Abstract
Despite decreasing prevalence, rotavirus infections still rank among the most important viral infections in colonies of laboratory mice. Although the disease is characterized by low mortality and a relatively short and mild clinical period, the infection has the potential to alter the outcome of experiments substantially. For animal facilities, it is therefore essential to eradicate the virus. Here we report a successful sanitation of a rotavirus-infected mouse colony in an animal facility. Despite a high ratio of transgenic and partially immunodeficient strains, a permanent eradication of the virus was achieved by euthanasia of highly susceptible mice, a prolonged breeding cessation in areas containing immunocompromised mice and a strict hygienic management. The management of a rotavirus infection reported here is a feasible and inexpensive opportunity for sanitation that benefits from maintaining most of the animal population, even in today's mouse colonies comprising mainly transgenic mice with unknown or compromised immune status.
The mouse rotavirus or EDIM (epizootic diarrhoea of infant mice) virus is a non-enveloped, double-stranded and segmented RNA virus of the family Reoviridae. It is prevalent in both wild and laboratory mice, respectively. Rotavirus is shed in faeces (faecal–oral route); transmission occurs directly through contact with infected animals or by contaminated fomites. As rotaviruses remain infectious outside the body, show resistance to inactivation (e.g. low pH, non-ionic detergents, hydrophobic organic liquids, proteolytic enzymes), and are shed in high quantities (>1011 particles per gram of faeces), these viruses are highly transmissible within a given species. However, rotaviruses are destroyed by disinfectants containing phenols, chlorine or ethanol. 1 All ages of mice are susceptible, but clinical signs (mild-to-severe diarrhoea) can be detected solely in infants younger than 14 days old. Despite a high morbidity, death seldom occurs since infants continue to suckle; mortality seems to be associated with rectal obstruction. 2 Mice normally recover within two weeks, but animals with virus-related transient weight loss often fail to reach normal adult weight. Adult mice pass through an inapparent infection, but shed virus in faeces for at least 17 days. In clinically-affected mice, rotavirus can be diagnosed on the basis of age, clinical symptoms and pathology. However, EDIM must be differentiated from other diarrhoeal diseases such as Coronavirus infection, reovirus 3 infection, Tyzzer's disease and salmonellosis, which can be achieved by serological (immunofluorescence assay [IFA], enzyme-linked immunosorbent assay [ELISA], multiplexed fluorometric immunoassay) or direct (ELISA, reverse transcriptase-polymerase chain reaction [PCR]) tests.1,2
In animal facilities rotavirus infection of infant mice is a serious problem for both breeders and researchers, respectively. Although the prevalence of virus infections in animal facilities has decreased over the last 20 years, rotavirus infections repeatedly appear in animal facilities worldwide and rank among the four most frequent viral pathogens in laboratory mouse colonies. According to three large commercial laboratories, 0.6–9% of mouse sera from North American and European facilities tested positive for rotavirus;3–5 however, up to 30% of mouse colonies in the USA have been identified as affected in a survey performed in 2006,6,7 and an unusually high frequency of positive findings was observed in European facilities in 2007/2008 (M Mähler, personal communication). The impact of rotavirus infections on research can be tremendous,8,9 especially studies utilizing neonatal or infant mice or those investigating the gastrointestinal system or infectious diseases might be affected.10,11 A disease-induced stress-related thymus necrosis may occur and alter immunological experiments. 2 Therefore, rotavirus screening is part of routine health monitoring programmes 12 and sanitation of the infected colonies is inevitable.
The most effective way for eradication of a rotavirus infection is to replace the entire colony by uninfected animals that are either imported or generated by embryo transfer or by Caesarean rederivation.1,2 However, a procedure that bypasses euthanasia of all animals is a breeding moratorium for several weeks.13,14 This method provides the opportunity to build up immunity in adult mice and has been successfully applied to eradicate murine corona-viruses.15,16 Additionally, in the absence of susceptible neonates the virus has no chance to spread or even survive. 14 However, the breeding suspension is thought to fail in colonies of immunodeficient or transgenic mice since those animals often shed virus for extended periods. 17 Therefore, it was questionable as to whether this approach would be suitable for sanitizing a rotavirus infection in current experimental mouse colonies comprising a huge variety of transgenic mice with partially unknown immune status. We present in this report that this approach should be considered despite the presence of immunocompromised mice in affected areas.
In the Central Animal Facility of Hannover Medical School, a rotavirus infection was detected based on clinical findings in newborns of an affected litter in the first half-year of 2008 (Figure 1). Subsequent serological (IFA) and PCR testing confirmed this virus in an area that was otherwise free of pathogenic agents except for
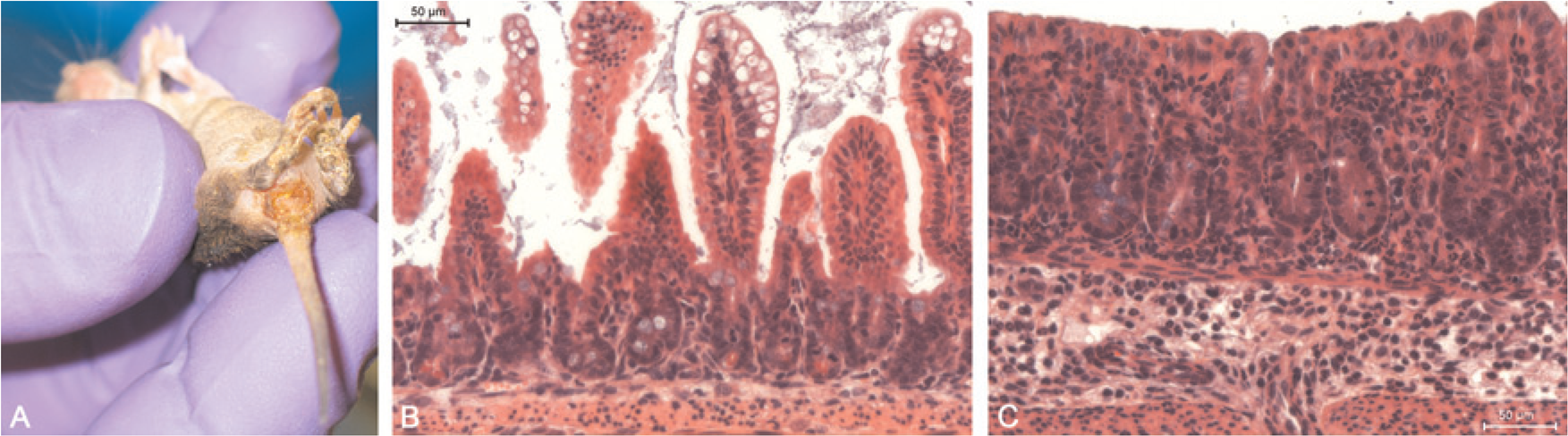
Clinical and histological presentation of EDIM at the Central Animal Facility. A rotavirus-infected suckling showing watery to oily and yellow faeces and an inflamed perianal region that appears wet and stained, as do the caudal extremities (A). Histology revealed vacuolation and cytoplasmatic swelling of villar epithelial cells in the small intestine (B) and mixed infiltration of leukocytes in mucosa and submucosa of the colon (C)
Results of serological surveys during and after rotavirus outbreak
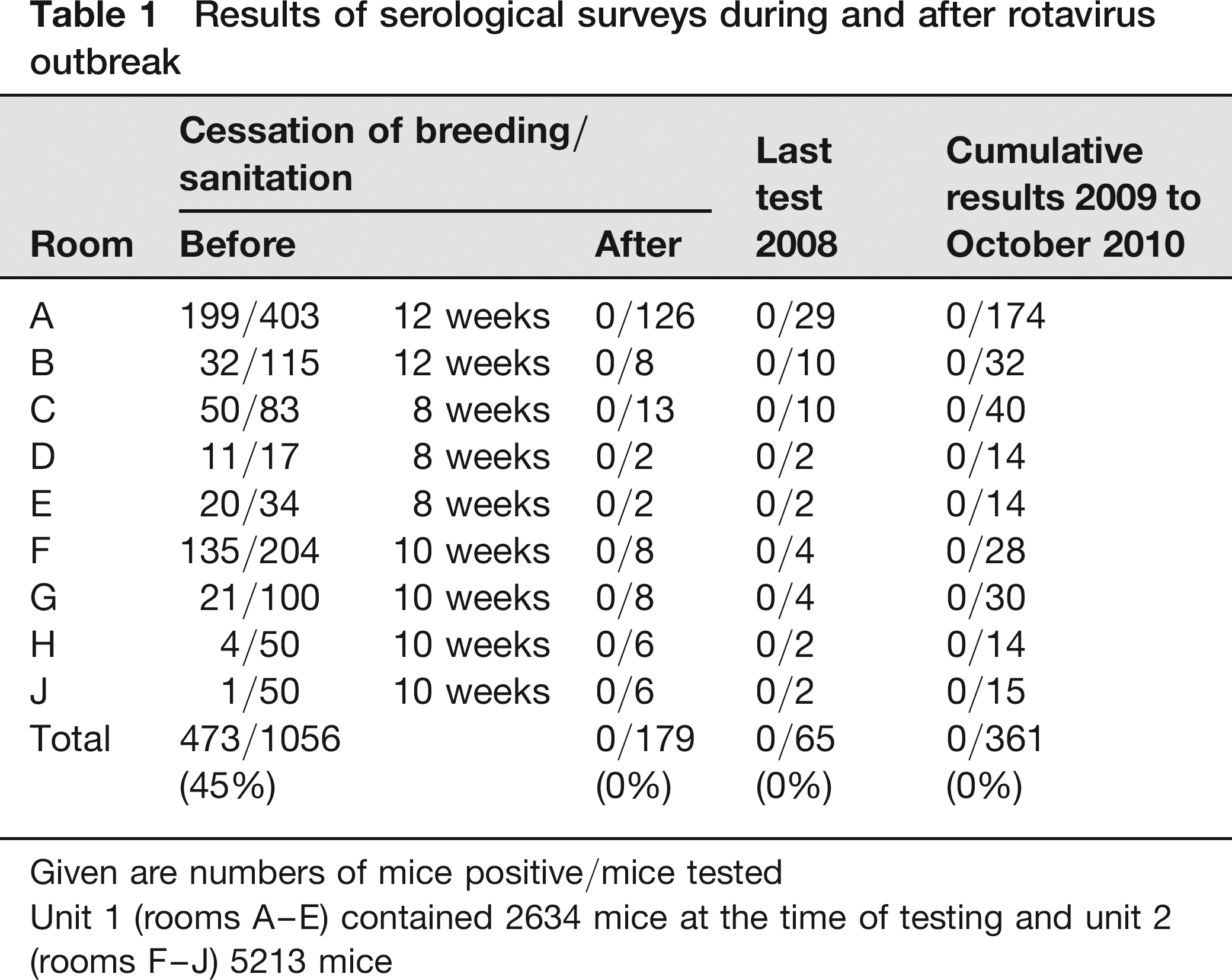
Given are numbers of mice positive/mice tested
Unit 1 (rooms A-E) contained 2634 mice at the time of testing and unit 2 (rooms F–J) 5213 mice
In the first step, the two areas were quarantined to prevent spreading of infection. In detail, the import as well as the export of animals were instantly stopped. Additionally, a strict hygienic management was performed, including entrance limitation to selected personnel, auto-claving of all materials (cages, protective clothing, etc.) that left the animal rooms, daily disinfection of affected units and adjacent areas using suitable germicides (formaldehyde; Lysoformin®, Lysoform Dr Hans Rosemann GmbH, Berlin, Germany) and application of negative pressure against adjacent areas.
For sanitation of the affected units it was planned to eradicate the virus by breeding cessation. In the first step those animals that were not absolutely needed were euthanized instantly. This included standard inbred strains that were easily available, and also mice that were not selected for breeding after the cessation period, as well as sucklings and pre-weaners. In case of breeding pairs that were not planned to be replaced after the cessation period, males and females were separated to prevent production of susceptible litters. Immunoincompetent mice were culled in case the strains were available for re-import or quarantined separately. However, one of the controlled rooms contained several immunocompromised transgenic strains (e.g. Fcγ-receptor, C5a-receptor or CD45-deficient strains) and the strategy of breeding cessation was also applied to this area. As shedding of rotavirus is known to be transient and rather short,11,18 we refused to distribute bedding material throughout the entire colony to promote serocon-version of possibly all mice present. In contrast, we used filter-top caging to prevent further spreading. Therefore, this strategy was a slight modification from what is generally recommended, e.g. by GV-SOLAS. 10
The outcome of this approach was controlled by testing sentinel (contact or dirty bedding sentinels) and experimental mice after eight to 10 weeks of breeding cessation (Table 1). Immunodeficient mice that had to be kept were directly socialized with contact sentinels. Sentinel mice (NMRIxB6) originated from a specified pathogen free area protected by a strict hygiene barrier system (with access limited to a few animal caretakers who had to pass through a water shower; bedding and diet were autoclaved, water was deionized, filtered and UV light treated; microbiological monitoring 12 did not reveal the presence of pathogens) and were introduced 3–4 weeks prior to testing. Because two contact sentinels tested positive for rotavirus at this time point, the breeding cessation was extended for an additional four weeks in areas with immunocompromised mice or transgenic mice with unknown immune status. In a second control phase that was started after the additional cessation period, all tests (IFA) were found to be negative for rotavirus (Table 1). By the end of 2008, a total of 244 samples were examined, none of which were rotavirus-positive. The intensive monitoring was continued for months to ensure the success of sanitation. From 2009 to October 2010, previously affected units were controlled by means of 361 probes that all showed negative for rotavirus (Table 1).
Sanitation of the rotavirus infection by breeding cessation proceeded successfully. The procedure has been previously proposed as an effective way to eradicate rotavirus in colonies of immunocompetent animals; however, case reports have not been published to our knowledge. Furthermore, some authors exclude the breeding cessation as a suitable procedure to eradicate rotavirus in colonies of immunocompromised mice.1,13,14 Therefore, effectiveness of this approach in current experimental mouse colonies with hundreds of different genetic modifications and often unknown immune status was doubtful. Owing to our experiences, it is reasonable to consider this method also for colonies containing genetically-modified and immunocompromised animals. Certainly, this strategy has to be well planned, communicated to scientists and technical personnel and tightly controlled. One has certainly to anticipate a prolonged cessation period in case mice of unknown immune status are present in an area, and this has also to be considered when prospective breeding pairs are selected before cessation has started. According to our experience, the suspension should last 8–10 weeks, and in areas with immunodeficient animals as long as 12 weeks. Continuous monitoring using sentinel mice can be helpful to ensure a positive outcome.
Breeding cessation provides the opportunity to sanitize colonies in a relatively short time preserving most strains; however, potential risks have to be considered. Infected and virus-shedding mice remain in the affected animal rooms and virus might spread into ‘clean’ units. Furthermore, the long interval of breeding interruption especially in immunodeficient animals must be considered. The breeding period and performance of mice differ individually and are dependent on transgene and background genetics,19,20 and time from mating to retirement varied even between inbred strains from 22 to 30 weeks in a recent study. 21 Therefore, breeding might fail after cessation, resulting in loss of the strain. Fortunately, we did not encounter this problem.
In the presented approach, we successfully sanitized a rotavirus-positive animal unit by breeding cessation despite the presence of a variety of transgenic and immunocompromised mice. Although cessation had to be prolonged up to 12 weeks in certain areas, all strains recovered. Therefore, this cost- and time-effective approach might be considered as suitable even in facilities that are faced with a tremendous and still growing variety of transgenic mice whose immune status is often not clearly predictable.
