Abstract
Summary
Inhalant anaesthetic agents are commonly used in studies investigating the electroencephalographic (EEG) effects of noxious stimuli in animals. Halothane causes less EEG depression than isoflurane, however, the EEG effects of halothane, isoflurane, sevoflurane and desflurane have not been compared in the same model. This study aimed to compare the EEG effects of these inhalational agents in the rat.
Forty male Sprague-Dawley rats were assigned to four groups and anaesthetized with halothane, isoflurane, sevoflurane or desflurane. EEG was recorded from the left and right somatosensory cortices for 5 min at three different multiples of minimal alveolar concentration (MAC) (1.25, 1.5 and 1.75). Median, 95% spectral edge frequency and total power were derived and a single mean value for each was calculated for the first 60 s of each recording period. When the raw EEG contained burst suppression (BS), the BS ratio (BSR) over 60 s was calculated. No BS was found in EEG recorded from the halothane group at any concentration. BS was present at all concentrations with the other anaesthetic agents. BS was almost complete at all concentrations of isoflurane, whereas BSR increased significantly with increasing concentrations of sevoflurane and desflurane. No significant differences were found between the BSR due to the 1.75 MAC multiple of isoflurane, sevoflurane or desflurane. Halothane causes significantly less depression of cortical activity than the newer inhalant agents at equivalent multiples of MAC. These data support the hypothesis that halothane has a fundamentally different mechanism of action than the other inhalant agents.
Minimal alveolar concentration (MAC) is commonly used to compare the potency of volatile anaesthetic agents (Eger 2002). MAC is defined as the concentration at which 50% of subjects respond to a supramaximal noxious stimulus with a purposeful movement response (Urban & Bleckwenn 2002). However, current evidence indicates that MAC does not primarily assess hypnotic properties and is more related to anaesthetic effects on the spinal cord rather than the effects in the brain (Antognini & Schwartz 1993, Rampil et al. 1993).
The central nervous system (CNS) effects of anaesthetic agents can also be assessed using electroencephalography, a direct measurement of drug effect on cerebral cortical activity. Volatile anaesthetic agents cause changes in the electroencephalogram (EEG) (Rehberg et al. 1999). Generally, as depth of anaesthesia increases with increasing anaesthetic agent concentration, the EEG slows towards high amplitude low frequency pattern (Clark & Rosner 1973). High concentrations of some anaesthetic agents cause burst suppression (BS), a pattern characterized by isoelectric EEG periods (2–10 s) interspersed with high voltage bursts usually lasting for 1–10 s (Yoshitani et al. 2003).
The EEG is also frequently used as a measurement tool in studies of noxious stimulation. Animals are commonly anaesthetized during these studies, so that the EEG effects of the anaesthetic agent must be taken into consideration during interpretation of changes caused by noxious stimulation. Studies have investigated the comparative EEG effects of noxious stimulation in rats anaesthetized with halothane or isoflurane and found that isoflurane blunted EEG changes to a greater extent than halothane (Barter et al. 2005).
Halothane is currently the anaesthetic agent of choice in the minimal anaesthesia model (Murrell & Johnson 2006). This model uses the EEG response to noxious stimulation as a tool to investigate analgesia for painful manipulations in applied veterinary research. Halothane is administered to a stable plane such that the animals are unconscious, judged on clinical assessment of anaesthetic depth, but still able to demonstrate EEG responses to noxious stimulation. A specified noxious stimulus is applied, such as velvet antler removal in deer, and changes in EEG variables used to compare the degree of cortical response. When compared with more traditional approaches to studies of potentially painful manipulations, this model has the advantage that a negative control group can be included without compromising the welfare of animals used in the study. It can also be used to compare the effectiveness of different analgesic regimens (Murrell et al. 2002, Haga & Ranheim 2005, Johnson et al. 2005, Murrell et al. 2005).
Manufacture of halothane is decreasing due to the decline in demand from human hospitals as newer agents such as sevoflurane and desflurane have become available. In the future, it is likely that halothane will no longer be readily obtainable as manufacture becomes commercially unviable. An alternative anaesthetic agent will have to be found for the minimal anaesthesia model, and models used by other groups investigating the EEG effects of noxious stimulation with halothane as the background anaesthetic agent. Inhalant anaesthetics are preferable to injectable agents for these models because the effect site concentration of the anaesthetic agent can be easily measured by monitoring the concentration of the inhalant agent in the expired respiratory gases. Manipulation and control of the effect site concentration of inhalant agents is also easier than for injectable agents because inhalant agents undergo little metabolism and are expelled from the body during respiration. In contrast, injectable agents must be metabolized, therefore control of drug concentration is more difficult, even when detailed pharmacokinetic information is available. The inhalant agent should also cause minimal depression of cerebral cortical activity.
The aim of the present study was to compare the EEG effects of halothane, isoflurane, sevoflurane and desflurane with the objective of identifying a suitable replacement for halothane in the minimal anaesthesia model. The EEG effects of these four agents have not been previously compared in a single investigation.
Materials and methods
The study was approved by the Massey University Animal Ethics Committee (approval 05/44). Forty male outbred Sprague-Dawley rats, aged between eight and 11 weeks old, weight (mean [SD]) 341.3 (56.7) g were studied. The animals were obtained from the Small Animal Production Unit, Massey University, NZ. Rats were housed in groups of six during rearing and were fed commercial rat chow ad libitum (rat diet 86, Feed Processing Unit, Massey University, NZ) up until the time of experimentation. Groups of six rats, housed in their original cage, were moved to the research laboratory daily and used for the experiments carried out that day. Free access to food and water were available up until the time of induction of anaesthesia. Animals were randomly allocated to one of four groups to receive halothane (Merial, Manukau City, NZ), isoflurane (Abbott laboratories, Queenborough, UK), sevoflurane (Abbott laboratories) or desflurane (Baxter Healthcare Corporation, Deerfield, IL, USA) for maintenance of anaesthesia during the experimental recording period. Induction of anaesthesia and electrode placement were carried out under halothane anaesthesia in all animals. Following electrode placement anaesthesia was maintained with the test anaesthetic agent only and a 15 min stabilization period allowed.
The rat was placed into a perspex custom-built anaesthetic chamber and anaesthesia was induced with 4% halothane vaporized in oxygen (3 L/min). Once visual inspection of the rat indicated adequate depth of anaesthesia, demonstrated by lateral recumbency and an absence of a withdrawal reflex to a toe pinch, orotracheal intubation was carried out with an 18-gauge cannula (Advantive, Viuret, Morelos, Mexico). The tracheal opening was visualized through the mouth using a transtracheal illumination technique. A guide-wire was threaded into the upper part of the trachea, over which the cannula was threaded. Following confirmation of successful intubation using capnometry (Hewlett Packard M1025B gas monitor, Palo Alto, CA, USA), the tracheal cannula was connected to a T-piece breathing circuit and anaesthesia was maintained with halothane vaporized in oxygen. Ventilation was controlled using IPPV (V valve ventilator, Vetronics, Bioanalytical Systems Inc, W La Fayette, IN, USA), adjusted to maintain end-tidal carbon dioxide concentration (ET [CO2]) between 5 and 5.5 kPa. The halothane concentration was adjusted to maintain adequate anaesthesia to prevent movement responses during electrode implantation. Monitoring during anaesthesia comprised end-tidal inhalant agent concentration, and ET [CO2] using the side stream agent monitor. The monitor was calibrated daily according to manufacturer's instructions. Rectal temperature was monitored continuously using a digital thermometer (Q 1437, Dick Smith Electronics, NZ). Body temperature was supported throughout anaesthesia with a circulating warm water blanket-heating device (T pump, Gaymar Industries Inc, NY, USA). The body of the rat was wrapped in the blanket (Mul-T-Pad, Gaymar Industries Inc) and the temperature of the water was set to 38°C.
The head of the rat was secured in a stereotaxic apparatus (Dual ultra precise small animal stereotaxic instrument, David Kopf Instruments, Tujunga, CA, USA) using a standard technique. The dorsal surface of the skull was shaved and the periosteum exposed by a midline skin incision. The periosteum was scraped back from the surface of the skull to expose the underlying bone and the bregma was located. Four silver-silver chloride electrodes (custom built, Massey University, NZ) were placed durally through 2 mm diameter holes drilled through the skull. Two active electrodes were placed over the left and right primary somatosensory cortices (2.5 mm caudal to bregma, 5.5 mm left and right of midline [left primary somatosensory cortex, S1L and right primary somatosensory cortex, S1R]). The reference electrode was placed over the frontal sinus (10 mm cranial to bregma, 1 mm left of midline) and a ground electrode was placed over the right frontal sinus (10 mm cranial to bregma, 1 mm right of midline). The electrodes were held in place with cold curing dental cement (Pro Base cold, Oral B Laboratories Pty Ltd, Maukau City, NZ) placed on the surface of the skull. The EEG was recorded continuously from the left and right somatosensory cortices. Signals from each channel of EEG were fed via custom-built ‘break-out’ boxes in a shielded cable to separate but identical amplifiers (Iso-Dam isolated biological amplifier, World Precision Instruments Inc, FL, USA). The signals were band-pass filtered between 0.1 and 100 Hz, amplified 100 times and fed to Powerlab 4/20 data recording system (Powerlab Data Acquisition System; AD Instruments Ltd, Sydney, Australia), which digitized the signal online at 1.0 kHz. The digitized signal was recorded on an Apple Macintosh personal computer.
Five minutes of EEG was recorded at three different multiples of MAC (1.25%, 1.5% and 1.75% MAC end-tidal concentration) for all anaesthetic agents (see Table 1). The order of administration (ascending or descending concentrations) was randomly divided between the animals in each anaesthetic group. Once the new anaesthetic concentration was attained a 10 min stabilization period was allowed before data collection started.
ET(hal) refers to end-tidal halothane concentration. iso, sevo and des refer to isoflurane, sevoflurane and desflurane, respectively
At the end of the experiment, rats were euthanized with an overdose of pentobarbital (Pentobarb 500, National Veterinary Supplies Ltd, Auckland, NZ).
Raw data from the EEG were inspected manually and any artefacts were excluded from further analysis. When no BS was found in raw EEG data, the total power (Ptot), F50 and F95 for 1 s epochs were calculated using specialized software (FFT program Craig Johnson). Data from each channel of EEG recording were analysed separately. For the purposes of statistical analysis, EEG data from the first 60 s of the five-minute block of EEG data recorded at each MAC multiple were compared. A single mean value for each EEG parameter was calculated for each of these time periods in every rat.
When the raw EEG data contained BS, the BS ratio (BSR) was calculated instead. Suppression was recognized as periods lasting longer than 0.5 s during which EEG amplitude was less than 5 mV. Time in the suppressed state was measured and BSR reported as the fraction of the epoch where the EEG was suppressed. Because of the non-stationary nature of EEG data, BSR was averaged over 60 consecutive epochs (60 s) of raw EEG to give a single percentage value for each five-minute block of EEG data where BS was found.
Statistical analysis
Statistical analysis of data was carried out with Prism 4 for Macintosh Version 4b (Graphpad Software Inc, San Diego, CA, USA). Statistical significance was assumed when alpha <0.05. Data from power spectral analysis (F50, F95 and Ptot) or BSR were compared between the different MAC multiple concentrations for each anaesthetic agent. Power spectral analysis data were normally distributed (Kolmogorov-Smirnov test) and were analysed using repeated measures analysis of variance (ANOVA) with Bonferroni post-tests when significant differences were identified. BSR data were not normally distributed and were analysed using the Friedman test with Dunn's post-tests when significant differences were found. The BSR for different anaesthetic agents at equipotent concentrations was compared using a one-way ANOVA test.
Results
No BS was found in the raw EEG recorded in the halothane group at any concentration. The mean (SD) values for F50, F95 and Ptot recorded during the different halothane concentrations from both EEG channels are shown in Table 2. F50 recorded from the S1L significantly increased with increasing halothane concentration (P < 0.01) (Figure 1), whereas changes recorded from S1R were not significant. Ptot decreased significantly with increasing halothane concentration in the S1L channel (Figure 2), whereas changes in the S1R channel were not significant. Halothane concentration did not significantly affect F95 in either EEG channel.
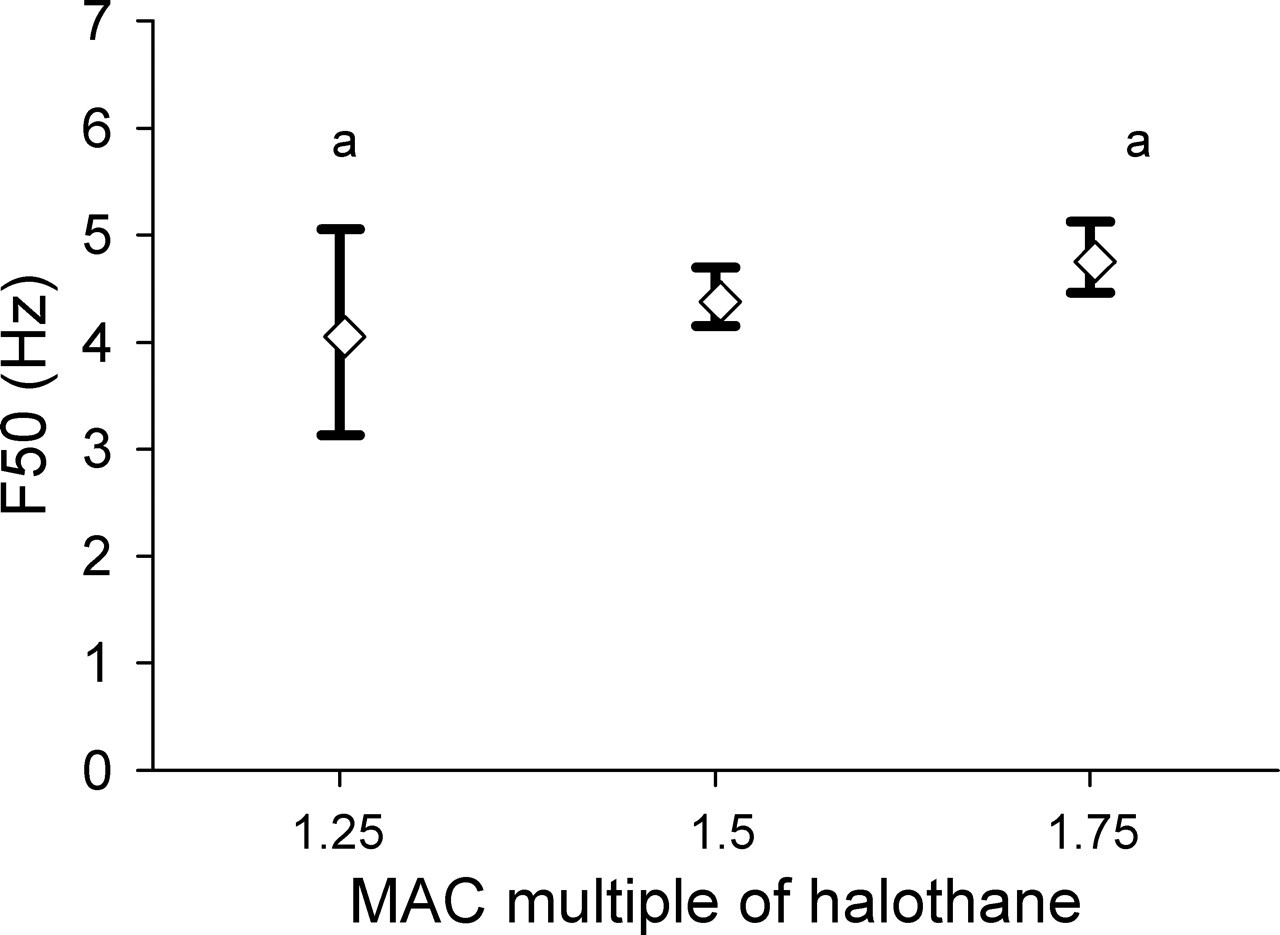
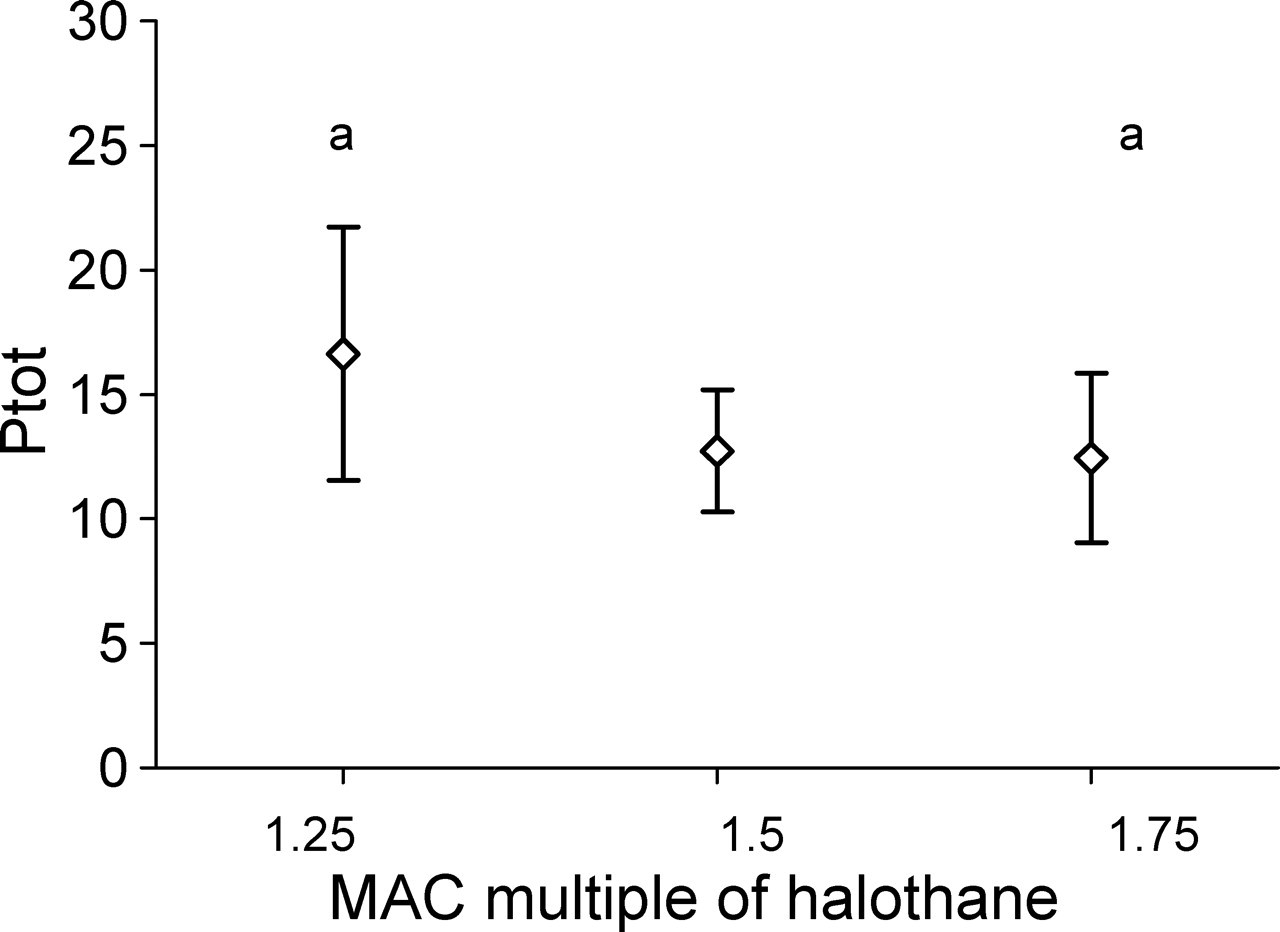
Paired symbols represent the values that are significantly different from each other
S1 refers to primary somatosensory cortex
F50, F95 and Ptot refer to median frequency, spectral edge frequency and total power, respectively
BS was recorded continuously at all concentrations with all other anaesthetic agents. The BS pattern was identical for both S1R and S1L channels, therefore the BSR was calculated for the S1R EEG channel only. The BSR was almost 100% and not significantly different between the different isoflurane concentrations, whereas the BSR increased significantly with increasing anaesthetic concentration with sevoflurane and desflurane (P ≤ 0.0001 and P = 0.0001, respectively) (Figure 3). Sevoflurane was associated with a significantly lower BSR when compared with either isoflurane or desflurane at 1.25 and 1.5% MAC (P = 0.0002 and P < 0.0001, respectively). There was no significant difference between the BSR caused by 1.75% MAC multiple of any anaesthetic agent except halothane.

Discussion
This is the first study to compare the EEG effects of equivalent MAC concentrations of halothane, isoflurane, sevoflurane and desflurane in the same model. We have shown that at equipotent concentrations (measured by MAC), cerebral cortical depression caused by these inhalant agents is very different, supporting the concept of MAC as a spinal rather than central phenomenon (Rampil et al. 1993). Halothane caused the least cortical depression, with no BS occurring at any test concentration. All the other agents caused BS, an EEG pattern characteristic of deep anaesthesia (Lukatch et al. 2005). The BSR increased with increasing concentration of sevoflurane and desflurane, whereas the BSR was almost 100%, at all concentrations of isoflurane, indicating that there was profound cortical depression at the lowest concentration of isoflurane studied (1.25% MAC).
Previous studies have compared the EEG effects of halothane with other inhalational agents, particularly isoflurane, and found halothane caused less EEG suppression than isoflurane at equipotent concentrations (Ono et al. 1997, Johnson and Taylor 1998, Antunes et al. 2003, Orth et al. 2006). The MAC of an anaesthetic agent is related to the ability of that agent to prevent response to a noxious stimulus. Johnson and Taylor (1998) suggested that the halothane is more antinociceptive than isoflurane because the response to a standard nociceptive stimulus was prevented by a concentration of halothane that induced less CNS depression than the required concentration of isoflurane. This argument can also be extended to the present study. Our results suggest the following order of antinociceptive potency for the test inhalational agents: halothane > sevoflurane > desflurane > isoflurane. The position of isoflurane in this schema is at odds with the relative potency as defined by MAC value, a measure determined by antinociceptive efficacy in a rigorous test model (Urban & Bleckwenn 2002).
Ogawa et al. (1992) studied background electrical activity in midbrain reticular cells during volatile agent anaesthesia in cats as well as reactive capability by evoked responses in the visual neuronal pathway. Halothane caused less depression of background electrical activity compared with isoflurane and enflurane, a finding similar to the present investigation, whereas reactive capability was suppressed by halothane to a greater extent than isoflurane. The comparative effects of halothane and isoflurane on reactive capability are in accordance with their relative MAC values. The suppressive actions of halothane, sevoflurane and isoflurane on the response capability of the midbrain reticular formation to peripheral nerve stimulation in cats have also been compared (Tsushima et al. 1998). In accordance with the previous study (Ogawa et al. 1992), halothane caused less suppression of basal EEG activity, but had a greater effect on the response capability to peripheral stimulation than isoflurane or sevoflurane.
Increasing the concentration of halothane had comparatively little effects on the EEG, suggesting that the dose–response curve for the EEG parameters investigated was relatively flat over the concentration range investigated. The increase in F50 and decrease in Ptot recorded from the left somatosensory cortex was statistically significant between 1.25 and 1.75% MAC, however, these changes were numerically small and unlikely to be of biological significance. These changes are also opposite to the expected change, as increasing anaesthetic agent concentration is usually associated with an increase in EEG amplitude and decrease in frequency (Johnson & Taylor 1998). Changes in F50 only occurred in one cerebral hemisphere, and F95 remained stable in both hemispheres, also supporting the minimal biological significance of these changes.
Johnson and Taylor (1998) also studied changes in F50 and F95 with increasing halothane concentration and found that F95 decreased progressively as concentration increased. However these authors investigated 0.8% to 1.2% MAC halothane concentrations, lower than the present study. It is probable that the dose–response curve for F95 plateaus around 1.25% MAC. BS is rarely reported during halothane anaesthesia (Lloyd-Thomas et al. 1990), even at high concentrations (Antunes et al. 2003). The relatively little depression of cerebral electrical activity during halothane anaesthesia when compared with the other test agents emphasizes the suitability of halothane for studies investigating EEG changes during noxious stimulation and for use in the minimal anaesthesia model.
Although BS is commonly recognized during deep anaesthesia, the underlying neuronal events that cause this EEG pattern are not fully understood. Steriade et al. (1994) found hyperpolarization of cortical neurons reliably preceded overt EEG BS and attributed the hyperpolarization to an increase in GABAergic inhibitory processes at both thalamic and cortical synapses. Others have suggested that both glutamate-mediated excitatory transmission and GABAergic activity are required for the generation of BS during anaesthesia (Lukatch et al. 2005). Volatile anaesthetics are recognized to depress glutamate transmission via a pre-synaptic mechanism, and enhance gamma-aminobutyric acid (GABA) inhibition (MacIver et al. 1996). Therefore the presence or absence of BS will depend partly on the relative balance of glutamate and GABAergic activity, influenced by anaesthetic agent concentration. The precise neurological mechanism leading to BS is also likely to vary with anaesthetic agent (Akrawi et al. 1996).
In terms of BS, the dose–response curves for isoflurane, desflurane and sevoflurane were different over the concentration range tested, although BS was almost complete with all agents at 1.75% MAC concentration. Despite the association between BS and deep anaesthesia, the cortex remains responsive to stimuli during BS induced by volatile anaesthetic agents (Porkkala et al. 1997, Hartikainen & Rorarius 1999, Huotari et al. 2004). Somatosensory and auditory stimuli cause evoked potentials and trigger bursts during EEG suppression with isoflurane or sevoflurane (Porkkala et al. 1997, Hartikainen & Rorarius 1999, Rytky et al. 1999). These studies indicate that the brain retains the ability to respond to external stimuli during deep anaesthesia. Decreasing cortical responsiveness due to increasing anaesthesia is also not a linear process through all stages of anaesthesia. Deep anaesthesia with volatile agents seems to cause generalized depression of background activity and a facilitation of stimulation-induced responses (Hartikainen and Rorarius 1999). As hypothesized by Johnson and Taylor (1998), this paradoxical effect may also explain the disparity between the degree of EEG suppression and the relative MAC values of the agents tested in the present investigation.
Other studies have found the EEG effects of isoflurane, sevoflurane and desflurane to be similar (Rampil et al. 1988, Hoffman & Edelman 1995, Schwender et al. 1998, Rehberg et al. 1999, Freye et al. 2004). However some of these investigations were clinical studies, where other anaesthetic drugs were given concurrently, which may have masked differences between volatile agents (Hoffman & Edelman 1995, Schwender et al. 1998, Freye et al. 2004). The numbers of individuals included in some studies were also small (Rampil et al. 1988, Hoffman & Edelman 1995, Schwender et al. 1998, Freye et al. 2004). The present investigation used 6% end-tidal concentration of desflurane as MAC was not determined in the present investigation (Dimaculangan et al. 2006), whereas many other studies used 5.7% (Eger & Johnson 1987). When compared with studies that used 5.7%, we administered a higher concentration of desflurane relative to isoflurane and sevoflurane, which may have resulted in relatively greater cortical depression with desflurane in the present investigation. However, it would not have influenced the relative EEG data for sevoflurane and isoflurane. Aberrations in physiological parameters, such as body temperature and carbon dioxide concentration, can also influence the EEG (Paulson & Sharbrough 1974, Kochs 1995), but these were controlled within normal physiological limits in all animals. Each end-tidal concentration was maintained constant for at least 10 min in an attempt to reach equilibration between alveolar gas (end-tidal concentration), arterial blood and brain before EEG recording started. Recovery from halothane anaesthesia is slower than after the other test anaesthetic agents, particularly desflurane, which is associated with a very rapid return to consciousness after cessation of administration. Intubation of the rats took approximately 3 min, during which time administration of an inhalant agent by face-mask was not possible. It is likely that rats induced with desflurane would have regained consciousness before intubation was complete. Therefore, in order to facilitate intubation and standardize the experimental procedure halothane was used for induction of anaesthesia in all animals. A 15 min washout period was allowed between halothane, used for induction of anaesthesia and electrode placement, and the test volatile agent (Quasha et al. 1980). EEG data recorded in animals that received an ascending concentration schema were not different from animals assigned a descending schema, suggesting that the 10 min period was adequate. However, a possible interaction between residual halothane following induction and the test anaesthetic agent cannot be completely discounted.
The results of this study highlight the differences between the EEG effects of halothane compared with other modern inhalant agents. In the unstimulated resting state, background cortical electrical activity is minimally depressed by halothane compared with the other agents, rendering it ideal for use in the minimal anaesthesia model. The other agents all caused BS at concentrations necessary to provide surgical anaesthesia. However, other studies suggest that despite the greater suppression of background EEG, cortical responsiveness to stimulation may be facilitated with the newer agents compared with halothane. Caution must therefore be exercised when comparing EEG data recorded during studies using different volatile anaesthetic agents. If halothane becomes commercially unavailable in future, an alternative model may comprise EEG recordings made under deep volatile agent anaesthesia, creating an environment of enhanced cerebral responsiveness to noxious stimuli. However, the clinical relevance of studies carried out under very deep anaesthesia may be questionable. Differences between neuronal responsiveness and background EEG suppression with different volatile agents at equipotent concentrations supports a multisite action hypothesis as the mechanism of action of this class of anaesthetic agents.
Footnotes
Acknowledgement
This project was funded by the Massey University Research Fund (MURF).
