Abstract
Sentinel exposure to soiled bedding is frequently used for health monitoring of mice housed in individually ventilated cage systems (IVCS). Despite its advantages, the use of soiled bedding sentinels (SBSs) is far for being a reliable method. Two studies were conducted to evaluate the sensitivity of immunodeficient SBSs NOD.CB17-Prkdc scid /NCrHsd (NOD SCID) against two immunocompetent outbred strains, Hsd:ICR (CD-1) and RjOr1:Swiss (Swiss) to pinworm detection in IVCS-housing. Four different diagnostic methods were used: perianal tape test, fecal flotation, plate method and histology. Positivity was considered if at least one of the techniques used was positive. In the first study NOD SCID were more sensitive than CD-1 SBSs (P < 0.05), and except for the fecal flotation test performed at week 6, all the diagnostic methods were more sensitive with NOD SCID mice (P < 0.05). In the second study differences between the Swiss and NOD SCID mice were less obvious (P = 0.08). When compared separately, the different diagnostic methods, except for the fecal flotation test, were all more sensitive in the NOD SCID mice (P < 0.05). In addition, the anal tape test in the Swiss SBSs was more sensitive at week 7 than at week 15 (P < 0.05). In conclusion, combining various diagnostic techniques and samplings at week 7 post-exposure with non-invasive methods increases the rate of pinworm detection. Immunodeficient SBSs showed higher sensitivity than immunocompetent ones. Thus, use of immunodeficient SBSs is highly recommended in health control protocols.
Pinworms of the order Oxyurina (Syphacia obvelata and Aspiculuris tetraptera) are the two most common parasites found in laboratory mice. 1 Although rarely associated with clinical symptoms, they have been associated with rectal prolapse, intestinal impaction, intussusception and mucoid enteritis.2–4 Pinworm infestation affects behavior, growth, intestinal physiology and induces Th2-associated immune responses. These biologically altered responses in infected animals may confound research results.5,6
Oxyurids are monoxenous parasites that are transmitted through ingestion of embryonated eggs.2,6–8 Various methods are used to detect pinworm infection, the most frequently used are: (i) perianal tape test,9,10 (ii) fecal flotation, 8 (iii) plate method (maceration),8,11 and (iv) histological examination. 6 Other methods such as the anal swab technique and fecal concentration technique are less frequently used.12,13 In addition to traditional methods, polymerase chain reaction (PCR) techniques have been used in recent years with accurate results.5,6,14,15
The use of individually ventilated cage rack systems (IVCS) reduces the risk of infection transmission, limiting the number of cages affected when outbreaks occur. 16 As random sampling or the use of contact sentinels may not be compatible with breeding programs or experiments, the exposure of sentinels to dirty soiled bedding (soiled bedding sentinels [SBSs]) has been developed as a technique.17–19
Different susceptibilities have been reported in outbred and inbred strains, and athymic mice have an increased susceptibility to pinworm infection.7,20,21 In regard to susceptibility to pinworms, mouse resistance to pinworm infection depends on their major histocompatibility complex class II and not on their innate immune system. Interleukin (IL)-13 seems to have a dominant role in diminishing the population of S. obvelata in the cecum of mice.22,23
Few studies have compared the efficacy of different monitoring methods for pinworms, and only two of them have used immunodeficient sentinels.3,19,24–28
The purpose of this study was to compare the sensitivity of NOD SCID mice to that of two outbred strains in pinworm detection under field conditions, using four diagnostic methods.
Materials and methods
Animals and husbandry
Three different batches of animals were used. As immunocompetent mice, 23 outbred female specific pathogen-free (SPF) Hsd:ICR (CD-1) mice (Harlan Laboratories Models, SL, Barcelona, Spain), aged four weeks were employed in the first study. Four months later, 29 SPF female outbred RjOr1:Swiss (Swiss) mice (Janvier SAS, Saint Berthevin, France) weighing 15–19 g were purchased for the second study. As immunodeficient mice, 16 female SPF NOD.CB17-Prkdc scid /NCrHsd (NOD SCID), aged 6–16 weeks old were used for the first study, and another 16 female SPF NOD SCID (both groups from the institutional SPF breeding colony, originally purchased from Harlan Laboratories Models), aged 5–7 weeks old were used for the second study. Before being randomly housed in the sentinel cages, two animals of each batch (4 outbred and 4 NOD SCID) were euthanized and samples were collected to confirm their SPF status. All of them were free from Clostridium piliforme, Bordetella bronchiseptica, Citrobacter rodentium, Corynebacterium kutscheri, Helicobacter spp., Mycoplasma pulmonis, Pasteurella spp., Salmonella spp., Streptobacillus moniliformis, Streptococci beta-haemolytic, Streptococcus pneumoniae, ectoparasites, Giardia muris, Spironucleus muris, Syphacia spp., Aspiculuris tetraptera and Hymenolepis spp. Outbred mice were also confirmed to be free of epizootic diarrhea of infant mice, mouse hepatitis virus, mouse parvovirus, minute virus of mice, pneumonia virus of mice, respiratory enteric orphan virus, Sendai virus, Theiler's murine encephalomyelitis virus, ectromelia virus, lymphocytic choriomeningitis virus, Hantaan virus, murine adenovirus types 1 and 2, and murine cytomegalovirus. All batches were allowed to acclimatize for at least one week. Animals were housed in groups of five (3 outbred with 2 NOD SCID) per cage (SealsafeTM 1145TCP; Tecniplast, Buguggiate, Italy) in individually ventilated 64-cage rack cages (TouchSLIMLineTM 2T64MAC30CP; Tecniplast, Buguggiate, Italy) receiving 70 air changes per hour. Two additional cages with three Swiss mice were used in the second study. Sterile bedding (HBK 1500--3000; Harlan Laboratories Models, SL) and water and food (Global rodent diet 2018S, Harlan Laboratories Models, SL), both sterilized and provided ad libitum, were supplied. Staff were dressed in clean suits with disposable coats, hats, face masks and gloves. Cages were changed weekly in Class II laminar flow stations (LabGard ES 602; NuAire, Plymouth, MN, USA). Mice were maintained on a 12:12 h dark:light cycle (lights on at 08:00 h), with ambient temperature of 22 ± 2 ℃, and 50 ± 10% relative humidity.
Under Spanish law the study was considered to be a husbandry procedure, and therefore not subjected to ethical review. All animals were handled according to the guidelines set in the ‘Guide for the Care and Use of Laboratory Animals’ (National Institutes of Health, Bethesda, MD, USA).
Experimental design
Seven ventilated racks located in four different rooms in our conventional animal facility were health monitored twice in a four-month interval. Three of the four rooms, with two racks in each room (numbered 1 to 6) were detected positive to A. tetraptera and S. obvelata in previous health controls. The fourth room, where rack 7 was located, was restocked and treated with a medicated diet with fenbendazole at 150 ppm (Global rodent diet 2016S, Harlan Laboratories, Horst, The Netherlands) three months before the beginning of the study. One sentinel cage with five SBSs (3 outbred + 2 NOD SCID) was used for each of the seven ventilated racks in both studies; and in the second study, two additional cages with three Swiss mice in each cage were settled on racks 4 and 6. These two cages were used to detect differences in pinworm infection between the Swiss mice housed alone or with the NOD SCID mice. Any sanitary measures were taken between both studies.
Sentinel exposure to dirty soiled bedding was carried out as follows: every week, half (32 cages) of the index mice used cages on a rack were alternatively sampled. Aliquots of a handful of dirty bedding, approximately 40 cc, were taken from each cage and mixed in a sterile cage with an equivalent amount of new sterile bedding, and excess bedding was discarded in order to obtain an approximate final volume of 700 cc. Sentinel mice were moved to this sampling cage weekly.
After the exposure period all surviving sentinels, except three from the first study that were analyzed in an external laboratory, were euthanized with an isoflurane overdose (Isoba vet; Schering-Plough, Middlesex, UK) and sampled for health monitoring.
First study: CD-1 mice vs. NOD SCID mice
Seven cages housing three CD-1 mice and two NOD SCID mice per cage were exposed to dirty bedding for a period of 15 weeks. At week 6 of dirty bedding exposure, one CD-1 mouse per cage was euthanized and sampled for pinworm detection with the four methods previously mentioned. On the same day, one NOD SCID mouse per cage was sampled using anal tape and fecal flotation tests. Of the 21 surviving SBSs at the end of the study, 18 were euthanized and sampled for health monitoring. The remaining three, from racks 3, 6 and 7, were analyzed in an external laboratory for quality control.
Second study: Swiss mice vs. NOD SCID mice
Seven cages housing three Swiss mice and two NOD SCID mice per cage and two more cages housing three Swiss mice were exposed to dirty bedding for a period of 15 weeks. At week 7 of dirty bedding exposure, all of the 41 SBSs were sampled using anal tape test and 37 using the fecal flotation test. At the end of the study all 41 SBSs were euthanized and sampled for health monitoring. Results of the plate method were quantified, and in the plate and histology methods, results of colon and cecum contents were registered separately.
Pinworm examination
Four different methods were used to detect pinworms:
Modified perianal tape test was performed. Briefly, a slide covered with a thin transparent adhesive layer sized about 2 × 1 cm dispensed by a device (Pelikan Roll-Fix; Pelikan AG, Hanover, Germany) was applied over the perianal area. Here, a drop of Giemsa solution was placed over the adhesive area and thereafter mounted with a coverslip and observed under a microscope.
10
Fecal flotation was carried out by mixing one fecal pellet with saturated NaCl solution in a vial. By adding drops of the float medium a meniscus bulged above the rim of the vial. Fifteen minutes later a coverslip was placed on the top of meniscus, mounted on a slide and examined microscopically. Plate method: after necropsy, cecal and proximal colon contents were squeezed into Petri dishes with sterile phosphate-buffered saline for inspection under a stereoscope microscope for the presence of pinworms. Histological examination: the cecum and proximal colon previously examined and the remainder large intestine were rolled, immersed in buffered formalin, and processed for histological examination on hematoxylin and eosin (H&E) stained 4 µ sections.
29
In both studies, mice were considered to be positive for pinworm infestation if at least one of the tests conducted gave positive results.
Statistical analysis
The proportion of pinworm-infected mice was calculated for all of the assays using the results obtained for each laboratory technique. The statistical significance of the differences in the proportion of mice infected with pinworms in each cage as a function of the mouse strain, was assessed through a Z-test for the difference between two proportions and the Fisher's exact test, with a confidence level of 95% (α = 0.05) and the calculus of the respective Z value.
Assessment of the pinworm detection
In order to determine the assessment of the laboratory tests used in the pinworm detection, results obtained by histological examination, anal tape test and fecal flotation test were compared with those obtained by the gold standard method (plate method), by the construction of two-way contingency tables and a comparison of the relative specificity, sensibility and positive and negative predictive values as described.30–32
Results
First study: CD-1 mice vs. NOD SCID mice (Table 1)
First study: pinworm detection in CD-1 and NOD SCID soiled bedding sentinels using different diagnostic methods.
Died before the end of the study.
Analyzed in an external laboratory.
Mice No. 14 not included. S = Syphacia obvelata.
A = Aspiculuris tetraptera; NT = not tested; ¿ = unknown.
In CD-1 mice the plate method was less sensitive than the histological examination, with detection in only one of the three positive mice using the histological technique. On the other hand, all the NOD SCID mice were tested positive using the plate and histological methods. In three of the six cages tested positive to pinworms all the CD-1 sentinels were negative (cages 1, 2 and 6), and in the other three cages only one out of three tested positive (cages 3, 4 and 5). CD-1 mice from cages 3 and 6 analyzed by the external laboratory gave negative results.
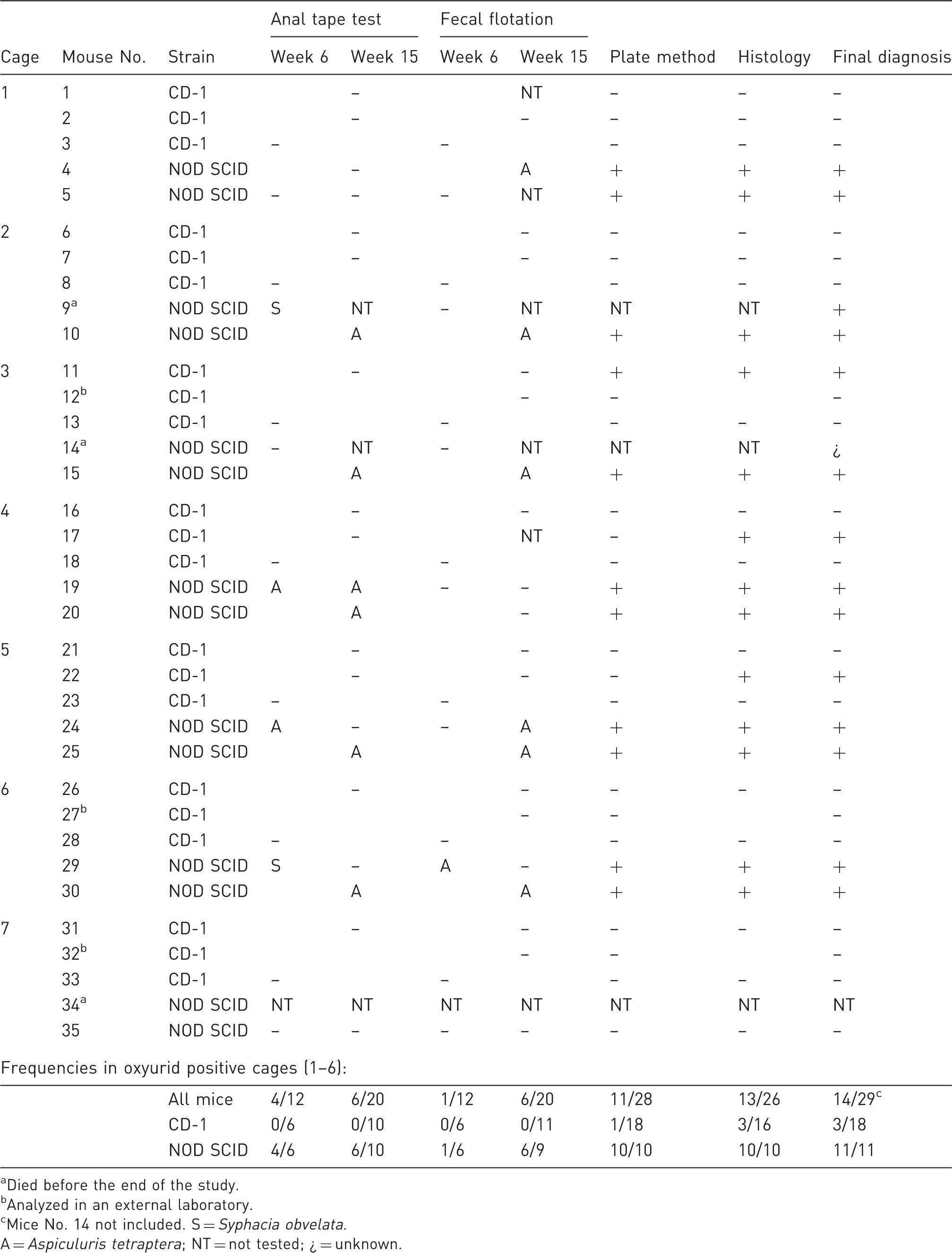
NOD SCID mice were more sensitive, using all the diagnostic methods except for the fecal flotation test performed at week 6, than CD-1 mice (P < 0.05). In this regard, the relative sensitivity of pinworm detection using CD-1 mice was 100% for the histological examination and 0% for the anal tape and fecal flotation tests (Figure 1). With NOD SCID mice the relative sensitivities in pinworm detection using the different laboratory techniques were 95.65% for histological examination, 86.96% for the anal tape test and 72.27% for the fecal flotation test (Figure 2).
CD-1 mice: Relative parameters of histological examination, anal tape test and fecal flotation test compared with the plate method. NOD SCID mice: Relative parameters of histological examination, anal tape test and fecal flotation test compared with the plate method.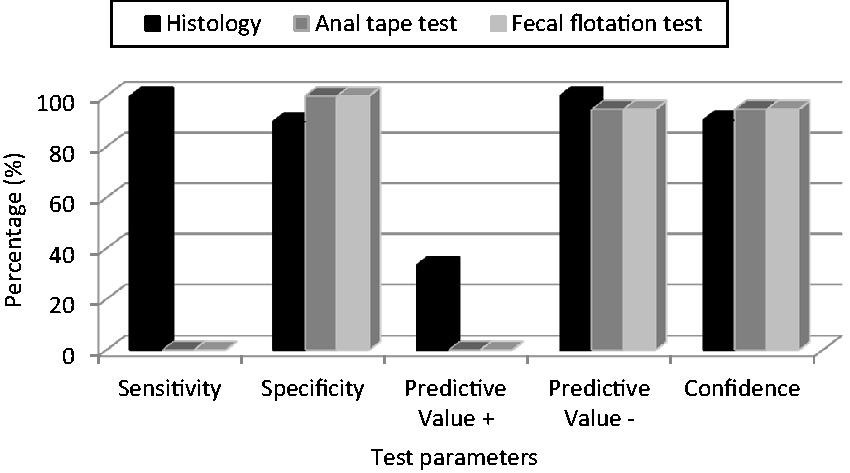
In cage 7, all the sentinels, including those analyzed by the external laboratory, showed negative results.
Second study: Swiss mice vs. NOD SCID mice (Table 2)
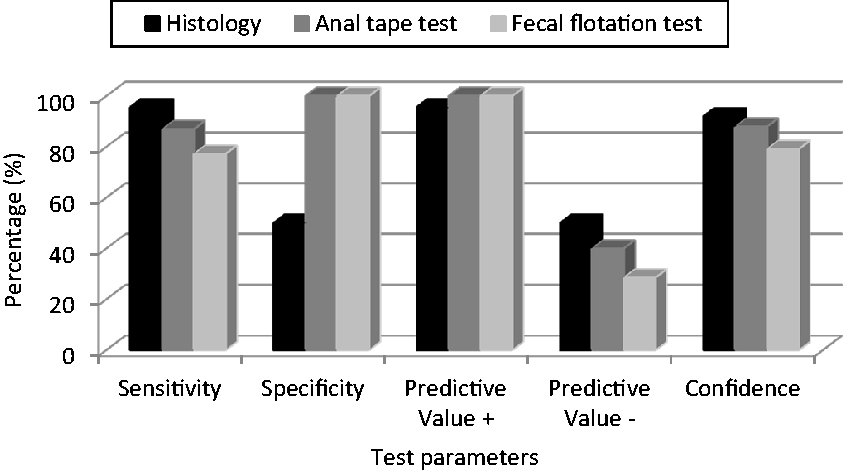
Differences between both strains at the final diagnosis were not statistically significant (P = 0.08). Comparing the different diagnostic methods separately, except for the fecal flotation test, the NOD SCID mice were more sensitive with all the other methods than the Swiss mice (P < 0.05). Pinworms were detected in both strains using all methods, with differences in their relative sensitivity. In this regard when the detection of pinworms was compared with the gold standard method (plate method), the sensitivity of pinworm detection in Swiss mice was 38.89% using histological examination, 72.22% using the anal tape and 22.22% using the fecal flotation test (Figure 3). A higher sensitivity was obtained with the NOD SCID mice (Figure 2).
Swiss mice: Relative parameters of histological examination, anal tape test and fecal flotation test compared with the plate method. Second study: pinworm detection in Swiss and NOD SCID soiled bedding sentinels using different diagnostic methods. (1)+ = 1 pinworm; + + = 5–20 pinworms; + ++ = >20 pinworms. Not included into the frequencies below. S = Syphacia obvelata; A = Aspiculuris tetraptera; NT = not tested; Col = Colon; Ce = Cecum.
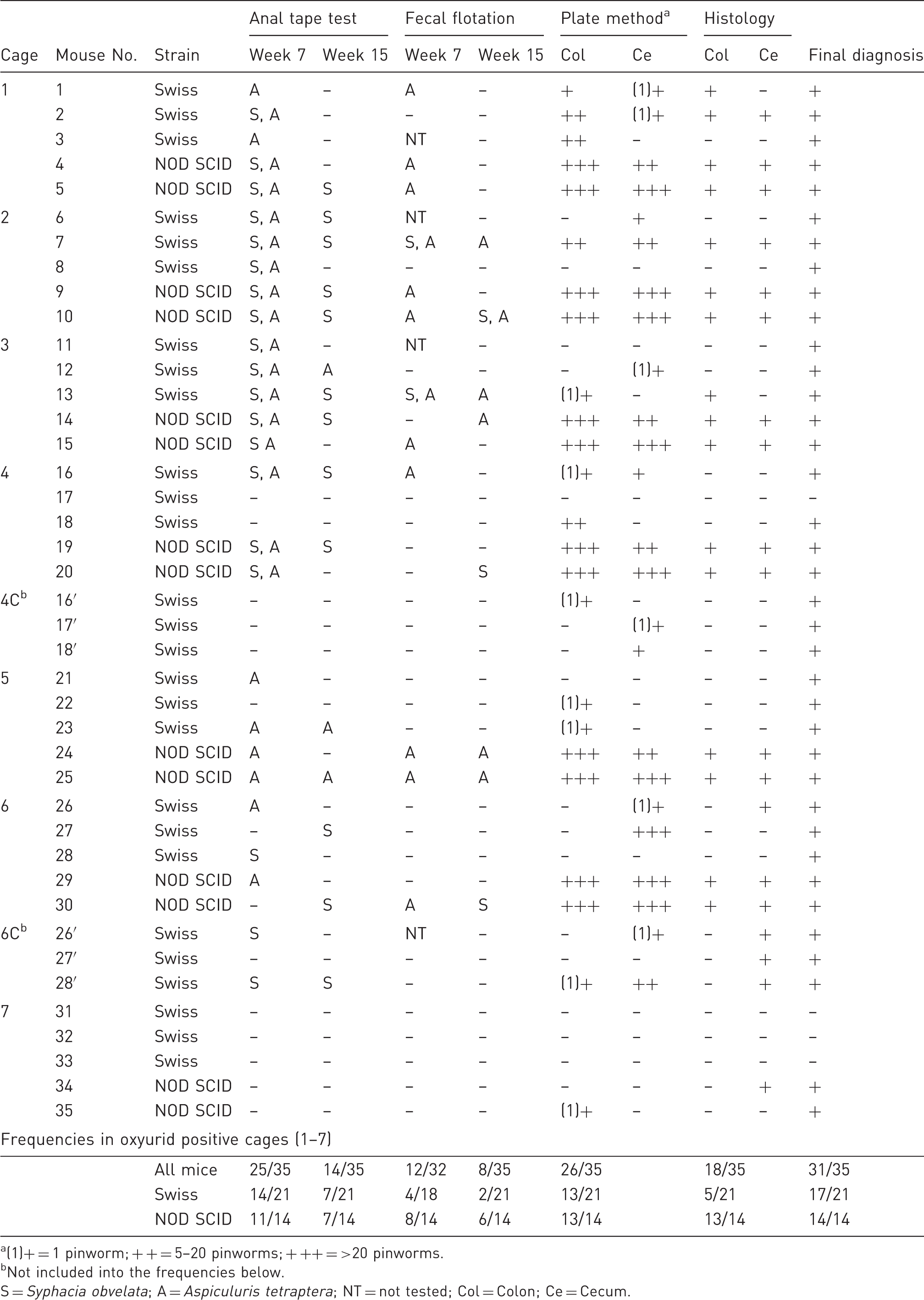
Of the 41 mice tested, only four Swiss mice yielded negative results. In NOD SCID mice, combining the results of the anal tape and fecal flotation tests at week 7, pinworms were detected in 12 of the 14 NOD SCID mice tested.
Swiss mice 8, 11, 21 and 28 were positive only using the anal tape test at week 7 post-exposure, while the rest of the analysis yielded negative results. The anal tape test in the Swiss SBSs was more sensitive at week 7 than at week 15 (P < 0.05), while these differences were not observed in the NOD SCID mice.
Detection of only one pinworm with the plate method was the unique positive result in Swiss mice 16′, 17′ and 22 and in the NOD SCID mouse number 35. Again there were two mice, Swiss mouse number 27′ and NOD SCID mouse number 34, in which pinworms were detected only by using histology. Numbers of pinworms observed using the plate method were consistently higher in the NOD SCID than in the Swiss mice.
Positive results, confirmed with index mice (data not shown), in the two NOD SCID mice in cage 7 suggested a contamination of rack 7 between the two studies.
Swiss mice in cages 4C and 6C all tested positive, and no apparent differences were seen with those of cages 4 and 6 co-housed with NOD SCID mice.
Significant differences were observed in pinworm detection between both CD-1 and Swiss immunocompetent strains (P < 0.05).
Discussion
Use of SBSs is effective in detecting infectious agents like pinworms which are transmitted by the fecal–oral route.19,20,26–28 The reliability of this approach is highly variable, therefore sampling design, diagnostic methods and sentinel-related parameters must be considered.19,26,27
Several factors may affect the efficiency of pinworm detection in SBSs: (i) infectious agent loads to which the sentinel mice is exposed, (ii) quantity of bedding transferred, (iii) frequency of sampling, (iv) time elapsing between bedding transfer and sampling, (v) diagnostic methods, (vi) sentinel age and (vii) sentinel genotype.4,6,26 All these aspects should be considered when interpreting the variability of results, even using the same methods in the same populations.
Infectious agent loading is an uncontrollable factor. 26 Pinworm eggs are shed intermittently and the age and immune status of the host determine the worm burden.2,3,22 Sustained detection over time has been previously reported in immunodeficient mice; however these studies were done with S. obvelata.3,22,23 The higher parasite burdens observed in our second study in NOD SCID SBSs, except those of cage 7, facilitated an early and more sensitive detection than in Swiss SBSs.
The use of non-invasive diagnostic techniques, such as anal tape and fecal flotation tests, allows for more frequent sampling and therefore a greater probability of detection. In a CD-1 S. obvelata infested colony, the tape test performed weekly reached 85.5% of sensitivity. 4 In our study, pinworm could not be detected in CD-1 mice by using any of these two methods. In another study with nu/nu and Swiss Webster SBSs, S. obvelata could be detected using the anal tape test in both strains from weeks 3 to 13 post-exposure, and in the nu/nu mice until the end of the study at week 19. 3 In our Swiss SBSs we obtained a sensitivity of 72.22% using this method.
In our second study, except those of cage 7, pinworms could be detected with non-invasive methods from week 7 in all NOD SCID SBSs. In addition, in four of the Swiss mice the only positive result was observed with the anal tape test at week 7. The anal tape test in the Swiss SBSs was more sensitive at week 7 than at week 15 (P < 0.05). This may be due to a contamination of the perianal area with pinworm eggs caused by its high concentration in the cage or by the immune response developed by these mice. In this regard, in immunocompetent mice, pinworm clearance is produced by an immune Th2 response. This phenomenon was reported in S. obvelata infections in which the parasite load falls in mice dramatically from 12 weeks of age.3,22,23 Immunodeficient NOD SCID mice are unable to develop this response and consequently maintain a high parasite burden over time, showing no differences between week 7 and week 15.3,24,25 At these sampling times no differences were observed in the fecal flotation test in either of the two immunocompetent strains tested (P > 0.05). When mixed infections occurred, more Syphacia than Aspiculuris are usually seen. 11 Lower burdens of the latter species make it more difficult to detect using the fecal flotation test, and this could explain the lack of differences observed using this technique in the Swiss SBSs.
Detection rates of the different diagnostic methods are variable and are strongly related to the parasite load at the time of examination. For instance, sensitivities of the anal tape method have been reported as being between 20% and 85.5%.1,4,7,19 In our studies this ranged from 0% in CD-1 mice to 86.96% in the NOD SCID mice. The fecal flotation test sensitivities reported have ranged from 20% to 75%, while ours varied from 0% in the CD-1 mice to 77.27% in the NOD SCID mice.1,7 The plate method has traditionally been considered the gold standard, but sensitivities from 80% to 91% have been reported when used in combination with other classical techniques.1,19 In our first study, the detection rate with this method ranged from 5.5% in immunocompetent SBSs to 100% in immunodeficient SBSs, while in the second the ranges varied between 62% and 93%, respectively. Despite its relative low use as a routine diagnostic method, and used in only about 25% of institutions, histological examination sensitivity has been reported as 75%, which is comparable to that observed with the plate method. 1 We observed a relative sensitivity from 38.89% to 100% in immunocompetent SBSs and 95.65% in immunodeficient SBSs, and in four of the mice it was the only positive result. As previously reported, pinworms are occasionally only detectable by histological analysis, especially in subtle infestations.1,19 In our study this happened in four (8.8%) of the 45 positively-diagnosed mice. This apparent inconsistency may be due to a failure in the removal of the contents of the large intestine with the plate method. When the parasite load is very low, some pinworms may remain in the intestinal tissue, making them undetectable using the plate method but visible by histological examination.
These data indicate the relative ease with which false-negative results can be produced. The fact that infected and uninfected mice can co-exist in the same cage favors a misdiagnosis of pinworm detection.3,4,6,15,21 The results underline the recommendation that different diagnostic methods should be used in order to minimize false-negative results, especially when Swiss mice or another immunocompetent strain are used as SBSs.1,3–6
Our first study showed clear differences between immunocompetent and immunodeficient SBSs. In the second study, due to the high sensitivity of the Swiss SBSs (80.1%), no statistically significant difference was observed in the final diagnosis when compared with NOD SCID mice.
Striking differences were observed in pinworm detection between both immunocompetent strains (P < 0.05). All experimental conditions were the same except for the periods, May and September, at which the studies were done. Thus, only genotypic variations between sentinel strains or seasonal fluctuations in pinworm burdens can explain this significant difference.21,33
In view of these results, the use of immunodeficient SBSs is highly recommended in health control protocols.
Footnotes
Acknowledgements
The authors thank their colleagues in the Animalario Convencional – CNM (ISCIII): Miriam Pérez, Eduardo Martorell, Sheila Ortega, Andrés Aguilera, Luis Fernández, Vicente Saavedra, Fernando Menor and Raquel del Cerro; and in the Animalario SPF – CNM (ISCIII): Daniel Baizán and Teresa González.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
