could be explained assuming that some of the mutations found are actually “passenger” mutations. Currently, consensus exists for the classification of cancer mutations into the 2 categories of “drivers” and “passengers”: the former are those casually implicated in cancer development which confer cells a growth advantage and hence are positively selected; the latter are those that have been acquired at some point during clonal evolution but do not provide any substantial advantage. 32
According to our data, 5 tumors presenting membranous Kit immunostaining (Kit pattern I) had c-Kit mutations (exon 9, n = 3; exons 8 and 11, n = 1). A possible explanation may be that those tumors acquired the mutation only recently and the changes in Kit localization may not have occurred at the time of surgical excision. Furthermore, 4 cutaneous MCTs in this study had aberrant Kit protein localization with no c-Kit mutations. In those cases, other factors may be responsible for aberrant Kit localization, including other c-Kit mutations or protein trafficking abnormalities. Finally, these findings may also suggest that c-Kit mutations and changes in Kit localization can represent separate events occurring independent of one another in the progression of FeCMCTs. The more complexity of Kit biology in FeCMCTs when compared with the canine counterpart could be also emphasized by the lack of correlation between c-Kit mutations and mitotic activity or prognosis. Due to the pivotal role of Kit receptors in mast cell growth and differentiation, it is unlikely that its constitutive activation could not play a role in cancer progression. Conversely, the coexistence of different subsets of neoplasm characterized by different pathogenesis could be hypothesized. To further clarify the role of Kit in feline MCTs, additional studies must be performed on a larger number of cases and include a mutation analysis of the whole nucleotide sequence of c-Kit. A deeper understanding of the biological effects of c-Kit mutations on the protein structure by in vitro experiments is also warranted.
Multiple cutaneous MCTs, either synchronous or asynchronous, are quite a common finding in cats; a negative prognostic significance of multiple (5 or more) nodules is reported, 22 and although in the present study cats with multiple nodules at diagnosis were not at higher risk of MCT-related death, all the deceased subjects had disseminated cutaneous nodules at the terminal stage. In these cases, determining whether the new MCTs truly arise de novo or whether they are a form of metastasis is a clinical dilemma. In one report, PCR assays were used to support the hypothesis of a clonal origin for multiple distant cutaneous MCTs in 2 dogs. 39 In this study, multiple MCTs from the same cat were characterized by different mutational statuses in 67% of cases, including 2 disseminated forms. Although it seems unlikely that each nodule may represent a separate event, these findings provide further evidence that the role of c-Kit mutations in feline MCTs is more complex than expected and open intriguing questions about the nature and pathogenesis of multiple cutaneous neoplasms. Additionally, the fact that in cats with multiple MCTs every nodule may potentially harbor a different mutation is a limit for the assessment of their prognostic relevance and of their response to kinase inhibitors.
In summary, the frequent occurrence of mutations in the IgD5 of Kit observed in this study provides further evidence of the strong instability of the c-Kit gene in FeCMCT, although these mutations do not appear to be strictly correlated with biological behavior. Additionally, multiple nodules from the same cat do not necessarily show identical mutational statuses, suggesting that Kit mutations may not be critically involved in tumor development. Conversely, aberrant Kit protein localization was significantly correlated with both mitotic activity and survival, and it was not regularly associated with c-Kit mutations. These findings need to be confirmed in a larger series, and exploration of further genomic regions of the c-Kit proto-oncogene is warranted to define its role in feline MCT and ultimately assess the utility of molecular targeted therapies in the treatment of these neoplasm.
Footnotes
Authors’ Note
This study was presented in a preliminary form at the annual congress of the European Society of Veterinary Pathology (September 2011, Uppsala, Sweden) and at the 2nd World Veterinary Cancer Congress (March 2012, Paris, France).
Acknowledgements
A special thanks to Drs Fabio Barbieri, Guido Pisani, Concetta Silvestre, and all the other veterinary clinicians who provided clinical histories for the cases in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
