Abstract
Objective:
Lung ultrasonography (LUS) is a safe, quick, and portable diagnostic tool, which can accurately detect postoperative pulmonary complications, postsurgically, without ionizing radiation. The aim of this scoping review was to map the evidence base regarding the use of LUS to assess cardiac surgery patients.
Materials and Methods:
The JBI methodology was used to conduct this particular scoping review.
Results:
In total, 90 publications were identified and of those, 73 were research studies, six were narrative reviews, and 11 were narrative, opinion, and text articles. The studies that were included were predominantly observational cohorts and aimed to determine or compare LUS diagnostic ability, prognostic ability, or both. The LUS methods used with patients were heterogeneous and variably reported.
Conclusion:
Despite an increasing number of studies since 2014, standardized protocols for the use of LUS are yet to be widely adopted and remain an important area for further work. Future research should consider exploring perceptions and experiences of LUS, the use of LUS in treatment outcome measurement, and use by nonphysician health care professionals.
Keywords
Patients undergoing cardiac surgery are subjected to prolonged anesthesia, supine position, ventilation, and compression, which increases their susceptibility to developing postoperative pulmonary complications (PPCs), such as atelectasis, pleural effusion, and pneumothorax.1 –4 Up to 30% of patients who develop PPCs die within 30 days of major surgery, a considerably higher mortality rate than those without such complications (0.2%–3.0%). 3 Consequently, timely identification of PPCs is crucial for effective management and improved patient outcomes.
A chest radiograph (CXR) and auscultation are commonly used to assess for PPCs following cardiac surgery, yet these common diagnostic assessment tools have limitations. For instance, while an erect CXR has a sensitivity of 92% for pneumothorax, a supine CXR (which is common postoperatively) has only 50% sensitivity. 5 Furthermore, a “white-out” on a CXR could indicate pleural effusion or one of 15 other causes. 6 Studies indicate that the intrarater reliability of auscultation can be as low as 28%. 7 Using CXR, auscultation, or both, to differentiate between pulmonary pathologies can be difficult and result in misdiagnosis. This can decrease confidence in the management plan and may result in delayed recovery.
In recent years, point-of-care ultrasound (PoCUS) has become increasingly popular due to its high accuracy, portability, and lack of radiation emissions.2,8,9 Lung ultrasonography (LUS) is a type of PoCUS, which focuses on the pleura and parenchyma, and has been studied globally in the medical field. Lung ultrasonography has the ability to detect common pulmonary pathologies, such as pneumothorax, pleural effusion, lung consolidation, atelectasis, and pneumonia,2,10,11 making it a valuable tool to identify PPCs. Lung ultrasonography compares well with the standard tools used in the cardiac surgery population. Senniappan et al 12 found LUS to be comparable with CXR both immediately postoperatively (k = 0.652) and 1-day postoperatively (k = 0.740) with a near-perfect agreement for pneumothorax (k = 0.931). Several other studies have tested the sensitivity and specificity of LUS against computed tomography (CT), the gold standard for identifying pulmonary pathology, and demonstrated high sensitivity, specificity, and diagnostic accuracy of LUS.13 –15
There is a growing body of evidence on the use of LUS within cardiac surgery patients. Lung ultrasonography’s comparability to both CXR and CT in addition to its lack of ionizing radiation has made it a diagnostic tool of interest, within cardiac surgery research. It will be useful to both the clinical and research community to map that body of evidence, to identify what is currently known about LUS, when used with cardiac patients, and to guide future research investment in the field. A preliminary search of the International Prospective Register of Systematic Reviews (PROSPERO), Open Science Framework (OSF), the Cochrane Database of Systematic Reviews, JBI Evidence Synthesis, Medline, and the Cumulative Index to Nursing and Allied Health Literature (CINAHL) was conducted, and no planned, underway, or completed systematic reviews or scoping reviews on the topic were identified. Systematic and scoping reviews conduct a more comprehensive search of the literature than literature reviews, using a systematic approach. While systematic reviews have a more focused research question and are explanatory or analytical in nature, scoping reviews broaden the research question and are more exploratory or descriptive in nature. Scoping reviews aim to assess and understand the extent of the knowledge in an emerging field, often identifying, mapping, reporting, or discussing literature characteristics using a systematic and methodologic approach. 16
Review Questions
The objective of this scoping review was to map the evidence base on the use of LUS within cardiac surgery patients. The primary review question was: What has been reported on the use of LUS within cardiac surgery patients?
The following sub-questions were explored:
What types of studies have been published on LUS and cardiac surgery?
How and why has LUS been used with cardiac surgery patients?
What anatomic structures and artifacts have been investigated with LUS with cardiac surgery patients?
Which professionals are reportedly using LUS with cardiac surgery patients?
What LUS protocols are reportedly being used by professionals undertaking LUS with cardiac surgery patients?
Inclusion Criteria
This scoping review considered literature including or concerning both adult and pediatric patients undergoing cardiac surgery irrespective of participants’ demographic profile. Types of cardiac surgery included, but were not limited to:
Coronary artery bypass grafting
Heart valve repair or replacement
Coronary angioplasty and stenting
Atherectomy
Cardiomyoplasty
Heart transplant
Catheter ablation
Literature that included a mixed demographic was considered if cardiac surgery patients contributed 75% or more of the total cohort or the cardiac surgery patient outcomes could be extracted separately.
Concept
This scoping review considered literature concerning LUS use by any qualified health care professional on a cardiac surgery patient. Lung ultrasonography could have been used as a diagnostic assessment tool, outcome measure, during treatment, or for any other purpose. For this review, LUS is defined as a tool for investigating the pleura and parenchyma ultrasonically. For this reason, literature sources investigating the diaphragm were excluded.
Context
This scoping review considered literature from any health care setting where cardiac surgery is conducted. Studies were not limited by geographical location. Non-English records were considered if they could be reliably translated using Google Translate.
Type of Sources
This scoping review considered: primary research of any type (e.g., qualitative, quantitative, case reports); literature reviews of any type (e.g., systematic, narrative); narrative, opinion and text (e.g., editorials, opinion pieces, commentaries); and conference abstracts reporting any of these types. Trial registrations and protocols were excluded; however, they were retrieved to identify additional published studies from their reference lists.
Materials and Methods
This scoping review was conducted in accordance with JBI methodology for scoping reviews 17 and followed an a priori open access protocol registered on OSF in March 2022. 18 This methodology was chosen as the research team has expertise in and experience with the methodology. JBI, a global organization, supports and promotes evidence-based health care decision-making, which considers feasibility, appropriateness, meaningfulness, and effectiveness. 19 The JBI methodology is a well-known and accepted methodology within health care.
Search Strategy and Information Sources
The search strategy aimed to locate both published and unpublished studies. An initial limited search of Medline and CINAHL (EBSCOhost) was undertaken using the keywords (TX lung ultrasound) AND (TX cardiac surgery) to identify articles on the topic. The text words contained in the titles and abstracts of relevant articles, and the index terms used to describe the articles were used to develop a full search strategy for Medline. The search strategy, including all identified keywords and index terms, was adapted for each included database and information source (see Supplemental Appendix 1). A second search for all databases was undertaken on April 6, 2022 and for gray and unpublished literature on August 3, 2022.
The databases searched included: Medline and CINAHL (via EBSCOhost), Embase (via Ovid), Cochrane Reviews and Trials, Scopus, and Web of Science. Sources of unpublished studies and gray literature included: Google Scholar and university repositories. The full search strategy including the search terms and hits from each database and gray literature source are provided in Supplemental Appendix 1.
The reference list of all included reports was then searched for additional eligible reports.
Source of Evidence Selection
Following the searches, all identified records were collated and uploaded into the reference manager Zotero (v6.0.13; Roy Rosenzweig Center for History and New Media, Fairfax, VA, USA) and duplicates removed. Remaining records were uploaded into Covidence (Veritas Health Innovation, Melbourne, Australia) with further duplicates detected and removed. Titles and abstracts were initially screened independently by two reviewers (two of CF, CW, KC) against the inclusion criteria for the scoping review. Excellent agreement (90%) was demonstrated after 10% of title and abstract screening, and as this scoping review formed part of an unfunded doctoral research program, one reviewer (CF) conducted the remainder of title and abstract screening, with regular review and discussion with members of the review team (CW and KC). This process was repeated for full-text screening, with 90% agreement following screening of 20% full texts.
Data Extraction
A data extraction form was constructed prior to database searching 18 based on the Covidence Data Extraction Form 2.0 and extraction from 10% of reports was piloted independently by two reviewers (CF and KC). Minor amendments were made to the data extraction form to focus more on LUS methods and characteristics. After good agreement was reached on independent screening by two reviewers (85%), one reviewer (CF) extracted data from the remaining reports, with regular review and discussion with the review team (CW and KC). Data were extracted on the aim, study design, setting, participant characteristics, and key findings. LUS methods were also extracted regarding the profession of the operator, the number of operators, the reason for use, time of use in relation to surgery, anatomic features or artifacts of interest, LUS findings, and any protocols used.
Results
Study Inclusion
Initial screening of databases retrieved 10 806 records, with an additional 172 records identified from other sources. After the removal of duplicates, 9626 records remained for title and abstract screening. Three hundred and seventeen records proceeded to full-text screening. Ninety reports met the inclusion criteria and were included in the review. The PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram (see Figure 1) illustrates the number of records and reports at each of these stages and the distribution of reasons for exclusion.
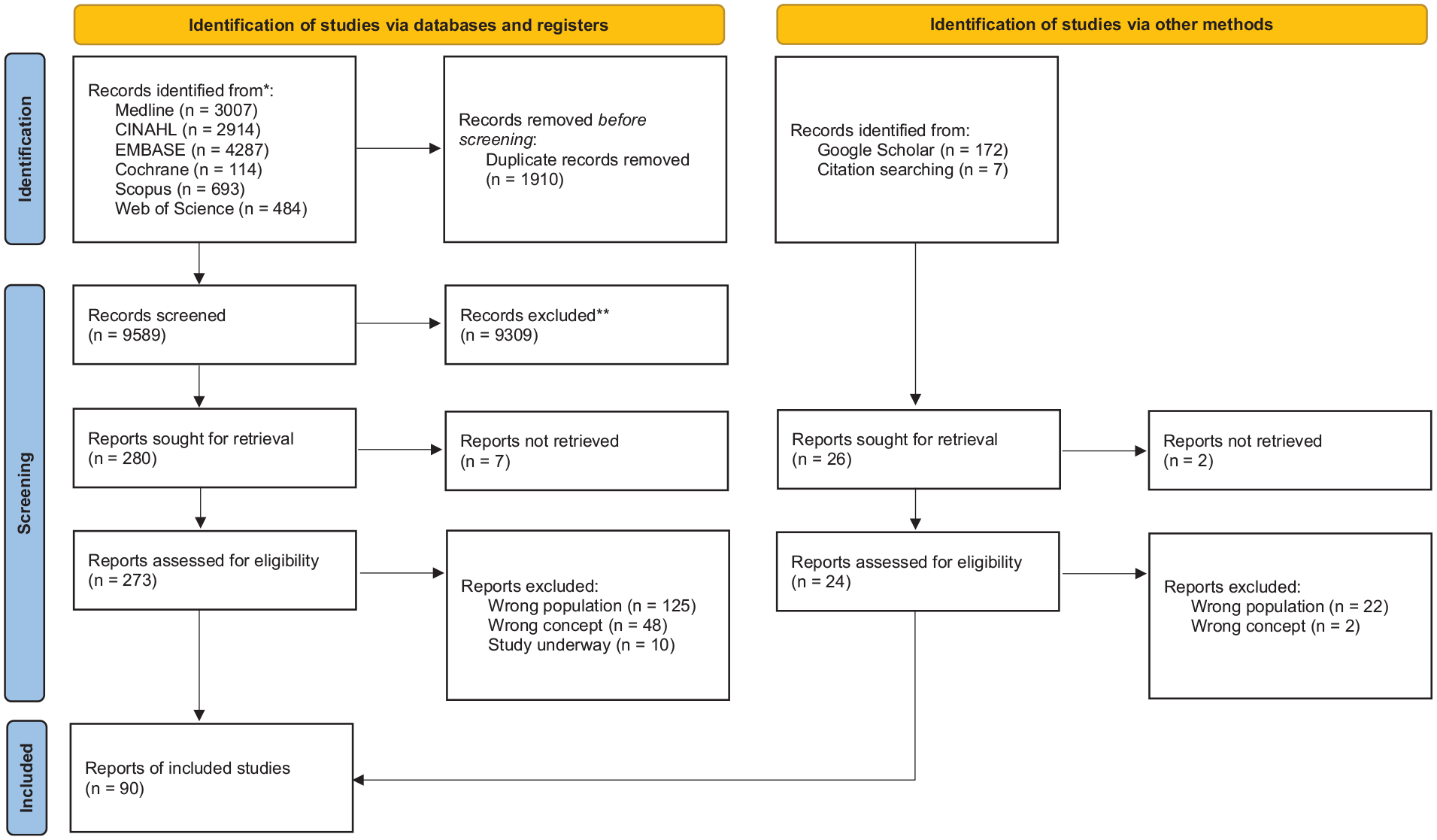
The PRISMA flow chart for the search results and the study inclusion process. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Characteristics of Included Reports
A summary of general characteristics of all included reports is reported in Table 1. The scoping review included 90 reports: 73 research studies, six narrative reviews, and 11 narrative, opinion and text. The reports were published between 1994 and 2022, with a sharp increase in publication rates from 2014, peaking in 2020 (n = 16) (see Figure 2). Reports were published in 27 different countries but most originated from Italy (n = 29, 32.6%) followed by China (n = 7, 7.9%). Five reports were translated successfully by Google Translate.15,20 –23
The characteristics of the included studies for this review.
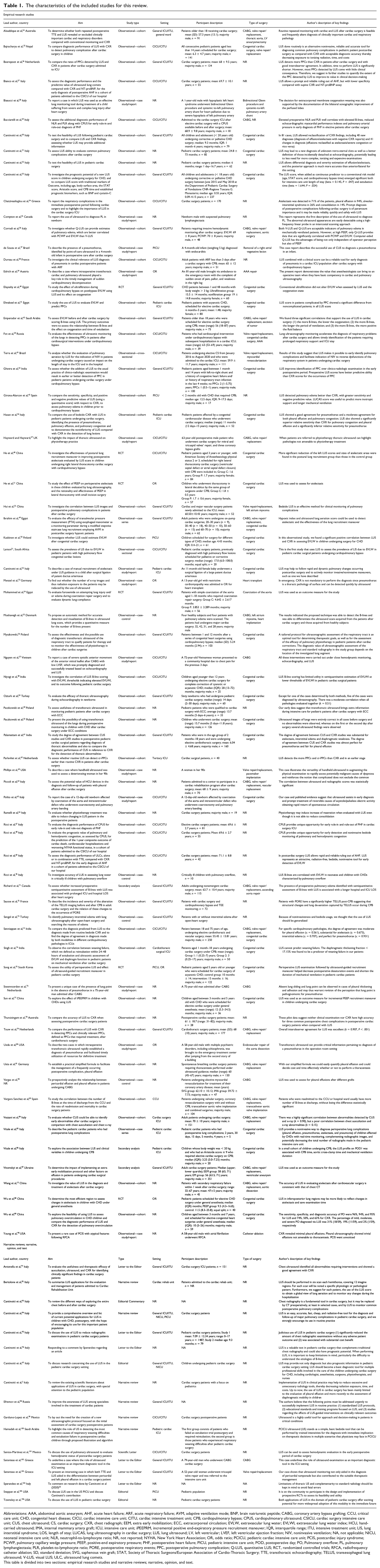
Abbreviations: AAA, abdominal aortic aneurysm; AHF, acute heart failure; ARF, acute respiratory failure; AVM, adaptive ventilation mode; BNP, brain natriuretic peptide; CABG, coronary artery bypass grafting; CCU, critical care unit; CHD, congenital heart disease; CICU, cardiac intensive care unit; CITU, cardiac intensive treatment unit; CPB, cardiopulmonary bypass; CPUS, cardiopulmonary ultrasound; CSICU, cardiac surgery intensive care unit; CUS, chest ultrasound; CS, cardiac surgery; CXR, chest radiograph; EEM, extra early mobilization; ECC, extracorporeal circulation; EVLW, extravascular lung water; EVLWI, extravascular lung water index; HCU, hand-carried ultrasound; IMA, internal mammary artery graft; ICU, intensive care unit; iPEEPRM, incremental positive end-expiratory pressure recruitment maneuver; IQR, interquartile range; ITU, intensive treatment unit; LIS, lung interstitial syndrome; LOS, length of stay; LUCAS, lung ultrasonography in cardiac surgery; LUS, lung ultrasound; LV, left ventricular; LVEF, left ventricular ejection fraction; NIV, noninvasive ventilation; NA, not applicable; NICU, neonatal intensive care unit; NLR, neutrophil-to-lymphocyte ratio; NR, not reported; NYHA, New York Heart Association; OR, odds ratio; PCICU, pediatric cardiac intensive care unit; PCIS, postcardiac injury syndrome; PCWP, pulmonary capillary wedge pressure; PEEP, positive-end expiratory pressure; PHF, postoperative heart failure; PICU, pediatric intensive care unit; POD, postoperative day; PO, Pulmonary overflow; PL, pulmonary lymphangiectasia; PLR, platelet-to-lymphocyte ratio; PORE, postoperative respiratory events; PPC, postoperative pulmonary complication; Q-LUS, quantitative LUS; RCT, randomized controlled trials; RFCA, radiofrequency catheter ablation; SD, standard deviation; SVG, saphenous vein graft; STAT, Society of Thoracic Surgeons/European Association of Cardio-Thoracic Surgery; TTE, transthoracic echocardiography; TELUS, transesophageal lung ultrasound; V-LUS, visual LUS; ULC, ultrasound lung comets.
This table is divided into two sections: empirical research studies and narrative reviews; narrative, opinion, and text.
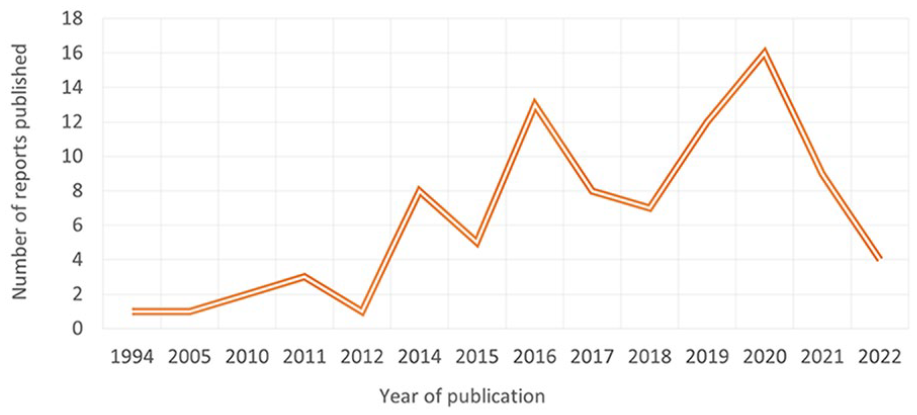
A graph of the publications by year, based on all included 90 reports.
Empirical Research Studies
Most of the included literature in this scoping review comprised empirical research (n = 73, 81.1%). This included 64 observational studies (71.1%), six randomized controlled trials (RCTs) (6.7%), and three secondary analyses (3.3%). Of the observational studies, the most common sub-type was cohort study (n = 43, 67.2%). Twenty of the studies were in the form of conference abstracts (22.2%). The most common setting that studies were conducted in was intensive care (n = 48, 88.9%). All studies included patients, with a large range in sample size (n = 1–351). Age was well-balanced between pediatric (n = 34, 51.5%) and adult patients (n = 32, 48.5%). Of the 55 primary studies that reported patients’ sex, 38 included mostly male patients (52%) while seven studies included only males (12.7%). Many primary studies included a mixed surgical population. Of the 55 primary studies, which reported type of surgery, the most common were congenital cardiac surgery (n = 31, 56.4%) followed by coronary artery bypass grafts (n = 19, 34.5%) and valve repairs or replacements (n = 19, 34.5%).
Narrative Reviews
This review included six narrative reviews (6.7%). All narrative reviews aimed to summarize LUS applications in cardiac surgery, with half focusing on the pediatric population (3.3%). The specific focus of the reviews varied from improving awareness of LUS among other specialists 98 to discussing the need for a new LUS protocol 99 to highlighting the role of LUS in weaning and extubation in pediatric cardiac patients. 100 The earliest narrative review was published in 2016. 2 Countries of origin included Italy2,92,94 (n = 3), Russia 98 (n = 1), Mexico 99 (n = 1), and Saudi Arabia 100 (n = 1).
Narrative, Opinion and Text
Other evidence types (n = 11, 12.2%) included in the scoping review consisted of letters to the editor (n = 6, 6.7%), editorials (n = 3, 3.3%), editorial commentary (n = 1, 1.1%), and a scientific letter (n = 1, 1.1%). Some reports underlined cases in which LUS played an important role in pathology identification and treatment.91,95,102 Other reports carried a discussion between researchers regarding studies conducted using LUS,96,105 including one letter to the editor 104 in which use of LUS in another included study 33 was questioned.
How and Why has LUS Been Used With Cardiac Surgery Patients?
Ten reasons for using or discussing LUS were identified from included reports (Table 2). Primary studies most commonly aimed to compare or determine the diagnostic (n = 27, 37%) or prognostic (n = 22, 30.1%) ability of LUS. In narrative reviews and within narrative, opinion, and text, LUS was most commonly suggested to be a prognostic tool (n = 8, 47.1%) or used as a standard protocol (n = 8, 47.1%) for cardiac surgery patients. Other suggested reasons for use across all included reports were using LUS to monitor pathology progression (n = 18, 20.2%), to assess a deteriorating patient (n = 16, 18%), and either to confirm or rule out a suspected pathology (n = 16, 18%).
The reported LUS methods.
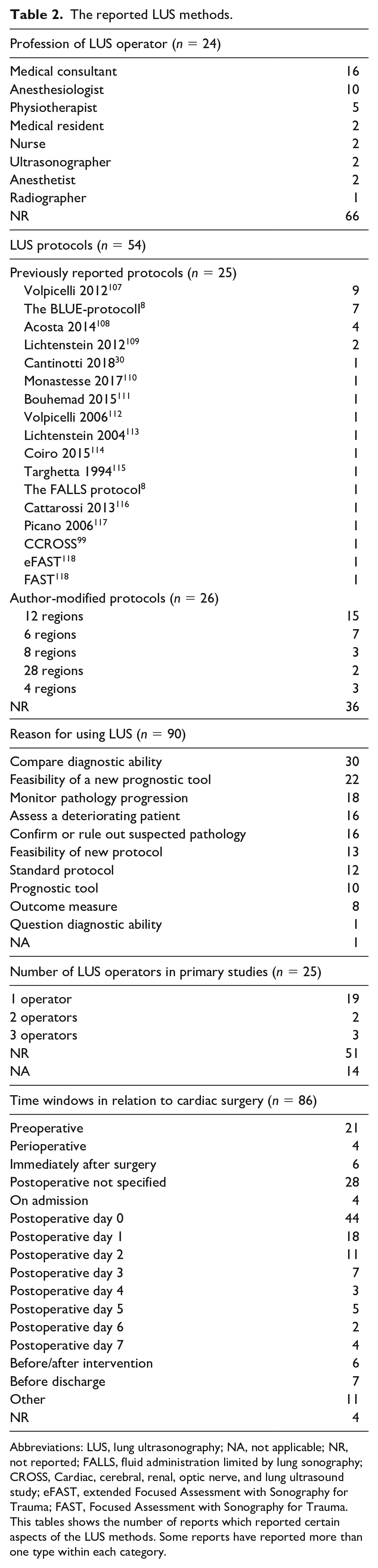
Abbreviations: LUS, lung ultrasonography; NA, not applicable; NR, not reported; FALLS, fluid administration limited by lung sonography; CROSS, Cardiac, cerebral, renal, optic nerve, and lung ultrasound study; eFAST, extended Focused Assessment with Sonography for Trauma; FAST, Focused Assessment with Sonography for Trauma.
This tables shows the number of reports which reported certain aspects of the LUS methods. Some reports have reported more than one type within each category.
Within primary studies, LUS was most commonly performed on postoperative day (POD) 0 (n = 40, 55.6%) followed by preoperatively (n = 20, 27.8%), POD1 (n = 17, 23.6%) and POD2 (n = 10, 13.9%). In the narrative reviews and narrative, opinion, and text, most reports suggested that LUS should be used postoperatively, but did not specify a day or time (n = 9, 64.3%). The frequency of scanning windows for up to POD7 and discharge can be seen in Figure 3 while a more extensive list can be seen in Table 2.
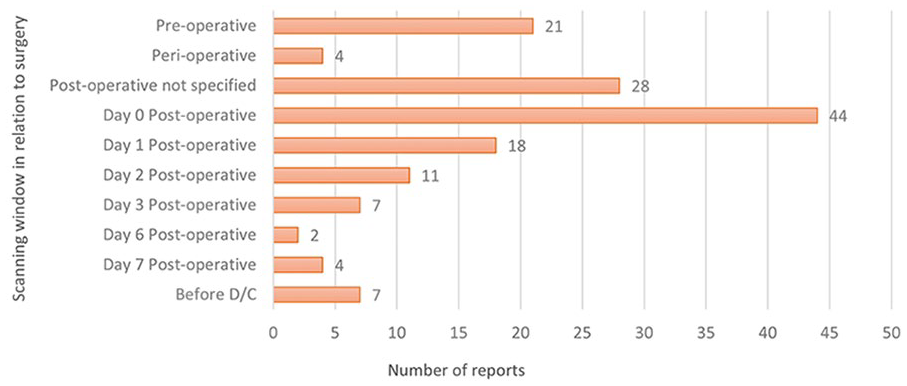
The scanning windows, used for lung ultrasonographer, in relation to cardiac surgery from preoperative to 1 week and D/C. D/C, discharge.
Which Anatomic Structures, Artifacts, and Pathologies Were Investigated?
The included reports found, sought, or discussed several anatomic structures, artifacts, and pathologies that can be investigated with LUS with many reporting more than one of each. Across all reports, B-lines were the most identified and sought artifact (n = 45, 81.8%) followed by lung sliding (n = 23, 41.8%) and A-lines (n = 18, 32.7%) (see Figure 4). Narrative reviews and narrative, opinion, and text discussed using B-lines to assess for extravascular lung water (EVLW) (n = 7, 63.6%). Eight primary studies used B-lines for this purpose (18.2%). Of the 82 reports, which reported pulmonary pathologies (91%), the most common pathologies of focus were pleural effusion (n = 42, 51.2%), atelectasis (n = 33, 40.2%), pneumothorax (n = 28, 34.1%), pulmonary edema (n = 23, 28%), and consolidation (n = 21, 25.6%) (see Figure 5).
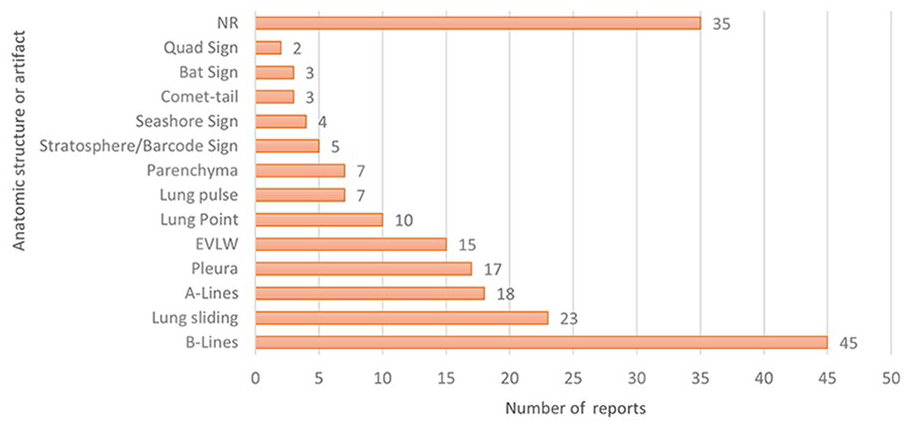
The anatomic features and artifacts found or sought, using lung ultrasonography (LUS), across all included 90 reports. EVLW, extravascular lung water; NR, not reported.
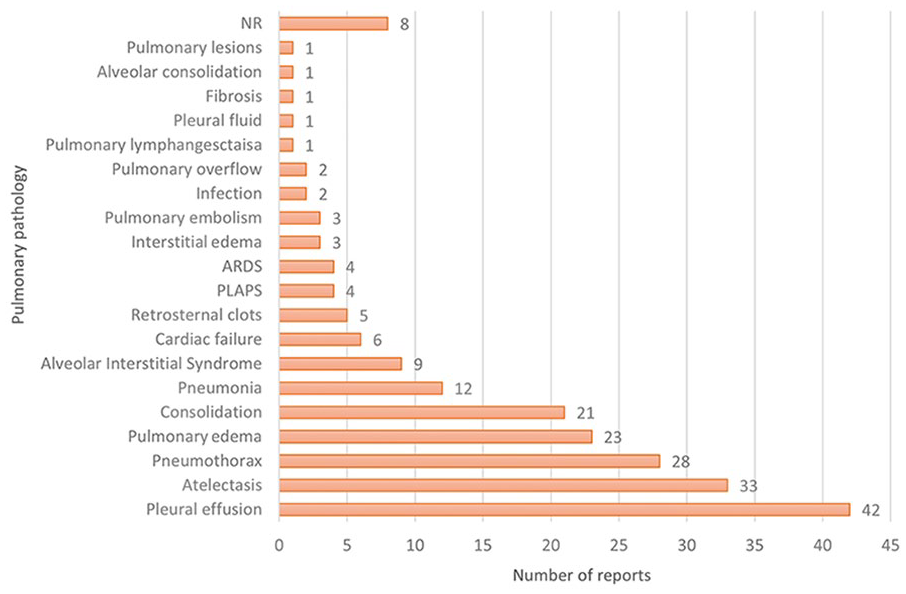
The pulmonary pathologies found or sought, using lung ultrasonography (LUS), across all included 90 reports. ARDS, acute respiratory distress syndrome; NR, not reported; PLAPS, posterolateral alveolar and/or pleural syndrome.
Who is Conducting LUS?
The profession of the LUS operators in primary studies was underreported with only 23 studies reporting a profession (31.5%). Of those 23 studies, the most common professions were anesthesiologists (n = 9, 39.1%) and medical consultants (n = 9, 39.1%). In the narrative reviews (n = 4, 4.4%) and editorials (n = 2, 2.2%), which discussed operator profession, physiotherapists (n = 3, 50%), and nurses (n = 2, 33.3%) were the most common nonphysician health care professions. Nonphysician LUS operators were first identified in an editorial in 2017, 97 with the first primary study involving a nonphysician LUS operator being published in 2019 and featuring a physiotherapist in a single patient case study. 47 An observational cross-sectional study included a physiotherapist as one of the two LUS operators along with a cardiologist in a study the following year. 43 Of the 25 primary studies that reported the number of LUS operators, it was most common to have one LUS operator for the study (n = 19, 76%). There were never more than three operators involved.
What LUS Methods Were Utilized?
There was a lack of reporting regarding LUS methods and protocols. Only 25 of all reports, where applicable, reported how many LUS operators were involved (43%), only 24 reports (26.7%) mentioned the profession of the LUS operator, and only 25 (27.8%) reported a previously reported LUS protocol. Of the primary studies, only 20 reported using a previously reported LUS protocol (27.4%).
There was a large variety of protocols used or discussed across all reports; a total of 18 different previously reported protocols were mentioned. Twenty-five primary studies used an author-modified protocol—a protocol which involved scanning either 4, 6, 8 12, or 28 regions of the chest, but did not cite a protocol previously reported (34.2%). The specific regions scanned varied across all author-modified protocols. The most commonly reported protocol was the international evidence-based recommendations for LUS by Volpicelli et al 107 (n = 7, 35%) followed by Acosta et al 108 (n = 4, 20%) for the pediatric population. Protocols were infrequently discussed in narrative reviews or narrative, opinion, and text (n = 5, 29.4%). Nonetheless, the most commonly reported protocol in these five reports was the BLUE-protocol 8 (n = 3, 60%).
This review also found a wide variety and combinations of scanning windows. While it was most common for patients to be scanned in the combination of preoperatively and POD0 (n = 6, 6.7%), there were a total of 69 unique combinations for when LUS was performed in relation to cardiac surgery. The most common scanning windows have been synthesized and reported individually by number of reports in Table 2 with several unique time windows grouped as “other,” for example, after 1 week of birth, before or after imaging, or POD18.
Discussion
In this scoping review, literature concerning the use of LUS with cardiac surgery patients was identified and explored, providing a comprehensive map of the evidence base to date. Lung ultrasonography has gained significant traction over the last decade, with a growing number of publications since 2014 exploring its use in this patient group. The reporting of LUS methods, however, was found to be inconsistent; this needs to be addressed in future research to better interpret and generalize findings to be applied in clinical practice.
Types of Sources
A total of 90 reports were included, none of which were systematic reviews. The reporting of LUS methods in primary studies, however, was variable and heterogeneous, which makes formal synthesis of the current evidence base challenging. Evidence syntheses are valuable resources for clinicians and are used to inform clinical practice guidelines. Therefore, there is a need for further high-quality research to be conducted and adequately reported to better facilitate future synthesis of the evidence base to provide robust practice recommendations. Moreover, no qualitative studies were identified with the search strategy, highlighting the need for further research to investigate the facilitators, barriers, and perspectives of patients and clinicians regarding the use of LUS in the cardiac surgery population. While it is vital to conduct quantitative research, it is equally valuable to investigate how best to integrate LUS into practice through qualitative exploration of patients’ and clinicians’ experiences.
The Pediatric Population
Just over half of the reports were centered on the pediatric patients, with the most prevalent surgery type being congenital cardiac surgery. Lung ultrasonography has become popular in this patient group, due to its noninvasive and nonionizing nature and is being used more often for the diagnosis and surveillance of pediatric pulmonary conditions. 119
The Uses of LUS
The use of LUS as a diagnostic tool has been extensively studied in both cardiac surgery patients and other groups. Lung ultrasonography use to identify PPCs has been seen in this review as monitoring pathology progression, assessing deteriorating patients, and confirming or ruling out suspected pathologies. Furthermore, LUS has also gained popularity as a prognostic tool. Pulmonary edema became a pathology of interest within the included reports in 2012, 23 and in 2014, B-lines began to be used to assess for EVLW to predict and diagnose pulmonary edema. 70 Another way LUS is used as a prognostic tool is using a LUS score to attempt to predict a variety of conditions. Lung ultrasonography score can be used to predict death, intensive care unit (ICU) admission, endotracheal intubation, and weaning failure with moderate accuracy. 120 Some authors have described the potential for LUS to go beyond its role as a diagnostic tool, specifically as a way of measuring the effectiveness of recruitment maneuvers.121,122 Only eight studies in this review (8.9%) explored using LUS as an outcome measure for intervention. This potential ability of LUS therefore requires more exploration through further high-quality research.
The Use of LUS by Nonphysician Health Care Professions
The evidence base regarding nonphysician health care professionals using LUS, with cardiac surgery patients is limited. Studies with noncardiac patients have explored this concept: Le Neindre et al 123 explored the influence of LUS on physiotherapists; for example, clinical decision-making when managing critical care patients. Further studies have begun to assess the ability of nurses to operate traditional124,125 and handheld LUS 126 following surgery or to assess cardiogenic dyspnoea. In addition, several narrative reviews have discussed the potential for nonphysician health care professionals to use LUS within their scope of practice.10,127 Considering the rate of mortality in postcardiac surgery patients, it is important to detect PPCs early on; Increasing the accessibility of LUS for other professions within the multidisciplinary team may aid in detecting PPCs earlier and allow for more appropriate treatment sooner. Further primary research involving these professional groups is required if this potential is to be realized.
LUS Techniques and Methods
This review found a lack of reporting regarding LUS techniques and methods. This is consistent with other LUS evidence syntheses: Heldeweg et al 128 raised the same concern for methodological inconsistencies in a systematic review evaluating the impact of LUS on clinical decision-making in the emergency department, intensive care, and in the general ward; Hayward and Janssen 129 found it difficult to compare studies with the numerous different scanning techniques in their scoping review exploring the use of thoracic ultrasound by physiotherapists; new international recommendations by Demi et al 130 suggest extensive studies to define the optimal imaging settings for LUS. At most, only nine reports cited the same previously reported protocol. This could be attributed to the lack of available protocols to replicate in research. The protocols most cited, Volpicelli et al 107 international recommendations and the BLUE-protocol, 8 were not introduced until 2012 and 2014, respectively. The first instance of a protocol cited in the included primary studies was in 2014 with the BLUE-protocol. 53 It is possible the sudden rise in the popularity of LUS has not allowed time for more standardized protocols to be developed and evaluated, which may be contributing to the inconsistent reporting.
The methodological inconsistency among studies could have an impact on the advancement of LUS research. For instance, the 27 primary studies assessing the diagnostic ability of LUS contained a variety of eight different protocols with a mix of previously reported protocols and protocols unique to the study. The diversity in scanning techniques and time windows may introduce confounding variables, making it difficult to generalize findings. In addition, this can pose challenges to clinicians looking to incorporate evidence-based LUS practice.
Furthermore, the protocols which have been previously reported and replicated vary in purpose and technique. The recommendations by Volpicelli et al 107 include a variety of methods. While an eight-region LUS examination is recommended for general patients with interstitial syndrome, a two-region approach is recommended for the critically ill. Other recommendations suggest what region to begin scanning and which direction to travel in without specifying a number of zones, for example, the technique for lung consolidation should begin at the area of interest and progress to the entire lung as needed. The BLUE-protocol 8 is specifically for patients with acute respiratory failure and consists of six regions, or “points.” Literature in other populations suggest otherwise: a prospective cohort study evaluating the impact of different LUS protocols in the assessment of lung lesions in COVID-19 patients found a 12-region method to improve diagnostic power compared with a 10- or an eight-region method; 131 a retrospective cohort found similar results in their secondary analysis finding a 12-region method to be superior to an eight- or six-region method. 132 The protocol used by Acosta et al 108 does consist of a 12-region method for children with anesthesia-induced atelectasis; this method was used solely by studies with a pediatric population in this review, but only two were assessing for atelectasis.21,48
Challenges for Clinical Practice
With so many variations in protocols for varying patient groups and pathologies, conducting research with consistent LUS methods and selecting the most appropriate protocol to use clinically remains a challenge. Standardizing protocols and methods will improve consistency in the research, facilitating more effective evidence synthesis. This, in turn, can improve generalization of diagnostic findings to cardiac surgery patients. Once best practices for LUS is established, future research can more effectively investigate other queries, such as the indications for LUS, effective scanning windows, alternative LUS applications, and who may be able to use LUS in other fields. Improving clinical practice and the ability to detect PPCs in a timely and efficient manner begins with improving consistency of LUS methods in the research.
Criticism
Despite its growing popularity, there is some remaining doubt and skepticism regarding the use of LUS. One letter to the editor by Sperandeo 104 questioned the diagnostic capability of LUS when using B-lines as a pathognomonic marker of lung disease. Owing to the growing interest in LUS over the past decade, there remains a significant amount of research to be conducted on both its established and novel applications, making it a challenging task to comprehensively investigate all aspects of the tool. As further research is needed to fully understand the potential of LUS, researchers and clinicians may continue to approach its use with caution and in conjunction with other tools.
Recommendations
Standardizing LUS protocols would be advantageous for future research, and qualitative studies could shed light on the facilitators, barriers, and perspectives of LUS operators in this population, as well as the experience of patients themselves. Once LUS best practice is established, exploring alternative applications of LUS, such as its potential as a measure of treatment effectiveness, would be beneficial. Further investigation of the use of LUS by other health care professionals, including physiotherapists and nurses, is encouraged before any recommendations for scope of practice can be made.
Limitations
This review involved a comprehensive search strategy and protocol which was developed by an experienced review team. Despite the rigorous approach, it is possible that some relevant articles may have been missed. This scoping review was limited to records which were either in English or could be translated by Google Translate. Nonetheless, the five included reports translated using Google may have a degree of inaccuracy. Illegible or untranslatable reports were excluded; therefore, mapping the entire evidence base was not possible. While the methodological quality of the literature was not assessed, this was in keeping with methodological guidance for scoping reviews, 16 which aim to map available literature rather than assess the quality.
Conclusion
This scoping review has comprehensively mapped the current literature exploring the use of LUS with cardiac surgery patients. While LUS has garnered significant attention in the field of cardiac surgery, this scoping review has identified areas requiring further investigation to fully harness its potential. Further research is needed to establish best practices for LUS, including standardizing methods, exploring its use by other professions, and conducting qualitative studies. Lung ultrasonography has the potential to improve patient outcomes by enabling early identification of PPCs. With continued research, LUS may prove to be a valuable tool for clinicians and researchers to assess cardiac surgery patients.
Supplemental Material
sj-docx-1-jdm-10.1177_87564793231198521 – Supplemental material for Exploring the Use of Lung Ultrasonography to Assess Cardiac Surgery Patients: A Scoping Review
Supplemental material, sj-docx-1-jdm-10.1177_87564793231198521 for Exploring the Use of Lung Ultrasonography to Assess Cardiac Surgery Patients: A Scoping Review by Casey Farrell, Kay Cooper, Simon Hayward and Craig Walker in Journal of Diagnostic Medical Sonography
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
