Abstract
Objectives
Most epidemiologic studies on soft tissue sarcomas (STS) and bone sarcomas (BS) are performed in western countries, with few in the Middle East and North Africa region. We describe the epidemiology of sarcomas in Lebanon using the medical records database at the American University of Beirut Medical Center (AUBMC).
Methods
This single-center retrospective cohort study included patients with sarcomas registered in the database between 2015 and 2019. Their charts were reviewed for baseline characteristics, tumor biology and location, treatment modalities, recurrence, metastasis, and death.
Results
The cohort included 234 patients with STS and 99 patients with BS. Most tumors were <10 cm in size. The most common subtypes were liposarcoma for STS and osteosarcoma for BS. The most common location of STS was the thigh. The most frequent sites of STS metastasis were the lungs. Histological subtype, smoking status, and tumor size and grade were significant for progression-free survival (PFS) in patients with STS. By multivariable analysis, smoking was significantly associated with poorer PFS in STS. For BS, only tumor grade was significant for PFS.
Conclusion
The epidemiology of sarcomas at AUBMC is similar to that previously reported. Smoking history was associated with poorer survival in patients with STS.
Keywords
Introduction
Sarcomas are a heterogeneous group of tumors that arise at connective tissue sites throughout the body. They are divided into two large groups: soft tissue sarcomas (STS) and bone sarcomas (BS). Although STS account for only 0.7% to 1% of all adult malignant tumors, the World Health Organization has described more than 100 histological STS subtypes with distinct clinical, prognostic, and therapeutic features.1,2 BS are less frequent than STS and account for only 0.2% of all adult malignancies. 2
The diagnosis of sarcomas is based on histologic morphology, immunohistochemistry, and molecular testing. STS range from low metastatic potential disease to aggressive metastatic disease depending on the histopathological subtype, size, and grade of the tumor. 3 STS occur in both men and women of all age groups. Their incidence tends to increase with age, with the highest prevalence between 45 and 90 years. 4 This diverse spectrum of STS has necessitated a multidisciplinary approach in terms of diagnosis and treatment. Computed tomography (CT), magnetic resonance imaging (MRI), and ultrasonography remain the best imaging modalities for the evaluation of primary and recurrent STS. 5 Surgical resection is used for the treatment of localized tumors with the addition of chemotherapy and radiotherapy based on the risk of local and distant recurrence. 3 In patients with metastasized STS, cytotoxic chemotherapy is still the first-line treatment. 3
Primary bone tumors are classified into three main malignancies: osteosarcoma, chondrosarcoma, and Ewing sarcoma. Osteosarcoma is the most common primary BS. 6 Both osteosarcoma and Ewing sarcoma are highly aggressive and metastatic cancers. 7 BS have a relatively high incidence in children and adolescents and account for approximately 15% of childhood/adolescent cancers. 8 The treatment of BS depends on whether the tumor is localized or metastatic. First-line therapy consists of chemotherapy before and after local treatment (i.e., surgery or radiotherapy). 9
The heterogeneity of sarcomas poses significant diagnostic and therapeutic challenges, making their incidence extremely difficult to study. It also limits the establishment of effective national registries in countries worldwide, especially in the Middle East and North Africa (MENA) region. In Lebanon, the national database registry for sarcomas was founded in 1998. Tremendous efforts must be made to keep up with the current incidence of sarcomas, follow up with old cases, and identify better treatment modalities that are appropriate for the Lebanese population. The aim of this study was to describe the epidemiology of STS and BS in a single institution (the American University of Beirut Medical Center, AUBMC) in Lebanon and compare the current results with those of previous studies performed in the MENA region. The variables collected in this study were also analyzed to evaluate their association with progression-free survival (PFS) in patients with sarcomas.
Materials and methods
Study design
This is a single-institution retrospective cohort study based on chart review analyses. The data included all adult patients (≥18 years old) diagnosed with STS/BS and registered in the medical records database at the AUBMC between 31 December 2015 and 31 December 2019. Patients were consecutively selected.
The study was conducted in accordance with the ethical principles stated in the Declaration of Helsinki (2013). The Institutional Review Board at the American University of Beirut reviewed the study proposal and granted approval prior to data collection (BIO-2019-0042). Verbal informed consent was obtained during patient visits. Patients were informed that their data might be used in descriptive studies. The data collected were kept confidential, and no patient identifiers were used throughout the study.
The study is reported in accordance with the Equator Network Guidelines. The reporting of this study conforms to the STROBE guidelines. 10
Data collected
The data collected included baseline characteristics of the cohort, tumor biology (subtype, size, grade), tumor location, treatment modalities (chemotherapy, radiotherapy, and/or surgery), incidence of recurrence, metastasis, and death (if applicable).
Statistical analysis
Patient characteristics and categorical variables were reported using descriptive statistics. PFS was calculated with Kaplan–Meier curves using the time-to-event analysis between the date of diagnosis and the date of disease progression/metastasis or date of death if applicable. Patients who did not reach the event were censored at the date of the last follow-up visit up to 2.5 years. Survival or disease-free progression for different sarcoma types was compared using the log-rank test. Cox regression multivariate analysis that included time-to-event data was used to examine variables influencing overall survival. Using the backward elimination method, the hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated for variables that remained significant in the model. A value of p < 0.05 was considered significant in all analyses. All statistical analyses were performed using IBM SPSS Statistics for Windows, Version 25.0 (IBM Corp., Armonk, NY, USA).
Results
STS
The total number of patients analyzed in this study was 333, including 99 patients diagnosed with BS and 234 diagnosed with STS. Among patients with STS, 56% (n = 131) were men. The noted comorbidities included hypertension, a previous malignancy or sarcoma, dyslipidemia, cardiac diseases, diabetes mellitus, urogenital problems, gastrointestinal disorders, and thyroid disorders. Eleven patients had two concomitant comorbidities, 28 patients had three concomitant comorbidities, and 13 patients had four or more concomitant comorbidities. Fifty-four patients had a previous malignancy, and eight patients had concomitant myelodysplastic syndromes. Moreover, 129 patients (55%) had a previous history of smoking, and 24 patients (10%) had a history of alcohol use.
The diagnosis of STS was confirmed by biopsy in 157 patients. Among 124 patients with reported tumor grades, 32 tumors were low grade, and 92 were high grade. Imaging techniques were also used as part of the workup to diagnose STS: MRI for 95 patients, X-ray for 54 patients, CT scan for 15 patients, and ultrasound for one patient. Sixteen patients did not undergo biopsy or imaging as part of the workup because STS was either removed directly by surgical excision or found incidentally during another surgery. Table 1 shows the location, size, and histologic subtypes of STS in our patient population. Only five patients had an unknown primary tumor location. The most common primary tumor location was the thigh (n = 67, 29%), followed by the buttock and groin (n = 62, 27%). The torso, upper extremities, retroperitoneum, and head and neck were also primary tumor locations with percentages of 15%, 11%, 9%, and 8%, respectively. Only three reported cases were found in the pelvis, masticator space, and gastrointestinal tract. In addition, 68% of STS tumors were less than 10 cm in size. Regarding histologic subtypes, 14% (n = 25) were reported as “other” in Table 1, including myxoid sarcoma, epithelioid sarcoma, desmoplastic round cell tumor, Kaposi sarcoma, angiosarcoma, and pseudomyogenic hemangioendothelioma. Concerning metastasis, 38% of patients (n = 90) had a metastatic history. The most common sites of metastasis in decreasing order were the lungs, gastrointestinal tract, lymph nodes, bone, brain, and venous system, with an incidence of 64%, 47%, 17%, 12%, and 5%, respectively. In cases with multiple metastatic sites, 19 patients had recorded metastasis to the lungs and gastrointestinal tract, and five patients had metastases to their lungs and bones. Only two patients had metastasis to four sites: the respiratory tract, gastrointestinal tract, lymphatic system, and bones.
Location, size, and histologic subtypes of STS and BS.

STS, soft tissue sarcomas; BS, bone sarcomas; GIST, gastrointestinal stromal tumors.
In addition, 204 patients underwent surgical resection for STS. Furthermore, 107 patients had an Eastern Cooperative Oncology Group (ECOG) score of 0, 61 had an ECOG score of 1, one had an ECOG score of 2, one had an ECOG score of 5, and 34 did not have a reported ECOG score. Preoperatively, 131 patients received chemotherapy, and 132 received radiotherapy. Postoperatively, 74 patients received chemotherapy, and 131 received radiotherapy.
Figure 1 shows the PFS of patients diagnosed with STS. The mean PFS for patients with STS was 100.95 months (95% CI 82.9–119,) and the median PFS was 55 months (95% CI 17.68–92.32).

Kaplan–Meier curve of progression-free survival (PFS) for patients diagnosed with soft tissue sarcomas (STS).
Figure 2 displays four PFS graphs for patients with STS according to tumor size, grade, histologic subtype, and smoking status. In Figure 2a, the Kaplan–Meier curve of PFS for patients with STS according to histological subtype is displayed with an overall statistical significance (p < 0.001). The mean PFS was 112.25 months for spindle cell sarcoma (95% CI 79.32–145.18), 64.9 months for liposarcoma (95% CI 42.49–87.32), 142.24 months for undifferentiated pleomorphic sarcoma (95% CI 101.71–182.77), 136.66 months for gastrointestinal stromal tumors (GIST) (95% CI 108.14–165.19), 67.24 months for malignant fibrous histiocytoma (95% CI 24.17–110.32), and 21.11 months for leiomyosarcoma (95% CI 7.921–34.31). The median PFS for liposarcoma, malignant fibrous histiocytoma, and leiomyosarcoma was 91 months (95% CI 20.34–161.66), 22 months (95% CI 0–64.52), and 14 months (95% 3.79–24.2), respectively. The median PFS could not be obtained for spindle cell, undifferentiated pleomorphic sarcoma, and GIST subtypes. Other histologic subtypes were grouped together, and they included phyllodes, dermatofibrosarcoma protuberans, synovial sarcoma, myxoid sarcoma, and desmoplastic round cell tumors. These tumors had a mean PFS of 97.18 months (95% CI 63.57–130.74) and a median PFS of 46 months (95% 17.03–74.97).

Kaplan–Meier curves of progression-free survival (PFS) for patients with soft tissue sarcomas (STS) according to histologic grade (a), tumor size (b), smoking status (c), and grade (d).
In Figure 2b, patients with STS are compared according to tumor size. Tumor size was divided into <10 cm and ≥10 cm. Patients with a smaller tumor size had a mean PFS of 144.59 months (95% CI 110.37–178.8) compared with 88.13 months (95% CI 57.01–119.25) for patients with a larger tumor size.
In Figure 2c, patients with STS are compared according to their smoking status. Patients with no previous history of smoking had a mean PFS of 134.63 months (95% CI 109.36–159.89) compared with smokers who had a mean PFS of 80.9 months (95% CI 58.99–102.81) (p < 0.05).
In Figure 2d, the Kaplan–Meier curve shows that patients with a high-grade tumor had a mean PFS of 9.58 months (95% CI 187.23–221.77), whereas patients with a low-grade tumor had a mean PFS of 50.17 months (9% CI 29.41–70.93) (p < 0.001).
Cox multivariable regression survival analysis was performed for PFS as the dependent variable in patients diagnosed with STS. The variables included age at diagnosis, smoking, grade, tumor size, histologic subtype, chemotherapy, and radiotherapy. Using stepwise conditional backward elimination, only statistically significant variables were kept in the model. Grade and smoking status remained in the final model and were found to be statistically significant (p < 0.05).
BS
Ninety-nine patients were diagnosed with BS. Among these patients, 53% (n = 52) were men. The noted comorbidities were hypertension, a history of malignancy, dyslipidemia, cardiac diseases, diabetes mellitus, urogenital diseases, pulmonary diseases, cataracts, ear, nose, and throat diseases, gastrointestinal disorders, and thyroid disorders. Ten patients had a previous history of STS. Forty-four (44%) patients reported a previous history of smoking, and 27 (27%) reported a history of alcohol use.
All patients had a biopsy performed as part of their workup. Among 43 patients with reported tumor grades, 15 tumors were low grade, whereas 28 were high grade. Imaging techniques were also used as part of the workup: MRI for 35 patients, X-ray for 14, CT scan for 21, and ultrasound for one. Tumor location for BS was reported as either axial (n = 19) or appendicular (n = 77). Only three patients had an unknown primary tumor location, and 63% of tumors were less than 10 cm in size. Table 1 shows the size and histological subtype of BS in our patient population.
Concerning metastasis, 35% of patients had a positive metastatic history. The most common sites of metastasis in decreasing order were the lungs, bone, and internal organs. Most notably, in cases with multiple metastatic sites, five patients had recorded metastasis to the lungs and bones, and two patients had metastases to their internal organs and bones. In addition, 77 patients out of 99 underwent surgical resection for BS. Preoperatively, 50 received chemotherapy, and 50 received radiotherapy. Postoperatively, 25 patients received chemotherapy, and 51 received radiotherapy.
Figure 3 shows the overall PFS of patients diagnosed with BS. The mean PFS for patients with BS was 8 months (95% CI 74.25–104.54), and the median PFS was 98 months (95% CI 79.16–116.84).

Kaplan–Meier curve of progression-free survival (PFS) for patients diagnosed with bone sarcomas (BS).
For BS, there was no statistically significant difference in PFS when histologic subtype, smoking status, and tumor size were compared separately. Only tumor grade was statistically significant with a p-value of 0.043 (Figure 4). Patients with low-grade tumors had a mean PFS of 108.32 months (95% CI 73.56–143.07) compared with patients with high-grade tumors who had a mean PFS of 62.87 months (95% CI 42.66–83.08). The median PFS was 121 months (95% CI 28.96–213.04) versus 26.1 months (95% CI 31.84–134.16) for low-grade versus high-grade tumors, respectively.
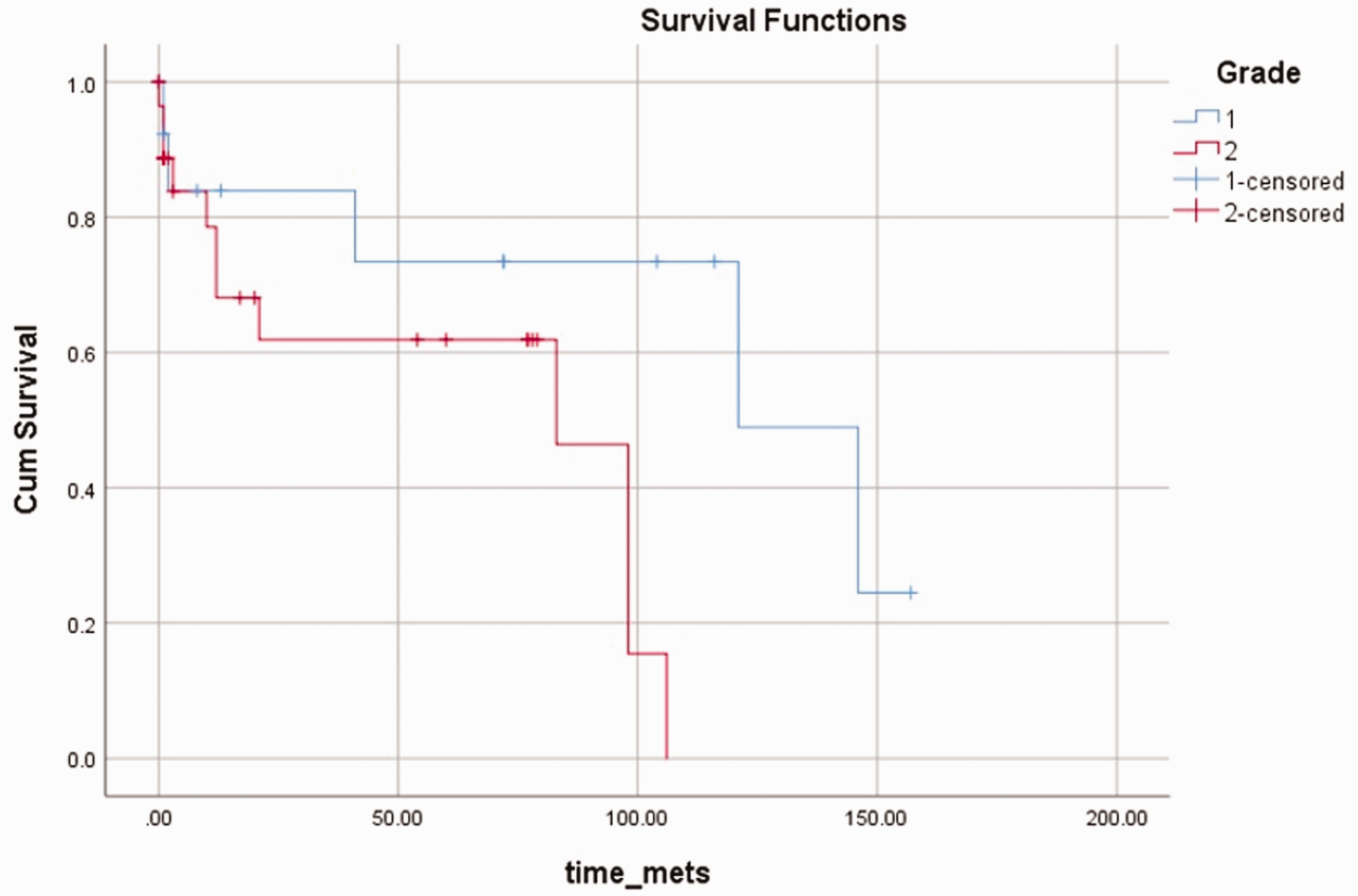
Kaplan–Meier curve of progression-free survival (PFS) for patients with bone sarcomas (BS) according to histologic grade.
A multivariable analysis was performed for patients with BS using PFS as the dependent variable. None of the variables included (age at diagnosis, smoking, grade, tumor size, histologic subtype, chemotherapy, radiotherapy) were statistically significant, regardless of the model used (backward or forward conditional).
Discussion
Sarcomas are a heterogeneous group of neoplasms arising from mesenchymal cells, and they can affect any anatomical site. Sarcomas are divided into STS and BS. According to the American Cancer Society, STS and BS account for <1% and <0.2% of all newly diagnosed malignancies, respectively. 2 In Europe, the retrospective RACECARE study from 1995 to 2002 stated that the incidence of all sarcomas was 5.6 per 100,000 per year, with STS accounting for 84% (4.7 per 100,000) and BS accounting for 15% (0.8 per 100,000). Regarding tumor registries or national cancer databases, the MENA region has been lagging, and there are only a few studies related to sarcomas in the literature.2,11 –13
To date, the most expansive observational study related to STS conducted in the Middle East is the MOON study. The MOON study was an observational, multicenter, retrospective patient chart review study that included 200 patients with metastatic STS in the final analysis.11 –13 In this study by Memon et al., the most frequent STS localizations were the lower extremities (74%), trunk (28.5%), and upper extremities (10.5%). 12 This is similar to the findings in our study in which the thigh, buttock, and groin were the areas primarily involved, followed by the torso and then the upper extremities. Despite the efforts of this study, it does not compare to parallel studies performed in the western world, such as the RACECARE project in 2012. 13 Aljuhani et al. conducted a retrospective study in Saudi Arabia and described 451 patients with primary BS. The most common primary tumor was osteosarcoma (55%), followed by Ewing’s sarcoma (35.5%) and chondrosarcoma (9.5%). 11 This is similar to the incidence of BS reported in our study in which osteosarcoma (42%) was the most common primary subtype, followed by Ewing’s sarcoma (17%) and chondrosarcoma (14%). Although our number of patients was substantially smaller than that in prior studies, the distribution of tumors among different histological subtypes was similar.
Many factors are associated with the survival of patients diagnosed with STS. Previous studies have shown that age, tumor grade, tumor size, and therapeutic response affect the outcomes of these patients.14 –17 In our study, patients with a smaller tumor size (less than 10 cm) had a mean PFS of 144.59 months (95% CI 110.37–178.8) compared with 88.13 months (95% CI 57.01–119.25) for patients with a larger tumor size (greater than 10 cm). This is similar to the findings of a cohort study in the Surveillance, Epidemiology, and End Results database performed by Cates that concluded survival decreases for tumors greater than 8 cm. 18
Although smoking is strongly correlated with the pathogenesis of several malignancies, 19 there is limited information on its association with the incidence and survival of patients with STS. In our univariate analysis, a history of smoking was associated with shorter PFS in patients with STS. Multivariate analysis also confirmed that smoking was a significant factor. This result is consistent with the findings from a retrospective study conducted by Gannon et al. in 166 patients with STS treated with pre-operative radiotherapy or chemoradiotherapy. Their analysis showed that current smokers and patients with a significant smoking history (>10 pack-years) had decreased distant metastasis-free survival (p-value = 0.0009) and PFS (p-value = 0.0036) but unchanged overall survival. 20 Similarly, Matsuoka et al. concluded that patients with STS and a positive smoking history had worse pulmonary metastasis-free survival (HR = 2.00, 95% CI = 1.12–3.60) but unaffected overall survival. 21 Moreover, a prospective cohort study by Zahm et al. showed significant increases in STS deaths among cigarette smokers (relative risk = 1.8, CI = 1.1–2.9). 22 The exact mechanism behind the association of smoking with decreased PFS is not clearly understood. Some studies suggest that carcinogens from cigarettes affect host defense mechanisms and increase the inflammatory response, predisposing patients to gene mutations and tumor suppressor gene silencing.23,24 By promoting angiogenesis, smoking also contributes to the metastasis of cancer to different locations.23,24 Therefore, smoking may compromise the immune system and lead to higher rates of progression and metastasis.
Our results suggest an association between smoking and lower PFS, whereas some studies that collected data on smoking history found that smoking was not significantly associated with survival in STS. For example, a retrospective cohort study conducted by Alamanda et al. showed that smoking is not predictive of sarcoma-specific death, distant metastasis, or local recurrence. 25 Another retrospective review demonstrated that smoking was not a risk factor for distant metastasis in STS but was associated with death from any cause. 26 This discrepancy in the literature regarding smoking and metastasis-free survival in patients with STS warrants further research with higher power and less confounding factors.
This paper presents data for both STS and BS from one Middle Eastern country. Although it is the first of its kind, it has several limitations that will be discussed below. In addition, we would like to highlight that more effort is needed to bridge the gap between the data from Western countries and data from the MENA region. From simple factors, such as the referring physician at the study center involved (i.e., a general practitioner, general surgeon, or medical oncologist), to whether a curative, palliative, or conservative procedure was performed on a specific body part, the data collected in different studies highlight the meticulous approach needed in targeting sarcomas. In the MENA region, there is a paucity of data on the epidemiology, treatment patterns, clinical management, and clinical outcomes of these tumors. Although the MOON study focused on the clinical and demographic characteristics, STS histological types or incidence were not obtained. Here, the authors would like to focus on the need for patient support programs in our countries. A multicenter collaboration or specialized center should be used when it comes to the management of sarcomas. These tumors are rare in incidence compared with other malignancies, and a dedicated approach to these tumors is needed. Not only is a database collecting information on every single patient needed, but these tumors should also be sequenced. Several studies have already explored a comprehensive and integrated approach to genomic characterization of adult STS and BS,27 –32 identifying distinct molecular subsets with potentially targetable alterations. By targeting patient-specific somatic copy-number alterations, a significant decrease in tumor burden was observed, and a roadmap for genome-informed treatment of sarcomas was established. This again highlights how far behind the MENA region is from achieving what is needed for optimal patient care.
Our study has several limitations. First, the retrospective nature of this study makes selection bias inevitable. The cohort size is modest, and some variables were missing from the electronic medical records of several patients. The heterogeneity of STS may also affect the accuracy of the results, especially when multiple subtypes were grouped together in the statistical analysis. Follow-up time was limited, and the effect of smoking may not have been documented during this short period. PFS in our study was defined as the time until disease progression or death. Further studies should address this limitation by differentiating between local recurrence-free survival, metastasis-free survival, tumor-free survival, and overall survival. Several confounding factors not addressed in this study may have affected the PFS of patients. Smoking history was reported as present or absent, with no differentiation between current or past smokers. The number of pack-years was also missing. This dichotomous description of smoking is a severe limitation to our study. Further research should focus on gathering a more comprehensive smoking history, such as status (current or past smoker), pack-years, and use of other tobacco products (vaping).
Conclusion
Overall, sarcomas are a heterogeneous group of tumors. Extensive research is still required to delineate the differences between each subset of STS and BS. With the increasing incidence of cancers in Lebanon, STS and BS cases are expected to continue increasing. 33 Our study shows that the epidemiology of sarcomas at AUBMC is similar to that reported in the literature. A history of smoking was associated with poorer survival in patients with STS. Further research is still needed to accurately estimate the incidence of sarcomas in the MENA region and study the effect of smoking, among other risk factors, on the survival of patients with sarcomas.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221082852 - Supplemental material for Descriptive epidemiology of soft tissue and bone sarcomas in Lebanon
Supplemental material, sj-pdf-1-imr-10.1177_03000605221082852 for Descriptive epidemiology of soft tissue and bone sarcomas in Lebanon by Maroun Bou Zerdan, Paul Meouchy, Nour Abdul Halim, Said Saghieh, Eman Sbaity, Rachid Haidar, Jaber Abbas, Amir Ibrahim, Mohamad Khalifeh and Hazem I Assi in Journal of International Medical Research
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
