Abstract
Objective:
Early recognition of acute coronary syndromes (ACSs) is crucial in the emergency department. This study was designed to determine the diagnostic value of echocardiography, which was performed by an emergency medicine (EM) physician, to diagnosis ACS.
Materials and Methods:
This prospective and cross-sectional study was conducted between June 2011 and December 2011. All patients who were admitted with chest pain, to the EM department, and fulfilled the inclusion criteria were enrolled in this study. The focused echocardiography was performed by the EM physician. All echocardiography videos recorded were shown to the cardiologist. The final comments were compared with EM physician’s early findings.
Results:
Out of 48 patients, four were diagnosed with ACS. Three out of four patients diagnosed with ACS were detected by the EM physician with echocardiography. The EM physician’s prediction value with echocardiography in ACS diagnosis was 75% (95% confidence interval [CI] range 59%–100%), and the negative predictive value was 97% (95% CI range 77,9%-100%). There was no major cardiac event in the patient in whom the EM physician failed to detect a wall motion defect.
Conclusion:
Echocardiography is a bedside, noninvasive test for diagnosis of ACS. The EM physician who participated in this study was able to recognize ACS with the absence of a segmental wall motion defect, as well as the cardiologist providing the final report.
Keywords
Admission to the emergency department (ED) with chest pain constitutes an important group of patients. In the United States, 5% of ED admissions are due to chest pain. 1 Chest pain can be a presentation with conditions such as aortic dissection, pulmonary thromboembolism, and cardiac tamponade. A rapid and prompt evaluation is needed for diagnosis, initiation of treatment, and avoiding morbidity and mortality.
Coronary heart disease (CHD) is the single largest cause of death in the developed countries and is one of the leading causes of disease burden in developing countries. 2 Coronary artery disease (CAD) is the most common form of cardiovascular diseases (CVDs), and CAD is associated with high mortality and morbidity. The clinical manifestations of CAD include silent ischemia, stable angina pectoris, unstable angina, myocardial infarction (MI), heart failure, and sudden death.
The evaluation of patients with symptoms of chest pain is a serious process in an ED. In the differential diagnosis of chest pain, acute coronary syndrome (ACS) is a special consideration. It has been determined that more than 1.5 million people in the United States have ACS each year. 3 CHD is the leading cause of CVD deaths throughout the world. Forty-three percent of all CVD deaths are attributable to CHD according to the global burden of disease estimates from 2001. Globally, CVD deaths represent about 30% of all deaths. 2
Retrosternal onset of chest pain forms the basis of the ACS clinic. For the diagnosis of ACS, clinical, laboratory, and electrocardiographic (ECG) changes were well established in literature. 2 In ACS diagnosis, at least 6 hours of observation, serial cardiac enzyme, and ECG follow-up are required according to the risk classification of the patient. With early diagnosis of the disease and timely treatment, morbidity and mortality will be positively affected, and complications that may arise during the follow-up period can be controlled more easily.4,5
Echocardiography at the patient’s bedside can be conducted noninvasively with a cost-effective device used in the ED. Echocardiography plays an important role in the diagnosis of fatal conditions such as ACS, cardiac tamponade, pulmonary thromboembolism, aortic dissection, and so on. The presence or absence of left ventricular segmental wall motion defects were evaluated and used to support or exclude the diagnosis of ACS. However, the diagnostic value will change according to specialty, which is the background of this study. Cardiologists may not always be available in EDs. Early diagnosis of life-threatening conditions such as ACS by echocardiography especially in the ED may decrease the mortality and morbidity. The context of this study was to compare the diagnostic accuracy of an emergency-department physician conducting echocardiography with the diagnostic cardiologist interpretation of the echocardiogram.
Materials and Methods
The study was performed at the Department of Emergency Medicine, Ege University Faculty of Medicine. The study was undertaken during June 2011 to December 2011. All patients were informed before the study, and written informed consent was obtained.
All patients admitted to the ED with chest pain were enrolled in this study. Patients younger than 18 years, patients with a history of previous CAD, patients with a coronary artery bypass graft/stent, patients with ST segment elevation on ECG (MI diagnosis) on admission, and patients with a low ejection fraction who needed resuscitation on admission were excluded from the study.
An ACS diagnosis was made according to the World Health Organization guidelines. Two of the following three criteria were needed for diagnosis 6 :
Ischemic type of chest pain and/or chest discomfort
Changes in serial ECGs
Characteristic elevation in serum cardiac markers
Demographic data, complaints, medical history, and physical examination findings of the patients were recorded in the case report form. A 12-lead ECG was completed on all patients. The blood samples were taken for a full blood count, kidney function tests, creatine kinase, creatine kinase muscle-brain (CK-MB), and troponin I.
A focused echocardiography of each of the patients was first performed by the emergency medicine (EM) physician, after the routine procedures were completed, interpreted, and recorded in the patient’s report form.
Echocardiography
The echocardiogram was performed in the ED. The EM physician and his assistant had a seniority of more than 2 years. The physician previously completed a certified basic emergency ultrasonography course and targeted emergency echocardiography training.
Echocardiographic evaluations were made with a portable ultrasound equipment system (Sonosite MicroMaxx; FUJIFILM Sonosite, Inc., Bothell, WA, USA). The ultrasound equipment system has a broadband transducer and a transmit frequency of 1 to 5 MHz, as well as a 17-mm aperture size. The ultrasound system also included a phased array transducer which allowed for processing tissue harmonics in abdominal, cardiologic, and transcranial evaluations. During the evaluation of the patients, routine patient care was not interrupted, and supine chest x-ray (SAG), serial ECG, and biochemistry samples were taken as indicated by the patient’s primary physician.
During echocardiography, a left ventricular regional wall motion defect was investigated. Focused echocardiograms of patients for a left ventricular wall motion defect were scored as suggested by Feigenbaum et al. 7 The patients were evaluated in the left decubitus position. Measurements and wall motion defects were evaluated using the parasternal long-axis (PSLA) view, the parasternal short-axis (PSSA) view, the apical four-chamber view, and the apical two-chamber echocardiographic view (see Figure 1). The quantitative wall motion score was evaluated in all patients. Patients with insufficient echocardiographic images were excluded from the study.
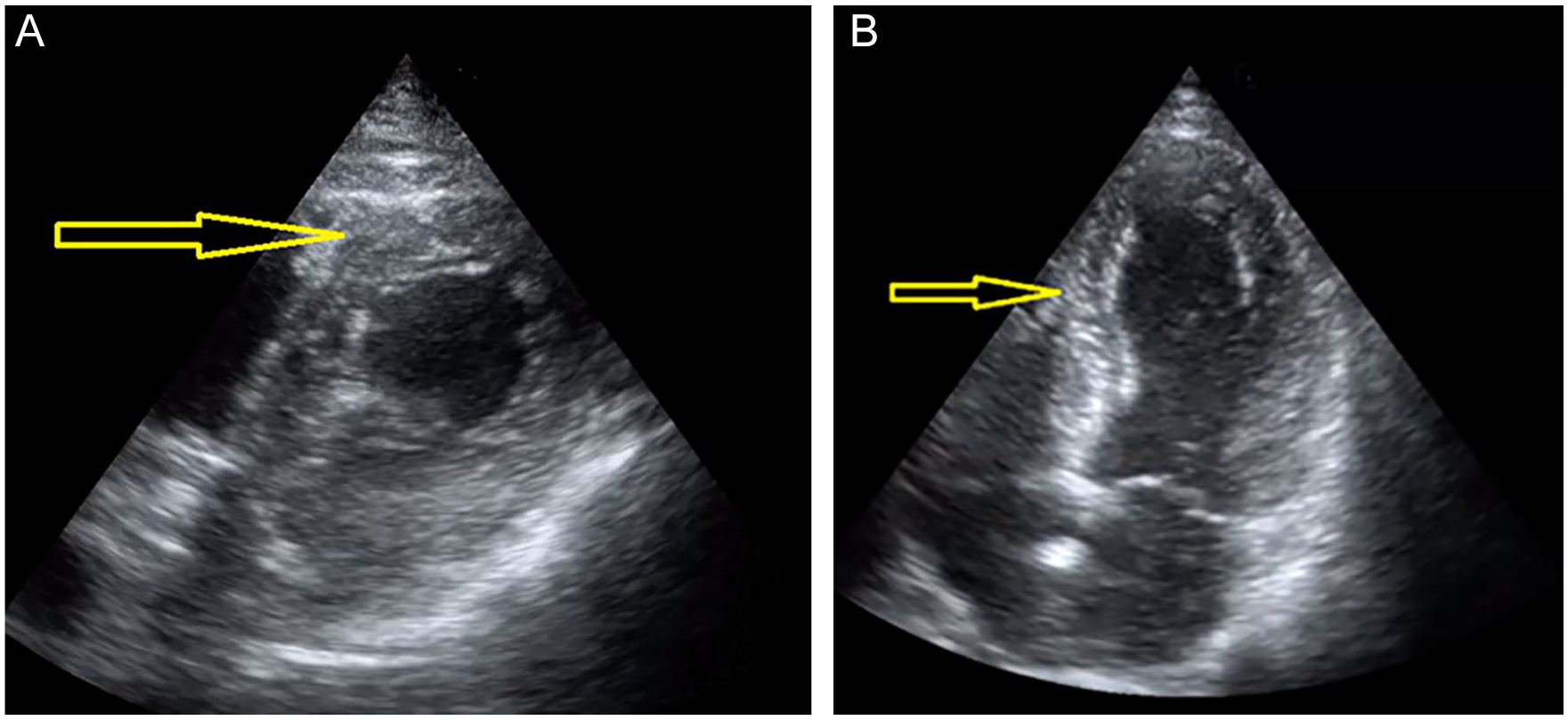
(A) A parasternal short-axis (PSSA) papillary-muscle-level, anterior hypokinesia identified by the yellow arrow. (B) An apical four-chamber window, hypokinetic septum identified by the yellow arrow.
Quantitative wall movements were evaluated over 17 segments, as recommended by the American Society of Echocardiography (ASE). A sonographic assessment was obtained from the PSLA, mitral valve, papillary muscle, apical four-chamber, and apical two-chamber images. In the quantitative evaluation, a scoring system was created using the following point system: normal = 1, hypokinesia = 2, akinesia = 3, dyskinesia = 4, aneurysm = 5. The total value from these 17 segments was divided by the number of segments. A segmental wall movement abnormality (SWMA) score was calculated. A value of one was considered as normal, and if the value exceeded one, it was regarded as significant for ACS. And the obtained regional wall motion defect score was evaluated in terms of ACS.
Focused echocardiography images taken by the EM physician were simultaneously recorded on the Sonosite MicroMaxx. A focused echocardiography evaluation and comments by the EM physician’s assistant, regarding wall motion defects, were recorded on the patient’s report form. The sonographic images were then transferred to a computer. The downloaded images were reviewed by a cardiologist, which was the institution’s accepted clinical practice, and the cardiologist’s review was captured in a separate patient report form. The cardiologist was shielded to the origin of the patients’ clinic. That is, the cardiologist did not know that the patient was being followed up with a preliminary diagnosis of ACS. The cardiologist just followed up the ASE’s recommendations specific to the wall motion evaluation. During the 6-hour follow-up, the patients’ ECGs were taken four times, and cardiac markers were sent at the sixth hour.
Based on the diagnostic findings, examination sensitivity and specificity for the EM physician conducting the echocardiogram were calculated. The final diagnostic assessment by the cardiologist was regarded as the clinical gold standard. Positive predictive values (PPVs) and negative predictive values (NPVs) were calculated by comparing the focused echocardiographic findings of the EM physician according to the sixth-hour troponin I results. ACS estimation was calculated according to the regional wall motion defect score and the 30-day serious cardiac event results.
Statistical Analysis
The statistical analysis was completed using SPSS for Windows 16.0 (SPSS Inc., Chicago, IL, USA) software program. Descriptive statistics (e.g., standard deviation, mean, frequency, ratio, minimum, and maximum) were used when evaluating the study data. A chi-square analysis test was used to determine the significance of the difference between rates. A Pearson product moment, a chi-square test, and kappa coefficient were used to compare qualitative data. The Fisher’s exact test was used to compare different data. The results were evaluated with a 95% confidence interval. The statistical significance was set at P < .05 and bidirectional. Compliance was graded using the following kappa coefficient values: <0.20 very weak, 0.21–0.40 weak, 0.41–0.60 moderate, 0.61–0.80 strong, and 0.81–1.00 perfect.
Results
In total, 62 patients were enrolled in this study. During the diagnostic workup, 5 out of 62 patients declined to continue with the study protocol. Nine out of the 62 patients were excluded due to a technical failure (i.e., based on a lack of echocardiographic visualization of the heart). All the patients’ diagnostic data were transferred to the cardiologist. In total, 48 patients were evaluated by both the EM physician and cardiologist (see Figure 2). Twenty-four (50%) out of the 48 patients were male, and 24 (50%) patients were female. The mean age of the patients was 49 ± 13 years (age range of 19–83 years).
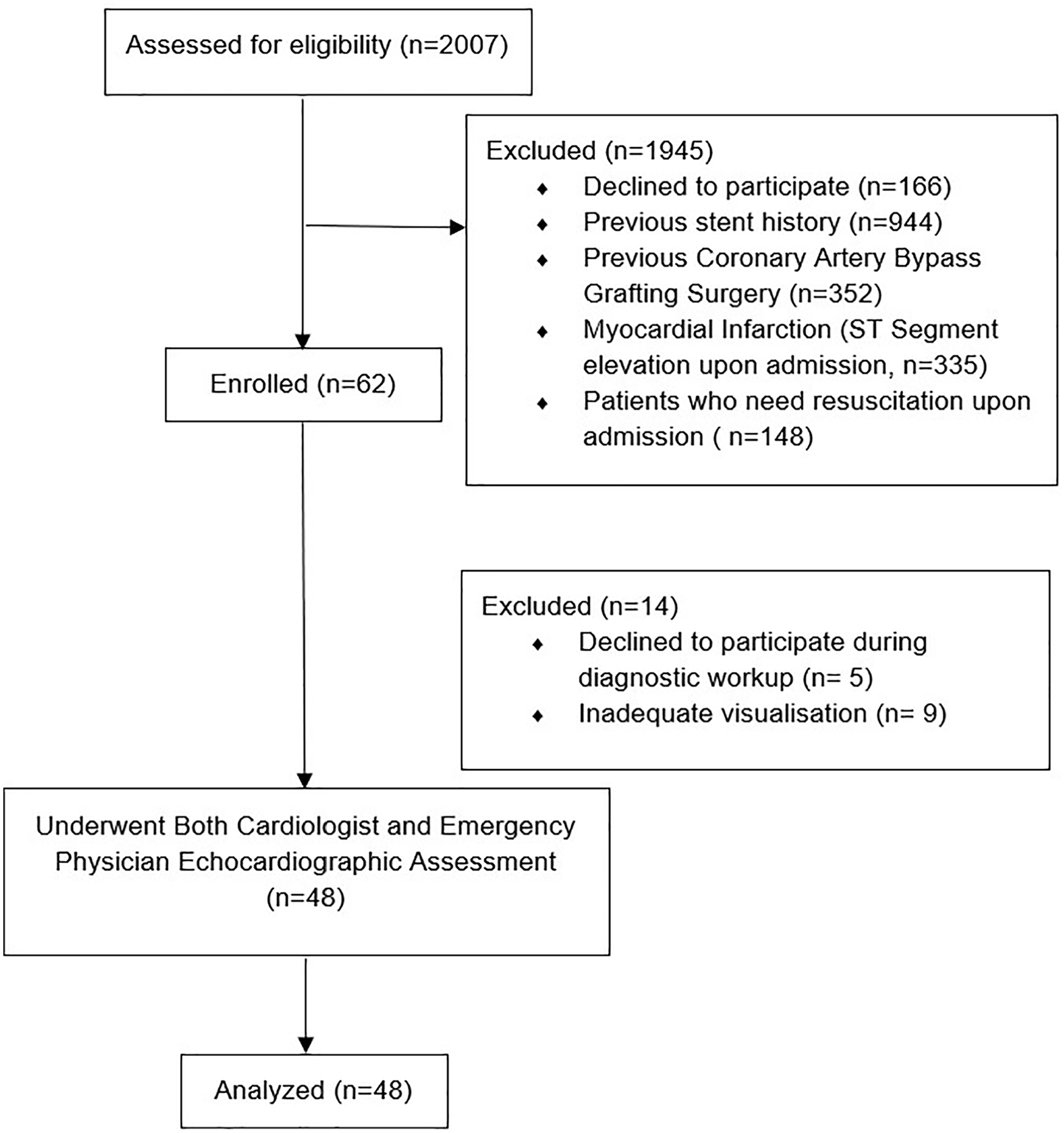
The flow diagram of the patient cohort study.
A perfect correlation (e.g., kappa coefficient range of 0.81–1.00) was observed for the PSSA, basal anterolateral, PSSA mid-anterolateral, and PSSA apex anterior, as well as the PSSA apex inferior segment comparisons (e.g., kappa coefficients of 1.000, 1.000, 1.000, and 0.846, respectively). A strong correlation (e.g., kappa coefficient range of 0.61–0.80) was observed in PSSA basal anterior, PSSA basal inferior, PSSA mid-anterior, PSSA mid-inferolateral, PSSA mid-inferior, PSSA apex lateral, PSSA apex septal, and PSLA apical cap (e.g., kappa coefficients 0.644, 0.633, 0.657, 0.727, 0.777, 0.657, 0.657. and 0.727, respectively) and segment comparisons. A set of moderate correlations (e.g., kappa coefficient range of 0.41–0.60) was observed in PSSA basal inferolateral and PSSA mid-anteroseptal segments (e.g., kappa coefficients 0.455 and 0.478, respectively). A weak correlation (kappa 0.21–0.40) was observed in PSSA basal inferoseptal, PSSA basal anteroseptal, and PSSA mid-inferoseptal segment evaluations (kappa 0.379, 0.231, and 0.379, respectively). No very weak correlations were observed within the data (see Table 1).
Summary of the Sensitivity, Specificity, PPV, and NPV of Emergency Medicine Physician-Driven Echocardiography (Cardiologist-Driven Echocardiography Was Regarded as the Gold Standard).
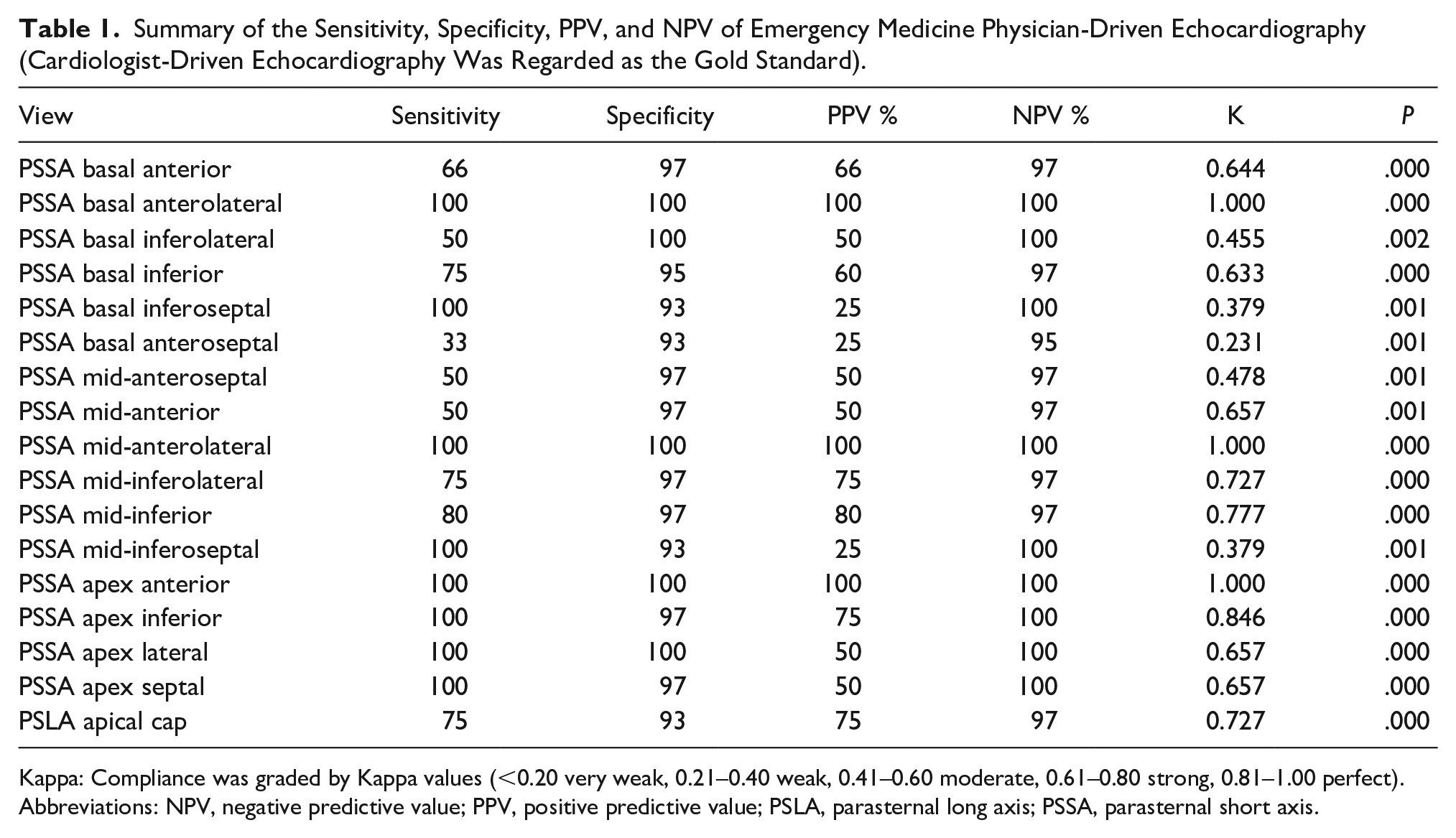
Kappa: Compliance was graded by Kappa values (<0.20 very weak, 0.21–0.40 weak, 0.41–0.60 moderate, 0.61–0.80 strong, 0.81–1.00 perfect).
Abbreviations: NPV, negative predictive value; PPV, positive predictive value; PSLA, parasternal long axis; PSSA, parasternal short axis.
The diagnostic sensitivity, specificity, PPV, and NPV based on the EM physician’s echocardiographic report for PSSA are provided in Table 2. The EM physician’s highest sensitivity was observed for the basal anterolateral and basal inferior views (e.g., sensitivity of 100%). His highest diagnostic specificity was noted for the basal anterolateral view (e.g., specificity of 100%).
Sensitivity, Specificity, PPV, and NPV of Emergency Medicine Physician-Driven Echocardiography (Cardiologist Assessment Was Regarded as the Gold Standard) for PSSA Views.
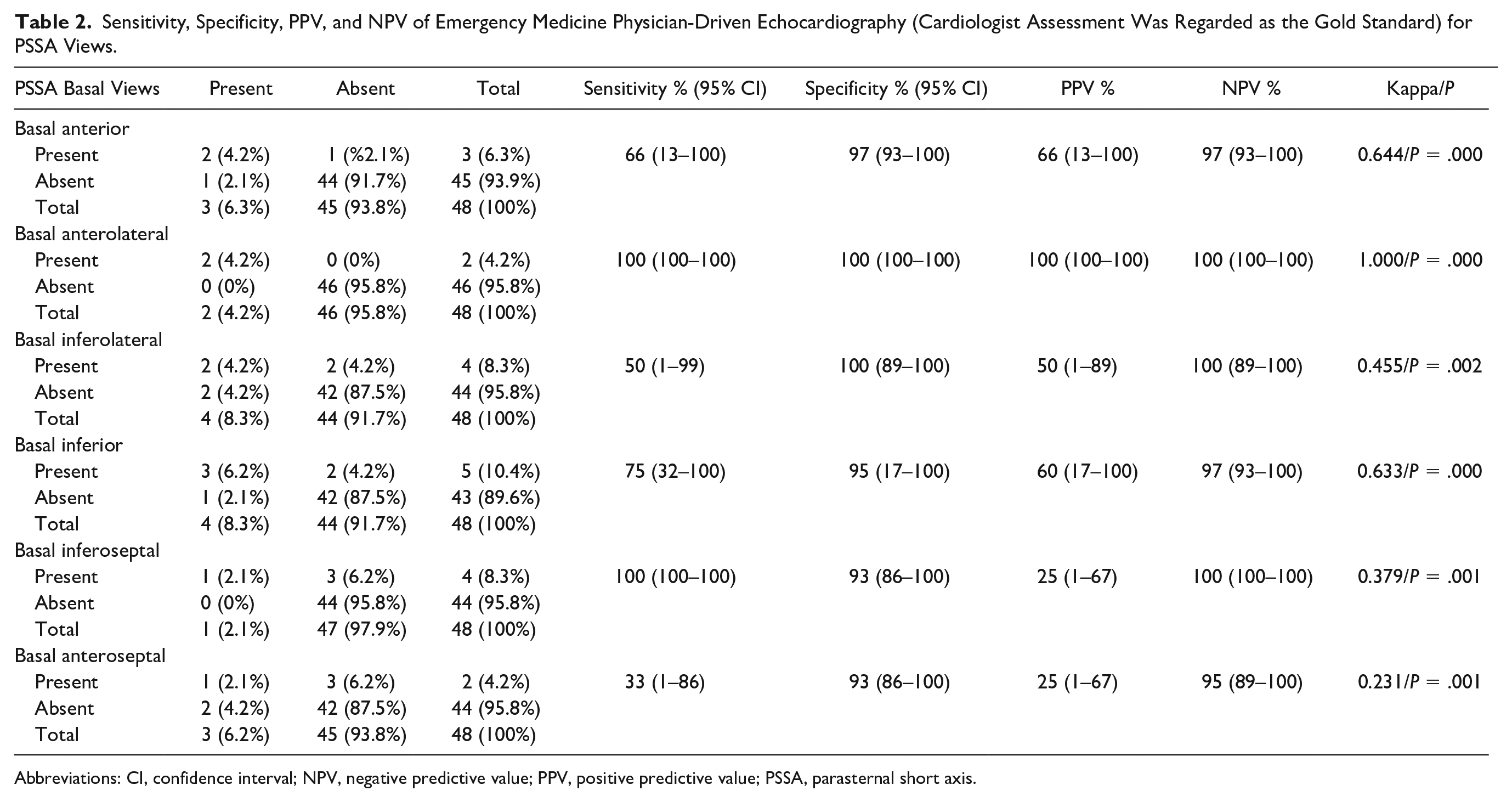
Abbreviations: CI, confidence interval; NPV, negative predictive value; PPV, positive predictive value; PSSA, parasternal short axis.
The diagnostic sensitivity, specificity, PPV, and NPV for the EM physician’s echocardiographic reporting on the PSSA mid-views are provided in Table 3. The highest diagnostic sensitivity was found when evaluating the reporting on the mid-anterolateral and mid-inferoseptal views (e.g., sensitivity of 100%). His highest diagnostic specificity was observed for reporting on the mid-anterolateral view (e.g., specificity of 100%).
Sensitivity, Specificity, PPV, and NPV of Emergency Medicine Physician-Driven Echocardiography (Cardiologist Assessment Was Regarded as the Gold Standard) for PSSA Mid-Views.
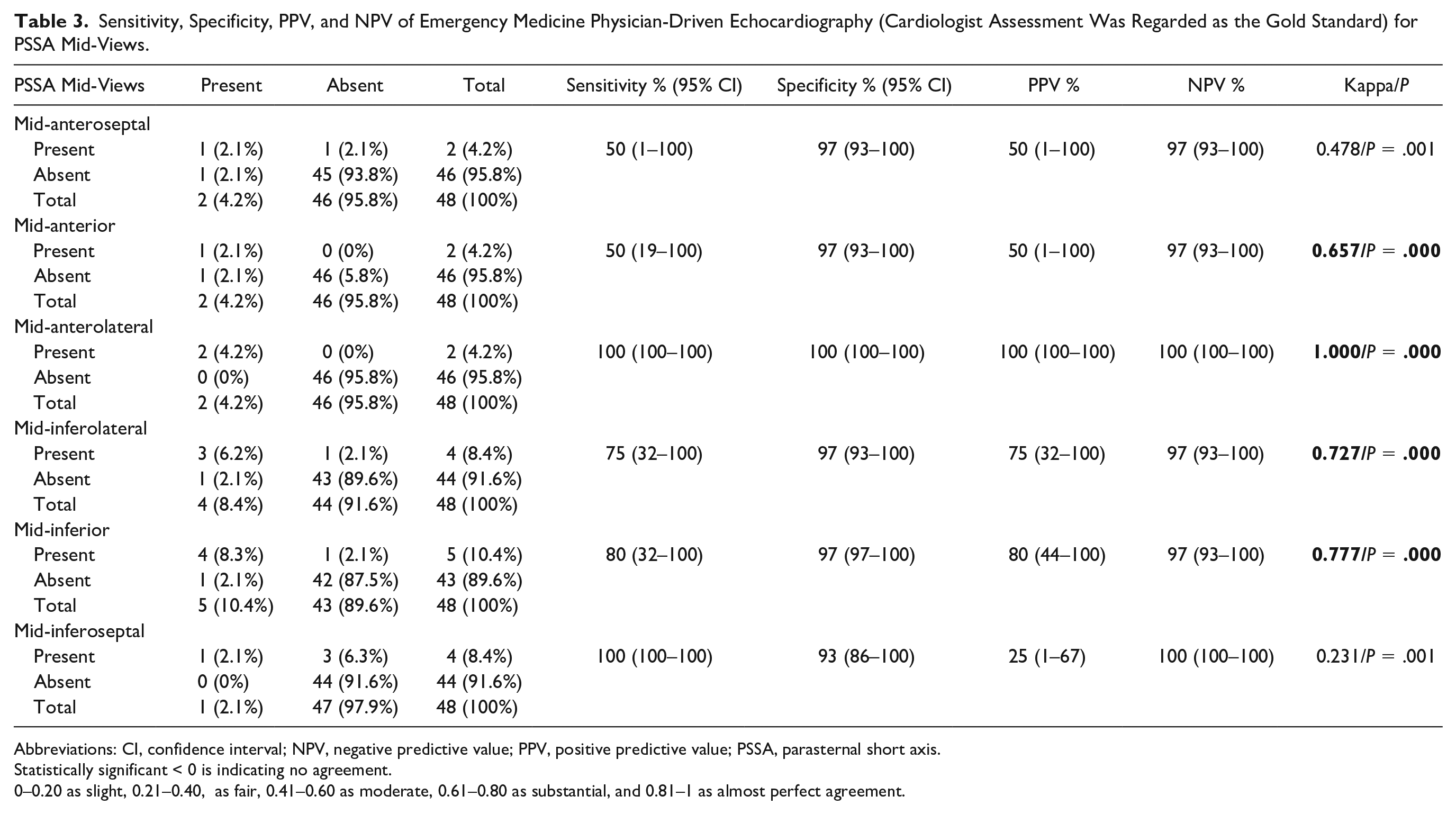
Abbreviations: CI, confidence interval; NPV, negative predictive value; PPV, positive predictive value; PSSA, parasternal short axis.Statistically significant < 0 is indicating no agreement.
0–0.20 as slight, 0.21–0.40, as fair, 0.41–0.60 as moderate, 0.61–0.80 as substantial, and 0.81–1 as almost perfect agreement.
The diagnostic sensitivity, specificity, PPV, and NPV for the reporting of the EM physician completing echocardiography for PSSA apex views and PSLA views are presented in Table 4.The PSSA apex views and all other views had a 100% sensitivity. The EM physician’s highest specificity was noted for the apex anterior views (e.g., specificity of 100%).
Sensitivity, Specificity, PPV, and NPV of Emergency Medicine Physician-Driven Echocardiography (Cardiologist Assessment Was Regarded as the Gold Standard) for PSSA Apex Views and PSLA Views.
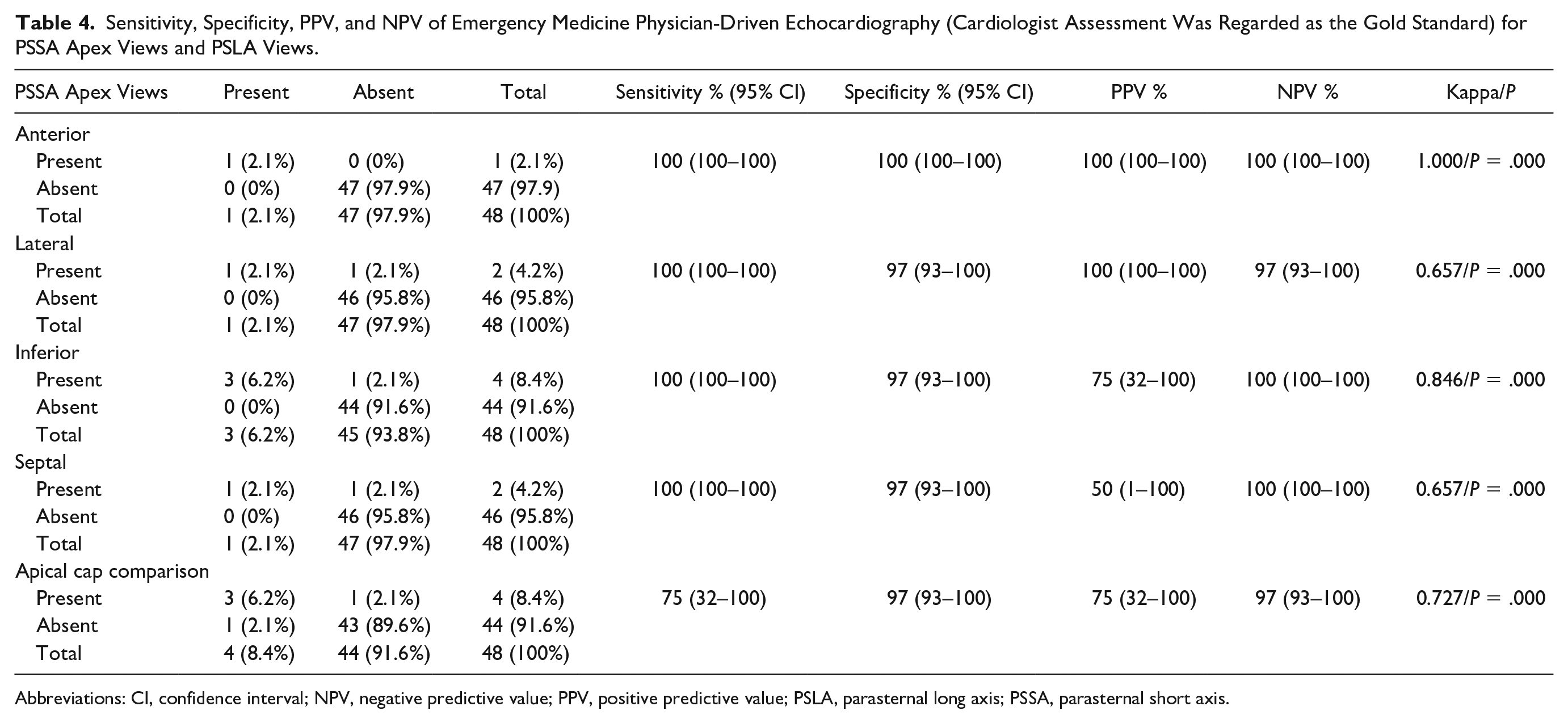
Abbreviations: CI, confidence interval; NPV, negative predictive value; PPV, positive predictive value; PSLA, parasternal long axis; PSSA, parasternal short axis.
The diagnostic value of the EM physician’s echocardiographic reporting had a very weak correlation with the troponin levels (e.g., kappa coefficient = 0.17). The diagnostic value of EM physician’s echocardiographic reporting had a mid-high correlation with the SWMA scoring (e.g., kappa coefficient = 0.86; P = .00). No serious cardiac event developed within the patient cohort’s 30 days. Therefore, the diagnostic value of EM physician’s echocardiographic reporting on predicting serious cardiac effects could be calculated (see Table 5).
Sensitivity, Specificity, PPV, and NPV of Emergency Medicine Physician-Driven Echocardiography (Cardiologist Assessment Was Regarded as the Gold Standard) for Predicting Troponin Levels, SWMA, and 30-Day Serious Cardiac Event.
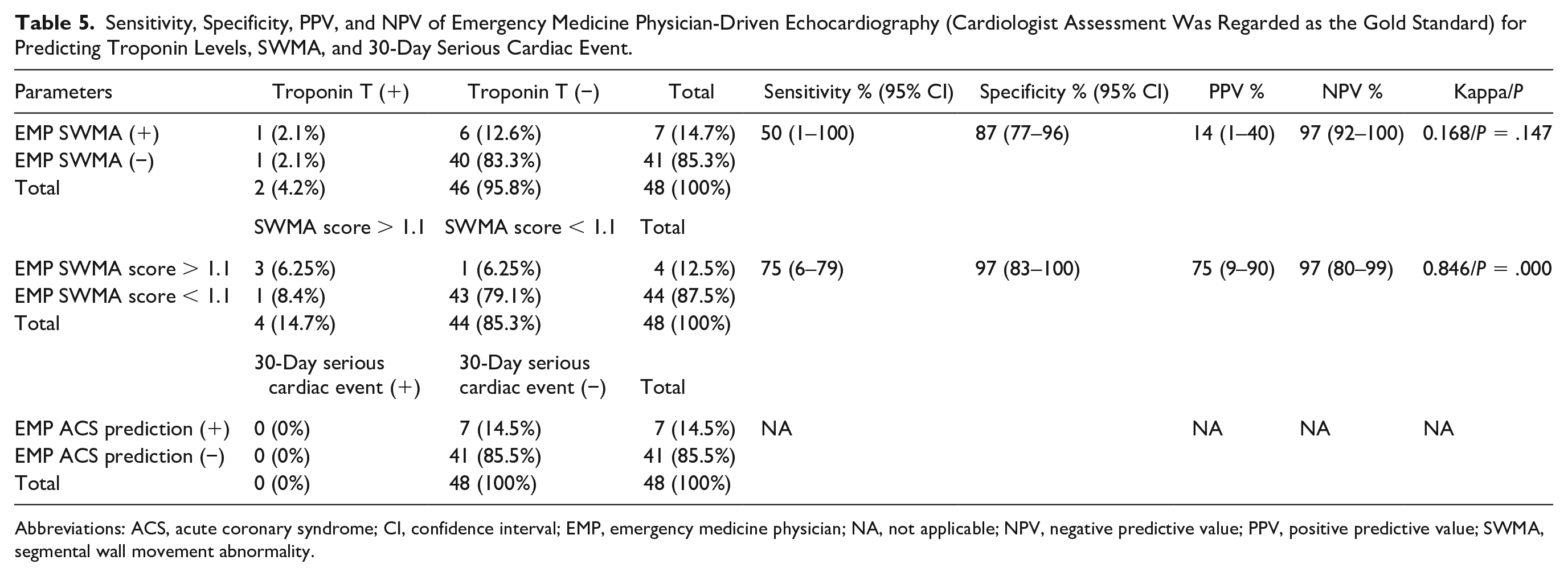
Abbreviations: ACS, acute coronary syndrome; CI, confidence interval; EMP, emergency medicine physician; NA, not applicable; NPV, negative predictive value; PPV, positive predictive value; SWMA, segmental wall movement abnormality.
Discussion
In the present study, the diagnostic value of EM physician’s echocardiographic reporting was assessed and compared with the cardiologist’s final assessment. When the focused echocardiographic results recorded by the EM assistant and cardiologist were compared, a good and very good agreement was found in 14 sections of the myocardial assessment. Unfortunately, a concordance was not found in three sections. A good to significant correlation was found between the EM physician’s reporting and the cardiologist’s assessment, with regard to detecting regional wall motion defects in the PSSA basal section anterior, inferolateral, and inferior segment wall motion evaluation. In the present study, ACS developed in one of the patients in whom ACS was predicted according to the left ventricular SWMA score. A study of 100 patients by Rasooli et al.8 showed an acceptable agreement between EM residents and cardiologists, in detecting regional wall motion defects, with echocardiography.
Chest pain is one of the frequent reasons for admission to the ED. The differential diagnosis of chest pain includes life-threatening conditions that need a rapid and strict evaluation. The ACS is one of the life-threatening conditions in which patients should be followed up for at least 6 hours. If the patient is discovered to have ACS after 6 hours and treatment is started, this could significantly decrease the patient’s morbidity and mortality. In contrast, unnecessary follow-up in the hospital increases the length of hospital stay, which can be a burden for the ED and the health system. 5
The ACS diagnosis was made according to anamnesis, clinical examination, serial ECGs, and cardiac enzymes. 4 Utilizing ECG, as a single conventional diagnostic tool, has a diagnostic accuracy of only 40%. 4 Echocardiography has been shown to be more sensitive in detecting cardiac ischemia than the ECG, which is a more definitive tool for infarction only. The sensitivity of echocardiography for predicting cardiac events within 4 hours of presentation was found to be 91%, compared with 40% for the ECG. 2 Cardiac biomarker tests permit accurate diagnosis and aid in early risk stratification, which is essential for guiding treatment. 2
Based on the “time is muscle theory,” if a patient has ACS and the diagnosis cannot be made by ECG and history, initiation of treatment is guided by the patient’s troponin levels. Initiation of treatment at the time when the troponin levels rise may increase the patient’s morbidity. This may lead to a lifelong low ejection fraction and frequent admission to emergency services with symptoms of heart failure. 9 Evaluation of segmental wall movements and calculation of SWMA with bedside echocardiography may be used to support the diagnosis. If a wall motion is detected, an early lytic therapy can prevent the patient’s myocardium from necrosis. Focused echocardiography may also be helpful to differentiate cardiac and noncardiac conditions. Aortic dissection, pulmonary thromboembolism (with enlarged right-sided structures), and cardiac tamponade can be diagnosed or excluded. 10 Focused echocardiography also may be helpful in diagnosing ACS when a newly formed wall motion defect is detected. In the past years, several studies have been conducted with the diagnosis of ACS by echocardiography.10,11
With focused echocardiography, an examiner needs to evaluate the left ventricular wall thickening (equally or not) especially in PSSA sections. Evaluation needs to be done separately for each segment. A delay in myocardial thickening during systole or a contractile defect in asymmetric narrowing of the endocardial cavity is the sign for ACS. According to Feigenbaum, the SWMA score was obtained by dividing the collected total wall motion defect scores from 17 segments by 17. 7 A normal value is one, which means all the myocardial walls are normokinetic. A score above one will indicate that one or more of the segments has a motion defect. If the score is over one, instead of waiting for the patient’s dynamic changes in the ECG or the increase in cardiac necrosis markers, the patient’s treatment can be started. 12
It is stated that at least 20 hours of training should be given to emergency physicians to properly evaluate critically ill patients with cardiac sonography. This training program may be enough for an EM physician to detect a segmental wall motion defect. 13 Echocardiography performed by cardiologists on patients with chest pain in EDs shows a very high NPV, especially in excluding ACS (e.g., NPV 95.1%). 14 Over time, the ability of EM physicians to diagnose ACS with echocardiography has gradually increased. In this context, Chandra et al. 15 showed that EM physicians performing a bedside cardiac echo had a sensitivity of 100% (95% confidence interval [CI] 40%–100%), a specificity of 77% (95% CI 54%–91%), a PPV of 40% (95% CI 15%–77%), and an NPV of 100% (95% CI 77%–100), for predicting cardiac events. The emergency services clinical practice guidelines, jointly published byAmerican Collage of Emergency Physicians and ASE, recommends 4 hours of theory and 4 hours of practice (based on 75 patients) for focused echocardiography and suggests that EM physicians can make the differential diagnosis for chest pain and start appropriate treatment.7,13
In the present study, it was possible for this EM physician to achieve a very high NPV, in the evaluation of patients, in terms of ACS. As Peels et al. 14 pointed out, this value is consistent with the results in which very high NPVs were reached in the evaluation made with the prediction of ACS, by calculating the regional wall motion score.
Gibler et al. 16 reported the NPV to be between 94% and 98% in echocardiographic evaluations, in terms of ACS in patients with chest pain. They reported a PPV between 31% and 50%. Based on the data of Gibler et al., specificity and PPV were found to be low as well. This situation can be overcome by repeating an echocardiogram, after 9 hours of follow-up for those patient’s with chest pain. 16 This diagnostic scheme was also recommended for evaluating whether ischemia has turned into necrosis. 16 Conversely, Farsi et al. 17 showed that the NPV of the focused echocardiography, performed by EM residents, was high and suggested that it can be used as a means to rule out a pericardial effusion, abnormal heart size, increased pressure in the right ventricle, and cardiac wall motion abnormalities. In that same study, the agreement between a focused echocardiography performed by an EM resident and an echocardiogram performed by a cardiologist was 100%, in detecting wall motion abnormality and pericardial effusion. 17
Cardiac segments in which compliance was not detected were revealed in the evaluation of the inferoseptum and anteroseptum in the PSSA basal section and inferoseptum walls, in the PSSA midsection. Although this may be due to the lack of perception technically, it may also be due to the lack of the EM physician’s experience, compared with the cardiologist. This deficiency can be overcome by the EM physician performing more focused echocardiography. It can be considered that images can be of better technical quality in the apex section. In the evaluation of SWMA, in the PSLA apical section, a good and significant agreement was found between the EM physician and the cardiologist in detecting regional wall motion defect.
In the present study, the diagnostic sensitivity rates were moderate (e.g., sensitivity 75%), while the specificity rates were high (e.g., specificity 97%). NPVs were also high (e.g., NPV 97%), and PPVs were moderate (e.g., PPV 75%). Kontos et al. 18 conducted a study on 260 patients with chest pain. Patients were evaluated with echocardiography for ACS, and SWMA was assessed. In this study, the sensitivity, specificity, PPV, and NPV were 91%, 75%, 44%, and 98%, respectively. They suggested that the low specificity may be due to the incomplete fit of the ischemic wall motion abnormality in patients presenting with chest pain, when they were first admitted. The authors noted that this situation can be overcome by repeating the echocardiogram at either the fourth or sixth hour, after the onset of symptoms. Echocardiography is an operator-dependent diagnostic imaging method. In most of the studies that exist in the literature, the physician performing the echocardiogram was a cardiologist. This may explain the high sensitivity rates in previously reported studies.
ACS developed in two patients within this cohort, whose SWMA was sought by focused echocardiography. In the present study, SWMA was detected in one of the patients with ACS, and the assumption was that this patient had ACS. The reason why this could not be identified in the other patient may be the absence of transmural involvement by that patient. In a similar study, Horowitz et al. 19 investigated the absence of SWMA despite having ACS and showed that SWMA occurs when the transmural thickness of the myocardium is more than 20%.
A weak correlation was found in the comparison of SWMA within the sixth-hour troponin levels. The reason for this may be the early recognition of SWMA. With early intervention, necrosis did not occur, and the troponin levels did not increase because the obstruction was recanalized. The present study results were in agreement with the results of the study of Kontos et al. 18 This situation can be overcome by repeating the focused echocardiography at the fourth or sixth hours as a diagnostic follow-up.
The relatively high specificity and NPV values may suggest that short-term focused echocardiography training allows the EM physician to gain the ability to recognize the normal, but not enough to recognize the pathologic one. In the present study, the number of patients with SWMA was low as focused echocardiography was only applied to patients without a history of CAD.
Limitations
The main limitation of this cohort study was the research design and threats to internal and external validity. In addition, this study had a limited number of patients and a short data-collection period. The status of the coronary arteries of the discharged patients, from the hospital, was not known due to the fact that coronary angiography was not performed for all those in the cohort. The large confidence interval was due to the small number of enrolled patients. A solution to narrowing the CI would be to increase the number of patients.
Conclusion
Bedside echocardiography could be useful for the differential diagnosis of ACS, in terms of aortic dissection, massive pulmonary thromboembolism, cardiac tamponade, and so on. In patients without a documented SWMA, the newly detected SWMA can be regarded as a strong predictor of ACS. The treatment of these patient could be started earlier, based on the focused echocardiogram, in terms of recanalization, and further morbidity and mortality may be avoided. With increased experience, the EM-physician echocardiographic reporting’s specificity and PPV will increase. The EM physician can develop the ability to recognize the absence of a segmental wall motion defect, as well as the cardiologist (e.g., sensitivity 97%, NPV 97%). If an EM physician cannot detect any wall motion abnormality, it lowers the chance for a 30-day serious cardiac event to occur.
Footnotes
Acknowledgements
We would like to thank Ege University emergency department staff for their technical support to our study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval for this study was obtained from the local Ethical Committee of Ege University (certificate no: 11–5.1/18). Clinical and laboratory information was collected during clinical workout.
Informed Consent
Written informed consent was obtained from all subjects before the study.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study0.
Trial Registration
Not applicable.
