Abstract
Feline cutaneous mast cell tumors (MCTs) have been histologically classified as mastocytic (well differentiated or pleomorphic) and atypical/poorly granulated. Their biologic behavior ranges from benign to malignant, but prognostic factors are not well defined. Histologic classification, number of tumors, mitotic index, cytoplasmic granularity, and infiltration by eosinophils or lymphocytes were evaluated retrospectively in 25 feline cutaneous MCTs. Immunohistochemistry was applied to assess KIT (CD117) pattern and immunoreactivity score, telomerase expression (human telomerase reverse transcriptase), and proliferation index (MIB-1/Ki67 index). Case outcome was obtained via telephone interviews. The tumors comprised 15 mastocytic well-differentiated, 7 mastocytic pleomorphic, and 3 atypical/poorly granulated MCTs. Immunohistochemically, CD117 was expressed in 13 of 25 tumors (52%), and telomerase reverse transcriptase was expressed in 15 of 22 (68%), with no correlation to histologic classification. Mitotic index, KIT immunoreactivity score, and Ki67 index were significantly higher in mastocytic pleomorphic MCTs than in the other 2 categories. Five cats (20%) died of tumor-related causes. Multiplicity of lesions, pleomorphic phenotype, KIT immunoreactivity score, and mitotic and Ki67-indices correlated with an unfavorable outcome. Mitotic index was the strongest predictive variable. These results suggest that histologic classification, CD117/KIT immunohistochemistry, and proliferation indices may help to identify potentially aggressive cases of feline cutaneous MCT. Aberrant KIT protein localization and telomerase immunoreactivity warrant further exploration as potential prognostic markers.
Mast cell tumors (MCTs) are among the most common skin tumors of cats, accounting for up to 21% of all feline cutaneous neoplasms. 22,25 The tumors most commonly arise on the head and neck, followed by the trunk and limbs, 3,18,22,25 although 1 study reported no site predilection. 36 Multiple tumors develop in 12% to 20% of cases 2,10,26,29 and may be clustered or disseminated. Larger tumors are often ulcerated. 10,11,34
Feline cutaneous MCTs are classified histologically as the more common mastocytic and less common atypical MCT. 36 The mastocytic type can be subdivided into well-differentiated and pleomorphic MCTs. 11 Well-differentiated MCTs are variably demarcated but unencapsulated dermal masses that may invade the subcutis. These tumors are composed of sheets of cells that resemble normal mast cells with negligible pleomorphism; mitotic figures are rare. Infiltrating eosinophils are few, but aggregates of lymphocytes may be observed. 10 –12 Pleomorphic MCTs are uncommon, tend to infiltrate more deeply into the dermis and subcutis, 11 and are composed of large cells with eccentric nuclei, prominent nucleoli, and variable presence of giant cells with a multilobulated nucleus or multiple nuclei. Some authors reported high mitotic activity in pleomorphic MCTs, 10,11 whereas the mitotic index (MI) was variable but usually low in another study. 15 Pleomorphic MCTs are often more heavily infiltrated by eosinophils. 10 –12 The atypical feline cutaneous MCT is rare and reported mainly in juvenile to middle-aged Siamese cats, in which it may spontaneously regress. 5,36 Atypical MCTs have also been called “poorly granulated” or “histiocytic,” 36 although the latter term has been discouraged to avoid confusion with true histiocytic proliferations. 11 In atypical/poorly granulated MCTs, neoplastic cells are large and polygonal to spindle shaped with abundant amphophilic cytoplasm. The nuclei are large and vesicular and may be slightly indented; mitotic figures are infrequent. Admixed eosinophils and lymphoid aggregates tend to be more numerous than in mastocytic MCTs. 10 –12 Cytoplasmic granularity may not be obvious in pleomorphic or atypical MCTs with HE stain, but granules are almost always present, at least in a few cells, in sections stained with Giemsa or toluidine blue. 10,11
Most feline cutaneous MCTs are considered benign, despite their tendency to recur locally or develop at additional cutaneous sites. 3,11 The rate of visceral involvement is usually low, but variable (0 to 22%). 3,4,18,34,36 In most reports, only a small proportion of tumors (3 of 42, 36 4 of 14, 3 0 of 32, 26 1 of 14, 15 2 of 30, 18 6 of 30 19 ) behaved aggressively or led to death or euthanasia of the cat. Disseminated nodules may be a component of generalized (cutaneous and visceral) MCT and have been associated with a worse prognosis. 19,29 However, cats with multiple cutaneous MCTs do not always develop generalized disease, and cats with solitary tumors may subsequently develop multiple cutaneous tumors without evidence of visceral involvement. 29
The morphologic features typically associated with malignancy, such as pleomorphism and infiltrative growth, have not correlated with clinical behavior in feline cutaneous MCTs, 3,11,15,26 with the exception of mitotic/proliferative activity. 1,15,18 The histologic grading system proposed by Patnaik et al in 1984 30 for canine MCT is not considered prognostically useful in cats. 3,26
In dogs, MCTs with aberrant cytoplasmic localization of the KIT receptor tyrosine kinase had a worse prognosis than that of MCTs with a membranous pattern of KIT localization. 17,35 Cytoplasmic expression of KIT has been demonstrated immunohistochemically in feline cutaneous MCTs, 27,33 but its relationship with clinical behavior of the tumor has not been investigated.
Upregulation of telomerase, a mechanism of immortalization of cancer cells, has been investigated in various canine and feline tumors. 23,28 Human mast cell precursors have transient induction of telomerase activity, 6 but telomerase expression in feline MCTs has not been assessed.
The identification of the few feline cutaneous MCTs with the potential for aggressive behavior remains a challenge. These considerations prompted a retrospective analysis of histologic and immunohistochemical features in feline cutaneous MCTs. All parameters were assessed in light of the clinical or gross appearance of the tumors and the outcome of the case to estimate their prognostic value.
Materials and Methods
Case Inclusion Criteria and Clinical Information
Surgical biopsy specimens with a diagnosis of feline cutaneous MCT from 2002 to 2007 were retrieved from the archives of the Diagnostic Pathology Service. All samples had been fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 4 μm, and stained with HE and toluidine blue. For cases with multiple excised MCTs, the largest specimen was chosen for subsequent analyses. Cases with concomitant visceral involvement at diagnosis were excluded.
Signalment, anatomic location, and multiplicity of tumors were recorded from submission forms. The following clinical information was requested by telephone from owners or veterinarians: tumor size, interval between initial observation and surgical excision, ancillary treatment, recurrence or metastasis, current health status, and survival. Cases with inadequate historical, clinical, or follow-up information were excluded. The minimal follow-up period was 2 years from diagnosis.
Histopathology
Each tumor was classified histologically as mastocytic well differentiated, mastocytic pleomorphic, or atypical/poorly granulated. 11,12,36 Additional histologic parameters—including MI, cytoplasmic granularity, and infiltration by eosinophils or lymphocytes—were assessed without knowledge of the clinical outcome. MI was calculated by counting the number of mitotic figures in 10 high-power fields (HPFs; 400×). Cytoplasmic granularity in toluidine blue–stained sections was scored as 1 (low density in most neoplastic cells), 2 (moderate density in most cells or high density in less than half the cells), or 3 (high density in most cells). The number of eosinophils was scored in sections stained with eosin (1:10,000 for 24 hours) as 0 (absent), 1 (few), 2 (moderate), or 3 (numerous). Lymphocytic infiltration was scored in HE (hematoxylin and eosin)-stained sections as 0 (absent), 1 (low), 2 (moderate), or 3 (high). The growth pattern (compact versus diffuse) 13 was not evaluated, because of inadequate tissue in the biopsy specimens.
Immunohistochemistry
Immunohistochemistry for CD117 (rabbit polyclonal antibody, 1:100, Dako, Glostrup, Denmark), human telomerase reverse transcriptase (hTERT; mouse monoclonal antibody, 1:50, Novocastra, Newcastle Upon Tyne, UK), and Ki67 (MIB-1 mouse monoclonal antibody, 1:30, Dako) was applied to assess KIT expression, telomerase activity, and proliferation index (PI), respectively. Immunohistochemistry was performed with a streptavidin–biotin–peroxidase technique. Endogenous peroxidase activity was blocked by incubation for 30 minutes with 3% hydrogen peroxide in distilled water (CD117) or 0.3% hydrogen peroxide in methanol (hTERT and MIB-1). Slides were then microwaved in citrate buffer (pH 6.0) for 2 cycles (CD117 and hTERT) or 4 cycles (MIB-1) of 5 minutes at 750 W for antigen retrieval. The sections were incubated overnight at 4°C in a humid chamber with the primary antibody diluted in phosphate-buffered saline (PBS; pH 7.4, 0.01M). Following washing in PBS, sections were incubated with secondary biotinylated anti-rabbit, anti-mouse, or anti-goat immunoglobulin G (LSAB, Dako) for 30 minutes at room temperature and then with the streptavidin–peroxidase complex for 25 minutes at room temperature. After incubation in DAB (diaminobenzidine 0.02% and H2O2 0.001% in PBS) for 12 minutes, sections were immediately rinsed in PBS and in running tap water, counterstained with hematoxylin, dehydrated, and mounted under DPX (Fluka, Riedel-de Haën, Germany). Appropriate positive controls were used to assess the specificity of the reactions. As negative control, an isotype-matched antibody of irrelevant specificity (NeoMarkers, Fremont, CA) was used in place of the primary antibody.
KIT protein localization was assessed in CD117-stained sections, as predominantly membrane associated or cytoplasmic (focal or diffuse). 9 Additionally, CD117 immunoreactivity was scored (KIT immunoreactivity score [KIT IS]) as the product of the percentage of positive cells (1, ≤ 10%; 2, 11 to 30%; 3, 31 to 60%; or 4, > 60%) and the intensity of labeling (1, low; 2, moderate; 3, high).
Telomerase activity was indirectly measured by determining immunoreactivity for hTERT. The MIB-1 antibody was used to determine the PI; it recognizes the Ki67 antigen, a nuclear protein expressed in all active phases of the cell cycle (gap 1, synthesis, gap 2, mitosis). The labeling indices for hTERT and MIB-1 were calculated in five 400× fields with a semiautomatic image analysis system (Lucia G, Nikon, Kingston Upon Thames, UK) and expressed as a percentage of positive neoplastic cells—respectively, telomerase reverse transcriptase (TERT) index and Ki67 index (or PI).
Statistical Analysis
The differences in parameters among histologic types of MCT and clinical outcomes were tested for significance with the Fisher exact test and the Kruskal–Wallis test for nonparametric analysis of variance. The Spearman rank test was used to correlate the MI, Ki67 index, KIT IS, and TERT index. Significance was set at P < .05. Tests were executed with STATISTICA analysis software (StatSoft, Inc, Tulsa, OK).
Results
Signalment and Gross Appearance
In total, 17 excisional and 8 incisional biopsies of feline MCT were selected. Sex, age, and breed were recorded in all cases (Table 1 ). There were 23 domestic shorthairs, 1 domestic longhair, and 1 Siamese cat; 36% were males (n = 9; all castrated) and 64% were females (n = 16; 11 spayed). Mean age was 9 years (range, 1 to 16).
Signalment and Clinical Course of 25 Cats With Cutaneous Mast Cell Tumor
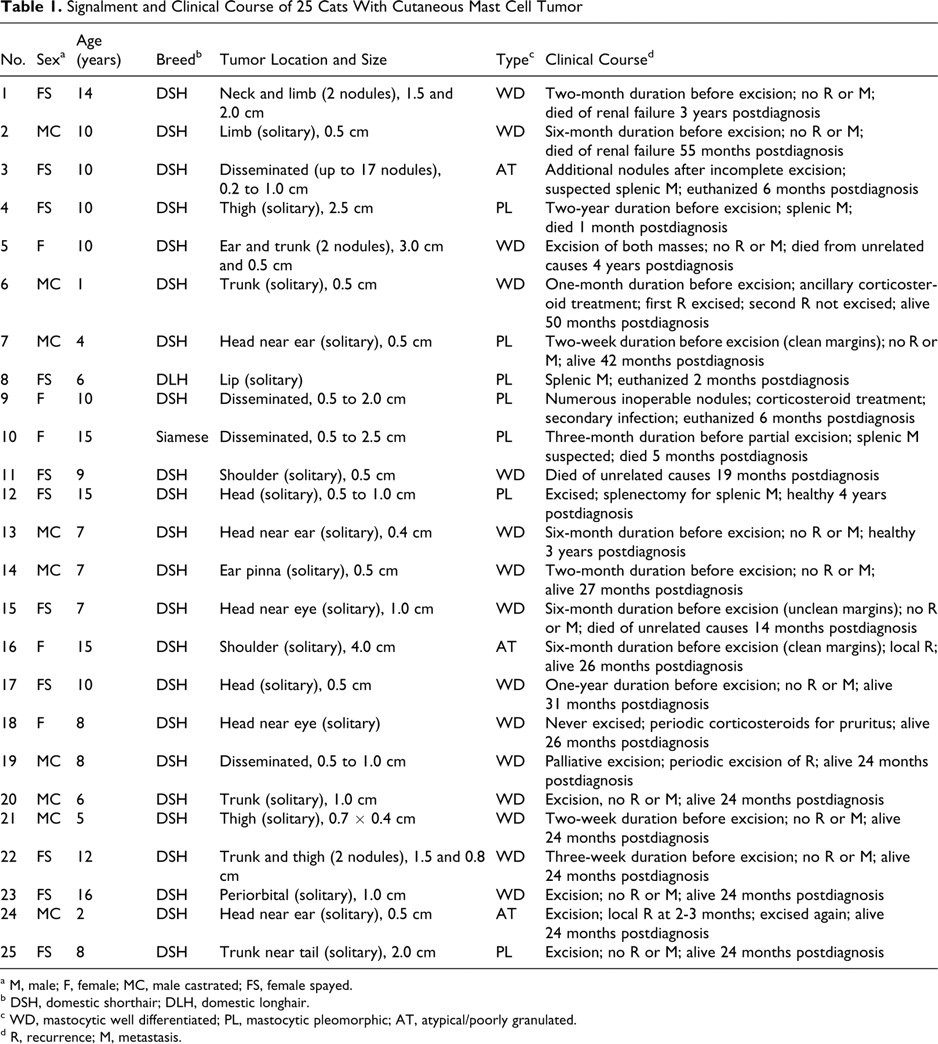
a M, male; F, female; MC, male castrated; FS, female spayed.
b DSH, domestic shorthair; DLH, domestic longhair.
c WD, mastocytic well differentiated; PL, mastocytic pleomorphic; AT, atypical/poorly granulated.
d R, recurrence; M, metastasis.
Twenty-one MCTs were solitary nodules. Among those, 18 were single presentation: 10 on the head and neck, 3 on the trunk, and 5 on the limbs. In 3 cases (Nos. 1, 5, 22), 2 regions were involved at presentation, with a single nodule in each location (neck and limb, head and trunk, trunk and limb). In the remaining 4 cases (Nos. 3, 9, 10, 19), disseminated nodules (n = 6 to 17) were scattered over the body. Tumor diameter was recorded in 23 cases and ranged from 0.2 to 4.0 cm.
Histopathology
Fifteen MCTs were classified as mastocytic well differentiated (Table 2 ). These tumors consisted of sheets and cords of uniform round cells with discrete cytoplasmic margins. Nuclei were round and central with stippled chromatin (Fig. 1A, 1B). Nuclear pleomorphism was mild with mostly inconspicuous nucleoli. Mitotic figures were rare (mean MI, 2; range, 0 to 5).
Histologic Findings in 25 Feline Cutaneous Mast Cell Tumors by Type, n (%)
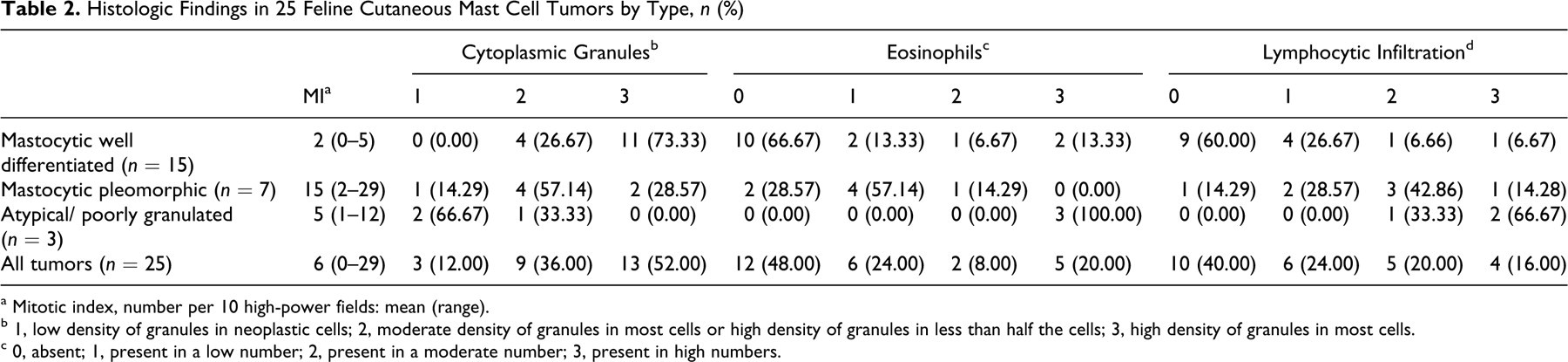
a Mitotic index, number per 10 high-power fields: mean (range).
b 1, low density of granules in neoplastic cells; 2, moderate density of granules in most cells or high density of granules in less than half the cells; 3, high density of granules in most cells.
c 0, absent; 1, present in a low number; 2, present in a moderate number; 3, present in high numbers.
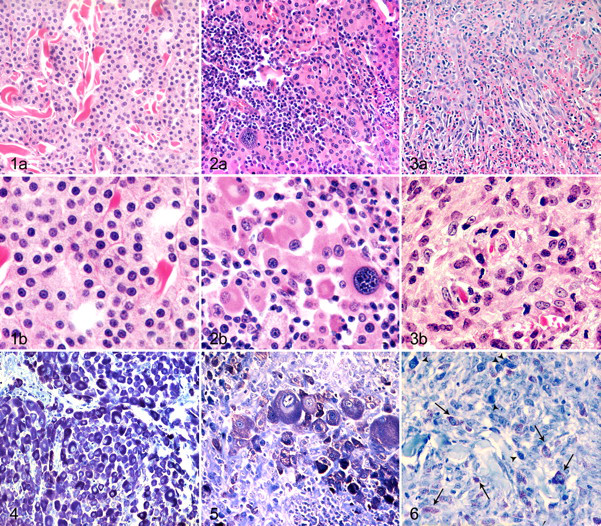
Seven MCTs were classified as mastocytic pleomorphic and had nuclear atypia with scattered multinucleated cells and prominent nucleoli (Fig. 2A, 2B). Mean MI was 15 (2, 6, 8, 16, 18, 28, 29).
Three MCTs were classified as atypical/poorly granulated. They comprised solid sheets of large polygonal to spindle-shaped cells with large vesicular nuclei, occasionally slightly indented (Fig. 3A, 3B). MI varied (1, 3, 12). Pleomorphic MCTs had significantly higher MI than that of well-differentiated or atypical MCTs (P = .0061).
Well-differentiated MCTs had a high (11 of 15; 73.33%) to moderate (4 of 15; 26.67%) density of metachromatic granules. Pleomorphic MCTs were highly granulated in 2 of 7 cases (28.57%), moderately granulated in 4 cases (57.14%), and poorly granulated in 1 case (14.29%). Atypical MCTs had either a moderate (1 of 3; 33.33%) or low (2 of 3; 66.67%) density of granules. The difference in the density of cytoplasmic granules among the 3 histologic types was significant (P = .0071). Even when strongly stained by toluidine blue, granules generally had low metachromasia (Figs. 4–6).
Infiltrating eosinophils were absent in 10 of 15 well-differentiated MCTs (66.67%) and were present in the remaining cases in low (n = 2; 13.33%), moderate (n = 1; 6.67%), and high (n = 2; 13.33%) numbers. Pleomorphic MCTs had no eosinophils in 2 of 7 cases (28.57%), a low number in 4 cases (57.14%), and a moderate number in 1 case (14.29%). All 3 atypical MCTs had numerous eosinophils, significantly more than in well-differentiated or pleomorphic MCTs (P = .0071) (Fig. 3A).
Nodular aggregates of small lymphocytes were detected in 6 of 15 well-differentiated MCTs (40.00%), in 6 of 7 pleomorphic MCTs (85.71%), and in all atypical MCTs (P = .0084) (Fig. 2).
Immunohistochemistry
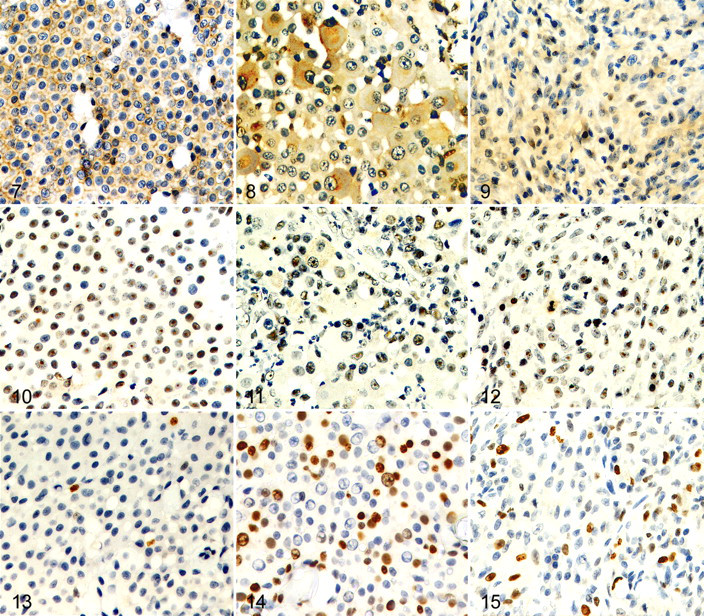
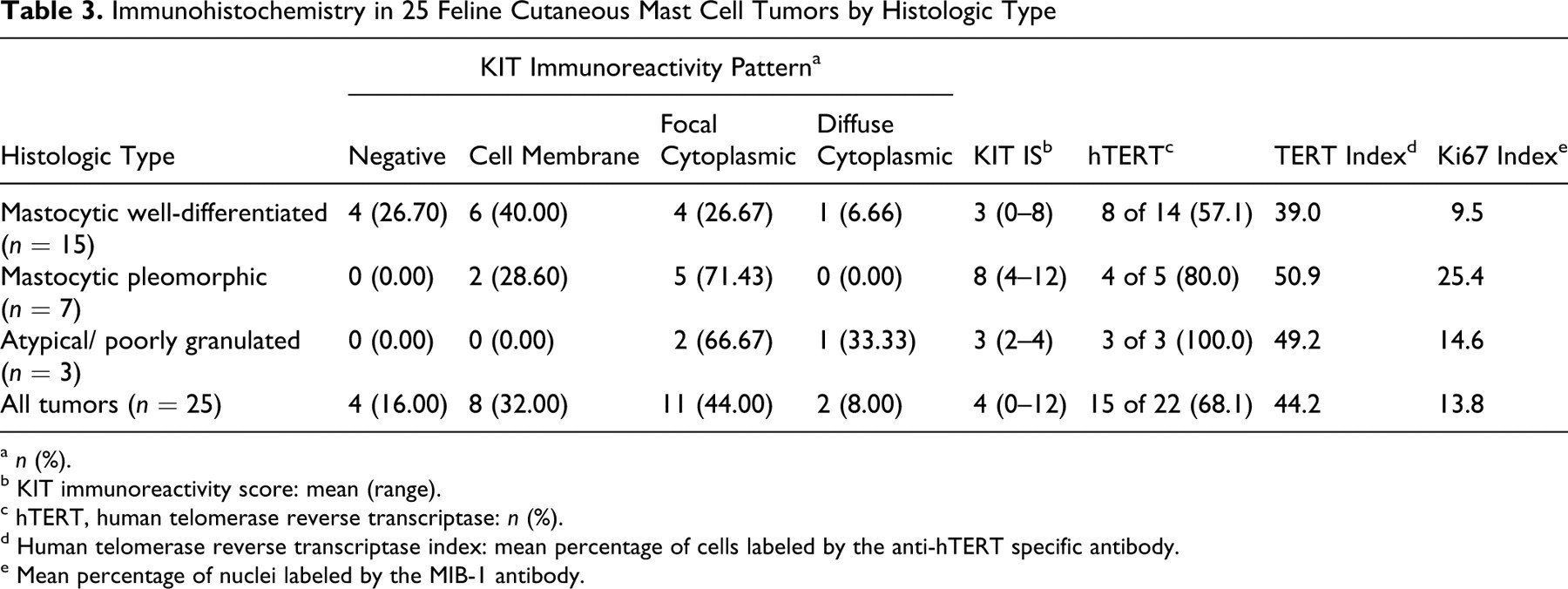
The assessment of KIT protein was performed in all cases (Table 3 ). Neoplastic mast cells were labeled by the CD117-specific antibody in 21 of 25 cases (84%). Eight had mainly membranous pattern (32%); 11 had strong, focal to stippled, mainly paranuclear cytoplasmic pattern (44%); and 2 had faint to moderate diffuse cytoplasmic pattern (8%) (Figs. 7–9). There were no significant differences in KIT expression pattern among the histologic types. However, KIT IS was significantly higher in pleomorphic MCTs (mean IS, 8) than in well-differentiated or atypical MCTs (mean IS, 3; P = .0014).
Immunohistochemistry in 25 Feline Cutaneous Mast Cell Tumors by Histologic Type
a n (%).
b KIT immunoreactivity score: mean (range).
c hTERT, human telomerase reverse transcriptase: n (%).
d Human telomerase reverse transcriptase index: mean percentage of cells labeled by the anti-hTERT specific antibody.
e Mean percentage of nuclei labeled by the MIB-1 antibody.
Immunohistochemistry for hTERT was performed in only 22 of 25 MCTs because case Nos. 8, 11, and 12 had insufficient tissue. Telomerase activity was demonstrated in 15 of 22 cases (68.18%); the TERT index in positive cases ranged from 10.00 to 87.42% (mean, 44.21%); labeling was nucleolar or nuclear with nucleolar prominence (Figs. 10–12). Neither the TERT index nor the percentage of MCTs that expressed hTERT correlated with the histologic type.
Cell proliferation activity was assessed with the MIB-1 antibody in 22 cases. Labeled mast cells ranged from 2.00 to 31.08% (mean PI = 13.77%); PIs were significantly higher in pleomorphic MCTs (mean PI = 25%) than in well-differentiated (mean PI = 9%) or atypical (mean PI = 15%) MCTs (P = .0025) (Figs. 13–15). Strong correlations were found between PI and MI (R = .8348; P < .0001), PI and KIT IS (R = .6688; P = .0007), and MI and KIT IS (R = .646; P = .0005). Additionally, PI correlated with the TERT index (R = .4419; P = .0395).
Clinical Course
Information on patient management and clinical outcome was available for all cats (Table 1). In 20 cats (80%), the tumors had been excised with wide margins; 3 cats with disseminated cutaneous MCT had only the largest masses removed (Nos. 3, 10, 19); in 2 cats, the tumors were not excised (Nos. 9, 18). Local recurrence and visceral involvement (splenic in both cases) were histologically confirmed in 4 cats (16%; Nos. 6, 16, 19, 24) and 2 cats (8%; Nos. 8, 12), respectively. In 2 other cats (Nos. 3, 10), visceral metastasis was suspected clinically. In the 4 cats with disseminated cutaneous MCTs (Nos. 3, 9, 10, 19), new nodules continued to develop. Five cats had died or were euthanized because of the tumor (20%; Nos. 3, 4, 8–10); 2 cats died from unrelated causes during the 2-year follow-up period (Nos. 11, 15). Eighteen cats (72%) were alive at the end of the study. Of the 5 cats that died of tumor-related causes, 3 (Nos. 3, 9, 10) had disseminated cutaneous MCTs with recurrent nodules, of which 2 had suspected visceral involvement (Nos. 3, 10); the remaining 2 had solitary cutaneous MCTs but died shortly after surgical excision with visceral involvement (Nos. 4, 8). Mean survival time in those 5 cats was 4 months (range, 1 to 6).
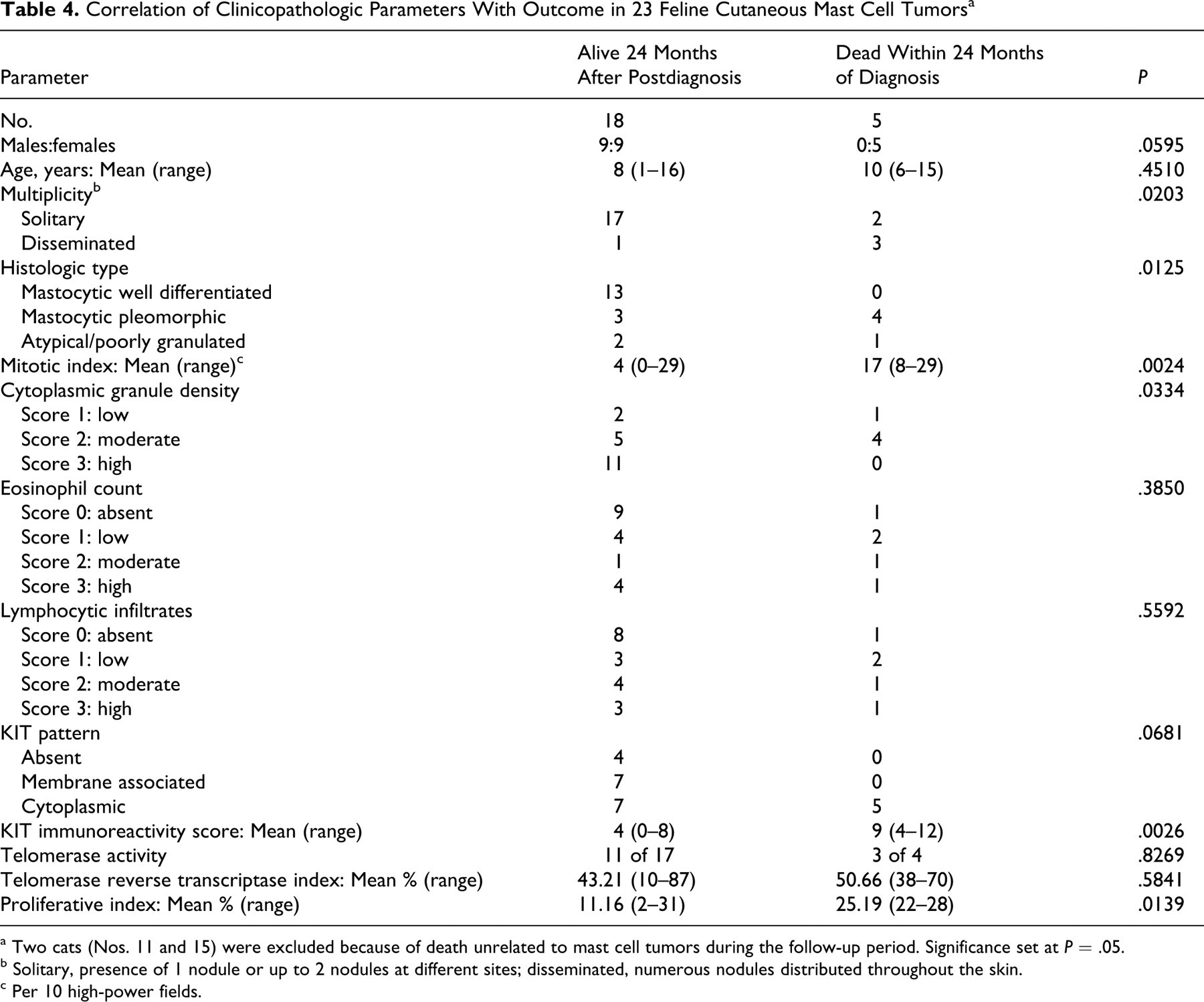
The parameters of live cats (n = 18) and dead cats (n = 5) are compared in Table 4 based on the 2-year follow-up period. Significant differences were evident for multiplicity of lesions (P = .0203), histologic type (P = .0125), MI (P = .0024), density of cytoplasmic granules (P = .0334), KIT IS (P = .0026), and PI (P = .0139). Of the 5 cats that died during the follow-up period, 4 had pleomorphic MCTs and 1 had an atypical MCT. Those 5 cases had higher MIs (mean, 17) and PIs (mean, 25.19%) and low to moderate density of cytoplasmic granules. KIT labeling was cytoplasmic with focal clustering in the 4 pleomorphic MCTs and faint and diffuse in the atypical MCT.
Correlation of Clinicopathologic Parameters With Outcome in 23 Feline Cutaneous Mast Cell Tumors a
a Two cats (Nos. 11 and 15) were excluded because of death unrelated to mast cell tumors during the follow-up period. Significance set at P = .05.
b Solitary, presence of 1 nodule or up to 2 nodules at different sites; disseminated, numerous nodules distributed throughout the skin.
c Per 10 high-power fields.
Discussion
Histologic and immunohistochemical features of feline cutaneous MCTs were evaluated to detect features that correlated with case outcome. The 20% of MCTs with malignant behavior in this series was similar to that observed in previous studies. 3,4,15,18,19,26,34,36 In contrast to studies in which male cats had higher prevalence of MCTs, 3,22 the male:female ratio in this study favored females, but sex was not correlated with outcome. The reported tendency of young Siamese cats to develop benign atypical cutaneous MCTs 36 was not evident; the only Siamese cat in this study was 15 years old and died with a disseminated mastocytic pleomorphic MCT. Our results confirm the predilection of feline cutaneous MCT for the head and neck region. 3,18,22,25 In 1 study, 19 cats with solitary MCT survived longer than those with multiple nodules (5 or more). Our findings support that result because 75% of cats with disseminated MCTs had an unfavorable outcome, compared to 10% of those with solitary MCTs.
As expected, most MCTs in this series were mastocytic well differentiated. Loss of differentiation and pleomorphism are recognized as prognostically useful features in canine MCTs 30 but not in cats. 3,26 In fact, in one study the outcome of feline pleomorphic MCTs was generally good. 15 In contrast, in the present study pleomorphic MCTs had higher proliferative activity and a less favorable outcome, whereas no cat with well-differentiated MCT died from the tumor. In addition, of the 3 atypical MCTs, which are generally considered benign and characterized by low mitotic activity, 5,11,36 1 had high proliferative activity, and the cat was euthanized 6 months after diagnosis because of suspected splenic involvement. Caution is therefore recommended in the management of any feline cutaneous MCT that is not well differentiated.
Loss of differentiation by neoplastic mast cells was generally accompanied by a reduced density of cytoplasmic granules, which correlated with unfavorable prognosis. The tendency of the granules in feline cutaneous MCTs to stain orthochromatically, rather than metachromatically, has been interpreted as a sign of anaplasia. 11,32,36
Eosinophils were uncommon in feline mastocytic MCTs but more numerous in atypical MCTs. Although this did not correlate with clinical outcome, it may have diagnostic value, especially when coupled with histochemical demonstration of cytoplasmic granules, in distinguishing atypical MCTs from histiocytic disorders or poorly differentiated sarcomas.
Lymphocytic infiltrates were more frequent in pleomorphic and atypical MCTs; the significance of their presence in feline MCTs is unknown. 3,13 Similar aggregates are seen in other feline disorders, such as the eosinophilic granuloma complex and injection-site sarcomas, and are prominent in the involutional phase of canine cutaneous histiocytoma and transmissible venereal tumor; as such, their presence could reflect an immunologic response. However, the lymphocytic infiltration did not correlate with outcome in this study.
MI is reportedly the best prognostic indicator in feline cutaneous MCT, 11,15,18 and it had the highest correlation with outcome in this study. Proliferation was also assessed by the Ki67 index. Both parameters were significantly higher in pleomorphic MCTs and in cases with unfavorable outcome. Furthermore, correlation between these 2 parameters was strong, suggesting that the same information may be obtained by determining the less expensive MI.
C-kit proto-oncogene product (KIT, CD117) is a receptor tyrosine kinase for stem cell factor (mast cell growth factor) produced by a number of cells, including hematopoietic stem cells, mast cells, germ cells, melanocytes, and interstitial cells of Cajal. Interactions between stem cell factor and KIT promote hematopoietic progenitors to develop into mast cells, which express the receptor within the cytoplasmic membrane. 24,27 Mutations in the c-kit proto-oncogene have been identified in 15 to 50% of canine MCTs and are probably involved in oncogenesis by inducing the constitutive activation of KIT in absence of ligand binding. 7,20,38 C-kit mutations in dogs are mostly associated with aberrant cytoplasmic KIT expression by neoplastic mast cells, 35,39 and dogs with tumors that have these mutations or KIT overexpression have a worse prognosis and are considered good candidates for treatment with kinase inhibitors. 9,17,21,31,35,39 Feline MCT is suspected to have similar carcinogenetic mechanisms, and c-kit mutation in the extracellular ligand-binding domain (exon 8) has been recently reported in a cat, which responded to treatment with the tyrosine kinase inhibitor imatinib mesylate. 14
KIT immunopositivity has been demonstrated in feline MCTs; 27,33 in one report, cytoplasmic expression of KIT was detected in 18 of 27 cutaneous MCTs (67%), without correlation with the histotype. 33 However, the authors did not correlate KIT expression with tumor proliferative activity or outcome. In the present study, 52% of tumors had aberrant (cytoplasmic) KIT expression without correlation with the histotype. KIT labeling was scored by combining the intensity of immunoreactivity and the fraction of positive cells (KIT IS) and was correlated with mitotic and PIs—and with unfavorable outcome. KIT expression merits further study as a prognostic indicator in feline MCTs and as a target for therapy with tyrosine kinase inhibitors.
Telomerase is a ribonucleoprotein complex that prevents the erosion of telomeres. Maintenance of telomere length by telomerase is a carcinogenetic mechanism for many neoplasms. 8,16,23,28,37 Mast cell precursors undergo transient growth factor–mediated induction of telomerase activity during differentiation. 6 Human MCT cell lines tend to maintain a persistently high telomerase activity, uninfluenced by protein kinase or cell cycle inhibitors. 6
In this study, telomerase activity was indirectly assessed by immunohistochemical detection of hTERT. TERT expression was observed in 68% of tested MCTs, suggesting that telomerase may play a role in the progression of these tumors. TERT expression was not correlated with KIT expression or with case outcome, and it was only weakly correlated with PI, supporting the hypothesis that telomerase activation may be independent of proliferation signals. 6
In conclusion, results of this study emphasize the prognostic value of MI in feline cutaneous MCTs. Future prospective studies on a larger population should evaluate whether multiplicity of tumors, histologic type, KIT immunoreactivity score, or tumor PIs are helpful in identifying which cases of feline cutaneous MCT are more likely to have aggressive behavior and therefore require closer monitoring. Aberrant KIT expression and telomerase activity in a significant proportion of MCTs warrant further exploration of these pathways as potential therapeutic targets.
Footnotes
Acknowledgments
We would like to thank Dr. Michele Sampaolo for collaboration and all veterinary clinicians who provided clinical information for the cats in this study.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
