Abstract
Varied diagnostic imaging techniques may often be used in tandem, to enhance and complement each other, when an initial imaging diagnosis appears equivocal. Diagnostic medical sonography and magnetic resonance imaging (MRI) are imaging techniques that often complement each another. Sonography is an excellent diagnostic technique to determine the consistency of a soft tissue mass (e.g., solid vs cystic), and its relationship to adjacent structures, size, and shape. Sonography is especially useful for evaluating palpable lumps and allows sonographers to perform targeted examinations around the area of interest. In addition, color and pulsed-wave Doppler enable further characterization of a lesion’s presence or lack of vascularity. Sonography can also guide core-needle biopsy of superficial lesions. However, sonography is less reliable for deep masses in large anatomical regions. The American College of Radiology recommends MRI for deeper lesions of this type. Magnetic resonance imaging is also recommended for soft tissue masses suspected to be localized to the peripheral nervous system.
Multiple diagnostic imaging tools are sometimes required for confirmatory diagnostic decisions in health care. The evaluation of soft tissue masses in various locations throughout the body are often performed with multiple diagnostic imaging techniques. Several authors have discussed the advantages and disadvantages of utilizing multiple diagnostic imaging techniques to diagnose soft tissue masses.1 –9 The American College of Radiology (ACR) has established a guide, known as the Appropriateness Criteria, to provide best practice recommendations for the choice of diagnostic assessments of these masses. It serves as a guide to determining whether, when, and in which order plain radiographs, diagnostic medical sonography, magnetic resonance imaging (MRI), computed tomography (CT), and/or fluorodeoxyglucose-positron emission tomography/CT should be used. 1 The Appropriateness Criteria includes specific guidelines and parameters for when to use sonography and/or MRI to evaluate soft tissue masses.
Soft tissue masses are estimated at around 3 per 1000 per year. 2 About 99% of soft tissue tumors are benign; they outnumber malignant tumors by approximately 150:1.3,4 Diagnosing soft tissue masses is often challenging for many clinicians because there is so much overlap in the clinical presentation of benign and malignant tumors. 3 For example, many conditions, including infection, trauma, and cellulitis, can appear as soft tissue masses.3,5 When soft tissue masses are superficial, they are often recognized quickly, yet deep soft tissue masses are usually not identified until the mass has grown large, which is denoted by >10 cm. 3
Church et al 3 recommend beginning diagnostic imaging with radiographs to determine the presence or lack of bony involvement for diagnosing soft tissue masses. They also underscored that sonography is a cost-effective imaging technique with a high negative predictive value for lesions of this type. Church et al 3 also stated that sonography is an excellent diagnostic choice to determine the consistency of the mass (e.g., solid vs cystic), and its relationship to adjacent structures, size, and shape. Sonography is especially useful for evaluating superficial, palpable lumps, which allows sonographers to perform targeted examinations around the area of interest.1,5 Also, color and pulsed-wave Doppler enable further characterization of the lesion’s vascularity or lack of vascularity.5,7 Sonography can also guide core-needle biopsy of superficial lesions. 6
If sonography is chosen as the primary diagnostic tool to evaluate a soft tissue mass, and the diagnosis remains unclear, further imaging should be performed. Church et al 3 submitted that MRI is the most sensitive and specific diagnostic imaging technique to evaluate soft tissue masses. They also suggested that MRI is the study of choice for localizing and staging soft tissue masses because it provides the best delineation of surrounding soft tissue structures and their proximity to neurovascular structures. 3
The ACR Appropriateness Criteria support the above diagnostic recommendations. 1 The Appropriateness Criteria state that sonography is most helpful in evaluating small superficial lesions, such as those superficial to the deep fascia. However, the diagnostic accuracy of sonography is diminished when lesions are found outside of the subcutaneous tissue. The Appropriateness Criteria highlight that sonography is less reliable for deeper masses in large anatomical regions. For such deeper lesions, the Appropriateness Criteria recommendation is the use of MRI. 1 However, the Appropriateness Criteria does not typically support MRI as the diagnostic imaging choice for the initial evaluation of soft tissue masses.1,3
A T1-weighted MRI is the recommended modality for determining anatomical relationships of masses. 3 To identify free extracellular water and tissue edema, T2-weighted MRI is the recommended imaging choice. 3 Fat suppression techniques can subtract such signals to highlight abnormal fluid collections and areas of tumor transition produced by adipose tissue. 3 Finally, Gadolinium-enhanced MRI demonstrates rim enhancement, which helps radiologists differentiate between cystic lesions and solid masses, such as myxomas or myxoid liposarcomas, as rim enhancement is absent in solid lesions and is present in cystic lesions. 3
The following case series showcase three patients whose lesions were initially diagnosed with sonography or MRI. Then, their diagnoses were confirmed or enhanced using the other diagnostic imaging technique. In the first two cases, sonography was used first to localize the superficial masses. In the third case, an MRI of the lumbar spine was ordered for lower back pain. While the lumbar spine was unremarkable, the MRI revealed an incidental finding of a soft tissue mass adjacent to the lumbar spine. Sonography was then used to investigate further the mass’s components and the presence or lack of vascularity.
Case Series
Patient Case 1
A 52-year-old woman presented to her primary care physician with a palpable mass in the posterior aspect of her right thigh. Her medical history included anemia, a body mass index of 25 to 29 (overweight), dysmenorrhea, fibrocystic changes of the breasts, joint pain, and a tear of the lateral meniscus of the left knee.
A superficial soft tissue sonogram was ordered. A LOGIQ E9 ultrasound equipment system (GE HealthCare, Wauwatosa, WI, USA) was used with an ML6-15 linear array transducer. The radiology report stated that sonographic evaluation of the posterior right thigh, the site of clinical concern, revealed a lobulated, circumscribed solid hypoechoic mass measuring 5.5 × 7.9 × 5.5 cm3. Multiple cystic components were present within this mass, with blood flow demonstrated within some solid components (see Figure 1). It was stated that the mass appeared to lie deep in the subcutaneous fat, possibly intramuscular. Precontrast and postcontrast MRI was recommended for further evaluation.
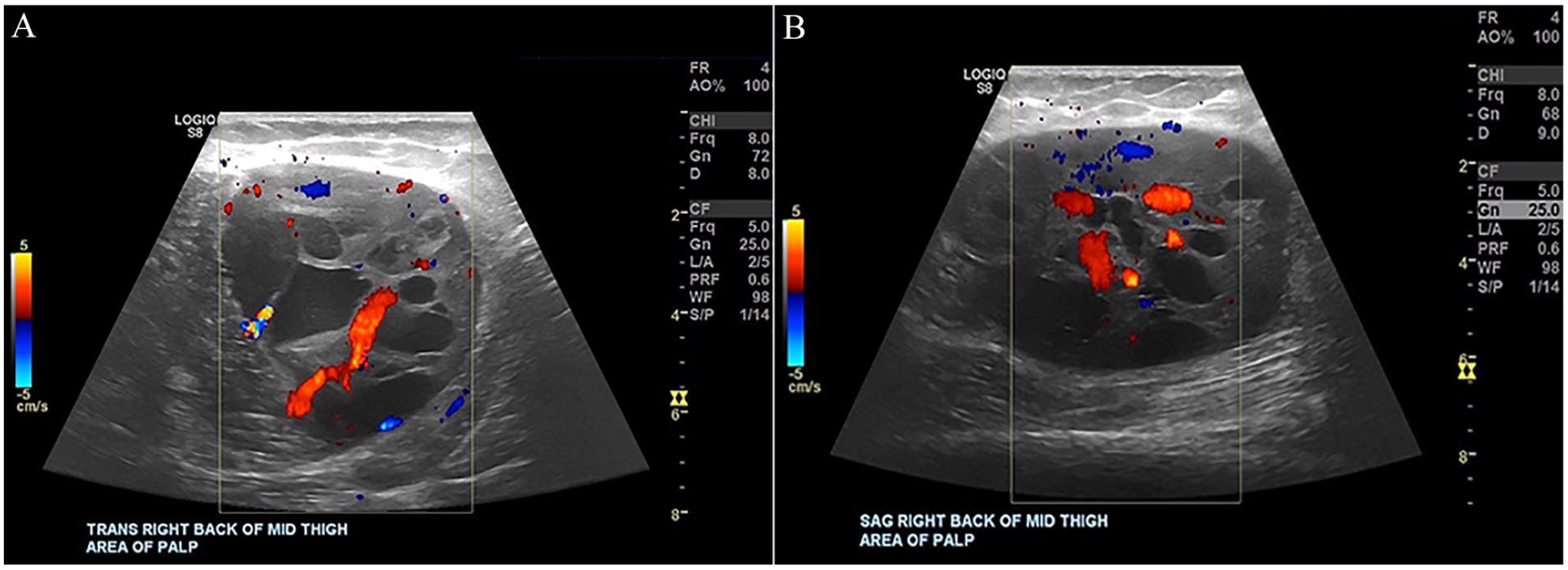
The following images demonstrate a sonographic transverse view (A) and sagittal view (B) of a complex mass with cystic internal components in the thigh with color Doppler.
About 4 weeks later, the patient underwent an MRI of the right femur. An OPTIMA MR450W 2014 MRI equipment system (GE HealthCare) was used. The technique included acquiring multiplanar, multisequential images without and with 7.5 cc of intravenous (IV) infusion of Gadavist. The radiology report indicated that a circumscribed mass within the biceps femoris muscle was present, correlating with the sonographic finding, measuring 8.0 × 5.7 × 6.5 cm3 (see Figures 2 and 3). This mass was intermediate in signal, containing well-defined pockets of increased signal and fluid-fluid levels. After IV contrast administration, there was enhancement of the solid components and no enhancement of the pockets of signal abnormality, which are likely associated with hemorrhage/necrosis. Increased T2 signal with enhancement extended along the posterior biceps femoris muscle superiorly inferior to the mass suspicious for tumor extension. No other masses were identified on the MRI images. There was no bone marrow signal abnormality, no evidence of fracture or focal osseous lesion, and no abnormal osseous enhancement. The overall impression was consistent with neoplasm with a recommendation to rule out sarcoma.
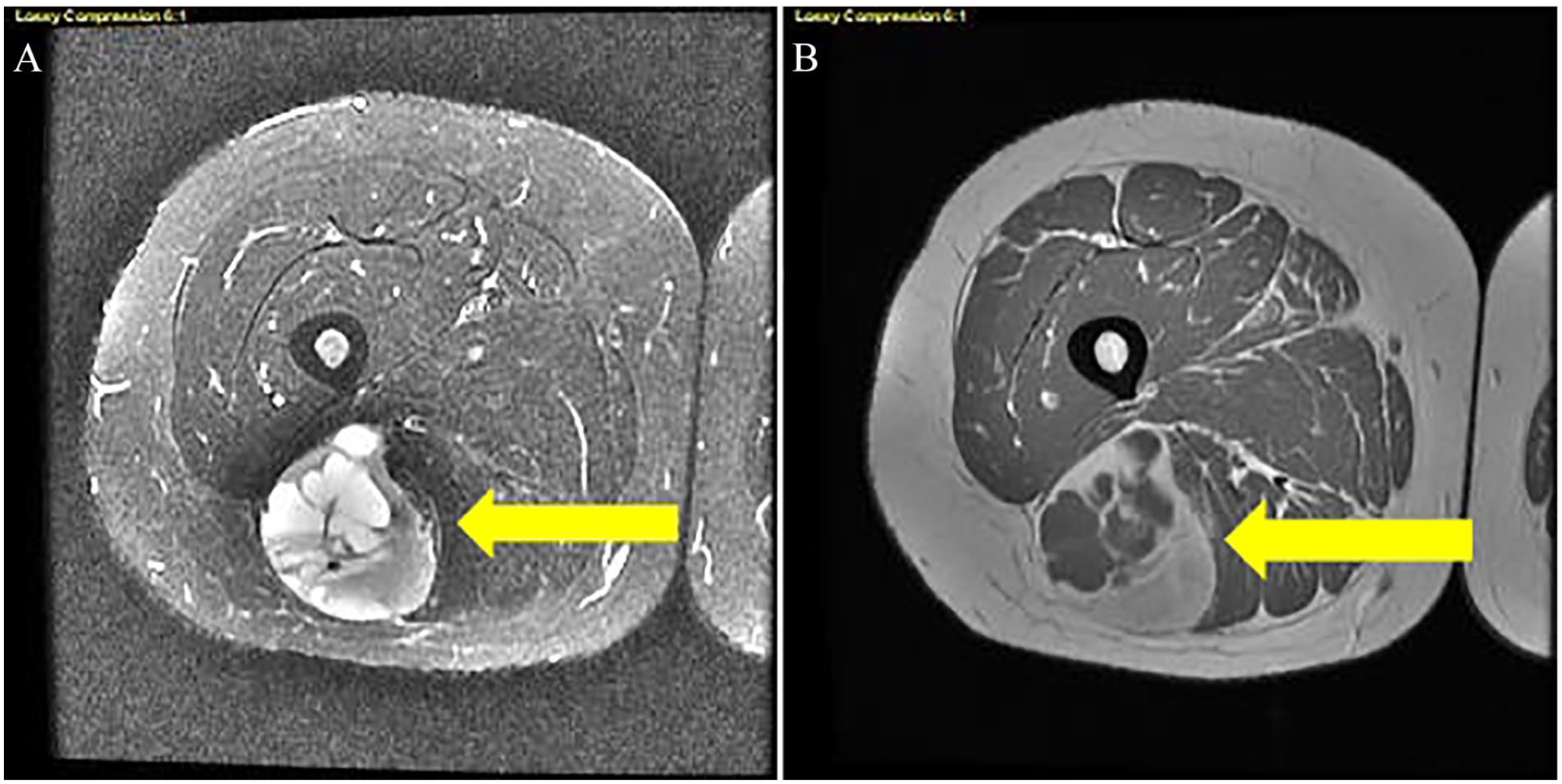
This set of images demonstrates an axial MRI image of the lower extremity precontrast (A) and postcontrast (B). The yellow arrows point to the complex lesion in images (A) and (B). MRI, magnetic resonance imaging.
The following image demonstrates a coronal MRI image of the lower extremity complex mass in the right lower extremity (yellow arrow). MRI, magnetic resonance imaging.
Patient Case 2
A 75-year-old man presented to his primary care physician with a palpable mass in his medial arm above the elbow. His medical history included dyslipidemia, essential hypertension, and a history of malignant thyroid neoplasm followed by a complete thyroidectomy. The patient discussed with the sonographer that he could palpate the mass, which had been there for several months, and it appeared to feel harder and softer at different stages of its development.
A superficial soft tissue sonogram was ordered. A LOGIQ E9 ultrasound equipment system (GE HealthCare) was used with an ML6-15 linear array transducer. Sonographic evaluation of the medial left arm above the elbow in the site of clinical concern revealed a circumscribed, solid, hypoechoic mass measuring 2.1 × 1.7 × 2.3 cm3 that demonstrated color Doppler flow, suspicious for neoplasm such as schwannoma (see Figure 4). However, a more aggressive lesion, such as sarcoma, could not be excluded with sonography alone. Further evaluation with precontrast and postcontrast MRI was recommended.
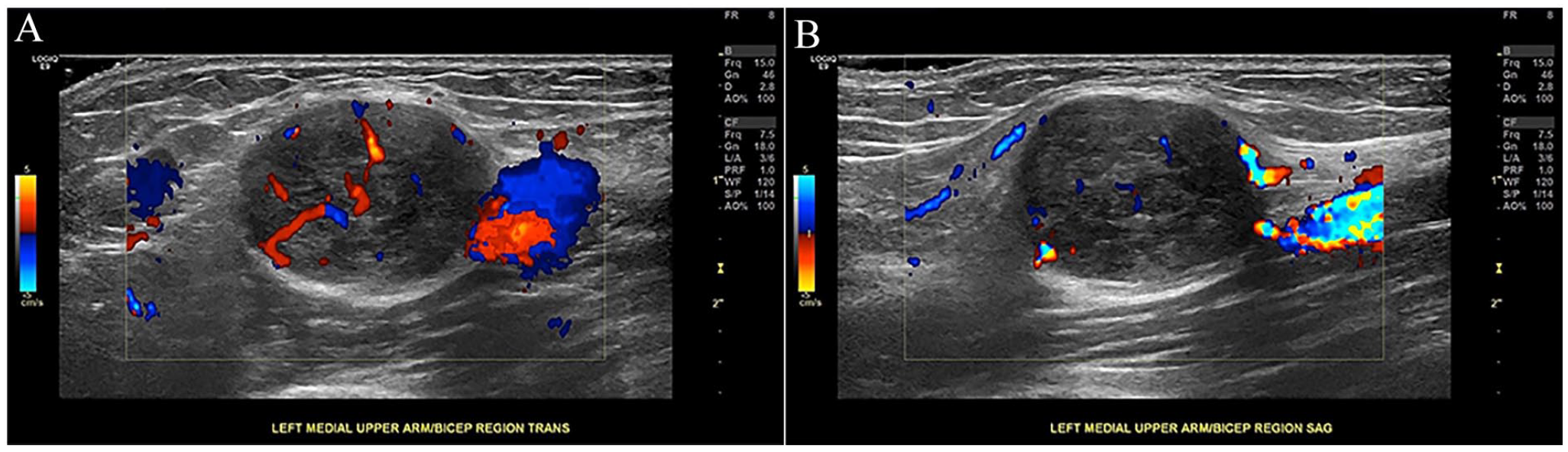
This image demonstrates a sonographic transverse view (A) with color Doppler and a sonographic sagittal view (B) of the lesion with color Doppler, near the brachial artery.
About 2 weeks later, the patient underwent an MRI of the right arm. An OPTIMA MR450W 2014 MRI equipment system (GE HealthCare) was used. The technique included acquiring multiplanar, multisequential images of the left mid to distal humerus without and with 5.5 cc of IV infusion of Gadavist. The radiology report described a circumscribed mass measuring 1.7 × 2.1 × 1.5 cm, 3 within the medial soft tissues abutting along the brachial vessels and median nerve adjacent to the brachialis muscle at the level of the distal humeral shaft (see Figure 5). This correlated with the sonographic finding and demonstrated intermediate T1 and increased T2 signal with relatively homogeneous postcontrast enhancement. It was stated that it likely represents a schwannoma arising from the median nerve. There was no signal abnormality or abnormal intramuscular enhancement, no bone marrow signal abnormality, no evidence of fracture or focal lesion, and no abnormal osseous enhancement.
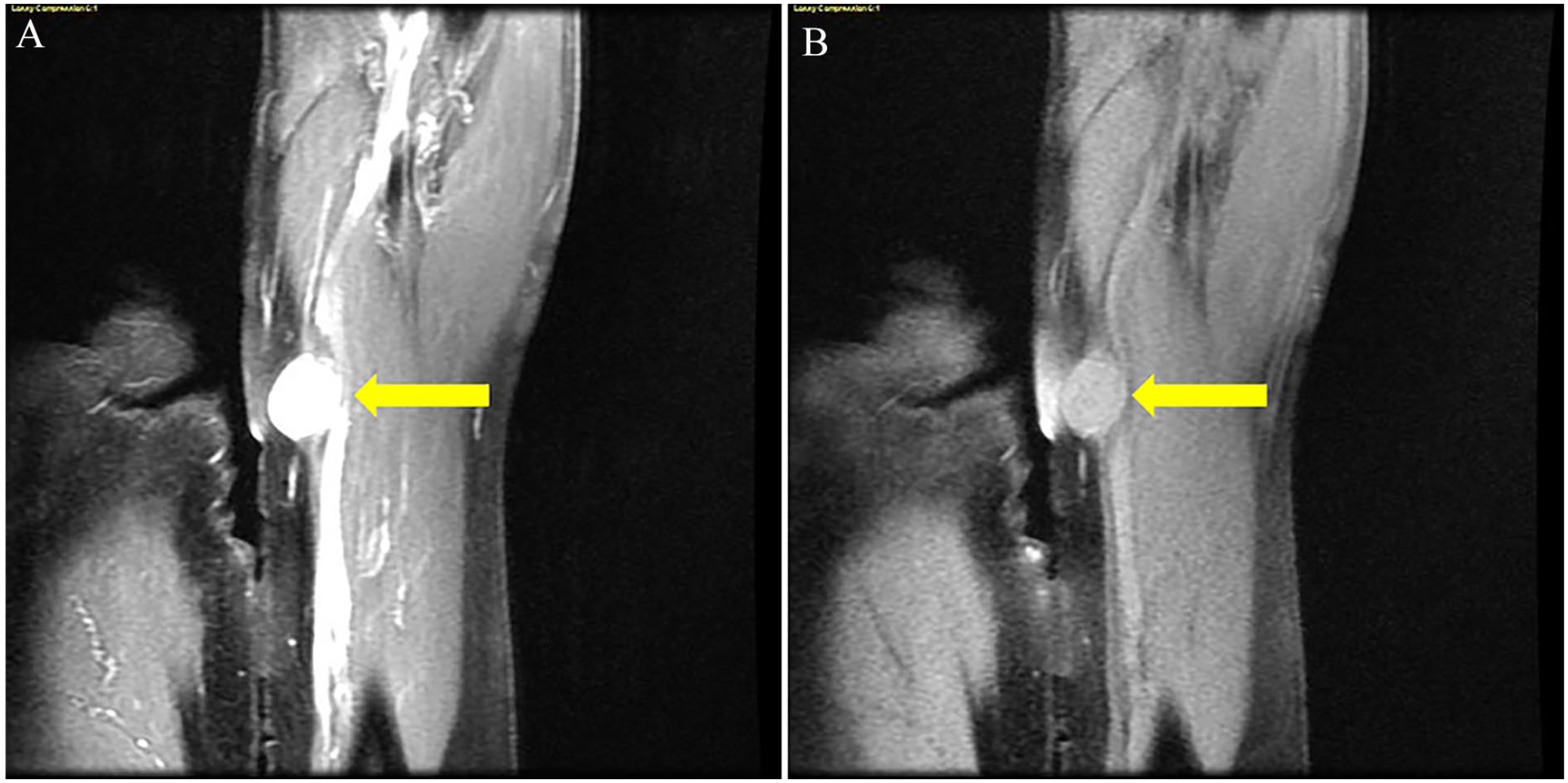
The following image demonstrates coronal MRI images of the lesion precontrast (A) and postcontrast (B). The yellow arrows point to the lesion in images (A) and (B). MRI, magnetic resonance imaging.
Patient Case 3
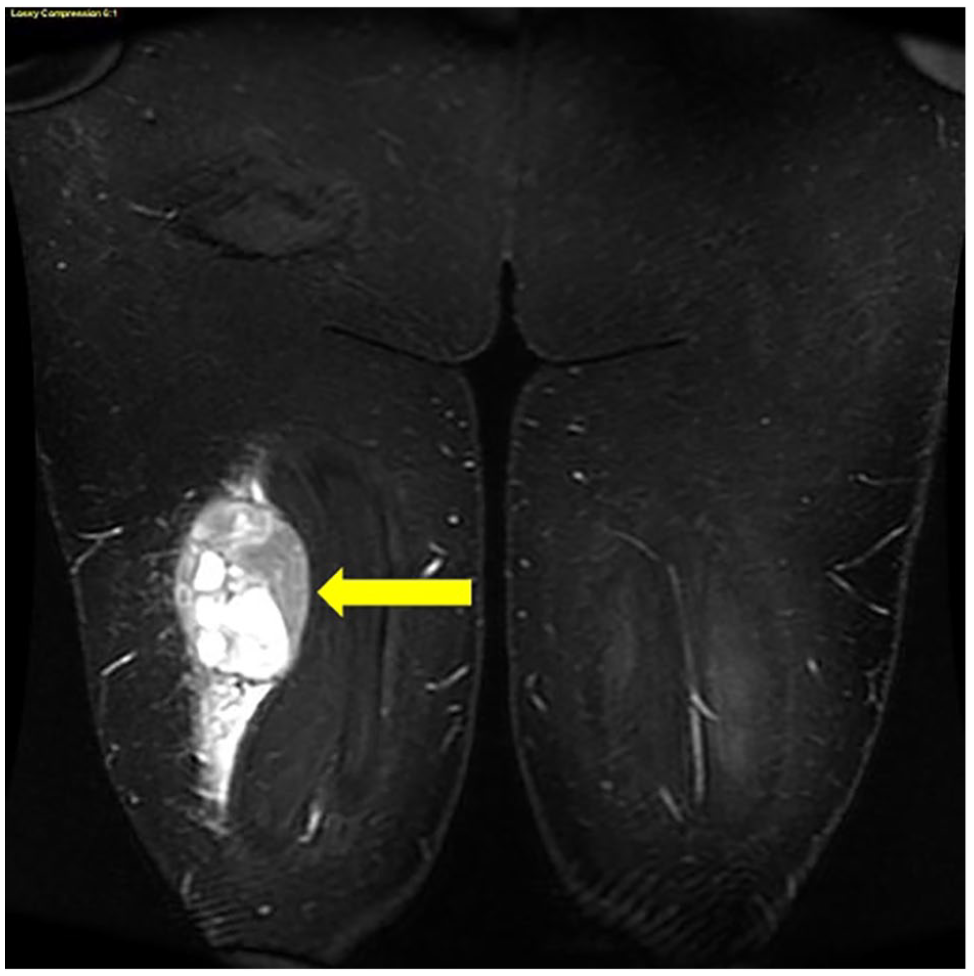
A 17-year-old boy presented to his primary care physician with lower back pain. The patient appeared athletic in build and had a history of playing sports in high school. His medical history included a traumatic injury while playing lacrosse, tendonitis of the right patellar region, and tibialis anterior tendinitis. A lumber spine radiograph yielded an unremarkable study. An MRI of the lumbar spine was ordered using an OPTIMA MR450W 2014 MRI equipment system (GE HealthCare) in which the radiology report described an unremarkable lumbar spine. However, an incidental finding was revealed as a 12.2 × 8.1 cm3 cystic structure in the left hemiabdomen that appeared to abut the undersurface of the left kidney (see Figure 6). Further evaluation with an abdominal sonogram was recommended.
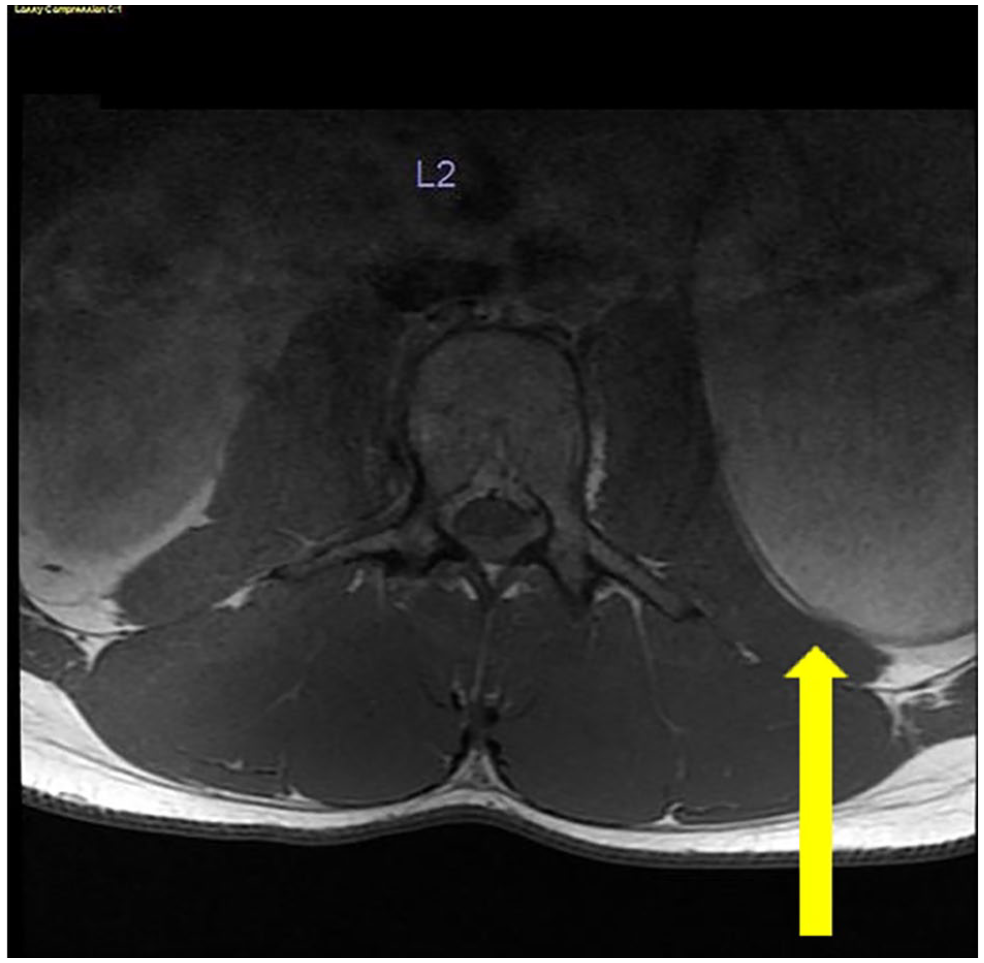
The following image demonstrates an axial MRI lumbar (L2) spine. The spine was considered unremarkable; the yellow arrow indicates the incidental finding. MRI, magnetic resonance imaging.
An abdominal sonogram was ordered. A LOGIQ E9 ultrasound equipment system (GE HealthCare) was used with a C1-6 curvilinear transducer. The sonographer invited the attending radiologist to examine the patient during the examination. After the standard abdominal sonography protocol was performed, images specifically targeting the mass in question were produced (see Figure 7). To differentiate the mass as a complicated cyst versus a solid structure, the radiologist asked the patient to lie in several positions, including supine, left lateral decubitus, and prone positions. Yet the radiologist could not influence the contents within the mass to move (see Figure 8A). The mass demonstrated extensive color Doppler flow (see Figure 8B). Yet it was initially unclear whether this was due to a network of arterials within the mass or, instead, a reverberation artifact. Pulsed-wave spectral Doppler could not demonstrate any arterial flow within the mass.
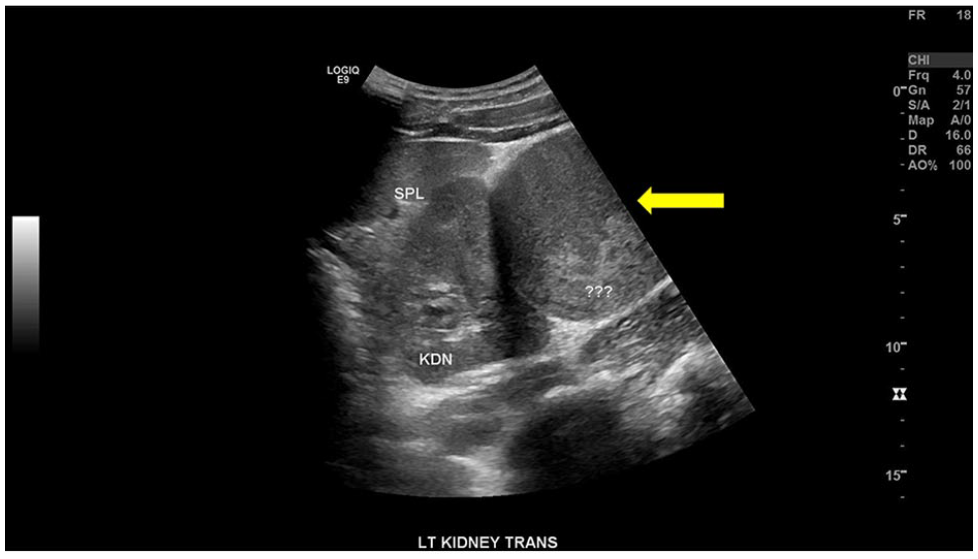
The image provided demonstrates a transverse retroperitoneal sonogram showing the spleen, left kidney, and a complex-appearing lesion (yellow arrow).
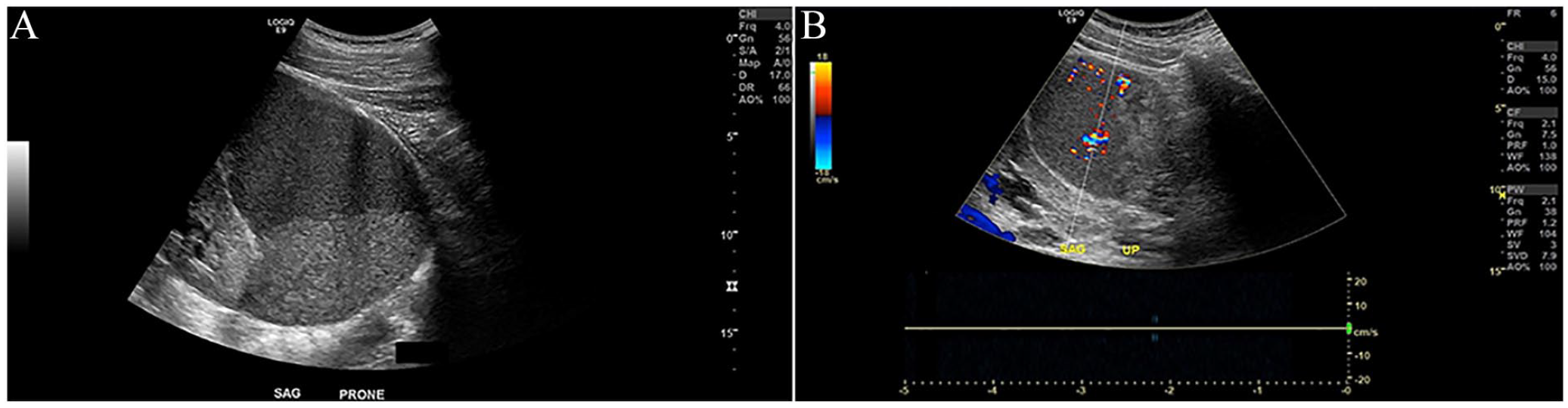
This image demonstrates a sagittal sonographic view (A) of the complex lesion after placing the patient in a prone position. The same complex lesion (B) using color Doppler demonstrates color, yet no arterial blood flow was detected when using pulsed-wave spectral Doppler.
The radiology report highlighted a left abdominal mass, lateral to the left kidney, ovoid and circumscribed, measuring approximately 15.0 × 7.5 × 9.6 cm3. This structure was also described as complex or solid, with a layered appearance of heterogeneous echogenic components and a mildly larger area of relatively homogeneous hypoechoic echogenicity. It was noted that no definite associated blood flow with color Doppler was seen. Upon prone and decubitus positioning, there was no definite change in the position of the content. Given the patient’s history of physical trauma while playing sports, the radiologist and sonographer speculated that the mass may be a hematoma that has not resolved. However, neither diagnostic imaging technique ultimately aided in a conclusive imaging diagnosis. After the two imaging examinations, the patient was then referred for a surgery consultation.
Discussion
Diagnostic choices to utilize sonography and/or MRI to evaluate soft tissue masses have been well-documented in the literature.1 –9 A comparison between sonography, MRI, and CT was conducted by Stojkovic et al and they evaluated 107 patients with cystic echinococcosis. They submitted that sonography was the cornerstone of diagnosis, staging, and follow-up for these cystic structures. 8 The one advantage CT and MRI had over sonography in that study pertained to imaging a mass in a location that was difficult to acquire using sonography, namely, a deep lesion. Testa et al 9 postulated that the cost of CT and MRI often render these examinations unfavorable for multiple and frequent utilization of gynecological cancers. Yet there is an advantage to recognizing the benefits of these imaging modalities used in tandem. Craig et al 10 suggested that while sonography remains the mainstay of diagnosing endometriomas, in their study, MRI is often added for its use as a problem-solving diagnostic tool.
The most common soft tissue masses are lipomas.2,5 They are found over a wide range of ages, but the most common age range in which they are seen is between 40 and 60 years. 2 Lipomas are rare in children and, in approximately 5% of patients in which they are found, they are often multiple. 11
A soft tissue sarcoma can be described as 50 different recognized entities, according to the World Health Organization classification. 12 These cancers demonstrate a variety of tumor types and represent less than 1% of cancers in the United States. 12 At least 70 different types of soft tissue sarcomas have been noted. The most common in adults are undifferentiated pleomorphic sarcomas, liposarcomas, and leiomyosarcomas; the most common in teenagers and young adults are rhabdomyosarcoma and Ewing sarcoma in soft tissue. 13
Rhabdomyosarcomas (RMS) are malignant tumors of mesenchymal origin, including small blue, round-cell tumors of childhood, neuroblastoma, lymphoma, and primitive neuroectodermal tumors. 14 Although they are most often seen in children, there are rare cases of RMS in adults, about 4% to 6%. 7 In children, the prognosis of rhabdomyosarcoma is relatively good. Yet the prognosis for adults with this disease is poorly documented due to its rarity.6,15 In adults, the prevalent histology of RMS is the pleomorphic subtype, accounting for more than 50%. 6 While they may arise in any anatomical site, RMS in adults is usually localized, with a prevalence of 40% in the extremities, 30% in the neck and head, and 20% in the genitourinary tract. 6 The location of RMS was also seen in a study by Kim et al, 16 which detailed a preventative screening technique called whole-body MRI (WB-MRI). In that retrospective study, they analyzed 7287 patients who underwent WB-MRI and it was found that most incidental cases of RMS were in the lower extremities. 16
Several interventions for RMS almost always include local therapy, consisting of surgery and/or radiation therapy and chemotherapy. 17 Little et al 15 performed a retrospective, actuarial univariate, and multivariate statistical analysis of 82 adults with RMS over 38 years. The histologic subtypes of RMS in this group were embryonal (34%), pleomorphic (43%), and alveolar (23%). 15 Tumors in these patients were observed in the head and neck (52%), trunk (26%), and extremities (7%). The treatments these patients underwent included radiation alone in 11%, radiation with surgery in 18%, radiation with chemotherapy in 34%, and all three diagnostic techniques in 37%. 15 With a median follow-up of 10.5 years, the 10-year actuarial disease-free and overall survival rates were 41% and 40%, respectively. The patients whose disease responded to chemotherapy had a significantly better metastasis-free period (72% at 10 years) than those whose disease failed to respond (19% at 10 years). 15
Schwannomas are mostly solitary masses commonly affecting the small peripheral nerves in the head, neck, and extremities. 18 They usually have a smooth nodular outline, occasionally with visibility of the nerve of origin. 19 They are the most common tumors of peripheral nerves. The incidence in adults is only 5%, and 19% are found in the upper limb. 20 Schwannomas are considered benign, which, in surgically accessible sites, respond well to excision and rarely undergo malignant transformation.19,20 The most common treatment for schwannomas is microsurgical resection. 21
The growth of schwannomas is slow, so they may remain asymptomatic for long periods. There is a higher incidence in the flexor surface of the upper limb since the concentration of nerve fibers is higher in this region. They are mobile in the longitudinal plane along the course of the involved nerve but not in the transverse plane. When symptomatic, they present with Tinel’s sign, characterized by shooting paresthesia in the distribution of the involved nerve, upon percussion of the schwannoma if the affected nerve is a sensory or a mixed nerve. 20 Chauhan et al 7 stated that MRI is a preferred imaging technique to diagnose tumors of the peripheral nervous system. So, a schwannoma initially detected with sonography will likely be followed up using MRI.
Conclusion
There are circumstances in which more than one diagnostic imaging technique may be needed to confirm the diagnosis of soft tissue masses. In the differentiation between cystic and solid components, sonography offers the ability to reposition patients to encourage the movement of cystic and solid components. The addition of color Doppler and spectral Doppler offers the ability to visualize internal vascularity within the borders of a lesion in real time. Yet deeper soft tissue lesions are better visualized using MRI because it provides the best delineation of surrounding soft tissue structures and their proximity to neurovascular structures. In addition, MRI is recommended for soft tissue tumors of the peripheral nervous system. Regardless of which examination is ordered first, the second diagnostic imaging technique may provide a complementary assessment that validates the suspected diagnosis and supports the use of a multi-imaging diagnostic approach.
Footnotes
Ethics Approval
Ethical approval was not sought for the present study because as a case report, this is not considered research, and it does not include interaction with human subjects.
Informed Consent
Informed consent was not sought for this study because all case data were de-identified and/or aggregated.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
