Abstract
Objective:
This literature review was designed to highlight the most up-to-date and evidence-based interventions, whenever available, regarding diagnostic imaging modalities and management strategies for patients, with the placenta accreta spectrum.
Materials and Methods:
A comprehensive review of the literature was conducted using database searches in Science Direct, Google Scholar, and PubMed to locate the most up-to-date information, related to this topic.
Results:
The placenta accreta spectrum has an unclear etiology, with cesarean delivery and placenta previa being the most implicated factors.
Conclusion:
There is a growing global consensus that the two main risk factors for placenta accreta are placenta previa and prior cesarean delivery. Up-to-date, grayscale sonography combined with Doppler interrogation is thought to be the best first-line assistance, for the identification of abnormal placental invasion. Cesarean hysterectomy, with placenta left in situ, is the classical surgical treatment.
The worst obstetric nightmare continues to be the placenta accreta spectrum (PAS), formerly known as morbidly adherent placenta (MAP). 1 This is due to the difficulties in diagnosing and treating the condition, as well as the strong correlation between it and maternal, fetal, and neonatal morbidity and mortality. 2 Maternal morbidity associated with PAS is highly significant, with reported overall rates ranging between 24% and 67%.3,4 Maternal mortality associated with PAS patients has been estimated at 7%. 5 There is a growing worldwide consensus that previous cesarean delivery and placenta previa, or low-lying placenta, constitute the major risk factors implicated, in the occurrence of PAS. The magnitude of this risk is directly proportional to the number of cesarean sections.6 –8 The most accepted theory explaining the occurrence of PAS states that the defect occurring at the endometrium-myometrium interface, probably due to endometrial damage, will lead to abnormal decidualization in this particular area of the uterine wall damage. This can lead to a deeper-than-normal invasion of placental anchoring villi with subsequent trophoblastic infiltration; however, this theory fails to explain the rare occurrence of the condition in nulliparous women with no previous uterine scarring or instrumentation. 9 Maternal vascular under perfusion, evidence of amniotic fluid infection/acute inflammatory pathology, and fetal obstructive vascular pathology were the three well-known pathological abnormalities identified by Ernst et al., based on placental histopathology data (fetal vascular necrosis, avascular villi). 10 Also, baseline chronic villitis was found, in which lymphocytes, histiocytes, or plasma cells moved into the anchoring chorionic villi. 10 Antenatal diagnosis of PAS and management of these patients by a multidisciplinary team are linked to a significant reduction in peripartum maternal and fetal complications, such as the need for blood transfusions, hysterectomies, and visceral damage during surgery, and, as a result, fewer maternal deaths.2,11 Accurate and properly timed antenatal diagnosis of PAS is crucial to improve the maternal and fetal outcomes regarding mortality as well as morbidity. Hence, it is important for sonographers to be well informed with the up-to-date progress in the role of diagnostic imaging techniques, especially sonography, as the first diagnostic choice involved in the management of patients with PAS.
Chaudhari et al., in a prospective study, conducted in India between January 2012 and November 2014, assessed the risk factors, evaluated different management strategies, and quantified the magnitude of adverse maternal, fetal, and neonatal outcomes, in terms of morbidity as well as mortality. 12 Hence, it is crucial for health care practitioners to be aware of the challenges associated with PAS in view of early diagnosis and proper management of patients with pregnancies complicated with this condition (see Figures 1 and 2).
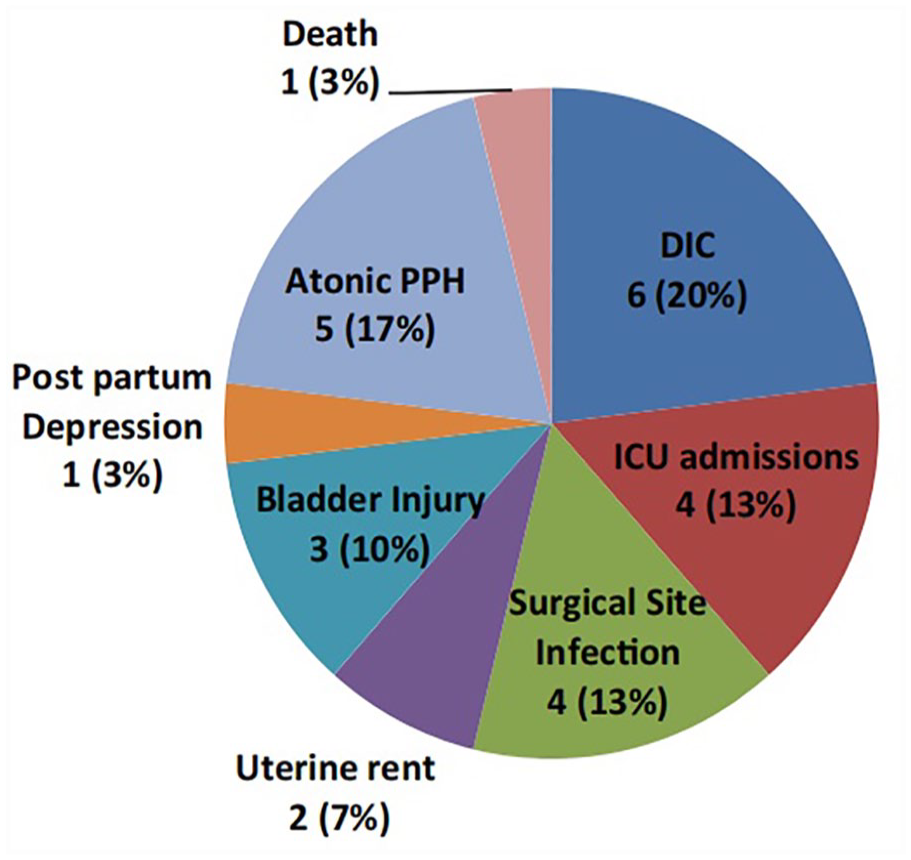
The magnitude of adverse outcomes associated with management of placenta accreta spectrum, for 30 patients enrolled in the Chaudhari et al. study. PPH: Post Partum Haemorrhage; DIC: Disseminated Intravascular Coagulopathy. 12
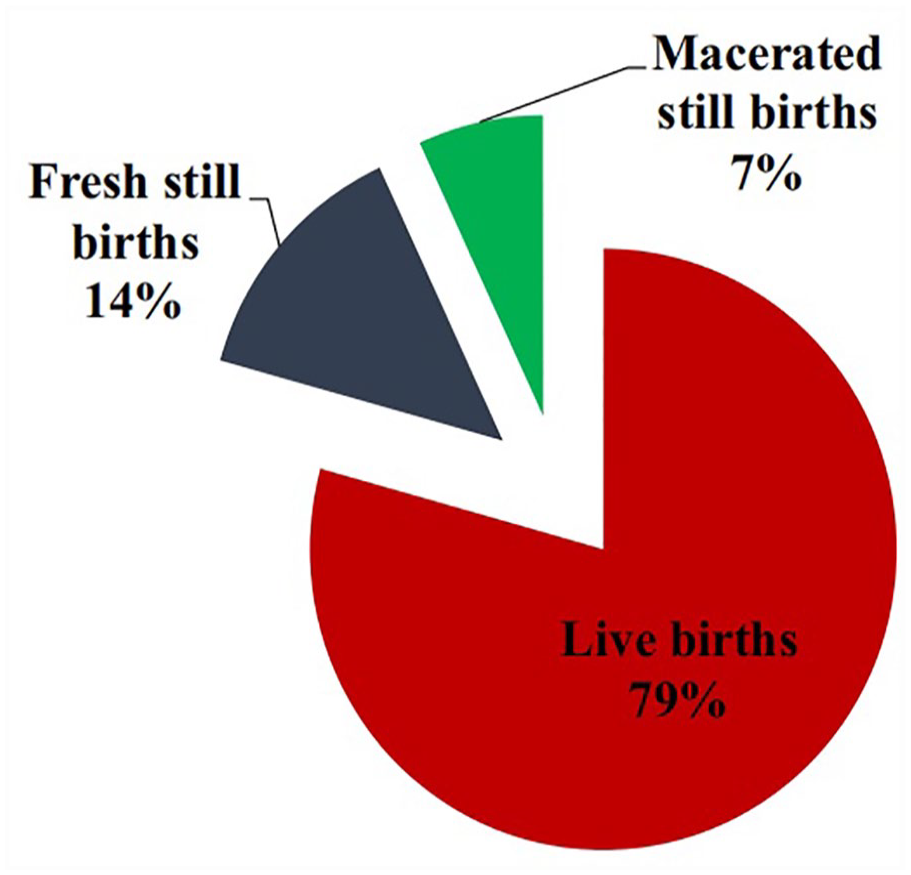
Further outcomes from the 30 patients, with placenta accreta spectrum, who were enrolled in the Chaudhari et al. study. 12
Materials and Methods
A comprehensive review of the literature was conducted using database searches in Science Direct, Google Scholar, and PubMed, to find up-to-date information related to this topic. The search strategy was developed, according to the PRISMA flow chart, and using the keywords placenta accreta spectrum, cesarean hysterectomy, maternal mortality, and cesarean hysterectomy. The titles and abstracts of the studies were retrieved and screened independently by two review authors (M.M. and F.H.). Duplicate articles were removed. The full texts of the potentially eligible studies were independently assessed for eligibility by the two authors and disagreements between the two parties were resolved through a third reviewer (Dg.M.). All authors shared in articles gathered and were involved in data revision and preliminary screening. Finally, a total of 382 papers were recruited; 77 of these were deemed suitable with an appropriate degree of rigor for inclusion in the literature review (see Figure 3). The inclusion criteria included articles addressing the topic, whether original or review articles, there were no constraints on publication dates; however, only research written in the English language was taken into consideration, while the exclusion criteria included articles not meeting keywords; articles written in non-English languages, for example, French or Spain; studies without an abstract available; and studies with a nonpertinent topic.
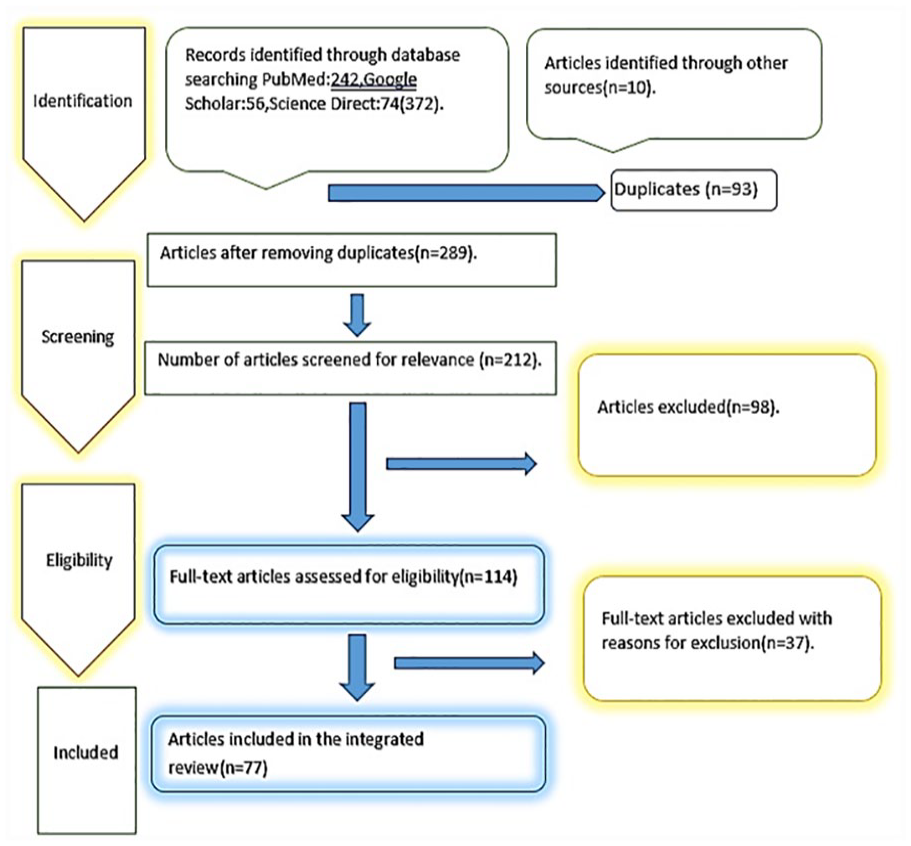
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow chart used to illustrate the process for retrieval, review, exclusion, and inclusion of published articles for this literature review.
Epidemiology
The reported incidence of PAS has shown a steady increase in the last century as well as during the first decades of the current one, which is directly proportionate to the rising rate of cesarean deliveries. This rise was in the range of 13-fold since the first time, the condition was described in the early 1900s. The most recent estimates quoted for the incidence of PAS were demonstrated in large cohort studies, such as 1 in 333-1 in 533 deliveries, rising from approximately 1 in 4000 deliveries in the 1970s to 1 in 2500 in the 1980s and 1990s.7,11,13 The incidence showed great variation from country to country according to the prevalence of cesarean section deliveries. In New Zealand and Australia, the recorded rate of PAS is in the range of 1:23 000 cases. 14 Maternal morbidity associated with PAS is attributed mainly to intractable hemorrhage with superimposed coagulopathy requiring massive blood as well as blood products (Disseminated Intra Vascular Coagulopathy [DIVC] regimens), including fresh frozen plasma, fibrinogen, and platelets. Pelvic organ damage such as urinary bladder, ureters, bowel, fulminant infections, and thromboembolic diseases are all possible peripartum complications. Postpartum hemorrhage, either atonic or coagulopathic, was one of the leading major causes of associated maternal morbidity and mortality.
Etiology and Risk Factors
A multi-centered observational study on maternal morbidity associated with multiple cesarean sections found a prevalence of placenta accreta (or PAS) in 0.24%, 0.31%, 0.57%, 2.13%, 2.33%, and 6.74% after the first, second, third, fourth, and, fifth, or more repeat cesarean deliveries, respectively. 7 Analysis of the sub-group of those women who had also superimposed placenta previa revealed that increased risks for developing MAP were 3%, 11%, 40%, 61%, and 67%, respectively. 7 This association between placenta previa and PAS especially in the presence of two prior cesarean sections was confirmed in a large maternal-fetal medicine network study published in 2006. In this study, a history of cesarean delivery, in a particular patient, would be associated with a significantly increased risk of developing PAS, particularly in the presence of placenta previa or low-lying placenta. An example of this strong association is for a patient with prior three cesarean sections, the presence of a placenta previa would put the patient at a 40% risk of acquiring accreta; however, the magnitude of that risk will be decreased to less than 1% in the absence of placenta previa. 15 Of note, cesarean sections with classical, T, or J uterine incisions were associated with a higher incidence of accreta (10.1%), compared with lower segment incisions. 16 The spectrum of the assessed gynecological procedures has included laparoscopies with uterine manipulation, operative hysteroscopies, surgical evacuation of the uterus by curettage, including suction curettage, endometrial ablation, and uterine irradiation. 8 This association proved to be dose-dependent with an increased risk to develop PAS in women subjected to prior three or more gynecological procedures.
Advanced maternal age is an independent risk factor for PAS; it was found that many of the women diagnosed with MAP were older than 35 years 16 (36.7%), and for every year increase in age beyond 20, there will be a considerable rise in the risk of PAS, especially with the further advancing of maternal age. 7 Esh-Broder et al. observed a 13-fold higher risk of PAS associated with in vitro fertilization (IVF) (0.16%) compared with women who conceived spontaneously (0.012).7,17 Kaser et al. demonstrated that the cryopreserved embryo technique has an independent strong risk factor for placenta accreta among women undergoing IVF (adjusted odds ratio [OR] = 3.20, 95% CI: 1.14–9.02). 17 Other factors associated with increased risk of PAS may include multiparity, smoking, and narrow pregnancy spacing, following cesarean delivery. 7 Most women diagnosed antenatally with PAS (95%) will have identifiable risk factors 16 ; however, many women did not have classical risk factors, with 18.4% of PAS patients having none, others (37.3%) with no history of previous cesarean delivery. 18
Histopathology
In a case-control study, Peng et al. discovered that PAS cases treated by hysterectomy were linked to a substantially observed placental invasive area >1/2 19 (see Figure 4). In their retrospective analysis of the baseline characteristics of PAS, El Gelany et al. discovered that the histopathological analysis of the removed, morbidly adherent portion of the placenta revealed deep chorionic villi invasion and the presence of myometrial fibers, thereby confirming the diagnosis of PAS. 20 Ernst et al. studied the pathological stages of myometrial fiber adherence at the basal plate in haematoxylin and eosin–stained slides from the placentae and hysterectomy specimens, of patients with PAS. 10 They defined the stages as follows: Stage 0: no evidence of myometrial fibers, attached to the basal plate; Stage 1: evidence of myometrial fibers, attached to the basal plate; Stage 2: basal plate with adhering myometrial fibers and two layers of decidual cells separating the myometrium from the anchoring villi and/or Rohr’s fibrin; Stage 3: the presence of alterations that are compatible with focal accreta-myometrium immediately on the anchoring villi and/or Rohr’s fibrin without intervening decidual cells; Stage 4: placenta increta with invasion into the myometrium 4A: 25%, 4B: 25%–50%, 4C: 50%–75%, and 4D: 75%–100%; Stage 5: placenta percreta but no involvement of surrounding organs; Stage 6: identification of neighboring organ invasion and adhesion 10 (see Figure 4).
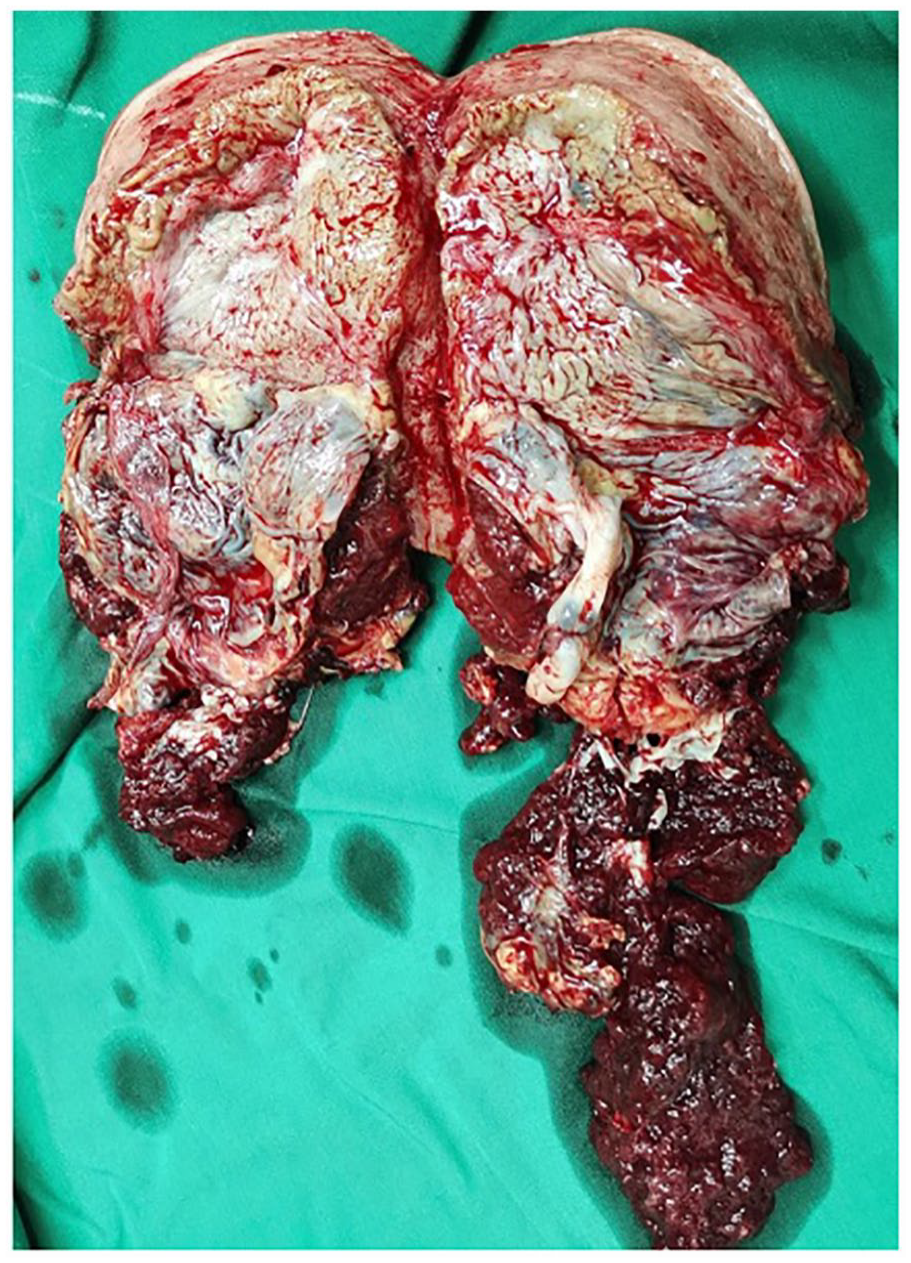
This specimen demonstrates the macroscopic appearance of a postoperative specimen from a uterus with a placenta previa and associated accreta. The patient delivered at 35 weeks and 2 days gestation and the placenta shows a large area of invasion into the uterine wall. It is also important to note that the invasion involves mainly the posterior uterine wall, which is thinner. 19
Pathophysiology
Korkmazer et al., in a retrospective case-control study, investigated the relationship, if any, between morbid placental adherence and the vascular cell adhesion molecule-1 (VCAM-1), which is a calcium-independent transmembrane glycoprotein, shown in several studies to increase invasion in tumor cells, and whether it may play a similar role in increasing the placental invasive capacity in PAS. The study showed a statistically significant increase in the expression of VCAM-1 in association with PAS, a result taken as a step for clarifying the up-to-date unknown pathogenesis of PAS. 21
A retrospective study from China investigated the role of zinc finger E-box-binding homeobox 1 (Zeb1), which acts as a promoter of the transformation of the epithelial cell mesenchyme. It is thought to have an important role in the pathogenesis of MAP, through examination of the protein expression levels of Zeb1 in placental tissues of patients with placenta accreta, others had placenta previa but no accreta, then, a third group of patients having placentas with normal implantation as well as invasion. The study observed a significant increase in Zeb1 expression levels in patients with placenta accreta compared with its levels in patients with normal placentas, which may suggest a possible role for Zeb1 in the pathogenesis of abnormal placental invasion. 22
Diagnostic Imaging Modalities
Up to date, grayscale sonography, supplemented by Doppler interrogation, is considered the recommended first-line aid for the diagnosis of abnormal placental invasion with overall sensitivity and specificity of 82.4% to 100% and 71% to 100%, respectively. The sonographic features enhancing the clinical diagnosis of the condition rest mainly on the absence of a clear interface between the posterior aspect of the placenta (uterine surface) and the myometrium and the presence of irregular interplacental lacunae, which can be detected, sonographically, as “swiss cheese” appearance.23,24 Of note, there is a little bit of difference, regarding the guidelines of sonographic features among different countries; the United States’s cardinal sonographic features for diagnosis of PAS include the detection of placental lacunae, the loss of the demarcation line between the placental and uterine wall as well as the coexistence of a placenta previa. 25 Magnetic resonance imaging (MRI) is only preserved for cases where sonographic findings are inconclusive, so, considered a second-line diagnostic modality. MRI diagnostic criteria of PAS include abnormal vascularity, placental bulge, placental heterogeneous intensity, dark bands within the placental tissue, focal villous interruption within the myometrium, and fibrin bands which help to assess the extent and depth of placental invasion and pelvic organ invasion by placental tissue.7,20 Nonetheless, the current concept in imaging techniques for diagnosis of PAS approved that MRI has neither higher sensitivity nor specificity and subsequently not superior to grayscale sonography in this text. 23
The placenta accreta index (PAI) was created to standardize PAS imaging characteristics and the patient’s previous cesarean delivery history. The index incorporates the number of prior cesarean sections, the position of the placenta as determined by the sonogram, the shape of the placenta (grading lacunae), the thinnest sagittal myometrium, and the presence or absence of bridging vessels. The index is calculated by adding the scores for the variables mentioned above, as shown in Table 1, and placental lacunae are graded according to the Feinberg and Williams classification, where grade 0 denotes the absence of lacunae, grade 1 represents small 1 to 3 lacunae, grade 2 represents large 4 to 6 lacunae, and grade 3 denotes numerous, large, and bizarre shaped lacunae.27,28 However, there were no objective criteria for either small or large lacunae; also, color Doppler was used for assessment of the flow in placental lacunae and detection of the bridging vessels. PAI values were interpreted because of two categories with values <5 as unfavorable for abnormal placental invasion while values ≥5 are favorable for invasion 29 (see Table 1 and Figures 5–7).
Shows the scoring system and value given to each of the PAI variables.following the recommendations of Rac et al. 26
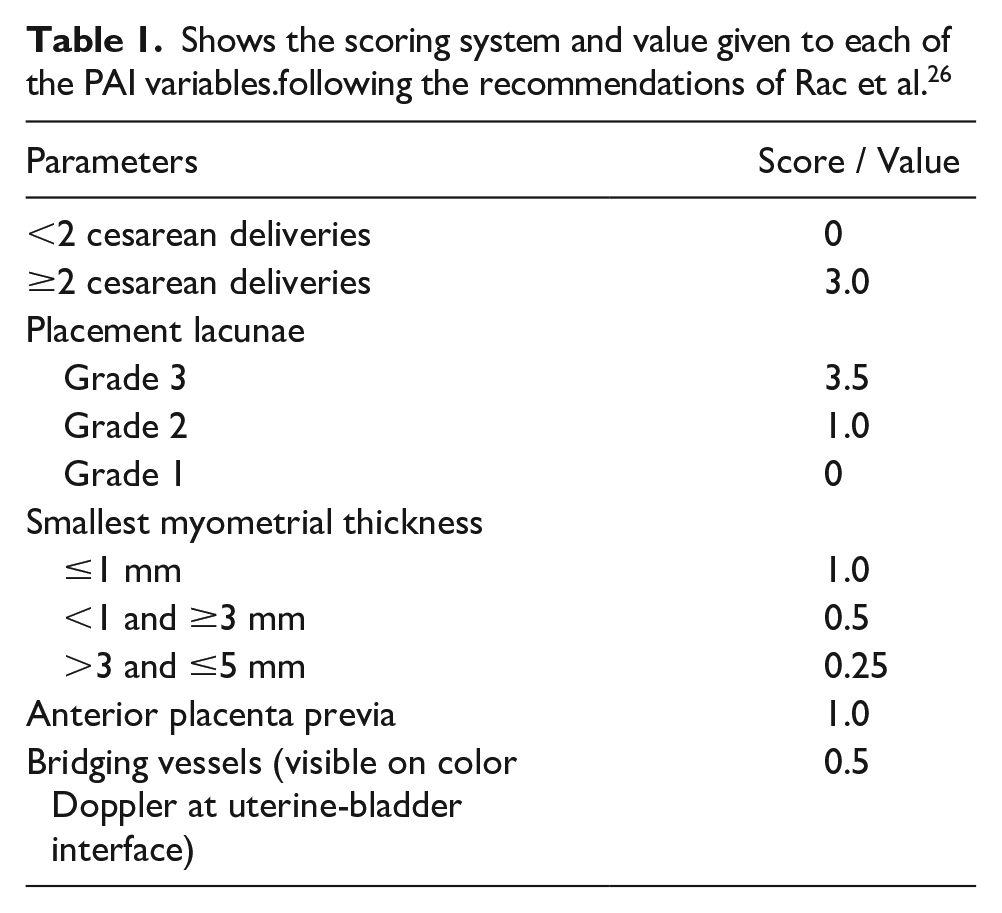
Sonographic images are provided that demonstrate the use of the Finberg-Williams classification of placental lacunae (marked with yellow arrows). (A) The sonogram is assigned a grade 0 with not lacunae. (B) The sonogram is assigned a grade 1, with 1 to 3 small lacunar spaces. (C) The sonogram is assigned a grade 2, with 4 to 6 large lacunae. (D) The sonogram is assigned a grade 3, with multiple (more than six), bizarre, shaped lacunar spaces. 29
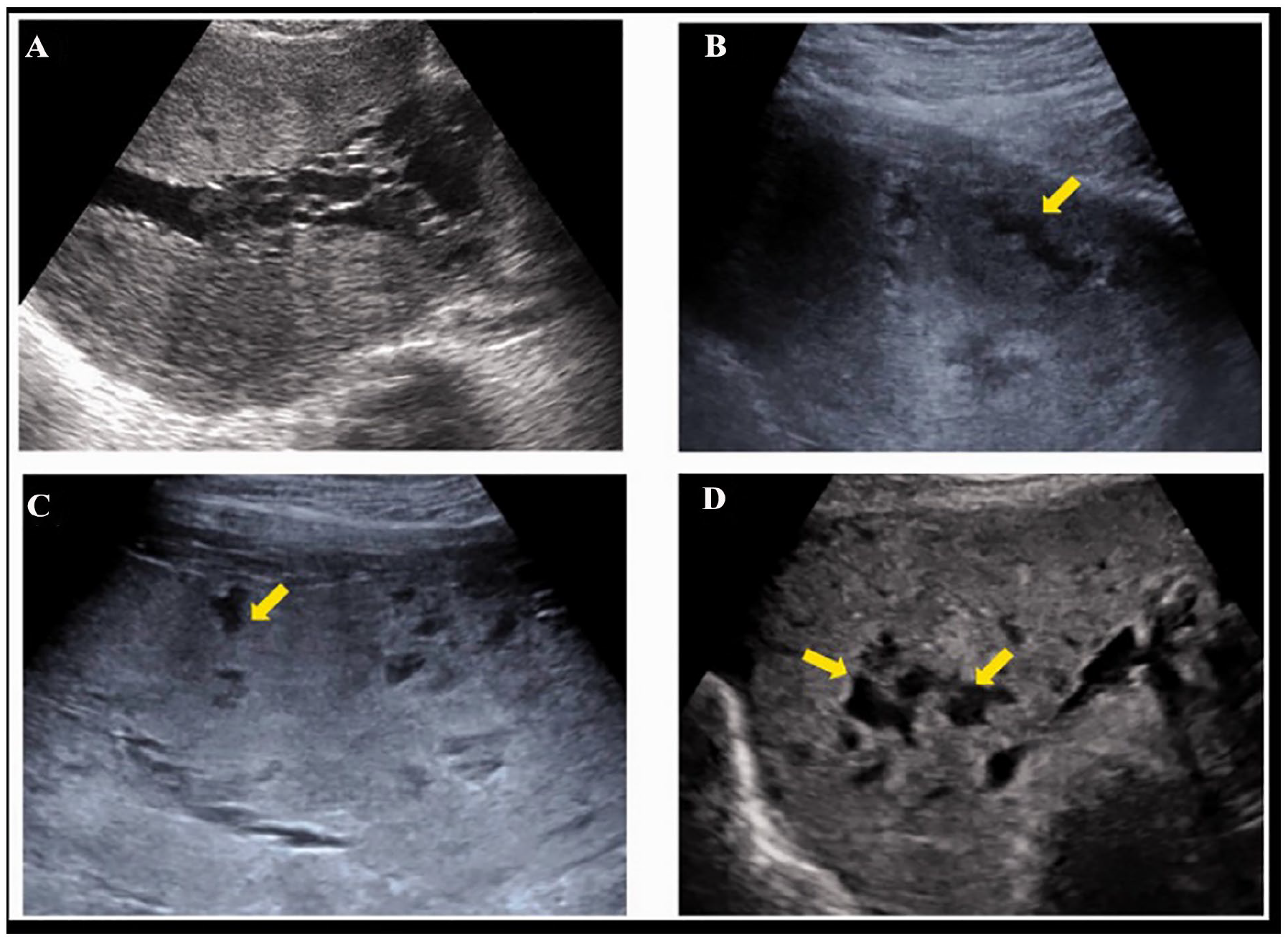
(A) A sonographic image of a placenta percreta case (PAI score > 5) showing total placenta previa, significant myometrial thinning (see yellow arrow), loss of the retroplacental zone, and grade 3 placental lacunae (multiple and throughout the placental substance). The loss of the interface with the urinary bladder is shown by the numerous bridging vessels (small white arrows). (B) A gross specimen of the uterus (fundus indicated by a black arrow) showing total invasion of the anterior lower uterine wall by placental tissue (white solid arrow), confirming placenta accreta. 29 PAI, placenta accreta index.
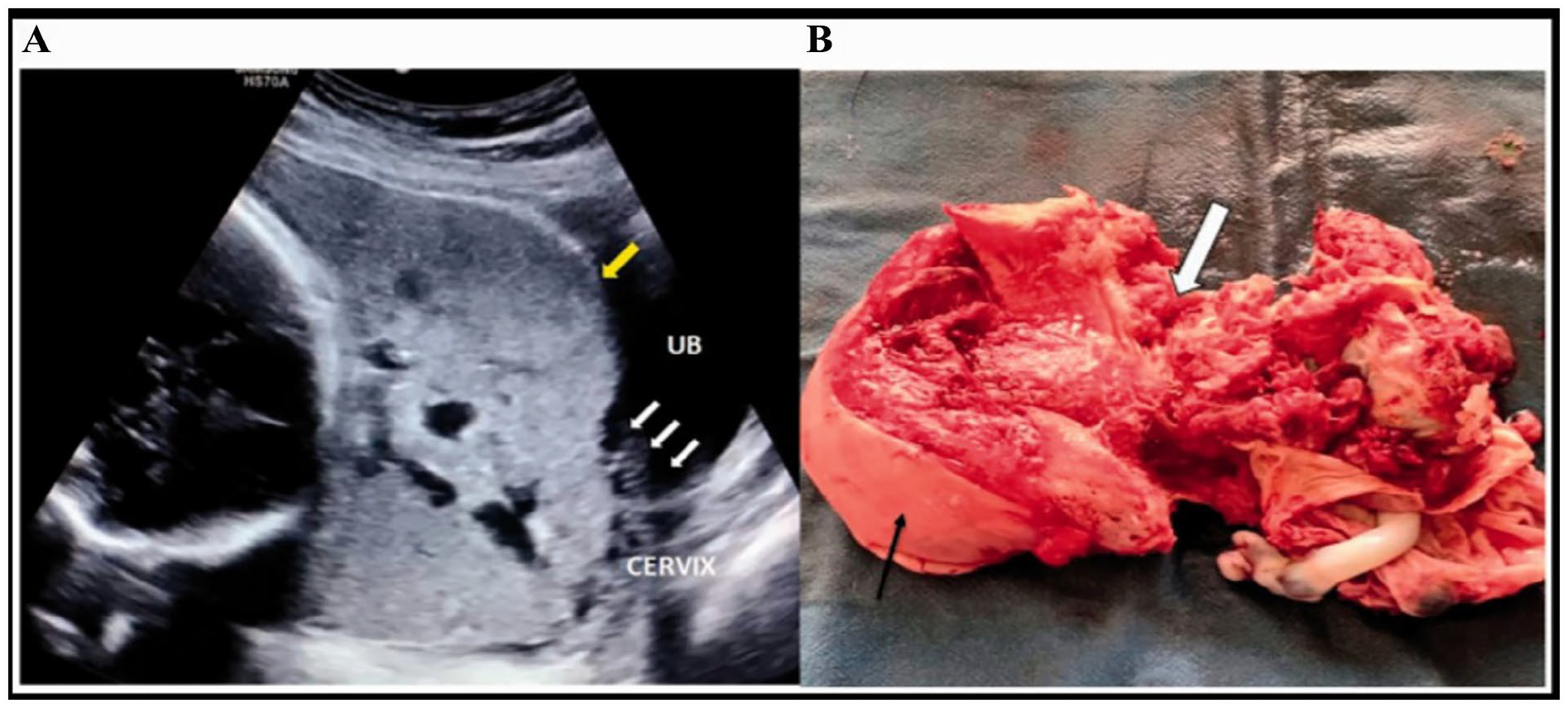
(A) A sonographic image of patient case that demonstrates an anterior marginal placenta previa with preserved utero-placental interface and no placental lacunae. There was no uniform myometrial thickness (see yellow arrow). (B) Color Doppler was applied and shows retroplacental vascular flow and a few bridging vessels. This is assigned a PAI score <5. 29 PAI, placenta accreta index.
Agarwal et al. evaluated the value as well as the reliability of PAI as a diagnostic aid for the existence of placental invasion and, if any, the magnitude of this invasion. The researchers found that the PAI can be employed as an easy, reliable, and cost-effective, sonographic-based, diagnostic modality for assessing the presence as well as the severity of placental invasion in PAS 29 (see Figures 8–13).
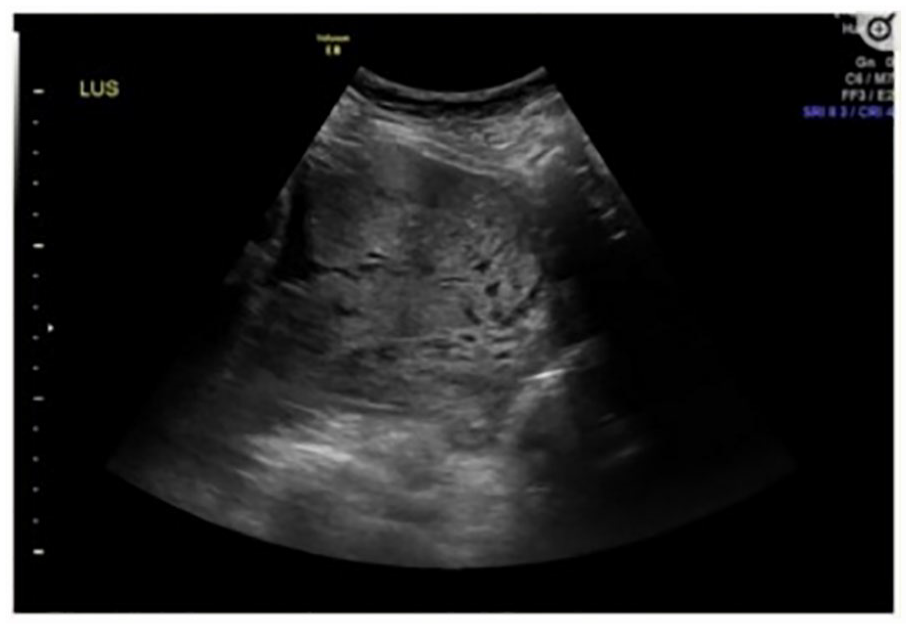
A transabdominal sonogram, which demonstrates a placenta accreta. It also is evident that there are irregular placental lacunae resulting in the characteristic “swiss-cheese” appearance and loss of the normal hypoechoic retroplacental zone. 30
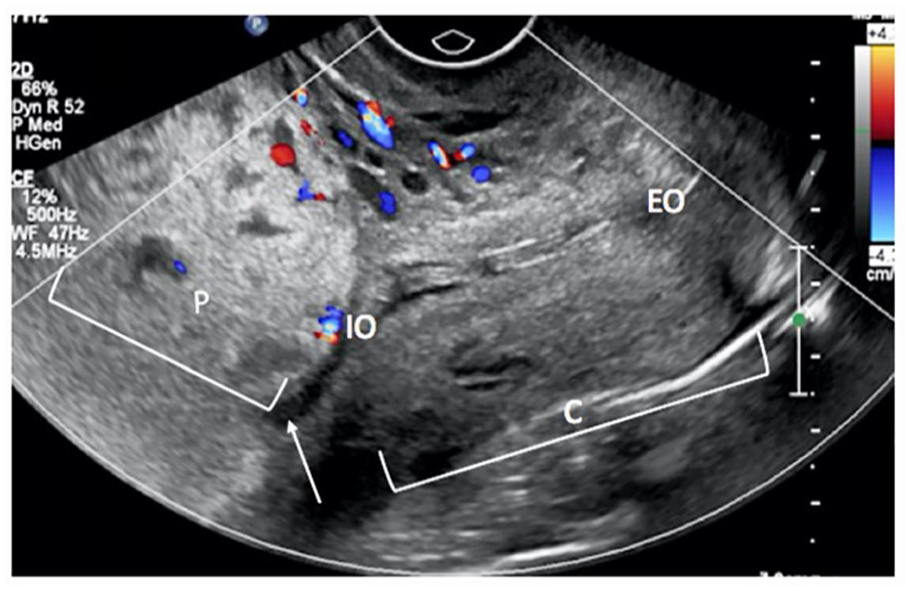
A sagittal view from a transvaginal sonogram conducted on a 32-year-old woman (29 weeks 3 days gestation) and demonstrates a complete placenta previa: the anterior placenta (marked by the letter P) extends from the lower uterine segment with the placental edge (see white arrow) covering the entirety of the cervix (denoted by the letter C) including the cervical os (marked by the letters IO). Color Doppler shows normal vascularity and no evidence of vasa previa. Retroplacental clear space is present and normal. The letters EO denote the external cervical os. 31
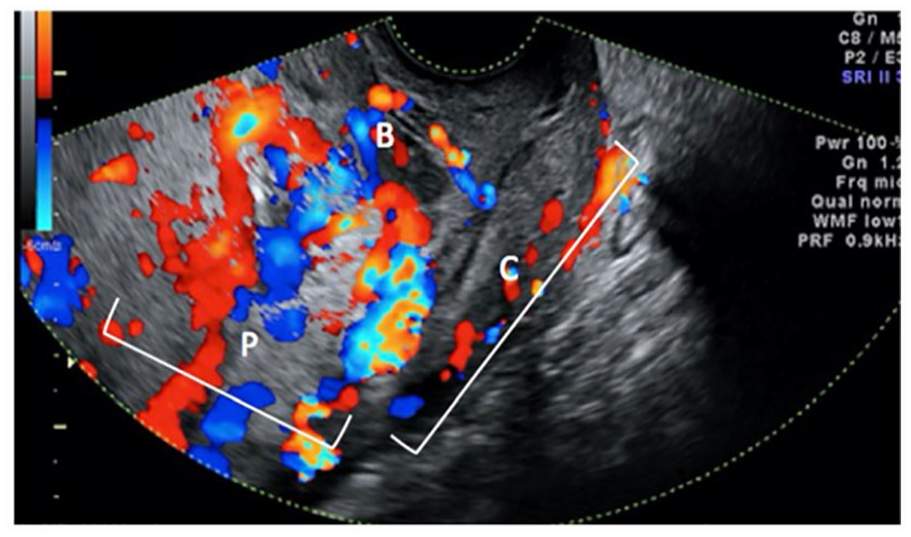
A sagittal view from a transvaginal sonogram conducted on a 29-year-old woman (31 weeks 2 days of gestation). A complete placenta previa is visualized: anterior placenta (marked by the letter P) with complete placenta previa. The color Doppler illustrates a significant amount of hypervascularity in the lower uterine segment bridging into the anterior wall of the cervix (denoted by the letter C); hypervascularity is defined as continuous retroplacental Doppler color flow “bridging” quite away from the retroplacental interface to the bladder (denoted by the letter B). 23
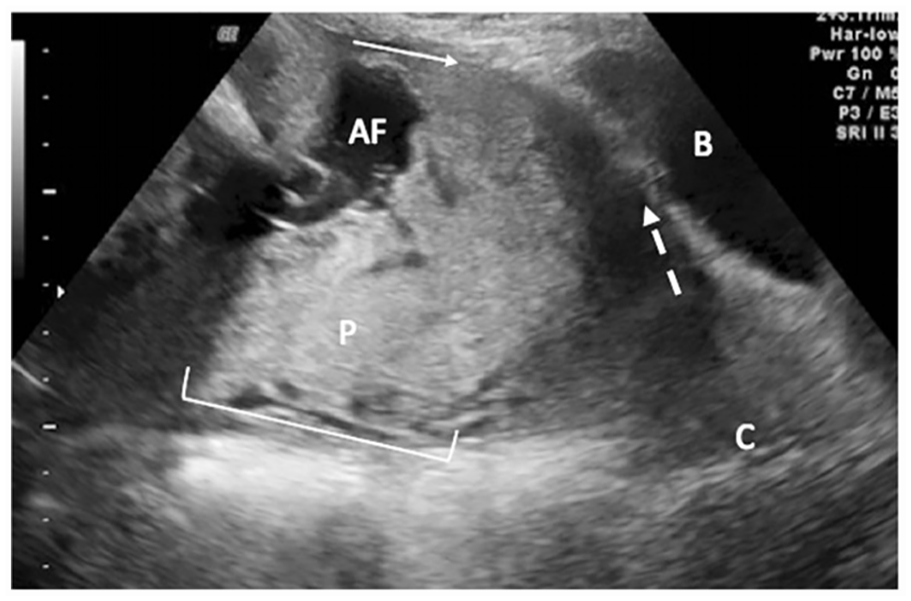
A sagittal view from a transabdominal sonogram conducted on a 36-year-old woman (30 weeks 4 days gestation). A complete placenta previa is noted: posterolateral placenta (denoted by the letter P) with complete placenta previa over the cervix (marked by the letter C); there is a loss of retroplacental clear space and the anterior portion of the placenta (see solid white arrow). Loss of retroplacental clear space was defined because the absence of the hypoechoic line between the maternal placental surface and the corresponding anterior uterine wall. The dotted arrow demonstrates a less defined uterine serosa/bladder (denote by the letter B) interface. The amniotic fluid is designed by the abbreviation AF. 12
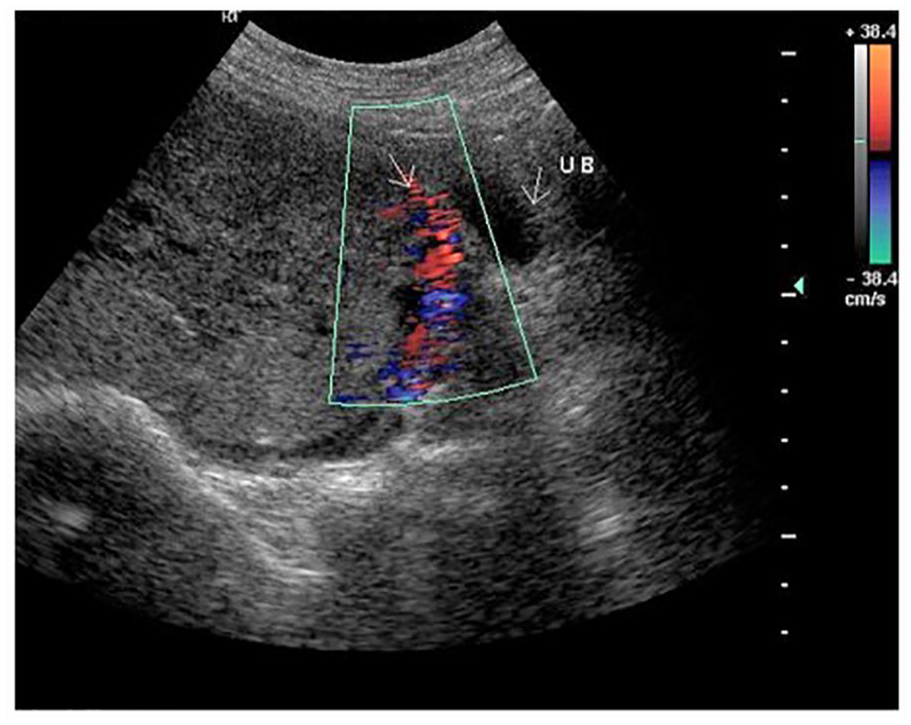
A sonographic image with color Doppler applied, which shows a placenta previa with accrete, urinary bladder (denoted by the letters UB) and hypervascularity (see solid arrow). 12
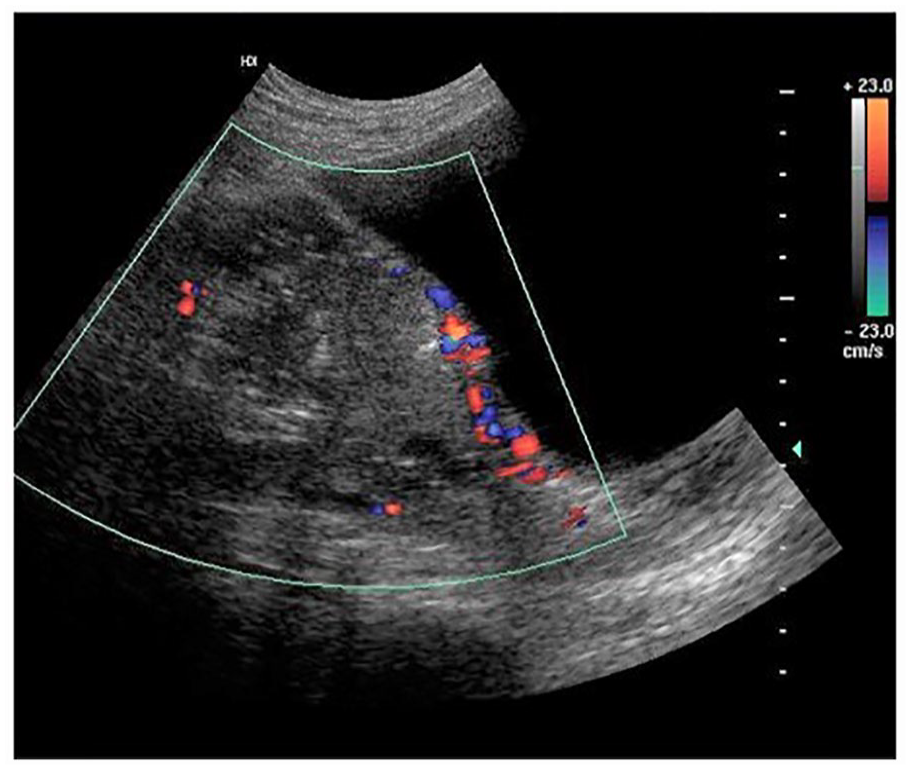
A transabdominal sonogram with color Doppler applied illustrates the vascularity of a placenta previa with increta. 12
MRI is only preserved for cases where sonographic findings are inconclusive, and most consider it a second-line diagnostic modality. MRI diagnostic criteria of PAS includes abnormal vascularity, placental bulge, placental heterogeneous intensity, dark bands within the placental tissue, focal villous interruption within the myometrium, and fibrin bands which help to assess the extent and depth of placental invasion and pelvic organ invasion by placental tissue.7,20 As mentioned earlier, the current concept in diagnostic imaging techniques, for the diagnosis of PAS, has not proved that MRI has neither higher sensitivity nor specificity and subsequently not superior to grayscale sonography in this context (see Figures 14–16).
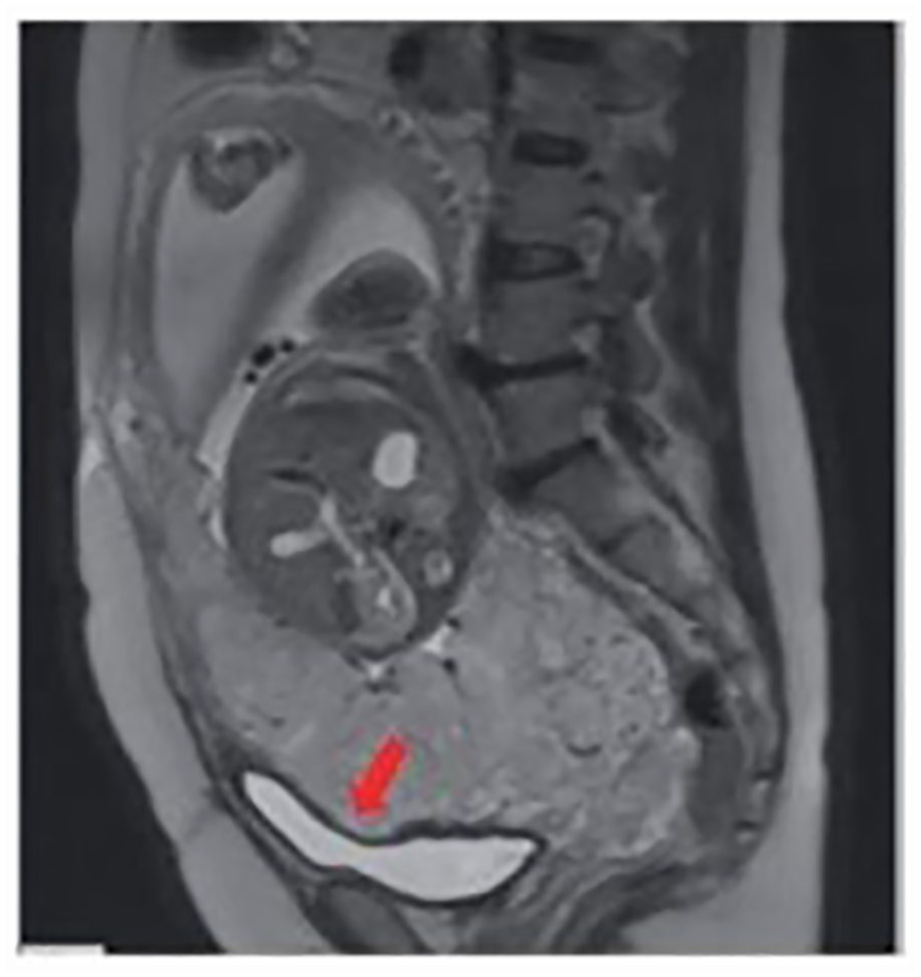
A sagittal view of an abdominal magnetic resonance imaging study (T2-weighted) showed a placenta which is completely covering internal os. The abnormal placental tissue was abutting the bladder wall (see red arrow). 32
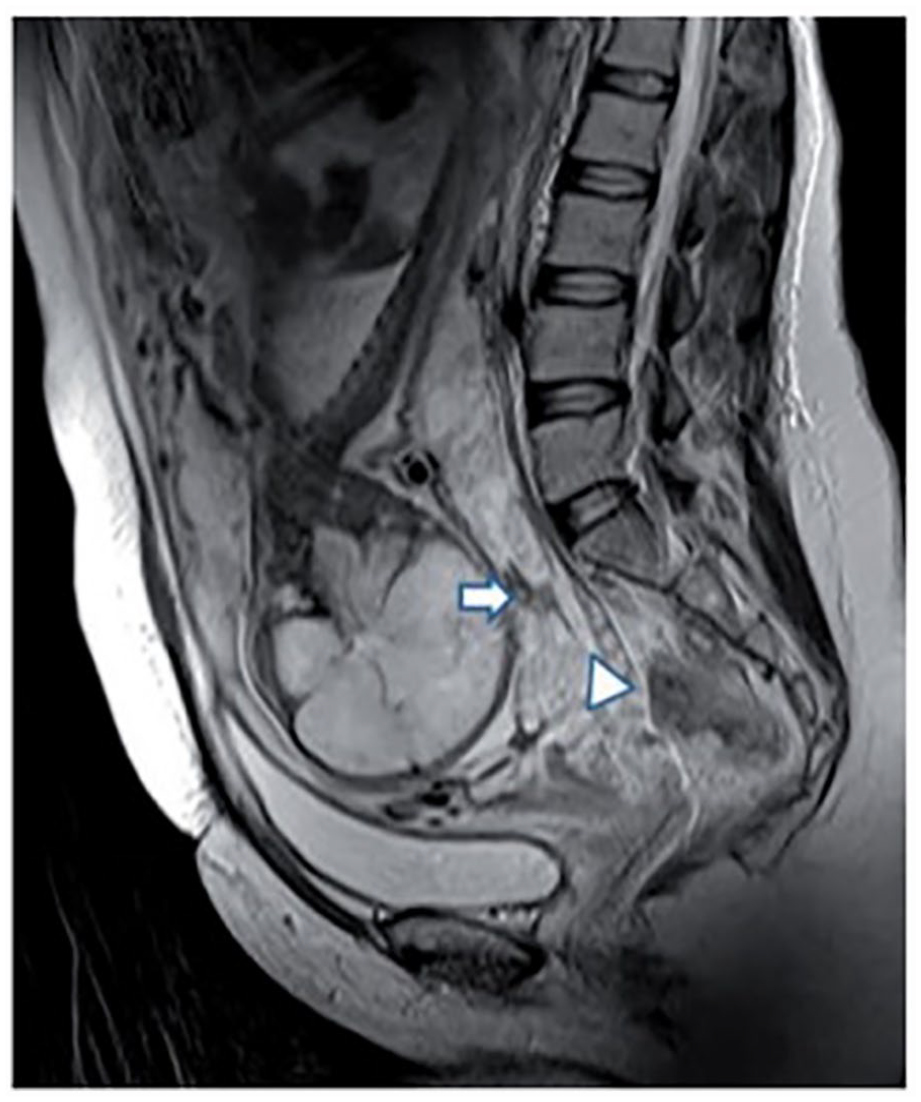
A sagittal view of an abdominal magnetic resonance imaging study (T2-weighted), which demonstrates an intraplacental dark bands (see white full arrow) and a loss of low-signal from the myometrium (see white arrowhead). 32
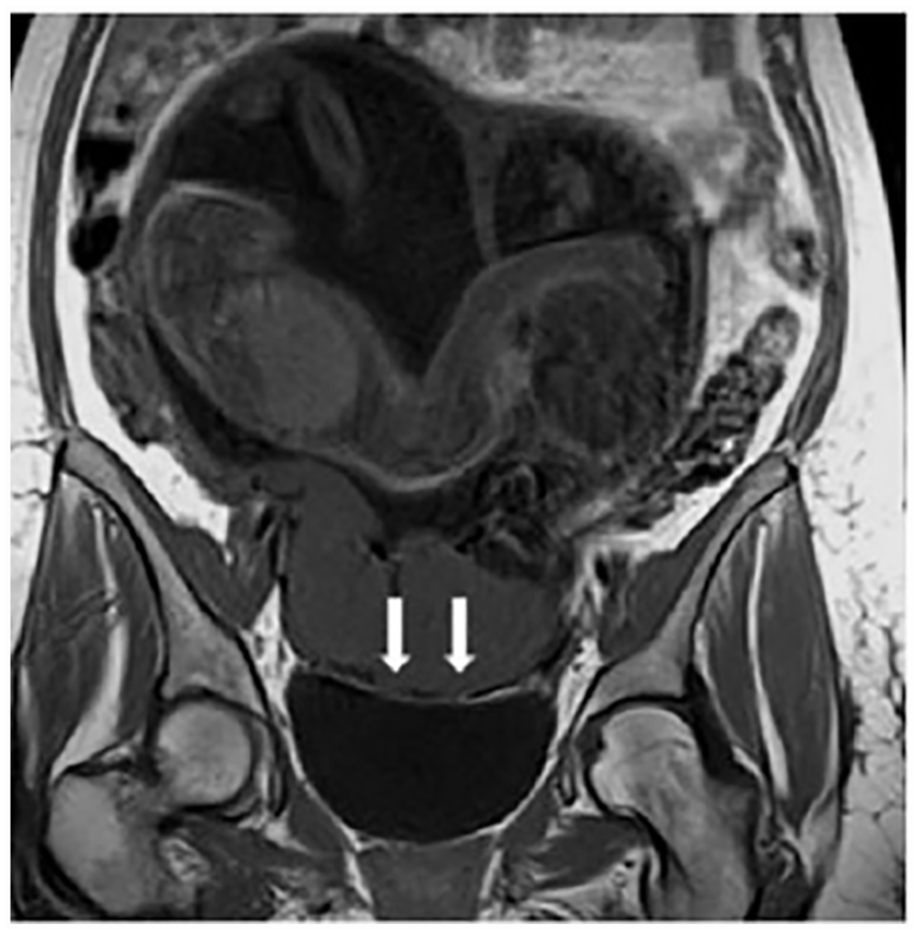
A coronal view of an abdominal magnetic resonance imaging study (T2-weighted), which demonstrates an interruption of the myometrial line (see white arrows). 33
Management Strategies
The protocols of management of patients, with PAS, have mainly employed a spectrum of approved approaches involving a comprehensive multidisciplinary team experienced in managing PAS cases. They mostly consist of senior obstetricians, general surgeons, intensivists, blood bank specialists, intervention radiologists, physicians, urologists, anesthesiologists, neonatologists, and whatever other diagnostic input, should the need arise. Hospital logistics also plays crucial roles in management plans, including midwifery and operating nursing teams with appropriate experience in handling such high-risk cases either antenatally, intraoperatively, and during the critical postpartum period. This would also include prompt transfusion facilities, capable of employing massive transfusion requirements, that have also vital roles in management plans.
Antenatal diagnosis of PAS is highly recommended because it allows prompt and timed referral of patients to a tertiary center, with high levels of maternal care, before the onset of labor or any bleeding episodes. The lack of early care is known to be associated with poorer outcomes 34 ; however, early action are associated with improved maternal, fetal as well as neonatal outcomes.
Preoperative Management
The preoperative management strategy should include all relevant considerations of value for optimization of the treatment plan and consequently improving the patient’s outcomes. Timing of delivery should be determined based on the delicate balance between the risks associated with the prolongation of pregnancy, namely, the possibility of bleeding from PAS and its sequelae and, on the contrary, the fear of iatrogenic immaturity and the associated fetal and neonatal morbidity. Discussion with the neonatologist is of utmost importance in determining the acceptable fetal age for delivery considering the available facilities and experience needed for neonatal support. There is a consensus agreed upon by most maternity centers that 34 weeks gestation is an acceptable compromise for delivery of MAP cases, after completion of the steroid course. 35 Delayed delivery beyond 36 + 0 weeks carries a higher risk of bleeding. 35
Anemia during pregnancy should always be investigated, and the patient’s hemoglobin levels should constantly be maintained at the highest normal levels. Iron deficiency, if any, should be corrected adequately, and all anti-anemia measures should be employed, including oral iron supplements, and parenteral iron preparations; even so, blood transfusions whenever indicated, erythropoietin-stimulating factors can, also, be used.35,36
The period of preoperative hospitalization, as well as the advice regarding bed rest (restricted activity), should be individualized and modified from patient to patient depending on the patient’s preferences and, otherwise, the patient’s logistics. 37
Classical Surgical Techniques
The intraoperative surgical strategies, as mentioned earlier, agreed on cesarean hysterectomy with placenta left in situ (PLIS), as the classical and widely accepted surgical approach for the treatment of patients with PAS. A total hysterectomy is recommended, rather than the subtotal one, due to the possibility of continuing bleeding from the cervical stump may be associated with the later one. 36
Delayed hysterectomy, being a recently introduced surgical intervention as an alternative to the traditional immediate hysterectomy in the treatment of patients with aggressive placental invasion, was evaluated in a retrospective chart review study in the United States in 2019 to assess the surgical outcomes associated with that technique, which entails deferring the hysterectomy with the placenta in situ, for 4 to 6 weeks following the cesarean delivery. The study aimed also at comparing the outcomes of different surgical parameters of the delayed hysterectomy management approach compared with those associated with the classical approach of immediate cesarean hysterectomy in patients with major placental invasion and extension, as in placenta percreta.
The proposed targets of the delayed hysterectomy technique in the management of severe PAS are to reduce the operating time and consequently, the amount of bleeding, and also, reduce requirements for transfusion as well as other surgical, medical, and anesthetic complications.
Fertility-Sparing Management
Fertility-sparing or conservative management strategies for PAS may be broadly classified into two groups of operations, depending on whether the placenta will be removed during cesarean delivery or will be left in situ. The first group of operations, which entails removal of the placenta and leaving the uterus behind after delivery, includes a wide spectrum of techniques and procedures and can be further divided into three main categories: The first comprises procedures with pelvic vessels occlusion or ligation; the second includes techniques entailing resection of the abnormal myometrium along with its related placental bed (constructive surgical management [CSM]); and last, entails compression sutures over the raw placental bed and abnormal myometrium related. These surgical techniques may be employed separately or in combination. 38
CSM, or resective surgical management, is a lesser-known alternative surgical intervention for uterine preservation techniques with placental removal in the context of fertility-sparing management of PAS. The technique entails a block resection of the placenta and affected myometrium (BRPM) with subsequent reconstruction of the uterus. The technique was evaluated in a prospective, nonrandomized study in Argentina (2011–2018) and published in 2019. The study concluded that CSM, being a third option in the treatment era of PAS (besides hysterectomy and uterine preservation with the placenta in situ), is considered a viable and safe intervention, among fertility-sparing management for PAS; in a condition, the procedure was included in an institutional program and employed in carefully selected patients by expert interdisciplinary teams. 38
The triple-P procedure entails the resection of the abnormally invading placenta, either partially (focal) or totally, along with the corresponding (involved) part of the uterine wall, followed by uterine wall reconstruction. 39 The procedure involves preoperative placental localization and delivery of the fetus through a transverse uterine incision above the upper border of the placenta; pelvic devascularization; placental nonseparation with myometrial excision; and reconstruction of the uterine wall. Blood supply is reduced immediately after delivery of the fetus by inflation of a pre-positioned occlusion balloon in the anterior division of the internal iliac artery. Following the removal of the whole myometrial wall covering the placental bed and the “myometrial defect,” blood loss from the separated and adherent portion of the placenta is controlled by under suturing. Hemostatic sutures are inserted along the line of the placental tissue’s invasion into the posterior wall of the bladder in cases of placenta accreta in which placental imaging shows trophoblastic invasion. This results in hemostasis. Myometrial defect closure in two layers follows this much like a cesarean section. 40
Another surgical approach for uterine sparing with placental removal is lower uterine segment folding, which was described and evaluated in a prospective study from Egypt in 2019. The study concluded that the lower uterine segment technique can also be employed to treat patients with PAS with fertility-sparing. The procedure was founded to yield appropriate hemostasis, reducing the intraoperative blood loss and, hence, the requirement for massive blood transfusions. The technique is simple, can be done in a relatively short time, and is consequently associated with improved all surgical outcome parameters compared with either radical or even other conservative surgical strategies employed for the treatment of PAS. 41 Bilateral uterine artery ligation, a relatively newly introduced surgical intervention for fertility-sparing management of patients with PAS, particularly suitable for low-resource settings, was the subject of a prospective, nonrandomized study. The study was conducted in Egypt, between January 2018 and November 2019, to evaluate the efficacy and safety of this technique among other alternatives employed for fertility-sparing management of PAS. The technique entails ligation of both uterine arteries, immediately after delivery of the baby, through dividing the broad ligament, then clamping and ligating the vessels just below the level of the placental bed; after that, gentle trials are started to separate the placenta from the uterine wall, preferable, in a piece-meal manner. The study concluded that the employment of bilateral uterine artery ligation techniques is a safe and effective intervention in the fertility-sparing management of PAS. The technique was associated with a high uterine preservation rate, less peripartum morbidity, and fewer blood transfusion requirements. It is important to note that these results are comparable with those obtained with the triple-P procedure. 42
B-Lynch suture, a newly introduced, hemostatic technique, useful in atonic postpartum hemorrhage, its efficacy in controlling postpartum hemorrhage in patients with PAS, was assessed in a retrospective cohort study, conducted in Israel, and published in 2018. The study found that, in view of bleeding control, B-Lynch suture is an easily applied, simple technique that does not require any particular logistics such as interventional radiology; however, it is effective in controlling massive obstetric hemorrhage.43 –45 However, the most employed technique for uterine conservation and fertility-sparing is one which entails leaving the placenta in situ (LPIS) after cesarean delivery to allow for spontaneous regression and autolysis. 46 A recent American survey demonstrated that the technique of uterine preservation with LPIS was the approach of choice for 32% of obstetricians as a fertility-sparing intervention. 47 This uterine sparing technique with placental retention will require a prolonged hospital stay, with the potential risks of secondary postpartum hemorrhage, the persistence of placental tissue in utero, requiring re-laparotomy and hysterectomy reported in more than 50% of cases.48,49 As there is always a high-risk of infection associated with this surgical approach, an umbrella of empirical broad-spectrum antibiotics, covering all the spectrum of organisms, is usually advisable and recommended.50,51
Intervention Radiologic Imaging Techniques
Endovascular interventional diagnostic radiologic techniques, for control of intractable obstetric hemorrhage, during the management of pregnancies complicated with morbid placental adherence have gained increased attention during the last few decades. An interventional radiologist, as already mentioned earlier, should be included in the multidisciplinary team managing PAS cases. Until lately, there has been no global consensus assessing the value and efficacy of these therapeutic interventions, their spectrum of them has been expanded greatly to involve most of the vascular tree supplying the pelvic organs. This spectrum may include the prophylactic balloon occlusion of the internal iliac arteries (PBOIIA), common iliac arteries (PBOCIA), abdominal aorta (PBOAA), and last the uterine arteries (PBOUA), with or without embolization. The spectrum includes, also, primary uterine artery embolization (UAE), pelvic collaterals, and the anterior divisions of the IIA.
The efficacy and safety of all these endovascular occlusion techniques for the treatment of patients with PAS were thoroughly assessed in a systematic review study, which included 85 full articles and the primary end point of evaluation was the estimated blood loss (EBL) volume. The reviews secondary endpoints included the total packed red blood cells (PRBC) units transfused, the number of patients requiring transfusion, number of patients subjected to hysterectomy, mean fluoroscopic time, maternal and fetal exposure doses of radiation, fetal complications including Apgar score, operating time, balloon occlusion time, duration of hospital stay, and postoperative complications related to the surgery itself or to the concurrent endovascular procedure.
The study concluded that, despite the conflicting evidence supporting the role of endovascular interventional radiologic techniques for perioperative bleeding control during the management of PAS patients, in previous studies, the available evidence obtained from this study supported the concept, in favor of use of these modalities for management of patients with PAS, as for both proven efficacy and safety. Among the endovascular interventional techniques, PBOAA was associated with the relatively least range of complications, as least perioperative bleeding, and the lowest radiation exposure doses compared with other endovascular interventions. 52
A multi-centered, retrospective cohort study conducted in the United States, during the period from May 2014 to June 2018, the objective of the study was to evaluate and compare the efficacy of endovascular balloon occlusion techniques of either the internal iliac arteries or abdominal aorta with the classical intervention of bilateral internal iliac artery ligation or without any adjunctive hemostatic procedures, in terms of controlling massive obstetric hemorrhage associated with the management of PAS. The study concluded that balloon occlusion of either the internal iliac arteries or the infrarenal abdominal aorta during the operative management of MAP is associated with improvement of all parameters of the surgical outcome, compared with vascular ligation technique or no adjunctive technique, including operating time, peripartum EBL, a necessity for critical care unit admission, and without any reporting of balloon occlusion-related complications. 53
Another retrospective cohort study from British Columbia, published in 2018, evaluated the internal iliac artery balloon tamponade in women who underwent emergency cesarean hysterectomy for the management of PAS in the context of controlling the massive peripartum bleeding, usually, associated with such cases. The study proved efficacy in the reduction of EBL and, hence, the requirements for massive transfusions in association with the employment of this intervention. 54
Prophylactic UAE, performed just after fetal delivery, by interventional radiologists, was included in the management of massive obstetric hemorrhage associated with PAS. The efficacy of the procedure was evaluated in 11 retrospective cohort studies and five case series in 2021, concluding that the available data derived from the study were promising in view of a positive rule for intraoperative UAE in managing massive obstetric hemorrhage 55 (see Figure 17).
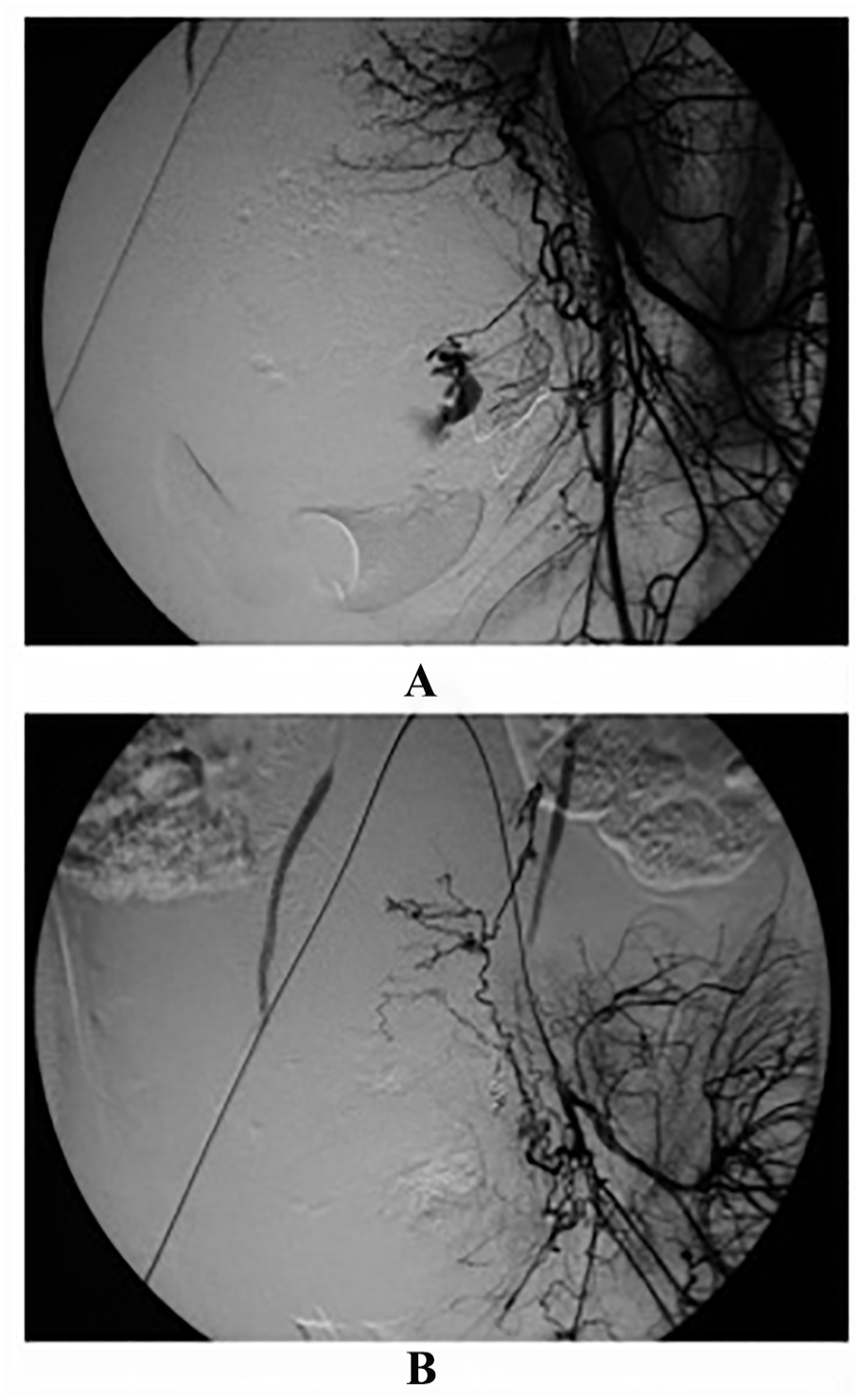
An angiographic study was conducted on a 20-year-old female patient with massive postpartum hemorrhage. (A) An angiographic image of the left internal iliac artery, during the arteriogram, with a left uterine artery branch which revealed extravasation of the contrast medium. (B) An angiographic image which highlights a super-selective arterial embolization with three micro-coils, the procedure aimed at complete occlusion of the bleeders. 56
The resuscitative endovascular balloon occlusion of the aorta (REBOA) is one of the known interventional radiological, minimally invasive techniques employed to control the intraoperative major obstetric hemorrhages associated with PAS. The procedure entails the introduction of a balloon occlusion catheter via the femoral artery, either percutaneously or via a cutdown in the groin, and pushed until it gains access to the abdominal aorta, to create an endovascular balloon occlusion.57,58
The REBOA is, in fact, not a new technique, as a multidisciplinary management team of a placenta percreta patient at 34 weeks gestation reported its prophylactic use in 1995 for a planned cesarean hysterectomy operation. The outcome of all management parameters was amazing; since no necessity for either massive transfusion or high critical care unit admission was raised, the patient was discharged 1 week after operation without any reported complications. 59
Operative Complications
The most often encountered complications during delivery of patients, with PAS, are intractable pelvic hemorrhage and urinary tract damage. 60
A retrospective cohort study from Egypt was conducted from January 2014 to December 2018 to assess the association between cesarean delivery, with or without hysterectomy, in patients with PAS and urinary tract damage. The study concluded that there is a significant relationship between the delivery of patients with PAS and the occurrence of urinary tract injuries. They estimated the incidence of these urinary complications when delivery is achieved by cesarean section per se (conservative surgery) is as high as 21.7% of them, the bladder was affected in 11.7%, ureter 4.7%, while combined damage of bladder and ureter occurred in 5.3%. Moreover, they found that this already high incidence of urinary tract damage associated with PAS treatment will jump to 40.5% when the condition requires a hysterectomy (cesarean hysterectomy). It is important to underscore that this study supported the employment of prophylactic preoperative ureteric stents to reduce the likelihood of urinary tract accidents in the context of the management of PAS patients. 61
Anesthetic Considerations
Markley et al. tried to address the most suitable anesthetic approach for the delivery of patients with PAS in the context of the proven advantages and the associated risks. They demonstrated that neuraxial anesthesia, which is the most commonly employed technique in obstetric anesthesia for cesarean delivery globally, included suspected cases of PAS, and, apart from the patient’s preferences, would require intraoperative additional intravenous analgesia supplementation such as I.V. opioids, anesthetic short-acting drugs (propofol and/or ketamine); the need for this analgesic supplementation may be attributed to the relatively long operating times, which is taken as evidence against a neuraxial approach being the anesthetic route of choice for delivery of patients with PAS. The authors also highlighted the known, potential hazards associated with general anesthesia, including the possibility of failed intubation, as in morbidly obese and short neck patients, as well as the likelihood of pulmonary aspiration of the patient’s vomitus; nonetheless, these complications are unlikely to happen except in emergencies, in view that, electively planned, high-risk PAS cases will be handled, as mentioned earlier, by a multidisciplinary management team, including a senior and experienced obstetric anesthetist as well as senior nursing staff expert in preoperative preparation of patients, including preoperative fasting Nothing By Orum (Oral). In addition, and considered, neuraxial anesthesia is not a procedure free of risks, especially following some published reports from the United Kingdom, attributing 20% of pregnancy-associated cardiac arrests, to the employment of neuraxial anesthesia. 62
Finally, the authors, based on the risk-benefit spectrum of both anesthetic approaches, were in favor to recommend general anesthesia as the primary anesthetic approach for the delivery of patients with PAS, provided that the operation will be managed by an experienced obstetric anesthetist with a well-prepared and fasted patient. 62
Postoperative Management
Broad-spectrum antibiotic prophylaxis and the use of prophylactic uterotonic agents (in case of uterine preservation) are widely accepted and practiced by most obstetricians despite there is no evidence-based literature supporting its use; however, it is worth noting, when it comes to operations involving uterine preservation with placental retention with considerable potential risk of infection. The antibiotic spectrum should cover all the possibly implicated pathogens. Culture and sensitivity tests should be resorted to once the necessity arises. The most common and worrying postoperative complication is bleeding; here, there is a place for the use of uterotonics for variable durations, if the bleeding occurred in the immediate postoperative and up to 24 hours postoperatively, so, uterine atony will be the most likely cause of bleeding. However, delayed postoperative bleeding, occurring in more than 24 hours, warrants full investigation for infective or coagulopathic backgrounds. 6 Close monitoring of the patient’s hemodynamic status either clinically or via laboratory, or even by imaging techniques, whenever indicated, is crucial for early detection and subsequently management when indicated. 6
Conclusion
The worst obstetric nightmare continues to be the PAS. Antenatal diagnosis of PAS as well as a multidisciplinary team approach for management of such patients are associated with significantly reduced peripartum maternal and fetal complications, including fewer requirements for blood transfusion, less required hysterectomies, and less intraoperative visceral damage and subsequently fewer maternal deaths. The reported incidence of PAS has shown a steady increase in the last century as well as during the first decades of the current one which is directly proportionate to the rising rate of cesarean deliveries. There is a growing worldwide consensus that previous cesarean delivery and placenta previa or low-lying placenta constitute the major risk factors implicated in the occurrence of PAS. The most accepted theory explaining the occurrence of the spectrum of PAS states that the defect occurring at the endometrium-myometrium interface, probably due to endometrial damage, will lead to abnormal decidualization in this area of the uterine wall damage, which will lead to a deeper than normal invasion of placental anchoring villi with subsequent trophoblastic infiltration. Up to date, grayscale sonography supplemented by Doppler studies is considered the recommended first-line aids for the diagnosis of abnormal placental invasion with overall sensitivity and specificity of 82.4% to 100% and 71% to 100%, respectively. Cesarean hysterectomy with PLIS is agreed as the classical and widely accepted surgical approach for the treatment of patients with PAS. However, the most employed technique for uterine conservation and fertility-sparing is one which entails LPIS after cesarean delivery to allow for spontaneous regression and autolysis. Endovascular interventional radiologic techniques for control of intractable obstetric hemorrhage during the management of pregnancies complicated with morbid placental adherence have gained increased attention during the last few decades.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
