Abstract
Male obese Zucker Diabetic Fatty (ZDF) rats develop type 2 diabetes around eight weeks of age, and are widely used as a model for human diabetes and its complications. The objective of the study was to test whether the complications manifested in the kidney and nerves of ZDF rats really correspond to human diabetic complications in their being related to the hyperglycaemic state. Four groups of ZDF rats were used. One lean (Fa/?) and one obese (fa/fa) untreated group served as non-diabetic and diabetic controls. In two further groups of obese (fa/fa) rats, diabetes was prevented by pioglitazone or delayed by food restriction. All rats were monitored up to 35 weeks of age with respect to their blood glucose, HbA1c and insulin levels, their kidney function (urinary glucose excretion, renal glucose filtration, glomerular filtration rate, albumin/creatinine ratio), and their nerve function (tactile and thermal sensory threshold and nerve conduction velocity). Pioglitazone prevented the development of diabetes, while food restriction delayed its onset for 8–10 weeks. Accordingly, kidney function parameters were similar to lean non-diabetic rats in pioglitazone-treated rats and significantly improved in food-restricted rats compared with obese controls. Kidney histology paralleled the functional results. By contrast, nerve functional evaluations did not mirror the differing blood glucose levels. We conclude that the ZDF rat is a good model for diabetic nephropathy, while alterations in nerve functions were not diabetes-related.
Microvascular late complications of diabetes mellitus represent an important unmet medical need. Diabetic nephropathy is the most common cause of end-stage renal disease in developed countries. 1 It starts with microalbuminuria and progresses to macroalbuminuria, decreased glomerular filtration rate (GFR) and finally renal failure. 2 Up to 50% of diabetic patients develop peripheral polyneuropathy. 3 They suffer from pain, paraesthesia, hypersensitivity or sensory loss and numbness in the distal extremities. 4 Tight glycaemic control reduces the risk of late complications, 5–8 although it is not able to eliminate it. Symptomatic treatment provides improvement of quality of life, but cannot prevent progression of disease. Treatments targeting the pathophysiological alterations underlying microvascular complications are needed, but so far, no approach has proven fully satisfactory.
For the development of new therapeutic options for the microvascular complications, animal models that mirror the pathogenesis in humans are essential. Male Zucker Diabetic Fatty (ZDF) rats with
Treatment of prediabetic ZDF rats with a PPARγ agonist normalizes blood glucose through an insulin-sensitizing effect and preserves beta cell function.
18,19
As obesity of
The aim of the current study was to evaluate whether nephropathy and neuropathy found in male diabetic ZDF rats are linked to the hyperglycaemic state of the animals and are therefore real diabetic late complications, or rather related to other characteristics of the
Materials and methods
Animals
The study was performed under the terms of the German Animal Protection Law and in accordance with the Sanofi-Aventis Company Charter on the Humane Care and Use of Laboratory Animals. Male lean (genotype
Experimental design
At the age of seven weeks, rats were allocated to one of four groups (
Blood parameters
Tail tip capillary blood was collected weekly for determination of glucose and HbA1c. Every four weeks, blood was sampled from the retrobulbar venous plexus under light isoflurane anaesthesia for analysis of serum insulin and creatinine. All metabolic parameters were determined under fed conditions.
Urinary parameters
At four-week intervals, rats were placed individually in metabolic cages for 24 h and urine was collected. Urine volume was determined and urinary glucose, albumin and creatinine concentrations were analysed. Urinary glucose excretion (UGE), renal glucose filtration (RGF), urinary albumin/creatinine ratio (ACR) and GFR were calculated.
Analytical procedures of blood and urinary parameters
Standard procedures were used to determine blood glucose, HbA1c, serum creatinine, urinary glucose, urinary albumin and urinary creatinine. 21 Serum insulin concentrations were assayed with an enzyme-linked immunosorbent assay obtained from Mercodia AB (Uppsala, Sweden).
Sensory thresholds and nerve conduction
Every four to six weeks, tactile and thermal sensory thresholds were assessed. Tactile withdrawal thresholds were measured with a Dynamic Plantar Aesthesiometer (Ugo Basile, Comerio, Italy). Rats were placed in a cage with wire mesh floor and allowed to acclimatize for several minutes. The testing device was placed under the plantar face of the hind paw and applied an incremental force (measured in grams) via a metal rod (diameter 0.5 mm), until the animal withdrew the paw. A cut-off value of 50 g was set to prevent tissue damage. Testing was repeated on the contralateral paw and the mean withdrawal threshold was calculated for each animal. Thermal withdrawal thresholds were measured using the tail flick test (Ugo Basile). Rats were gently restrained on the testing apparatus and the tail tip was placed over a radiant heat source (157 mW/cm2). Time from the onset of heat stimulus to tail withdrawal was recorded. Cut-off point was set to 16 s to prevent tissue damage.
Nerve conduction parameters of the tail nerve were measured every two to four weeks. Rats were lightly anaesthetized with isoflurane. Needle electrodes were inserted subcutaneously into the tail, with the recording electrode at the tail base and the stimulating electrode placed 5 cm distally. Supramaximal stimulation pulses of 0.2 ms duration were applied, and amplitude, duration and latency of the resulting whole nerve action potential were recorded. Maximum nerve conduction velocity was calculated from the latency between stimulus onset and beginning of the action potential and the distance between stimulating and recording electrode.
Kidney histology
At 35 weeks of age, rats were euthanized; kidneys were taken out, weighed and fixed in 10% buffered formalin. Tissues were processed by routine procedures. The specimens were stained with haematoxylin-eosin and periodic acid Schiff. A longitudinal section of one kidney per animal was examined. Absolute incidences of renal lesions were recorded, and the extent of lesions was assessed semi-quantitatively by an examiner blinded to the treatment of the animal. Damage to individual glomeruli and distribution of the damaged glomeruli were graded in a combined scheme of glomerulopathy, which reflects the experience in a semi-quantitative scoring system. The grading score describes severity (A) and quantity (B) in each slide according to the following definitions:
(A) (B)
A similar semi-quantitative scheme was applied to assess tubulointerstitial damage.
Statistical analysis
Data are expressed as means ± SEM. Statistical significance was determined only for endpoint values of each parameter by one-way analysis of variance with Bonferroni's multiple comparison post-test using the GraphPad Prism® 5 software.
Results
Kidney histology. Incidence and average grading of glomerulopathy, tubulopathy and Armani-Ebstein cells in kidneys of lean and obese untreated ZDF rats and obese ZDF rats treated with pioglitazone or food restriction
Metabolic parameters
Body weight of obese control rats was higher than that of lean rats at the beginning of the study, but then started to decline at around 16 weeks of age. At 25 weeks of age, lean rats reached the same body weight as obese rats and finally grew slightly heavier. Food restriction did not influence body weight of obese rats. Pioglitazone resulted in an enormous boost in body weight, which came to be more than 1000 g (Figure 1a). The three groups of obese ZDF rats showed hyperinsulinaemia at the start of the study. Serum insulin of obese untreated rats dropped to the level of control rats during the trial. Rats on food restriction showed prolonged hyperinsulinaemia and delayed decline. Pioglitazone preserved increased insulin secretion throughout the study (Figure 1b). Lean control rats had stable blood glucose values of around 6 mmol/L and HbA1c values of around 4%. Blood glucose of all three groups of obese rats was already elevated at the beginning of the study and rose to 28 mmol/L in obese control rats. Food restriction as well as pioglitazone treatment initially resulted in normoglycaemia, which was maintained during the study period in the pioglitazone group. In the food-restriction group, blood glucose started mounting again after four weeks of treatment and reached 20 mmol/L after eight weeks. Final blood glucose values were 22 mmol/L in this group (Figure 1c). HbA1c values started at 5% in obese control rats and ascended to around 9%. HbA1c of food-restricted obese rats stayed constant at 5% until 14 weeks of age and then increased to reach a level slightly below that of obese controls between 20 and 24 weeks of age. Pioglitazone treatment normalized HbA1c levels to that of lean control rats (Figure 1d).
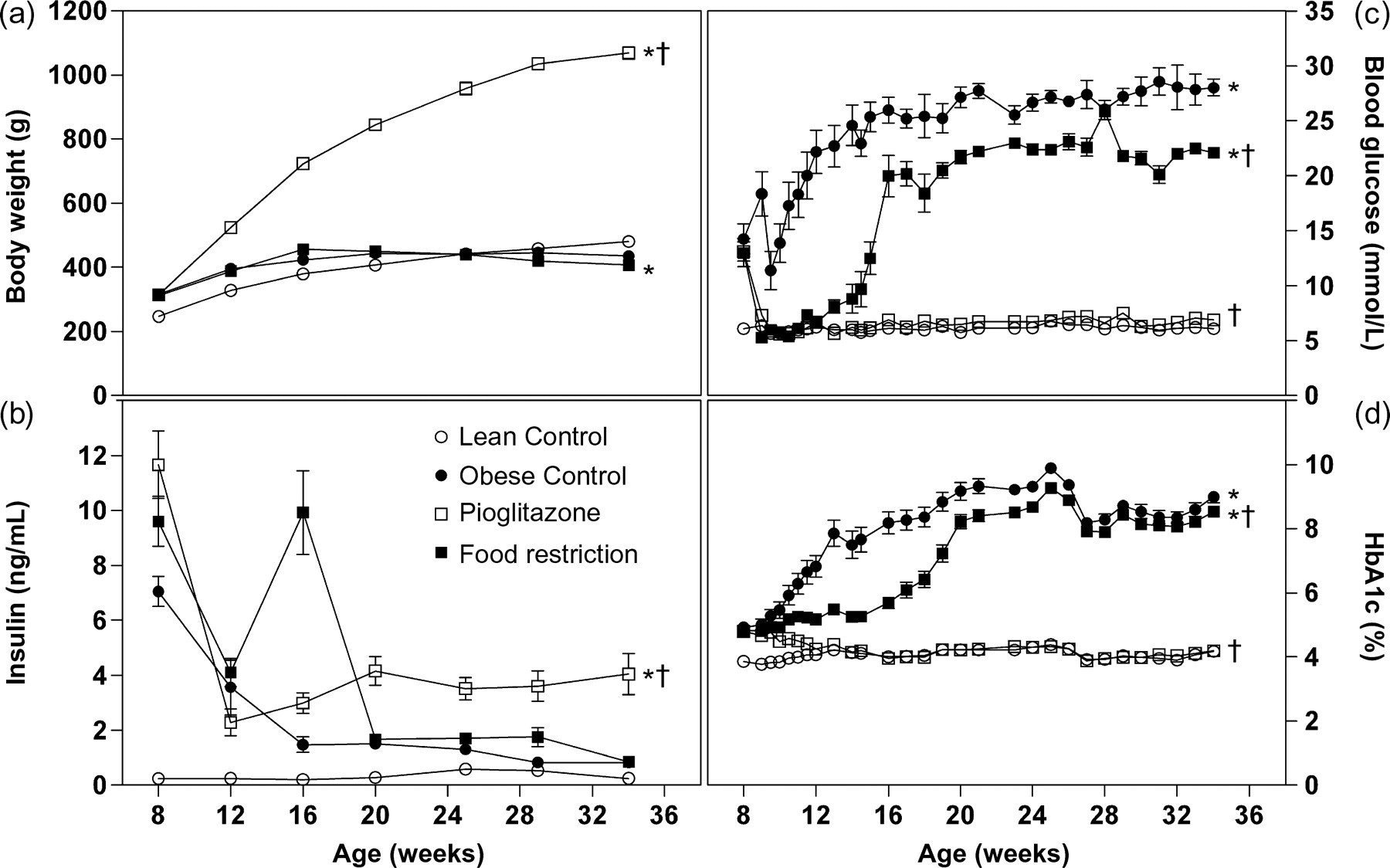
Body weight (a), serum insulin levels (b), blood glucose (c) and HbA1c values (d) of lean (white circles) and obese untreated ZDF rats (black circles) and obese ZDF rats treated with pioglitazone (white squares) or food restriction (black squares) (
Kidney function and histology
Lean control rats and rats treated with pioglitazone never showed UGE during the trial. Obese control rats already had high UGE at 12 weeks, while UGE of food-restricted rats augmented from week 12 to week 20, but did not reach the level of untreated obese rats (Figure 2a). RGF showed a similar time course, with high levels in untreated obese rats from week 12 onwards and an increase from near normal levels to clearly elevated ones in food-restricted obese rats. Pioglitazone treatment held RGF on normal levels (Figure 2b). Urinary albumin excretion rose massively and progressively in obese control rats and to a much smaller degree in obese rats on food restriction. Pioglitazone prevented urinary albumin excretion (Figure 2c). Urinary ACR showed progressive increase in obese control rats and – to a lesser extent – in obese rats with food restriction, while it was constantly low in lean controls and pioglitazone-treated rats (Figure 2d). GFR was augmented in obese controls and in the food-restriction group compared with controls, and slightly diminished in the pioglitazone group (Figure 2e). Serum creatinine was slightly reduced in obese control and pioglitazone-treated rats compared with lean controls and even more reduced in obese food-restricted rats (Figure 2f).
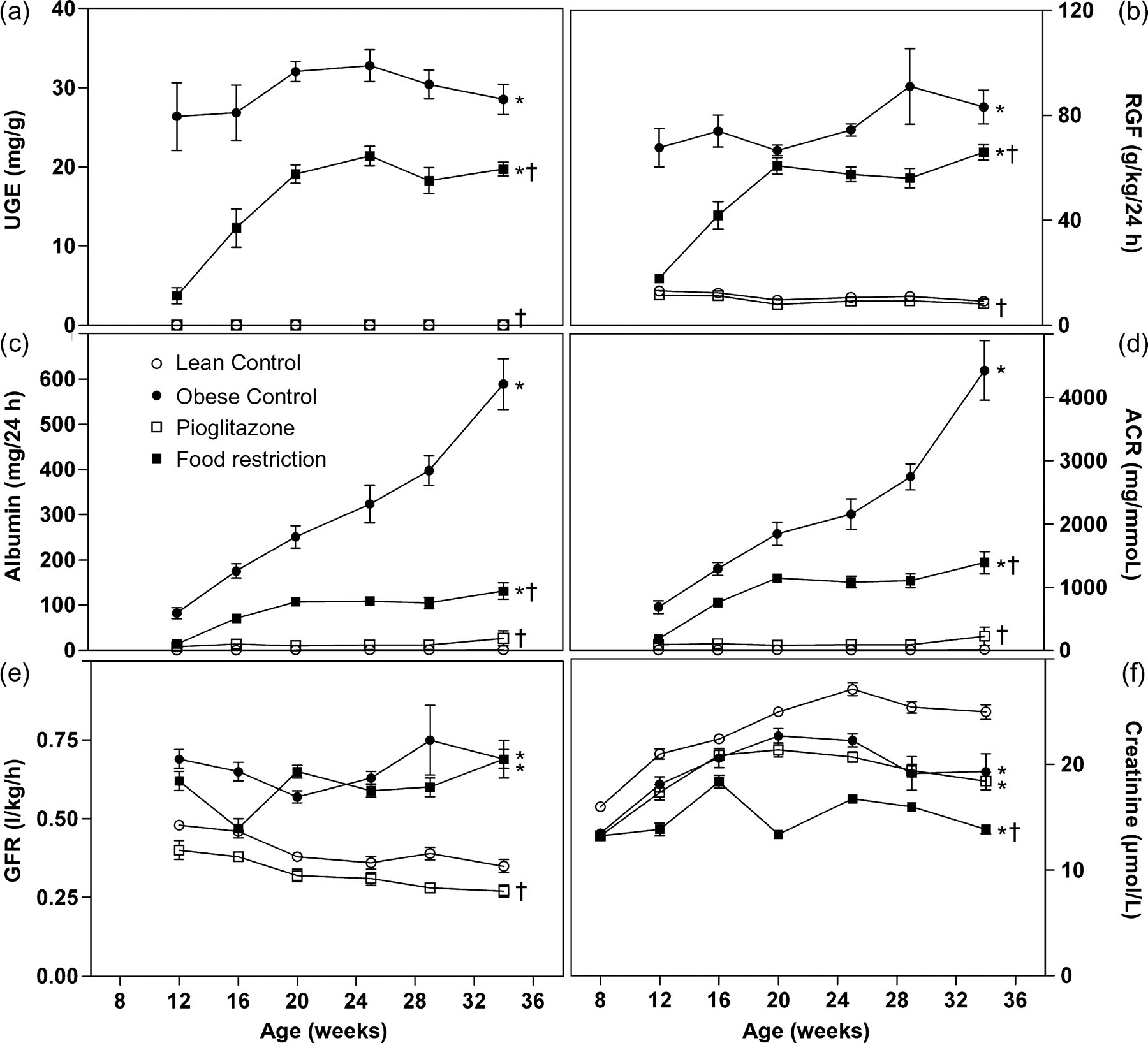
Urinary glucose excretion (UGE) (a), renal glucose filtration (RGF) (b), urinary albumin (c), urinary albumin/creatinine ratio (ACR) (d), glomerular filtration rate (GFR) (e) and serum creatinine (f) of lean (white circles) and obese untreated Zucker Diabetic Fatty (ZDF) rats (black circles) and obese ZDF rats treated with pioglitazone (white squares) or food restriction (black squares) (
Absolute kidney weight at autopsy was significantly elevated in the obese control versus lean control group. Both treatment groups had significantly lower kidney weight compared with obese controls, but kidney weight of the pioglitazone group was still significantly higher than that of lean controls (Figure 3a). Relative kidney weight related to body weight was also significantly raised in the obese control versus lean control group. Relative kidney weights of food-restricted rats were between those of lean and obese control rats. Pioglitazone-treated rats had lower relative kidney weights than lean controls (Figure 3b).
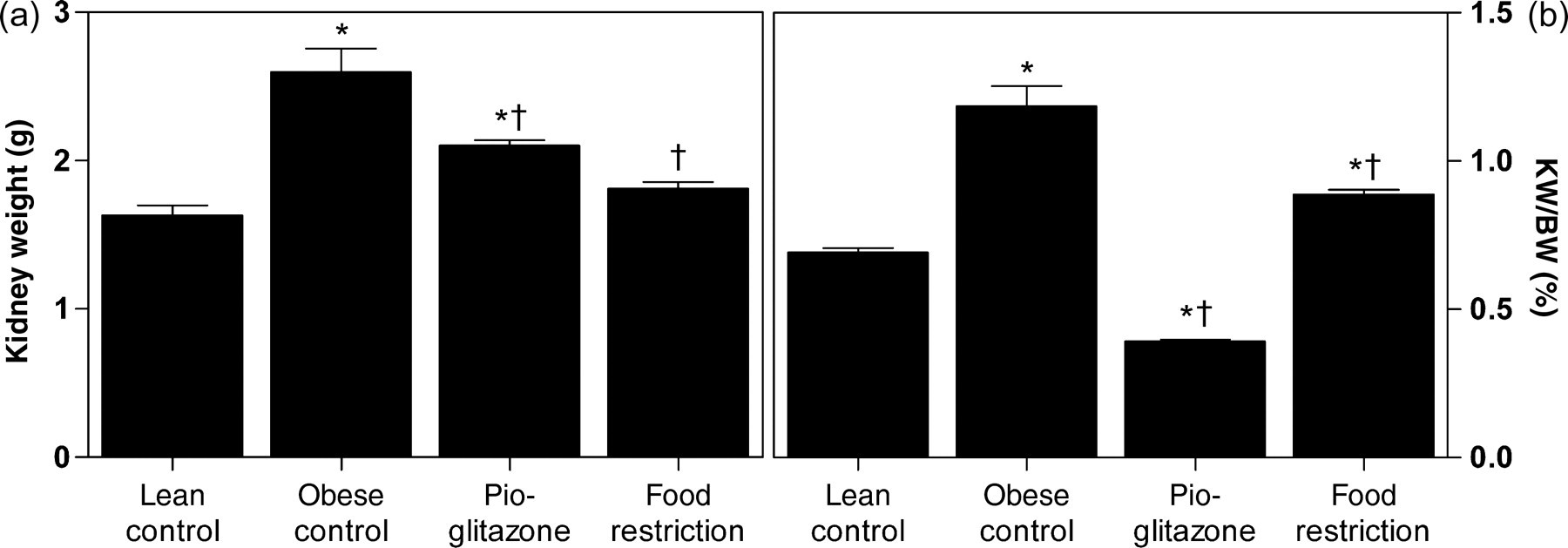
Absolute kidney weight (a) and relative kidney weight per body weight (b) of lean and obese untreated Zucker Diabetic Fatty (ZDF) rats and obese ZDF rats treated with pioglitazone or food restriction (
None of the lean control rats showed kidney glomerulopathy or tubulopathy, while 100% of obese untreated rats did, with an average scoring of 3.2 for both pathologies. Food-restricted rats also had 100% incidence for glomerulopathy and tubulopathy, but with milder appearance (average scores 1.4 and 1.5). Pioglitazone-treated rats did not show glomerulopathy and only 50% incidence of mild tubulopathy (score 1.7) (Table 1 and Figure 4). Armani-Ebstein cells as a correlate of glucosuria were present in all diabetic control rats (score 1.6) and in 88% of diabetic food-restricted rats (score 1.3), but not in lean controls and in pioglitazone-treated rats (Table 1).
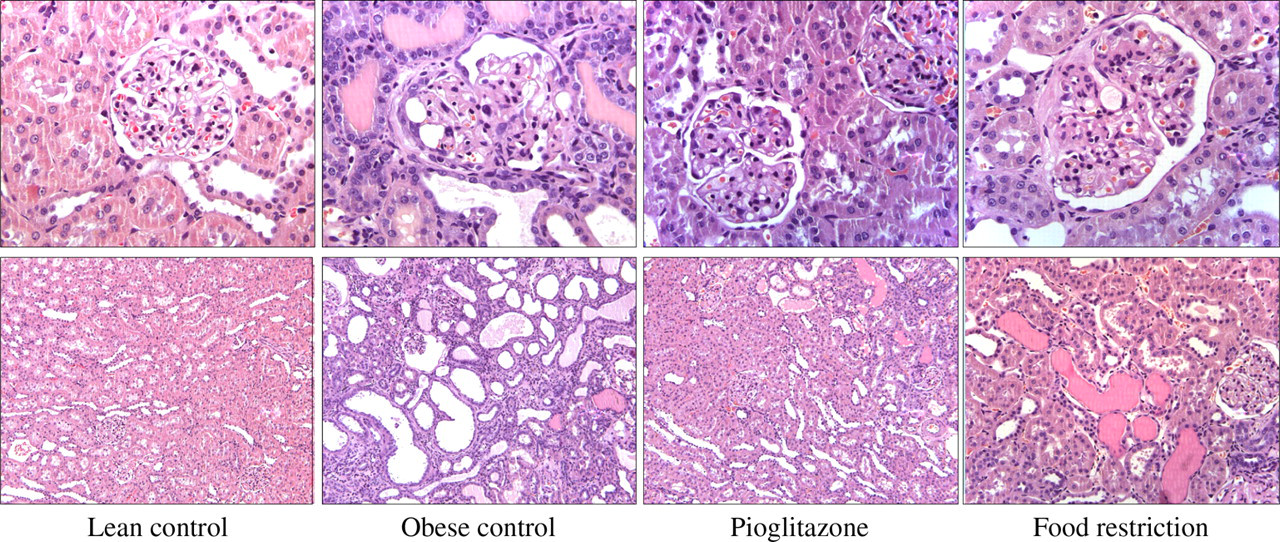
Kidney glomeruli (first row) and tubuli (second row) of lean and obese untreated Zucker Diabetic Fatty (ZDF) rats and obese ZDF rats treated with pioglitazone or food restriction. Representative histological slices of results are summarized in Table 1
Nerve function
Tactile withdrawal thresholds measured with the Dynamic Plantar Aesthesiometer were not different between lean and obese control rats and obese rats with food restriction. Only pioglitazone-treated rats showed slightly lower withdrawal thresholds in the beginning (Figure 5a). Thermal withdrawal thresholds measured with the tail flick test were lower in the three groups of obese rats, treated and untreated, compared with lean control rats (Figure 5b). Maximum nerve conduction velocities of the tail nerve were not different among the four groups (Figure 5c). The amplitude of the nerve action potential was significantly higher in untreated obese rats compared with the three other groups (Figure 5d), while the duration of the action potential did not differ between groups (data not shown).
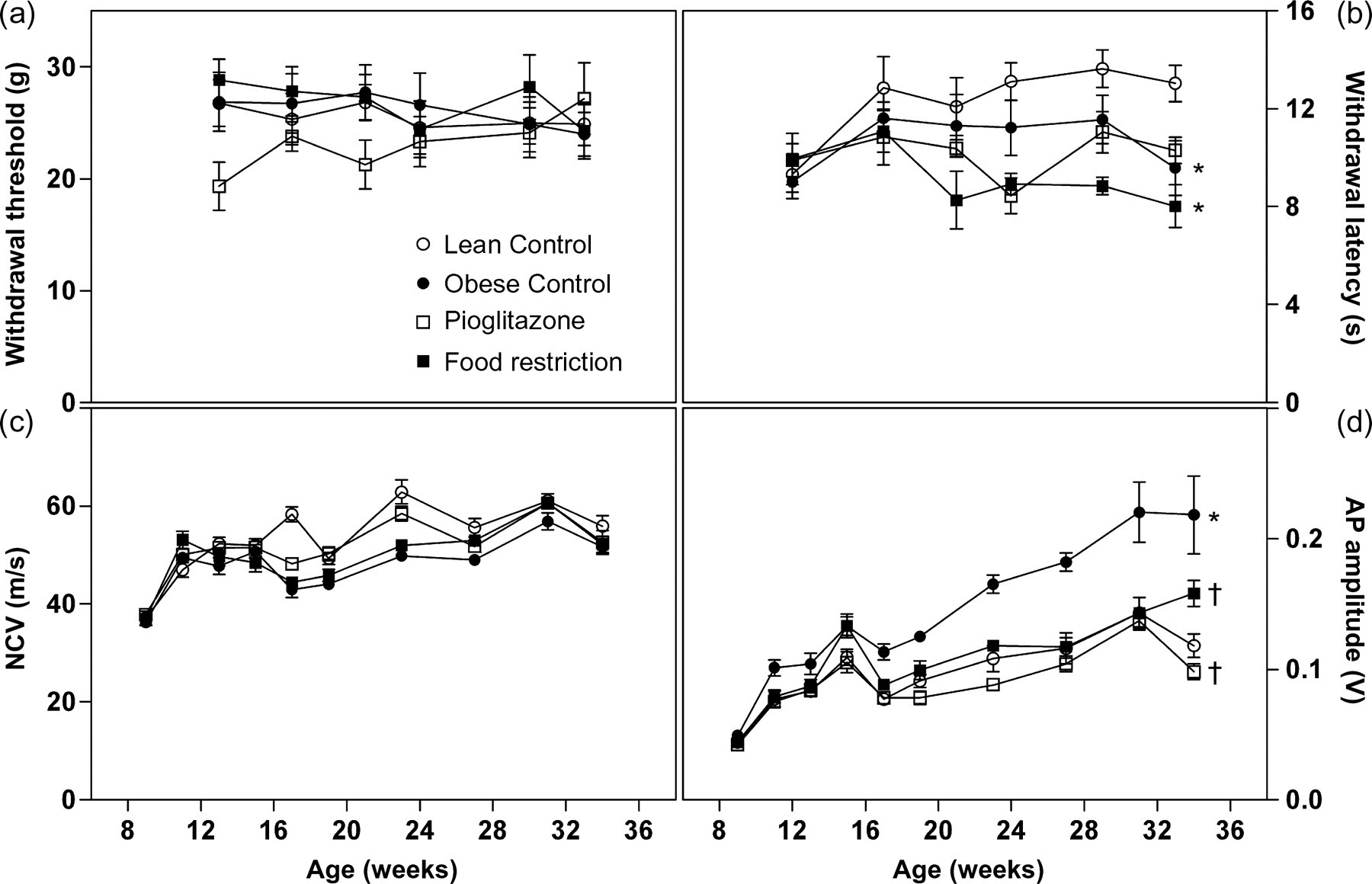
Paw withdrawal thresholds measured with the Dynamic Plantar Aesthesiometer (a), tail withdrawal latency in the tail flick test (b), nerve conduction velocity (c) and amplitude of the nerve action potential (d) at the tail nerve of lean (white circles) and obese untreated ZDF rats (black circles) and obese ZDF rats treated with pioglitazone (white squares) or food restriction (black squares) (
Discussion
Metabolic data of the present study confirm that treatment of prediabetic male ZDF rats with the PPARγ agonist pioglitazone completely prevents development from insulin resistance to overt diabetes due to its insulin-sensitizing effect.
18,19
Pioglitazone-treated animals remain hyperinsulinaemic but normoglycaemic. Food restriction to 70% of the amount consumed by
Taken together, metabolic data allow the three groups of obese male ZDF rats to be regarded as a non-diabetic group of
RGF and UGE augment in parallel with blood glucose levels during diabetes progression, and this was observed in untreated and in food-restricted obese ZDF rats. Lean control rats had constantly low RGF and no UGE, and so had pioglitazone-treated obese rats. The first clinical sign of nephropathy is progressive albuminuria with a rising urinary ACR. Both parameters showed major increase in obese control rats and a milder increase in the food-restriction group, but no change compared with healthy controls in pioglitazone-treated rats. If nephropathy progresses to renal failure, GFR decreases and serum creatinine increases. This did not occur in diabetic ZDF rats during the study period. On the contrary, GFR was higher and serum creatinine lower than in healthy controls. Glomerular hyperfiltration is commonly observed in early diabetic nephropathy 22,23 and is probably linked to poor glycaemic control. 24 Maybe osmotic effects of high RGF are involved in this phenomenon. Lower serum creatinine levels of diabetic rats might be explained by increased urinary creatinine loss due to the large urine volume excreted. In addition, obese ZDF rats have less muscle mass than lean ZDF rats, and serum creatinine levels are proportionally influenced by muscle mass. The lack of decrease in GFR and increase in serum creatinine indicate that ZDF rats display only mild nephropathy without progression to renal failure up to 35 weeks of age.
Kidney enlargement is another characteristic of nephropathy 25 observed in the untreated diabetic ZDF group. Absolute kidney weight was not increased in the food-restriction group, but correction to body weight showed an increase of the relative kidney weight in this group as well, although it was less pronounced than in the untreated group. Pioglitazone-treated rats had slightly elevated absolute kidney weights, but as their body weight was massively augmented, relative kidney weight was significantly lower than that of healthy controls. Correction to lean body mass would have been more appropriate for comparison, but this parameter has not been determined in the present study.
Histological evaluation of the kidneys revealed no sign of nephropathy in 35-week-old healthy control rats. Untreated diabetic ZDF rats showed pathological changes with an incidence of 100% in glomeruli and tubuli similar to those seen in human diabetic nephropathy. Changes were moderate to marked. In the food-restriction group, the same changes were observed with the same incidence, but only graded as minimal to small. In the non-diabetic group of pioglitazone treatment, glomerulopathy was fully prevented and only slight tubulopathy could be seen in 50% of the animals.
Taken together, these data clearly show that male diabetic ZDF rats develop clinical and histological signs of progressive nephropathy and that this is related to the hyperglycaemic state of the animals, as nephropathy development can be prevented by diabetes prevention and at least slowed down by a delayed onset of diabetes and reductions of blood glucose and HbA1c. Therefore, nephropathy seen in male diabetic ZDF rats can be regarded as a model of human diabetic nephropathy, which is also influenced by the quality of glycaemic control.
For neuropathy, results are far less conclusive. Measurements of sensory thresholds are widely variable between animals and between time points, clearly showing the importance of repeated measurements at intervals of several days when assessing sensory parameters. Only reproducible differences between groups can be considered reliable.
Tactile withdrawal thresholds showed no difference between diabetic obese and non-diabetic lean rats at all. By contrast, pioglitazone-treated non-diabetic obese rats seemed to show transient hypersensitivity from 13 to 21 weeks of age. It remains unclear if this was a random difference, or if it had any (patho-)physiological background. So far, only antinociceptive effects of pioglitazone have been described. 26,27
Although endpoint measurements of thermal withdrawal thresholds were significantly lower for obese diabetic rats compared with lean non-diabetic rats, this difference only became pronounced at the very last measurement for the untreated diabetic group. Food-restricted diabetic rats were most sensitive at most time points, and non-diabetic pioglitazone-treated rats showed thresholds varying between those of untreated diabetic and food-restricted diabetic rats except for the last time point. Thus, the level of hyperglycaemia did not influence tail flick thresholds. If the observed decrease in thermal sensitivity in obese ZDF rats is a sign of neuropathy, this neuropathy is not related to diabetes.
Our results regarding sensory thresholds are partly in contradiction to those of Brussee
Nerve conduction measurements at the tail nerve are frequently used, because the nerve is easily accessible without preparation, and measurements can therefore be repeated over time. Several authors have demonstrated decreased maximum tail nerve conduction velocity in streptozotocin-induced diabetic rats, 32–34 and this decrease has been shown to parallel changes in the sciatic nerve. 35 In our study, maximum nerve conduction velocity of the tail nerve was not different between the groups, but amplitude of nerve action potential was. This was the only nerve parameter, where diabetic untreated rats were significantly different from non-diabetic lean controls, and treatment or prevention of diabetes resulted in a significant convergence towards values of non-diabetic controls. However, amplitudes were increased in diabetic untreated ZDF rats, while in neuropathic patients a decrease in action potential amplitudes was observed. The amplitude of a whole nerve action potential is a measure of the number of activated myelinated fibres and is influenced by synchrony of activity. Therefore, a slowing of conduction velocity of distinct fibre populations creating asynchrony will result in decreased peak amplitudes as well as a reduction in the number of activated fibres. 36 Accordingly, improved synchrony of conduction velocity or a higher number of myelinated nerve fibres could underlie an amplitude augmentation. Both scenarios would rather reflect improvements of pathologies than their worsening, e.g. in cases where collateral sprouting during nerve regeneration occurs. 37 Although the cause for the observed amplitude augmentation remains unclear, it seems improbable that it is a sign of neuropathy in the diabetic ZDF rats.
Summing this up, neither sensory nor electrophysiological data support the assumption that male diabetic ZDF rats develop neuropathy linked to their state of hyperglycaemia. If there are pathologically relevant alterations in the nerves of ZDF rats, which cannot be excluded on the basis of our data, they are independent of the diabetic phenotype of the animals and are therefore not diabetic late complications.
In conclusion, the present study has shown that male obese ZDF rats develop progressive nephropathy causally linked to their diabetic state, and can therefore be seen as a model for human diabetic nephropathy. However, no clear signs of neuropathy could be detected in these animals and no connection to hyperglycaemia was found, rendering the male ZDF rat inappropriate as a model of human diabetic neuropathy. To make sure results of future animal studies on diabetic neuropathy are transferable to the human situation, other models than the male ZDF rat should be used.
Footnotes
Acknowledgements
We thank Elke Kleinschmidt, Marion Stein, Dagmar Fenner-Nau, Anja Wille, Jörn Wandschneider and Siegmar Kille for excellent technical assistance. We also thank G Biemer-Daub and her collaborators for the analysis of blood and urinary parameters.
