Abstract
Alveolar soft part sarcoma (ASPS) is an extremely rare type of soft tissue sarcoma. The primary sites of ASPS are mostly located in the extremities and trunk. Primary pulmonary ASPS is extremely rare. A search of the PubMed® database identified only five cases of primary pulmonary ASPS. This current case report describes the sixth case of ASPS in a 15-year-old male that presented with recurrent headaches. Head computed tomography showed space-occupying lesions in the left parietal lobe. Positron emission tomography–computed tomography confirmed the space-occupying lesions in the left parietal lobe and showed multiple nodules and masses in the two lungs and pleura, which were considered to be low-grade malignant mesenchymal tumours. The case report presents the clinical characteristics, diagnosis and treatment process. Programmed cell death protein 1 monoclonal antibody (sintilimab) combined with a tyrosine kinase inhibitor (anlotinib hydrochloride) achieved a good therapeutic effect, indicating that this combination therapy is worth exploring further. Large-scale prospective studies are needed to explore and develop standardized treatments for ASPS.
Keywords
Introduction
Soft tissue sarcoma (STS) is a malignant tumour originating from mesenchymal tissue that has a very low incidence, accounting for only 1.0% of all adult malignant tumours, while the incidence of STS in children is slightly higher than that in adults.1–3 There are numerous pathological types of STS and alveolar soft part sarcoma (ASPS) is a rare pathological type of STS, accounting for 1.0% of all STS.4,5 ASPS was first reported and named in 1952; and the name was based on the fact that the tumour cells are arranged in a pseudoalveolar or organ-like manner. 6 ASPS occurs mostly in the head and neck of children and adolescents and the lower extremities of adults; and because it is a vascularized tumour, more than 60% of patients have lung, brain and other organ metastases when diagnosed. 7 Pulmonary ASPS are mostly metastatic and primary ASPS is extremely rare. Primary lung sarcomas are a group of rare malignant tumours that account for less than 0.5% of malignancies. 8 A search of the PubMed® database identified only five cases of primary pulmonary ASPS.9–13 This current case report describes the diagnosis and treatment of primary pulmonary ASPS in a male adolescent and hopefully provides some insight into the diagnosis and treatment of ASPS.
Case report
In August 2019 a 15-year-old male was admitted to Ward 2, Department of Medical Oncology, Lixin People's Hospital of Bozhou City, Anhui Province, China with recurrent headaches. Head computed tomography showed space-occupying lesions in the left parietal lobe. Positron emission tomography–computed tomography showed the following: (i) space-occupying lesions in the left parietal lobe that demonstrated mild glucose metabolism and obvious peripheral oedema; (ii) multiple nodules and masses in the two lungs and pleura were considered to be low-grade malignant mesenchymal tumours. Posterior biopsy of the patient from the right lung revealed right lung malignancy. The postoperative pathology showed that the tumour cells were arranged in an organoid or glandular vesicular pattern. The tumour cells were uniform in size and morphology, large round or polygonal in shape, rich in cytoplasm, containing eosinophilic red granules and with clear cell borders. Immunohistochemical staining identified transcription factor enhancer 3 (TFE3) (Figure 1). The diagnosis of ASPS was made in conjunction with the histological images and clinical history. The patient was given systemic chemotherapy after a clear diagnosis of pulmonary primary ASPS using the specific regimen of 7.5 g/m2 isocyclophosphamide intravenous (i.v.) drip plus 60 mg/m2 doxorubicin i.v. drip, once every 3 weeks. At the end of one cycle of chemotherapy, the patient developed liver function impairment, which was evaluated as a grade 3 adverse reaction by the National Cancer Institute Common Terminology Criteria for Adverse Events version 5.0. 14 Later, the treatment regimen was changed to 12 mg anlotinib hydrochloride orally once a day on days 1–14, with one cycle every 3 weeks. Lung and brain lesions progressed slowly after two cycles of treatment. On the basis of anlotinib hydrochloride treatment, 200 mg sintilimab i.v. drip on day 1 was added every 3 weeks. After two cycles of treatment, the chest lesions were slightly larger than before, so considering the possibility of pseudoprogression due to the immunotherapy, the patient continued to receive anlotinib hydrochloride in combination with sintilimab. After four cycles of treatment, the lung and brain lesions of the patient gradually began to be alleviated. After six cycles of combination therapy, the patient's treatment regimen was changed to maintenance therapy for 1 year. The maintenance regimen was 200 mg sintilimab i.v. drip on day 1 every 3 weeks (Figures 2 and 3). At the time of preparing this manuscript, the patient was in remission, his progression-free survival (PFS) had reached 33 months and follow-up is ongoing.
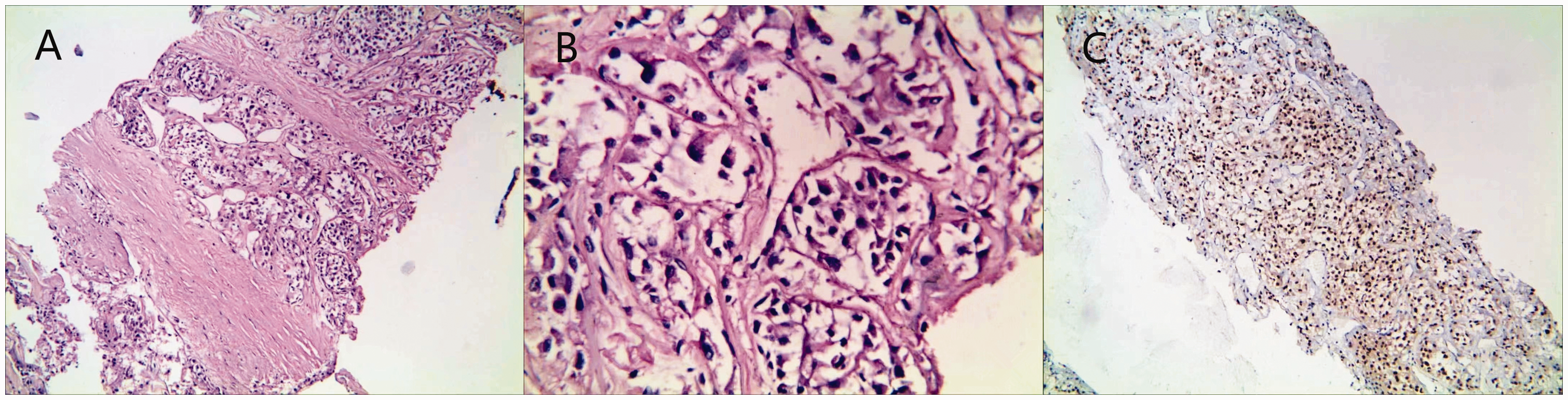
Representative photomicrographs of the postoperative pathology in a 15-year-old male that was admitted with recurrent headaches showed the typical features of alveolar soft part sarcoma: (A and B) adenoid structures of well-defined nests of cells. The cells were large, rounded or ovoid, eosinophilic, and the nests were surrounded by fibrous septa containing distinct blood vessels (haematoxylin and eosin) and (C) positive immunohistochemical staining for transcription factor enhancer 3. Sections were counter-stained with haematoxylin (a, magnification ×100; b, magnification ×400; c, magnification ×100). The colour version of this figure is available at: http://imr.sagepub.com.
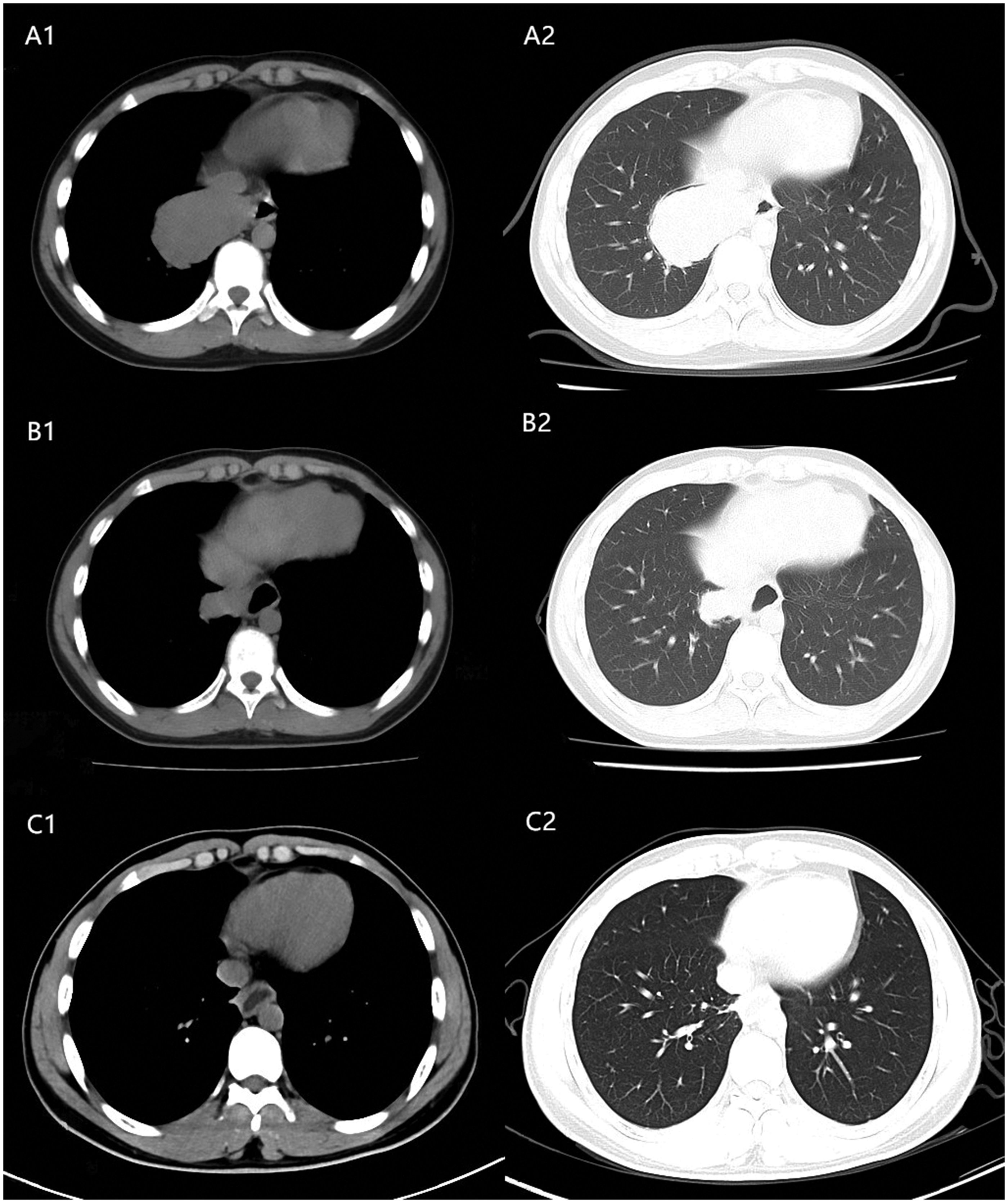
Representative computed tomography (CT) images of the lungs of a 15-year-old male that was admitted with recurrent headaches: (A1 and A2) a lung CT on 25 February 2020 before treatment with anlotinib hydrochloride combined with sintilimab showed that the maximum diameter of the primary lung lesion was 6.5 cm; (B1 and B2) a lung CT on 12 July 2022 after six cycles of anlotinib hydrochloride combined with sintilimab; (C1 and C2) the last lung CT on 29 November 2022 showed complete remission of the primary lung tumour.

Representative magnetic resonance imaging (MRI) scans of the brain of a 15-year-old male that was admitted with recurrent headaches: (A1 and A2) a brain MRI scan on 25 February 2020 before treatment with anlotinib hydrochloride combined with sintilimab showed that the maximum diameter of the metastatic lesions was 3.8 cm; (B1 and B2) a brain MRI scan on 12 July 2022 after six cycles of anlotinib hydrochloride combined with sintilimab; (C1 and C2) the most recent MRI scan on 29 November 2022 showed that the maximum diameter of the metastatic lesions was 0.9 cm (i.e. a 74% reduction after treatment) and the perifocal oedema had completely disappeared.
All patient details were deidentified for this case report, so informed consent was not considered necessary and ethical approval was not required for case report publication. Written informed consent for treatment was obtained from the patient. The reporting of this study conforms to the CARE guidelines. 15
Discussion
In this current case of primary pulmonary ASPS, the tumour cells showed a pseudoalveolar and nest-like arrangement in percutaneous pulmonary aspiration specimens, which is the pathological feature of ASPS. 16 In addition, the immunohistochemistry showed the characteristic positivity for TFE3. In 2001, the alveolar soft part locus–transcription factor E3 (ASPL-TFE 3) fusion gene was first detected and was shown to result in the onset and development of ASPS.17,18 This current patient was shown to have a positive TFE3 nuclear immune response by immunohistochemistry, but due to financial constraints, ASPL-TFE3 fusion gene tests were not undertaken for the patient. The diagnosis in the current case was confirmed as primary pulmonary ASPS based on the pathology and immunohistochemistry. In terms of treatment, surgical excision is still the main treatment for ASPS. A retrospective analysis found that surgery can significantly improve the prognosis of both patients with limited disease and in those with advanced distant metastases. 19 However, the patient in this current case had brain metastases at the time of diagnosis and multiple metastases were also seen in both lungs and pleura, so there was no indication for surgery. Based on the guidelines from the Chinese Society of Clinical Oncology (CSCO), 20 the first-line treatment offered to this current patient was a systemic chemotherapy regimen of 7.5 g/m2 isocyclophosphamide combined with 60 mg/m2 doxorubicin. Due to the young age of the patient and in consideration of the potential adverse effects of radiotherapy for the brain lesions on his future brain development, along with the uncertain efficacy of radiotherapy on ASPS, a decision was made not to undertake radiotherapy on the brain lesions. After one cycle of chemotherapy, the patient developed severe liver function impairment, which was evaluated as a grade 3 adverse reaction by the National Cancer Institute Common Terminology Criteria for Adverse Events version 5.0. A review of the published literature related has shown that the response rate of ASPS to chemotherapy is extremely low, with an objective remission rate of approximately 10%.21,22 In addition, the patient and his family members were unwilling to tolerate the severe toxicity and side-effects; and they subsequently refused follow-up systemic chemotherapy. Therefore, based on the patient's wishes and the finding from the relevant literature, we considered changing the regimen for the patient to anlotinib.
Alveolar soft part sarcoma is a malignant tumour with an abundant blood supply.5 Tyrosine kinase inhibitors (TKIs) are an effective therapeutic option for this tumour.5 The CSCO guidelines recommend anlotinib hydrochloride as the first choice of targeted therapy for ASPS.20 Anlotinib hydrochloride is an oral multi-receptor tyrosine kinase small molecule inhibitor developed in China, which mainly inhibits vascular endothelial growth factor receptor (VEGFR)2, VEGFR3, fibroblast growth factor receptors 1–4, platelet-derived growth factor receptor (PDGFR)α, PDGFRβ, c-kit proto-oncogene protein, RET and other targets.23 In addition, it can also reduce the expression of metastasis-related proteins and reduce the metastasis of tumours.23 Several studies have demonstrated that anlotinib hydrochloride has anti-angiogenesis effects comparable with that of sunitinib and sorafenib.24–26 A randomized, multicentre, double-blind phase IIb clinical trial that compared anlotinib hydrochloride with placebo demonstrated that in the ASPS subgroup analysis, PFS was 18.23 months and 3 months in the anlotinib hydrochloride and placebo groups, respectively. 27 The duration of PFS in patients was significantly prolonged by treatment with anlotinib hydrochloride. 27 Based on the recommendations of guidelines and the results of relevant clinical trials,20 the current case was administered 12 mg anlotinib hydrochloride orally once a day on days 1–14, with one cycle every 3 weeks as targeted therapy. In addition, sunitinib and pazopanib, two targeted drugs, have also demonstrated good efficacy in multiple clinical trials and were recommended by the 2019 CSCO guidelines.20,28,29 Compared with the two targeted drugs sunitinib and pazopanib, the evidence for anlotinib hydrochloride is stronger,23 so it was selected for the current case.
In the current case, a review after two cycles of treatment with anlotinib hydrochloride showed slightly progressive lesions in the lungs and brain. As a consequence of the progression of the patient's disease, the patient's treatment regimen was considered for adjustment. A review of the relevant guidelines identified that immune checkpoint inhibitors (ICIs) are also effective in the treatment of ASPS.30 The ICIs are one of the most important groups of tumour therapeutic agents developed in recent years.31 ICIs include programmed cell death protein 1 (PD-1), programmed cell death 1 ligand 1 (PD-L1) and cytotoxic lymphocyte antigen 4.31 At present, a considerable number of clinical trials have been conducted on PD-1/PD-L1 monoclonal antibodies in soft tissue sarcoma,30 so the rest of this discussion will focus on the introduction of PD-1/PD-L1 monoclonal antibodies. The main ligand for PD-1 is PD-L1 and the binding of PD-1 with PD-L1 produces negative regulatory effects that inhibit the activation of T cells.32 Malignant tumours express PD-L1 on their cell surfaces, which binds to the PD-1 on the cell surfaces of T cells, which facilitates the immune escape of malignant tumours.32 PD-1 monoclonal antibody drugs are targeted at this point of antitumour therapy. 33 Pembrolizumab, atezolizumab and geptanolimab have all been studied as monotherapy in ASPS with some efficacy.30,34,35 ICIs in combination with TKIs for ASPS have also been investigated, including a single-centre, single-arm phase II trial using axitinib in combination with pembrolizumab for STS with an objective response rate of 54.5% and a median PFS of 12.4 months in the ASPS subgroup. 36 In addition, with reference to a previous treatment for pulmonary primary ASPS reported in 2021,12 the current case’s disease developed rapidly 2 months after being treated with anlotinib hydrochloride so a PD-1 monoclonal antibody was added to anlotinib hydrochloride. A previous 24-year-old male with the same type of pathology and primary site as the current case had a good outcome with anlotinib combined with PD-1 monoclonal antibody (a PFS of 13 months), so this treatment regimen was attempted in the current case. 12 The previous case was similar to the current case in that they both had the lung as their primary target and both progressed rapidly with the use of anlotinib hydrochloride.12 Therefore, with reference to the previous case and the results of a previous clinical trial,12,36 sintilimab in combination with anlotinib hydrochloride was used as a treatment option in the current patient. Sintilimab is a fully humanized immunoglobulin G4 monoclonal antibody manufactured in China against PD-1.37 Sintilimab has a similar antitumour effect compared with nivolumab and pembrolizumab.37 Moreover, it has certain advantages in terms of cost. 37 After two cycles of treatment with sintilimab in combination with anlotinib hydrochloride, the current patient’s disease status was reviewed again and there was an enlarged lung lesion and a stable brain lesion. The current patient's performance status score and an assessment of his headache symptoms had improved and the possibility of pseudoprogression could not be excluded. Pseudoprogression is a common atypical reaction to the targeting of ICIs, with an incidence of 7.0–8.0%.38 It is mainly due to the activation and proliferation of large numbers of CD8+ T cells around the tumour at the initial stage of medication, which leads to the progression observed on a simple imaging assessment that can be checked by puncture biopsy of the tumour lesion.39,40 The current patient was recommended to have a lung lesion puncture to help select the next treatment option, but he refused percutaneous lung puncture. Meanwhile, a previous retrospective study on the onset time of PD-1/PD-L1 monoclonal antibody in 262 patients with malignant tumours (59.5% received PD-1 monoclonal antibody and 41.5% received PD-L1 monoclonal antibody) demonstrated that 76 of 262 patients had significant tumour shrinkage; while 28 of the these 76 patients began to see efficacy after 3 months of treatment. 41 Only three of the 21 complete responders occurred within 3 months. 41 The current patient only received 6 weeks of treatment and therefore may not yet have reached the critical point at which the disease begins to remit. Therefore, the current patient continued to receive sintilimab combined with anlotinib hydrochloride and four cycles later, the lung and brain lesions were reduced; and six cycles later, the lesions continued to be reduced. After six cycles of treatment, the patient was continued to receive maintenance therapy of 200 mg sintilimab i.v. drip on day 1 every 3 weeks for a total course of 1 year. The patient was not given subsequent antitumour therapy, but the tumour lesions continued to shrink. Re-examination on 29 November 2022 showed that the primary lung lesions were in complete remission compared with those observed before combined treatment with anlotinib hydrochloride and sintilimab. The brain lesions had a maximum diameter of 3.8 cm before combination therapy and a review on 29 November 2022 showed a maximum diameter of 0.9 cm, which was a 74% reduction from baseline. At the time of preparing this manuscript, his PFS had reached 33 months and follow-up is ongoing. PFS and overall survival were prolonged in this current patient compared with previously reported cases and clinical trials.12,28,29 It is not clear why this current patient achieved such good results from targeted combination immunotherapy. STS is generally considered to be a malignant tumour with less T cell infiltration and the effect of monotherapy of ICIs is poor.42 The combined use of anti-vascular endothelial growth factor (VEGF) drugs can enhance the efficacy of PD-1/PD-L1 monoclonal antibodies, mainly because VEGF has an immunosuppressive function.43,44 VEGF inhibits T cell activation, metastasis and infiltration; and the number of T cells is one of the key factors determining the therapeutic efficacy of ICIs.43,44 VEGFR2 is involved in the depletion of T cells through binding with CD8+ T cells. 45 In this current case, it is likely that the effect of VEGF and VEGFR on T cells was improved by the addition of TKI drugs, thus enhancing the antitumour effect of ICIs. In addition, this current patient has achieved a long-term response, which is one of the most significant features of immunotherapy. As a response to treatment with ICIs, some T cells are transformed into mature memory T cells and this can potentially facilitate an ongoing immune response even after drug withdrawal. 46 In this current case, treatment was stopped after 1 year of immunotherapy and no other antitumour therapy was given, but the tumour lesions were still shrinking and the patient has maintained long-term remission. There appears to have been no disease progress in the current case despite drug withdrawal. However, it is necessary to consider how long the immune response can last and how to choose the next antitumour therapy after the end of the durable immune response. Due to the low incidence of ASPS, the current sources of evidence for the treatment of ASPS are mainly case reports and retrospective studies,12,25,26 which provide little useful data to inform long-term treatment. The reporting of the treatment process of this current case should provide additional information about the successful treatment of ASPS.
In conclusion, this current case report has presented a case of pulmonary primary ASPS and a comprehensive review of the literature on the diagnosis and treatment of this rare tumour. The diagnosis of ASPS is made based on clinical manifestations and characteristic pathological and immunohistochemical findings. Unfortunately, most patients have already developed distant metastases and have lost the opportunity for surgical treatment when diagnosed. As patients with advanced distant metastases are not sensitive to chemotherapy, the application of conventional chemotherapy regimens is limited. Due to the small number of cases of ASPS, it is extremely difficult to organize large clinical trials to find a standardized treatment regimen, so the treatment of patients with ASPS is still in the exploratory stage. In this current case, PD-1 monoclonal antibody combined with a TKI achieved a good therapeutic effect, indicating that this combination therapy is worth exploring further. Large-scale prospective studies are needed to explore and develop standardized treatments for ASPS.
Footnotes
Acknowledgements
The authors thank their colleagues who have helped with this manuscript. The authors thank the anonymous reviewers and the editor for their valuable comments.
Author contributions
Feiyang Li and Haowei Lu: collection of materials and manuscript writing. Dong Zhao: guiding the writing of the manuscript. All authors contributed to the article and all read and approved the final version of the manuscript.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from funding agency in the public, commercial, or not-for-profit sectors.
