Abstract
Objectives:
Visceral leishmaniasis remains a deadly parasitic disease with diagnostic complexities. Currently, point-of-care chest imaging is gaining momentum in the diagnosis of infectious diseases. Respiratory symptoms are common in visceral leishmaniasis. Here we aimed to systematically synthesize the evidence on the utility of chest imaging on the diagnosis and management of patients with visceral leishmaniasis.
Methods:
We searched PubMed, Scopus, Web of Science, ScienceDirect, and Google Scholar databases for studies reporting chest imaging findings in patients with visceral leishmaniasis, published in English from database inception to November 2022. We used the Joanna Briggs Institute checklists to evaluate the risk of bias. The protocol of this systematic review was registered with the Open Science Framework: https://doi.org/10.17605/OSF.IO/XP24W.
Results:
Of 1792 studies initially retrieved, 17 studies with 59 participants were included. Of the 59 patients, 51% (30) had respiratory symptoms and 20% (12) were human immunodeficiency virus co-infected. Chest X-ray, high-resolution computed tomography, and chest ultrasound findings were available for 95% (56), 93% (55), and 2% (1) of the patients, respectively. The most common findings were pleural effusion (20%; 12), reticular opacities (14%; 8), ground-glass opacities (12%; 7), and mediastinal lymphadenopathies (10%; 6). High-resolution computed tomography was more sensitive than chest X-ray and detected lesions that were lost on chest X-ray, 62% (37) versus 29% (17). In almost all cases, regression of the lesions was observed with treatment. Microscopy of pleural or lung biopsy detected amastigotes. Polymerase chain reaction yield was better in pleural and bronchoalveolar lavage fluids. A parasitological diagnosis from pleural and pericardial fluid was possible in AIDS patients. Overall, the risk of bias was low.
Conclusions:
Visceral leishmaniasis patients frequently had abnormal findings on high-resolution computed tomography. Chest ultrasound is a useful alternative in resource-limited settings to aid in diagnosis and subsequent treatment follow-up, especially when routine tests yield negative results despite clinical suspicion.
Keywords
Introduction
Visceral leishmaniasis (VL), a parasitic disease caused by Leishmania donovani and Leishmania infantum,1–4 is almost always fatal if left untreated and is the second leading cause of parasitic deaths after malaria.5,6 It is estimated that 50,000–90,000 new cases of VL occur worldwide each year, with many cases going unreported. 5 The highest disease burden and human immunodeficiency virus (HIV)-VL co-infection rate is found in East Africa, India, and Brazil.5,7,8 East Africa, the worst affected region, accounted for 57% of VL cases reported to WHO in 2020. 7 The highest co-infection rate between 20% and 45% is reported from north-western Ethiopia.9–11 HIV-VL co-infection poses a challenge for early diagnosis and treatment, and negatively impacts efforts to control and eliminate VL. 7 So far, East African countries rely on early diagnosis and effective treatment to reduce disease prevalence and mortality rates due to a lack of robust vector control measures.5,9
In endemic settings, diagnosis of VL is made based on clinical criteria and serological tests, mainly rk39 immunochromatographic test.1,9–11 Definitive diagnosis depends on tissue detection of the amastigote form.12,13 Currently available serological tests are reliable for use in India.10,14 However, they have reduced sensitivity in East Africa, significantly less in HIV-VL co-infected patients.9,11,14–17 These tests cannot also be used to diagnose relapse and confirm response to treatment.13,14 More sensitive molecular tests such as polymerase chain reaction (PCR) are not currently available for routine use. 4 Therefore, microscopic examination of invasive specimens is required for diagnosis. 9 Tissue sampling, especially splenic aspiration, can lead to serious complications in inexperienced hands.20,28,29 Clinical findings in VL are nonspecific12,16 and immunosuppressed patients, AIDS patients, transplant recipients, and children with primary immunodeficiency can have atypical presentations.18–20 These all add to a complex diagnosis of VL.
The importance of ultrasound (US) in diagnosing tropical infectious diseases, particularly in resource-poor settings with high HIV infection, has been studied with remarkable detail.21–24 Studies found that a diagnostic US protocol focused assessment with sonography for HIV/tuberculosis (FASH), hastened treatment initiation, and provided exclusive support for TB treatment in 50–72% of study participants.22,25,26 In this protocol, patients are scanned in six different positions to identify pathologies in the chest, abdomen, and fluid collections in pelvis. 27 In the case of VL, studies have elaborated the abdominal US findings taking into account typical features of the disease.28–30 One study showed that abdominal US findings, hepatosplenomegaly, lymph node enlargement, and nodular splenic lesions, have unclear diagnostic accuracy. 21 A study conducted in Ethiopia reported that there is no evidence to support the routine use of abdominal US in the management of VL except to detect subclinical complications of splenic aspiration. 29
However, previous studies have given limited attention to chest imaging findings, which are not uncommon, with a sparse attempt to review the diagnostic yield of specimens from the identified lesions. To the best of our knowledge, this is the first systematic review done on this subject. This study evaluates chest imaging, namely chest X-ray (CXR), chest US, and high-resolution computed tomography (HRCT) findings in patients with VL and appraises the diagnostic yield of specimens collected from identified lesions. This could help identify the role of chest imaging in the diagnosis and management of VL patients.
Methods
This systematic review was reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA 2020) guidelines. 29 The protocol of this systematic review was registered with the Open Science Framework: https://doi.org/10.17605/OSF.IO/XP24W.
Eligibility criteria
Participants
Human patients with VL,
Diagnosed for VL based on laboratory criteria set by WHO, 9 and
No restrictions based on age groups, sex, immune, and co-infection status.
Intervention
Chest imaging with any of the diagnostic imaging modalities: CXR, chest US, or HRCT.
Radiology reports written or images available.
Comparator
With or without a comparator.
Outcomes
Early diagnosis and improved prognosis.
Study designs
No restrictions,
Published in English language.
Data sources
To find eligible studies, we searched PubMed, Scopus, Web of Science, ScienceDirect, and Google Scholar databases from their inception to November 25, 2022. Additional relevant studies were identified during forward citation searches and reference lists reviewed.
Search strategy
The search was run in incognito mode to reduce personalization bias. MeSH terms were identified from PubMed and supplemented with free text to enable a comprehensive search (Supplemental Material 1). The search strategies used in PubMed for the MeSH terms and text words was: “Visceral leishmaniasis” OR “Kala Azar” OR “kala-azar” OR “Fever, Black” OR “Black Fever” OR “Leishmaniasis, Visceral”[Mesh] AND Imaging* OR Radiography OR “X-Ray*” OR “X RAY” OR Ultrasound* OR Ultrasonographic OR ultrasonic OR CT OR Tomography OR “Diagnostic Imaging”[Mesh] AND “Thorax”[Mesh] OR “Respiratory System”[MeSH] OR “Heart”[MeSH] OR chest OR Pleur* OR Lung OR Pulmonary OR Pericardi* OR Trachea* OR Bronchi* OR mediastin*.
Study selection
The search results from the five databases were merged into an Excel spreadsheet and duplicates were removed. The title and abstract screening of the retrieved results was performed independently by three authors (TH, TA, and YM) based on the eligibility criteria. Papers for which the three reviewers did not reach a consensus were considered for full-text screening. Studies eligible for full-text review were retrieved and selection criteria were reapplied. The three reviewers performed the screening independently, and disagreements were resolved through discussion.
Data extraction
We developed a structured data extraction form and created a template in systematic review software (Covidence systematic review software, Veritas Health Innovation, Melbourne, Australia). The form was pilot tested by TH, TA, and YM in four randomly selected studies and then adjusted as appropriate. The three reviewers independently extracted data from eligible studies, and differences were negotiated through discussion. The information extracted are as follows:
The report: Author, journal, and year of publication.
Study characteristics: Study type, number of participants, and setting and country the study was conducted.
Patient characteristics: Age, sex, immune status, and comorbidities.
Clinical presentations: Clinical history and physical examinations, laboratory test results, and whether the case was new or relapsed.
Chest imaging findings: Details of chest imaging findings, the sample taken from pathologies as applicable, and results of tests performed.
Assessment of risk of bias
We assessed the risk of bias in the included studies using Joanna Briggs Institute (JBI) risk assessment tools for case reports and prevalence studies31,32 based on the study type. Three reviewers (TH, TA, and YM) independently scored individual items of the checklists. Any discrepancies were dealt with discussion. An overall score was calculated by adding the score given to each item, and calculating a percentage. The papers were then classified as low risk (score > 70%), medium risk (score: 50–69%), or high (score < 49%) risk of bias.
Results
Study characteristics and patient demographics
A total of 1792 search results were retrieved from the five databases and 27 duplicates were removed before screening. After title and abstract screening, 1734 papers were removed as they did not meet the selection criteria. Full-text screening was performed for 31 articles and 17 studies were eligible and thus included in the review (Figure 1).

PRISMA flow diagram of the study.
Most of the included studies were case reports and one study followed a cross-sectional design.33–49 The majority of the studies came from India, southern European countries, and Brazil. The studies included 59 patients with a mean age (standard deviation) of 40.3 (±5.9) years, with males predominating (74.5%; 44) and 20% (12) patients co-infected with HIV/AIDS and immunocompromised. Other reported comorbidities were hepatitis B virus, hepatitis C virus, diabetes mellitus, and tuberculosis, each present in two patients. Patient demographics and clinical presentation are summarized in Table 1.
Demographic characteristics and clinical presentation of patients in the included studies.
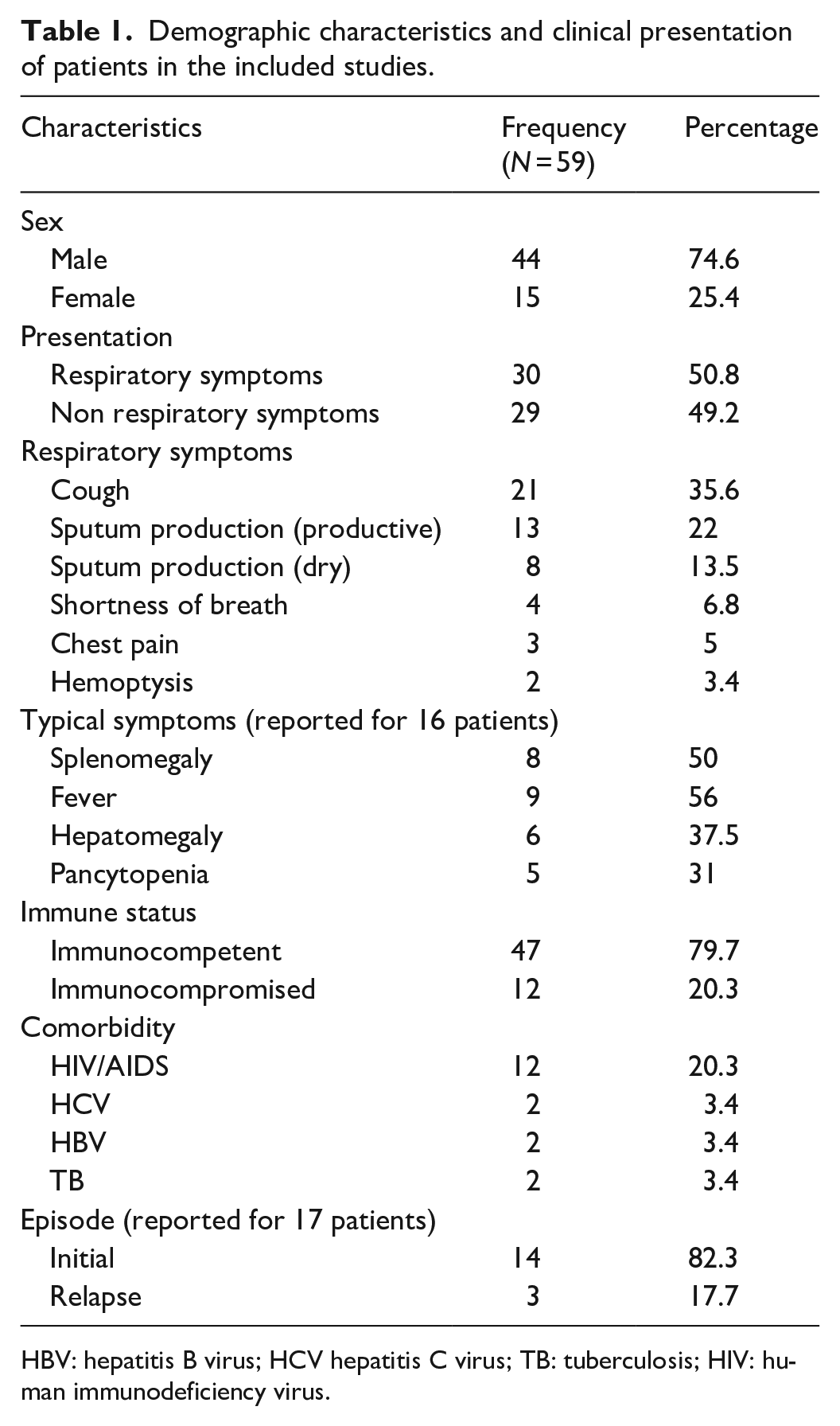
HBV: hepatitis B virus; HCV hepatitis C virus; TB: tuberculosis; HIV: human immunodeficiency virus.
Clinical presentation of patients
The number of patients with or without respiratory symptoms was comparable, 50.8% and 49.1%, respectively. Cough was reported by 21 (35.6%) patients, shortness of breath by 4 (6.8%) patients, chest pain by 3 (5%) patients, and hemoptysis by 2 (3.4%) patients (Table 1). In the 42 patients included in the cross-sectional study, splenomegaly was the most common finding. However, among the case reports with a full description of physical examinations, it was reported that splenomegaly was present in only eight (50%) of included patients, with five (62.5%) of these being diagnosed with VL-HIV co-infection.
Chest imaging findings
The studies used CXR, HRCT, or chest US as diagnostic imaging modalities. The cross-sectional study required participants to undergo both CXR and HRCT, and a full report of the results was available. Among the case reports, no CXR and HRCT findings were available for three and four studies, respectively. Echocardiography was performed on a single patient (Table 2).
Chest imaging modalities utilized and findings reported from included studies.

CXR: chest X-ray; HRCT: high-resolution computed tomography; US: ultrasound.
The most common chest imaging findings were pleural effusion in 20% (12), reticular opacities in 14% (8), ground-glass opacities in 12% (7), and mediastinal lymphadenopathies in 6(10%) of patients. Most of these findings, including but not limited to pulmonary interstitial changes and mediastinal lymphadenopathies, were diagnosed by HRCT. CXR of two patients showed an enlarged cardiac silhouette identified as a pericardial effusion on US in one patient and a pleuropericardial cyst on HRCT in another patient. Among patients with confirmed pulmonary VL, one patient had a bronchoapneumonia pattern on CXR and another patient had bilateral cavitary lung lesions on CXR and HRCT.
The prevalence of lung lesions detected by HRCT tends to increase with disease duration. The mean time from symptom onset to hospital admission was 58 days and 34 days in patients with and without HRCT pathology findings, respectively, in 71% (42) of patients. Interestingly, HRCT findings consistent with progressive lung injury were absent, even in patients who presented late. Overall, imaging findings were found to resolve with effective treatment. The chest imaging results reported from the included studies are summarized in Table 3.
A summary of HRCT and CXR imaging findings of patients included in the studies.

N: absolute frequency; %: percentage; No: number; CXR: chest X-ray; HRCT: high-resolution computed tomography.
VL diagnostic tests
In all, 12 studies have reported leishmaniasis tests, microscopy, culture, or PCR performed on clinical specimens collected from identified chest pathologies. Microscopic examination was the most commonly performed test, and mediastinal lymph nodes were examined relatively more frequently. The test results exclusively informed the diagnosis of VL in eight patients, two of them had culture-negative bone marrow aspirates, and microscopic examination or culture of the bronchoalveolar lavage (BAL) fluid was also negative. To make a diagnosis, either a PCR test was performed on the same samples or microscopic examination of more invasive samples was done. A pleural fluid or pericardial fluid microscopy examination was performed on three patients and tested positive. Notably, they were AIDS patients. Leishmania amastigotes have been identified at atypical sites, including BAL fluid, tracheal or bronchial tissue, mediastinal lymph nodes, and a cystic mediastinal mass in apparently immunocompetent patients. A list of reported leishmaniasis diagnostic tests is summarized in Table 4.
VL diagnostic tests done on samples taken from identified pathologies and their results.

BAL: bronchoalveolar lavage; VL: visceral leishmaniasis; PCR: polymerase chain reaction; IFAT: indirect fluorescence antibody test.
Test results were negative.
Quality assessment and risk of bias
Based on the JBI quality appraisal tools, five case reports were assessed to be high/moderate risk. The other included studies were low risk. Overall, the studies were deemed to be low risk (Figure 2).

“Risk of bias” summary: review authors’ judgment about each “risk of bias” item and overall appraisal for case reports.
Discussion
We included 17 articles in our review, 16 case reports, and one cross-sectional study. VL has heterogeneous imaging findings of the chest that occur more frequently than previously thought. 33 Pleura, lung parenchyma, mediastinal lymph nodes, airways, and the pericardium can be affected.33,44,46,49 Pleural effusion and pulmonary interstitial changes were the predominant imaging findings, and the pulmonary vessels were relatively spared. This is in contrast to chronic pulmonary schistosomiasis (CPS), in which pulmonary hypertension can develop over time, whereas the pleura and pericardium are spared.50,51 A possible explanation for this difference could be chronic inflammation in periarteriolar regions induced by migrating eggs in CPS and systemic spread of the parasite in VL.
Immune dysregulation associated with VL may predispose patients to develop pulmonary infections.43,52 However, respiratory symptoms should be considered signs of lung involvement in both immunocompetent and immunocompromised patients.38,39,43,45 In addition, patients with no respiratory symptoms may still have abnormalities on chest imaging, and the parasite can be detected.36,40,41,46 The sum of imaging findings combined with the clinical picture and epidemiology of the disease can help in reaching a diagnosis, even if individual imaging abnormalities are nonspecific and uncommon.50,53 A parasitologic and imaging response to treatment was observed in almost all cases, in line with response to treatment seen in CPS, including some cases of schistosomiasis-associated pulmonary hypertension.50,54 This observation suggests that follow-up imaging could confirm disease resolution and spare the patient from invasive testing.
HRCT is a more sensitive imaging modality and detects lesions that are lost on CXR, 62% (37) and 29% (17) of patients, respectively.33,34,39 This is consistent with what was found in previous studies on tropical lung disease, CPS and pulmonary hydatid cysts.50,55,56 CXR can serve as the primary imaging modality in low-resource settings, as only subtle or complicated imaging findings go undetected, and it is convenient for use in patient follow-up.50,57 However, our findings support HRCT as the imaging modality of choice in VL patients.
Chest US is a viable alternative to detect abnormal findings in the chest. 33 Man et al. 58 found that transthoracic lung US is a useful tool for the diagnosis and follow-up of patients with interstitial lung disease. Coley et al. 59 stated that US can reliably diagnose chest pathologies in children. US-guided biopsy can be safely performed in numerous pulmonary and mediastinal pathologies.60,61 Recently, it has been suggested that point-of-care lung US-based algorithms are useful for triage of patients with symptoms of COVID-19, especially when resources are limited.62,63 A study also found the importance of lung US to diagnose pneumocystis jirovecii pneumonia in HIV patients. 64 US is an appealing imaging modality for use in resource-constrained countries as it is safe, inexpensive, portable, and can be used by a range of users. 65
Diagnosis of VL can be made based on demonstration of the parasite in pleural fluid,35,40 BAL fluid, 47 transbronchial or open lung biopsy,39,42,45 pleural biopsy 45 mediastinal lymph node biopsy,36,43,45 pericardial fluid 46 tracheal, or bronchial biopsy.43,49 PCR on respiratory samples has also been used to diagnose leishmaniasis with increased sensitivity.40,44 Patients with lung involvement, lung biopsy, and BAL fluid have a better diagnostic yield for amastigotes than pleural fluid. 40 However, microscopic examination of pleural and pericardial fluid was particularly sensitive in AIDS patients.35,46 This can be due to a high parasite load seen in HIV-VL co-infected patients.9,66 It is important to note that negative microscopic exam results of any respiratory specimen cannot rule out pulmonary involvement.44,45,52 PCR detection of the parasite from pleural fluid and BAL fluid is a promising diagnostic test.40,44 Of note, newly developed PCR assays have improved diagnostic accuracy and potential to be used as point-of-care tests in the future. 14
We acknowledge the following limitations of the study. All the included studies, except one, have not utilized US, thus typical or suggestive US findings are not available. Most of the included studies were case reports, which naturally include a higher risk of publication bias 67 despite the overall low risk of bias assessment. The differential diagnoses commonly considered for the above imaging findings were not discussed. However, a diagnosis of VL should be suspected in a patient with abnormal chest imaging findings and a favorable clinical presentation, regardless of the patient’s immune status.
Conclusion
IHRCT was abnormal in more than half of the VL patients included in the review, although the findings were nonspecific and heterogeneous. Chest US may be developed for low-resource settings as an alternative to HRCT and used when routinely available diagnostic tests are negative despite clinical suspicion of VL. PCR has improved sensitivity to identify the parasite from respiratory samples, and parasitological tests give more positive results in AIDS patients.
Supplemental Material
sj-docx-1-smo-10.1177_20503121231177812 – Supplemental material for Utility of chest imaging in the diagnosis and management of patients with visceral leishmaniasis: A systematic review
Supplemental material, sj-docx-1-smo-10.1177_20503121231177812 for Utility of chest imaging in the diagnosis and management of patients with visceral leishmaniasis: A systematic review by Tesfahunegn Hailemariam, Yonas Mebratu, Tsion Andrias, Fikremariam Melkeneh, Abel Abebe, Biruk Mulualem, Zewdu Abadi, Wazema Desta, Selamawit Bedasso, Fekadu Belay, Abibual Sileshi, Elilta Desta, Felipe Velsaquez-Botero, Rahel Birhane, Francesco Marinucci and Tsegahun Manyazewal in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121231177812 – Supplemental material for Utility of chest imaging in the diagnosis and management of patients with visceral leishmaniasis: A systematic review
Supplemental material, sj-docx-2-smo-10.1177_20503121231177812 for Utility of chest imaging in the diagnosis and management of patients with visceral leishmaniasis: A systematic review by Tesfahunegn Hailemariam, Yonas Mebratu, Tsion Andrias, Fikremariam Melkeneh, Abel Abebe, Biruk Mulualem, Zewdu Abadi, Wazema Desta, Selamawit Bedasso, Fekadu Belay, Abibual Sileshi, Elilta Desta, Felipe Velsaquez-Botero, Rahel Birhane, Francesco Marinucci and Tsegahun Manyazewal in SAGE Open Medicine
Supplemental Material
sj-docx-3-smo-10.1177_20503121231177812 – Supplemental material for Utility of chest imaging in the diagnosis and management of patients with visceral leishmaniasis: A systematic review
Supplemental material, sj-docx-3-smo-10.1177_20503121231177812 for Utility of chest imaging in the diagnosis and management of patients with visceral leishmaniasis: A systematic review by Tesfahunegn Hailemariam, Yonas Mebratu, Tsion Andrias, Fikremariam Melkeneh, Abel Abebe, Biruk Mulualem, Zewdu Abadi, Wazema Desta, Selamawit Bedasso, Fekadu Belay, Abibual Sileshi, Elilta Desta, Felipe Velsaquez-Botero, Rahel Birhane, Francesco Marinucci and Tsegahun Manyazewal in SAGE Open Medicine
Footnotes
Acknowledgements
The authors thank the staff of CDT-Africa who facilitated the study.
Authors’ contributions
TH developed the protocol with the advice from TM. TH, YM, and TA reviewed the reference list and extracted the data. TH and RB conducted the analysis. TH, TA, YM, RB, FB, FM (Francesco Marinucci), and TM evaluated the quality and comprehensiveness of the study and underlying data. TH developed the first draft of the manuscript and TM critically reviewed it. TH, YM, TA, FM, AA, BM, ZA, WD, SB, FB, AS, ED, FV-B, RB, FM, and TM read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the Center for Innovative Drug Development and Therapeutic Trials for Africa (CDT-AFRICA), College of Health Sciences, Addis Ababa University, Ethiopia.
Ethics approval
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
The datasets analyzed during the current study will be available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
