Abstract
UK Health Security Agency is required to investigate the pathogenesis of emerging or re-emerging infections and to test novel interventions, such as vaccines and therapeutics against these and other diseases, such as tuberculosis and Ebola, that have a significant impact on human health world-wide. Research into the causative agents (mainly BSL 3 and 4) using a wide range of animal species as pre-clinical models brings a number of challenges in terms of effective biocontainment to address human safety whilst optimising delivery of scientific objectives and the welfare of the animals. Here we describe the strategies used for high containment of species that include mice, ferrets, hamsters, rabbits and macaques that have been infected with high consequence pathogens. To ensure relevance of these models we frequently challenge by the aerosol route and monitor the development of disease and protective or therapeutic efficacy by methodologies similar to those used in the clinic. We have devised methods of sampling that can inform on pathogenesis and immune function that include lung lavage and medical imaging such as computed tomography and positron emission tomography-computed tomography. Imaging assists our assessment of progression to disease whilst providing refinement in application of early humane endpoints. We have developed directional flow containment systems that provide quantifiable operator protection whilst allowing group housing and a wide range of enrichment strategies appropriate for each species. Furthermore, we have demonstrated our improvements in animal welfare through use of a software-based Animal Welfare Assessment Grid that was developed with help of NC3Rs funding and enables us to quantify the lifetime experience of animals.
Introduction
UK Health Security Agency (UKHSA) is required to conduct research on novel interventions for current and newly emerging infectious diseases that threaten human health. This mandate includes a requirement to respond to biothreat emergency situations that may result from natural occurrences or from deliberate release. In order to develop new therapeutics or vaccines it is currently necessary to use relevant animal models to characterize the disease and then to test immunogenicity, safety and the protective efficacy of interventions such as vaccines. To optimize the clinical relevance of animal models, reproducing the disease as seen in humans and applying the latest technology to monitor disease progression are essential. In addition, the application of technologies such as imaging addresses the ethical requirements to gain as much information as possible from an animal and to define the earliest intervention points that provide robust data on efficacy.
Pre-clinical and para-clinical models that support pathogenesis and efficacy data have the potential to speed up the regulatory progress of a vaccine or therapy to the clinic and may reduce the requirement for, or inform the design of, clinical trials in humans. Studies are conducted in a high containment facility that has suites of rooms that can operate at biocontainment levels 3 or 4 (BCL3, BCL4).
Containment strategies
At UKHSA we have developed animal models using a range of different species that provide the most relevance to the diseases seen in humans. The use of experimental animals infected with dangerous infectious agents poses a unique problem in terms of safe biocontainment that protects staff working with the animals and that prevents release of such agents into the environment. This level of operator protection can impact on the provision of enrichment and demands a creative approach to maximising animal welfare. The various operator protection and enrichment strategies used at a number of research establishments for a wide range of animal species have been reviewed in detail. 1
Facilities in the United Kingdom (UK) working with high consequence pathogens need to meet the stringent requirements of BCL3 or BCL4 as defined in the UK by the Advisory Committee on Dangerous Pathogens (ACDP) and monitored by the Health and Safety Executive.2,3 Containment strategies depend upon an organism’s hazard group (HG) classification and the risks associated with possible routes of transmission. The main strategy at UKHSA for containment of smaller laboratory species is to use flexible film isolators operating under negative pressure. These provide the advantage of a total microbiological barrier between the animal cages (plus ancillary equipment) and the staff, but they can present challenges in terms of access to all areas and dexterity for procedures. Ergonomics can be improved by use of integral half suits so that there is more ability for manoeuvre whilst retaining the ‘primary containment’ principle 4 (Figure 1).

High containment guinea pig housing using (a) half suit isolators and (b) directional flow system showing enhanced space and enrichment capability.
In addition to their use in experimental maintenance, portable variations of flexible film isolators can be used for transferring animals from one containment suite to another and for safe movement of subjects during medical imaging such as computed tomography (CT) and positron emission tomography-CT (PET-CT) 5 (Figure 2). The use of imaging technology has allowed us to monitor disease progression and vaccine or therapeutic efficacy by CT or PET-CT scanning of subjects sequentially over time. 6
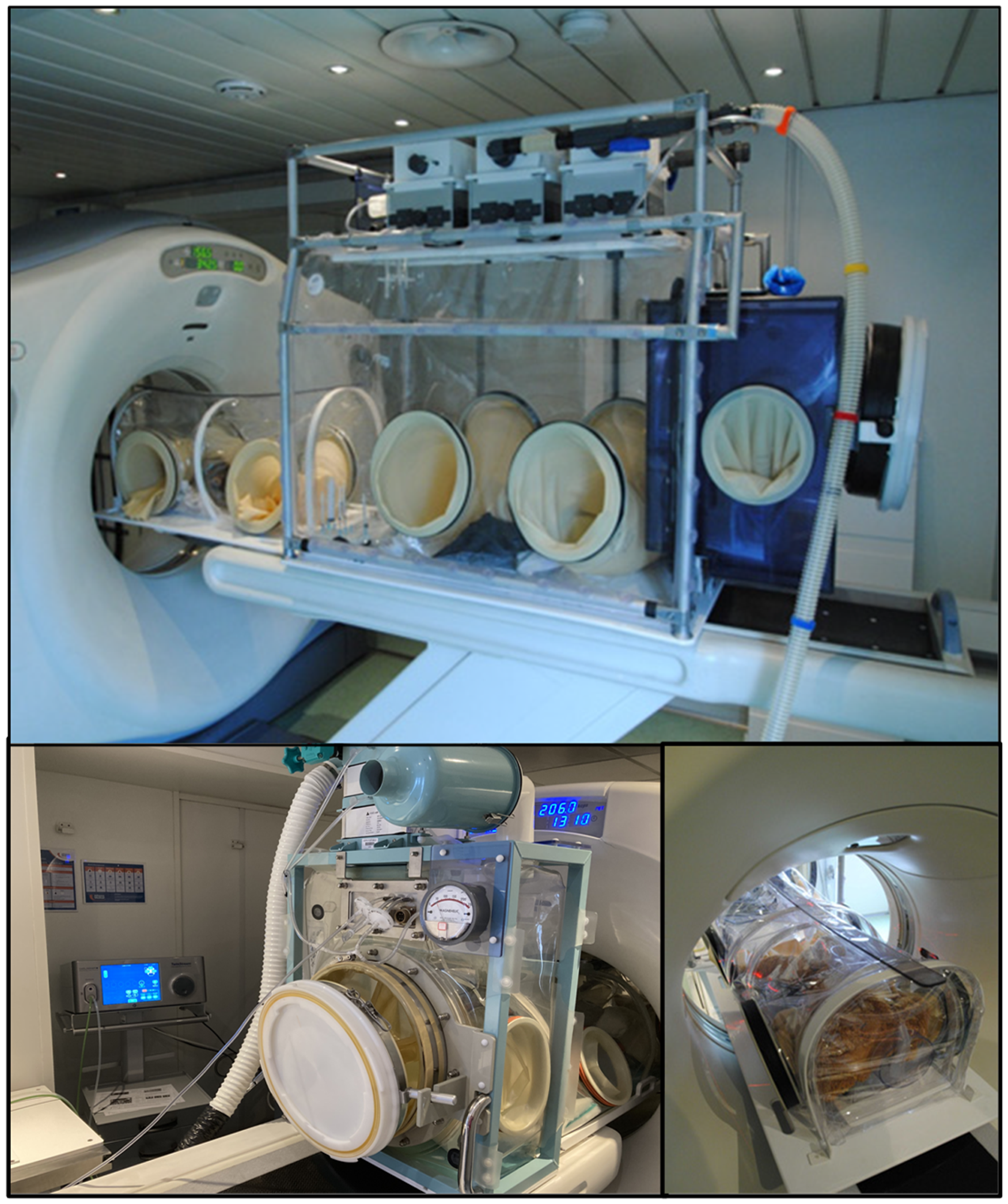
Imaging pod used for computed tomography and positron emission tomography-computed tomography scanning. Note high frequency jet ventilation, connected for improved image resolution.
In order to meet biosafety requirements for macaques infected with HG3 agents such as Mycobacterium tuberculosis we have developed a directional flow containment system that allows group housing of animals whilst providing quantifiable operator protection. 7 This system can be adapted to meet a variety of room and cage configurations (Figure 3(a)), has been used successfully for many years and meets welfare requirements by providing good usable space, clear observation of subjects, group housing and provision of a range of enrichment strategies 8 (Figure 3(b)).

(a) Two versions of directional flow containment systems, adapted for different room sizes, cage configurations and telemetry requirements, that can house social groups of macaques infected with level 3 pathogens. Note floor to ceiling height and use of full length of room to maximize available space and use of non-metal materials such as Trespa. The blue arrows (i) indicate flow of air through inlet vents. (b) Enrichment provision within group-housed accommodation.
The containment system provides staff protection by using the principle of directional airflow away from the operator towards the back of the cage system. A series of interconnected cages are placed inside modular booths that have a clear rigid plastic screen at the front of the system. This screen allows good observation of the macaques within the cages whilst providing protection from physical contamination. Each screen is fitted with flap valves (Figure 3(a) (i)) that control the velocity of the air into the system and each has a number of small access doors placed in strategic alignment with the cage front to allow animal care staff access to replace food and water, change bedding and sedate and remove the occupants for subsequent procedures. The number of airborne organisms is reduced by using this directional airflow away from the operator towards the rear of the cage. With correct operation, following protocols defining the number of access doors open at any one time, this flow is maintained at a minimum of 0.7 m/s and the air removed from the rear of the cage is extracted by a total-loss room air handling system via HEPA filtration. Further operator protection is provided by using a transfer box for sedated animals and a downdraught table for procedures such as blood sampling, bronchoalveolar lavage, X-rays, clinical assessment and necropsy.
Animal welfare needs are provided for by incorporating group housing into the study design, utilising full height cages that exceed the standards of the UK Home Office Code of Practice, Directive 2010/63/EU and Appendix A to Convention ETS 123, and providing foraging enrichment and toys such as balls, wooden dumbbells, chains, swings and mirrors. 8
Following the success of this type of containment for primates, the use of this strategy has now been extended to other species such as guinea pigs, ferrets and rabbits. Trials of the system with these species have now been conducted and feedback from staff on ergonomics, improved enrichment and welfare opportunities has led to investment in much larger cages that can now be accommodated (Figure 1). One of the main drivers for this change was the use of the Animal Welfare Assessment Grid (AWAG) in which welfare was quantified and compared between BCL3 studies using isolators or directional flow containment.
The AWAG was developed from an original concept by Honess and Wolfensohn. 9 The AWAG uses software to examine the welfare of the animal at key points throughout its life, taking into account the duration as well as the intensity of suffering producing numerical, as well as visual, representation of the animal’s overall quality of life. From the individual records maintained for each animal, a welfare assessment score is applied at each moment in the animal’s life when an intervention takes place. The system scores four parameters (Physical, Psychological, Environmental, Procedural (experimental and/or clinical)). These four parameters together contribute to the level of cumulative severity and are scored to obtain a quantitative measure, the cumulative welfare assessment score (CWAS). Within each parameter various factors are scored between 1 and 10 where a score of 1 indicates the best possible state (lowest possible impact on welfare) for the respective factor, whilst a score of 10 would be the worst possible state (highest possible impact on welfare). For each parameter the average of the factors scored is used to assess the level contributing to cumulative severity. The issue of observer bias can be mitigated by agreeing in advance, as much as possible, the scoring of factors within each parameter. Thus, agreed scores can be allocated to cage design, group size, particular procedural interventions and defined clinical responses. This approach has proved to provide consistent scoring in experimental and other arenas where animal welfare assessment is important.10 –18
AWAG was originally developed and tested in experimental macaque studies 10 (Figure 4(a)) but has been extended to other species11,12 and to other animal welfare arenas.13 –18 As an example of the utility of the AWAG we show how it was used in a comparison of two infectious disease studies using different pathogens in the same animal species: study Ant shows the welfare score for a cohort of guinea pigs housed initially in large cages in groups of six prior to challenge with an infectious agent and their subsequent experience after challenge and transfer to half suit isolator requiring paired housing. Study TB shows the welfare of guinea pigs before and after challenge, where use of the directional flow system allowed continued use of larger caging, groups of six and the potential for more enrichment strategies (Figure 4(b)). The CWAS for each study clearly shows the advantage of providing consistent larger group and cage size housing throughout a high containment study and how this can also have an impact on the procedural methodology and allow increased duration of the study and collection of more data without reducing the welfare.
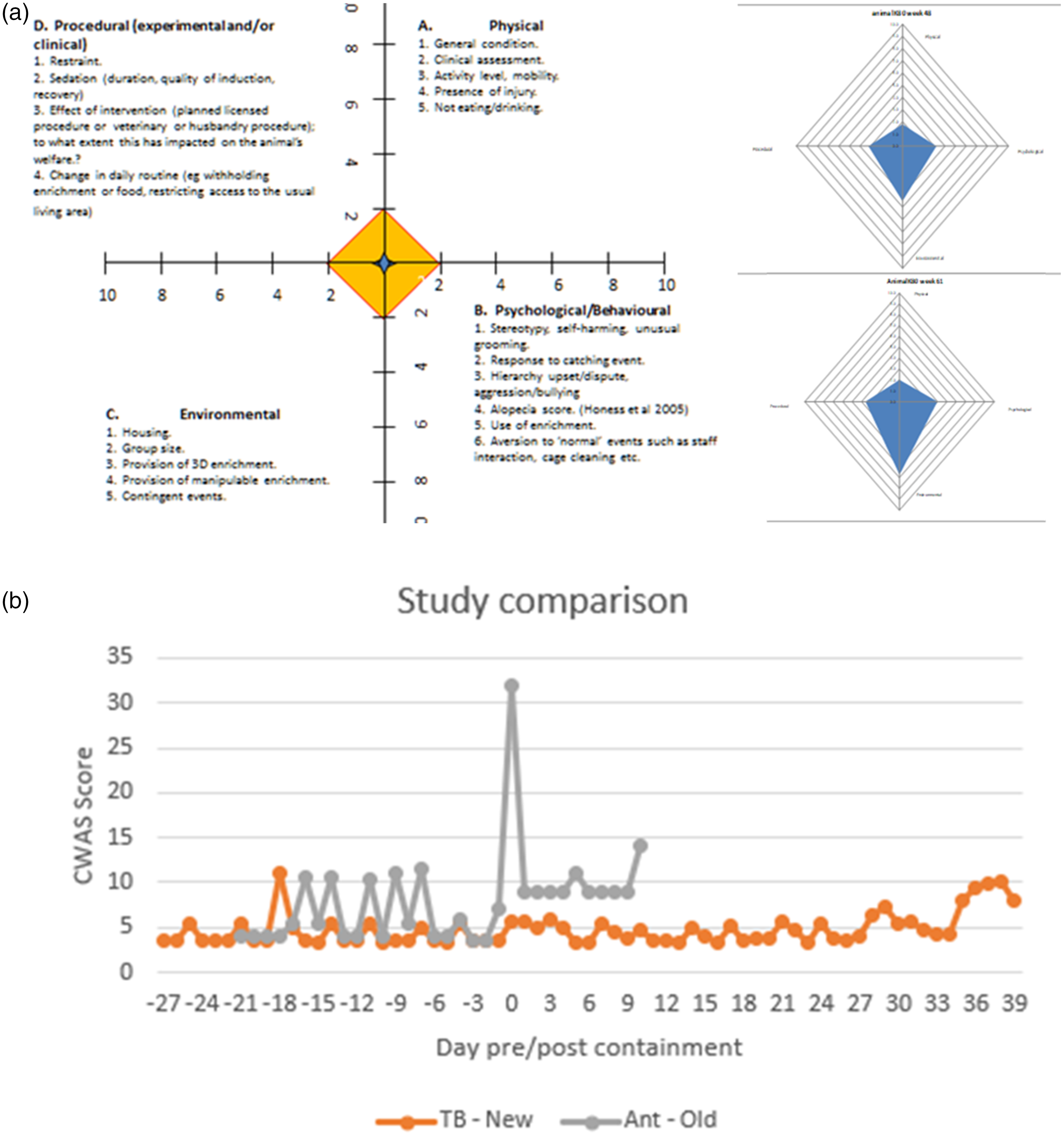
(a) AWAG assessment grid showing factors for each parameter (Physical, Psychological, Environmental and Procedural) and examples of radial plots of welfare. (b) CWAS of two comparable guinea pig studies, pre and post containment. The lower the score, the better the welfare. Pre-challenge housing for both studies is identical. Peaks indicate procedures such as vaccination or blood sampling. The large spike for study Ant indicates challenge with a pathogen, followed by pair-housing in an isolator compared with study TB where caging remains the same after challenge and transfer to higher containment, but has a better level of welfare. AWAG: Animal Welfare Assessment Grid; CWAS: cumulative welfare assessment score
The use of flexible film isolators for high level containment imposes considerable limitations on the size of caging that can be used and this impacts upon animal welfare in terms of group sizes and provision of enrichment. With the cooperation of research scientists, technical staff conducted a comparative trial of larger double tier caging within the directional flow containment originally developed for primates. These cages allowed groups of six animals to be housed together, compared with the isolator where they could only be housed in pairs in conventional wire-top boxes (Figure 1).
Technicians noted that the guinea pigs were able to exhibit a greater range of natural behaviours such as grooming, foraging in a deeper substrate and use of three-dimensional structures and hammocks.
This study showed that the guinea pigs made good use of higher structure that encouraged more exercise such as jumping. In addition, the animals were more relaxed in human presence and exhibited less stress when handled. In these larger cages staff were better able to assess and check the health of animals more efficiently as they could see behaviours more clearly in the home cage rather than the more stressful procedure of placing the guinea pig into a separate holding box for observation.
This was found to be of value in studies using respiratory pathogens when attributing clinical scores without the need for manipulation.
Advanced imaging
At UKHSA we use advanced imaging such as CT and PET to monitor the progress of disease in animals infected with HG3 agents and this provides powerful in-life data on disease pathogenesis and the efficacy of novel vaccines or therapeutic agents. Imaging not only provides additional experimental data but also addresses the 3Rs by reducing the number of animals required and by providing quantifiable assessment of disease that can inform on early humane endpoints, leading to refinement and the reduction of potential suffering. The use of advanced imaging modalities such as CT and PET-CT imaging presents additional problems in terms of safe containment during the transfer and imaging of subjects while also maximizing the use of such expensive equipment for different concurrent studies. Such equipment is complex and uses X-rays and radioactive materials that require specialized facilities with appropriate screening barriers that shield operators and adjacent facilities. In a multi-programme facility where more than one infectious agent may be in use at any given time, an additional problem must be considered.
This involves the decision to either avoid the contamination of imaging equipment or to decontaminate the equipment between scanning sessions or programmes of work. The challenge with such complex equipment is that many surfaces are either unreachable or may be sensitive to exposure to chemicals validated for successful decontamination by fumigation. Furthermore, investment in such technologies is considerable in terms of capital outlay, maintenance costs, and the hiring of specialized staff capable of designing suitable protocols for each species and operating the equipment successfully. Imaging strategies at establishments vary according to facility design and the species to be scanned.19 –23 In some, the scanning equipment is fully integrated into the containment facility, whereas others have adopted a strategy where the imaging equipment is located outside of, but adjacent to, the containment area and subjects are introduced for scanning through rigid containment tubes. To this end UKHSA has developed mobile flexible film isolators that operate under negative pressure and can be used to deliver subjects to a scanner and maintain them safely during the scanning process.5 Briefly, the mobile isolator or scanning pod has a main body for transportation of the subject with an integral rapid transfer port for docking with the containment suite and a narrower tubular section that fits into the tunnel of the scanning gantry (Figure 2). The scanning pod can be used to deliver subjects either to an in-house scanning suite or, as we have demonstrated, to a mobile scanning truck. When using PET-CT to scan subjects with respiratory pathogens it is essential to minimize respiratory motion during the relatively long process of image capture and we have achieved this through the use of high frequency jet ventilation. 24
Biosafety methods
For all our biocontainment strategies it is essential to provide rigorous testing data to quantify operator protection that will assure both the staff and the regulatory bodies that all equipment and processes are safe. For our directional flow systems and downdraught tables, the concept of operator protection factors (BS EN12469:2000) has been employed to indicate the performance, with a factor 10
4
being the target level; this level equates to a performance that is the same as that specified for Class I or II safety cabinets. UKHSA Porton has been using containment isolator technology for over a decade and has developed significant expertise in the design and validation of such systems. All of our flexible containment isolators undergo a full range of performance tests every six months, to ensure that continued high levels of containment and operator protection are provided. Tests outlined here ensure that the performance is in line with that of Class III glove boxes, as described in the current BS EN12469 standard (BS EN12469, 2000) and meets the current guidance from the UK regulator (ACDP, 1995, 1997).1,2 These tests include:
Tests of all HEPA filters using the DOP (dispersed oil particle) method, with 99.997% retention; Determination of airflow and air change rates, and maintenance of negative pressure; Positive pressure scan test, using DOP of the whole enclosed system, followed by pressure decay tests; Tests on the operation of alarms and indicators.
BS EN12469, 2000 is a European standard that sets the minimum performance criteria for safety cabinets for work with micro-organisms and specifies test procedures for microbiological safety cabinets with respect to protection of the worker and the environment, product protection and cross contamination.
Conclusion/discussion
This paper serves to illustrate the flexible approach required to adapt facilities to constantly changing experimental demands and technologies and the dependence on highly trained, skilled, adaptable and motivated staff to deliver meaningful scientific studies involving high consequence pathogens. Thus, there is an imperative for rigorous staff training and direct observational assessment at these levels together with a willingness by technical staff, scientific staff and management to promote new ideas on animal welfare improvement and evaluation. In addition, new biocontainment strategies can only evolve and become acceptable to regulators with robust evaluation by biosafety teams. The use of the AWAG to quantify animal welfare during the course of experimental studies has proved to be a powerful tool in demonstrating the value of improved caging, containment strategies and enrichment.
Footnotes
Ethics statement
All experimental studies referred to here were conducted under the UK Home Office Animals (Scientific Procedures) Act 1986 (ASPA) with authorization of project licences that had undergone local ethical scrutiny by the Animal Welfare and Ethical Review Body prior to approval by the UK Animals in Science Regulation Unit. All animals were housed in accordance with ASPA Guidance and Codes of Practice. 25
Supplementary material and availability of original data
A table of data contributing to the AWAG ![]() can be accessed as Supplementary material online, but further detail on this and on protocols regarding use of the containment system design, scanning modules and disinfection protocols can be accessed by interested parties using the contact
can be accessed as Supplementary material online, but further detail on this and on protocols regarding use of the containment system design, scanning modules and disinfection protocols can be accessed by interested parties using the contact
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
