Abstract
Objective:
Identify factors predictive of therapeutic success following ultrasound-guided sheath injection of the long head of the biceps tendon.
Materials and Methods:
A total of 162 cases were divided into three groups based on clinical responses: complete, partial, or no relief. The presence or absence of clinical symptoms, glenohumeral joint structural pathology, and treatment method was compared.
Results:
Of the 115 injections with follow-up, 19.1% reported no clinical response while 53.0% had a complete response. Patients with fibromyalgia or chronic spine pain were strongly associated with a poor outcome (odds ratio [OR] = 5.7, P < .001).
Conclusion:
Ultrasound-guided intra-sheath injection is an effective method of non-surgical management for biceps tendinopathy.
Keywords
Shoulder pain is a common cause of morbidity, accounting for the third most common cause for patients seeking musculoskeletal-related medical care.1–3 Various structures are involved in the complex shoulder joint anatomy and can serve as potential sources of pain. The muscles of the rotator cuff, glenoid labrum, long head of the biceps brachii (LHB) tendon, as well as accessory ligaments, osseous structures, cartilage, and bursae can each become pathologic. Generally, this produces pain and reduction in range of motion. It is common to see several of these structures concomitantly injured. In particular, the LHB is a primary generator of anterior shoulder pain, but isolated bicipital tendinitis is reported to be rare.4–6 The LHB has both intra-articular and extra-articular segments, the latter of which directly communicates with the glenohumeral joint. This allows inflammatory processes to affect additional and sometimes relatively distant structures, within the glenohumeral joint. 4
While acute injuries to the LHB do occur, chronic degeneration is more common. The pathology related to degeneration of the LHB ranges from tenosynovitis, of the surrounding tendon sheath, to injury of the tendon itself. As a consequence of chronic tendon injury, there is development of scar tissue and infiltration with mucopolysaccharides. This abnormal tendon structure can predispose patients to partial- and full-thickness tears. Moreover, during this progressive degeneration, adhesions form within the tendon sheath and bicipital groove, limiting LHB mobility. 4 Various management strategies have been proposed including non-surgical and surgical options. Non-surgical management typically includes behavior modification such as rest, non-steroidal anti-inflammatory drugs (NSAIDs), ice, and physical therapy. When these strategies fail to relieve symptoms, corticosteroid injection is recommended, within the tendon sheath or at the glenohumeral joint.4,7 The former injection site is seldom done as a positive association between tendon rupture and peri-tendon steroid injection has been previously reported. 8 Usually, the combinations of all non-operative interventions including either injection result in resolution of symptoms for 75% of patients. 9 The remaining 25% conversely are considered for surgery in the form of tenotomy or tenodesis.10,11 Given that intra-articular injection and extra-articular injections serve as the final non-surgical management strategy in most cases, it seems appropriate to characterize for whom this technique is best for as to avoid a more invasive procedure.4,12–16
For many years, there was inconclusive guidance on intra-articular steroid injection for LHB tendinopathy as no true long-term benefit had been identified. Short-term benefit of steroid injections has been typically characterized by less than 12 weeks in randomized control trials and subsequent meta-analysis. 9 Ineffectiveness of steroids has been anecdotally associated to patient-specific factors and no criterion has been developed to predict treatment response. No study reports symptomatic outcomes from LHB intra-articular injection and compared physiologic or provider technical differences. Yet, it is possible to formulate analogies from studies that explore other pathologies. Lee 17 demonstrated pain-related syndromes negatively affect pain reporting and functional assessment. Koh 18 reported the improvement in pain score as reported by visual analog scale (VAS) in patients with adhesive capsulitis but failed to document variation in glenoid and labral pathology. Age has been another interesting characteristic not thoroughly explored as some studies report long-term clinical benefits and others do not, and a key difference between studies being the median age of the population. 19 This nebulous understanding of steroid injection treatment response hampers physicians in providing effective recommendations. This procedure is now becoming more widely accepted, and has a GRADE “A” recommendation by the American Academy of Family Physicians (AAFP)* for short-term relief from chronic tendinopathies. 20 It is therefore of increasing importance to identify factors that predict the likelihood of therapeutic efficacy following ultrasound-guided LHB tendon sheath injection. The authors hypothesize that when applied to the correct patient population, LHB tendon sheath corticosteroid injections are efficacious and reduce the need for surgical management of shoulder pain. Based on review of studies, this would appear to be the first documented study specifically aimed at determining factors predicting the likelihood of treatment success or failure with direct LHB tendon sheath injection in patients presenting with clinical evidence of biceps-related pain.
Materials and Methods
Participants
After obtaining approval by the Institutional Review Board (protocol number 20130214), clinical records of all patients who underwent ultrasound-guided injection (USGI) of the LHB tendon sheath with a mixture of corticosteroid/local anesthetic from 2007 to 2013 were retrospectively reviewed. The retrospective review of clinical data without a need for additional patient follow-up precluded the requirement of necessitating informed consent for access to previous clinical records. These patients were referred for injection based on clinical signs and symptoms suggestive of biceps tendinitis as determined by three orthopedic surgeons using the treatment algorithm defined by Figure 1. Patients with a known history of ipsilateral malignancy or under 18 years of age were excluded. A total of 158 patients were identified and 4 of these patients underwent bilateral LHB USGI, totaling 162 procedures. Individual patient demographics were collected, including age, sex, and body mass index (BMI). Documentation was made as to whether patients had any of the following pain-associated diagnosis: fibromyalgia, rheumatoid arthritis, or other inflammatory arthropathies. All the study participants demonstrated abnormal sonographic appearance of the LHB tendon (increased size, hypoechoic appearance, hyperemia on Doppler interrogation or tenosynovitis, see Figure 2A–E).
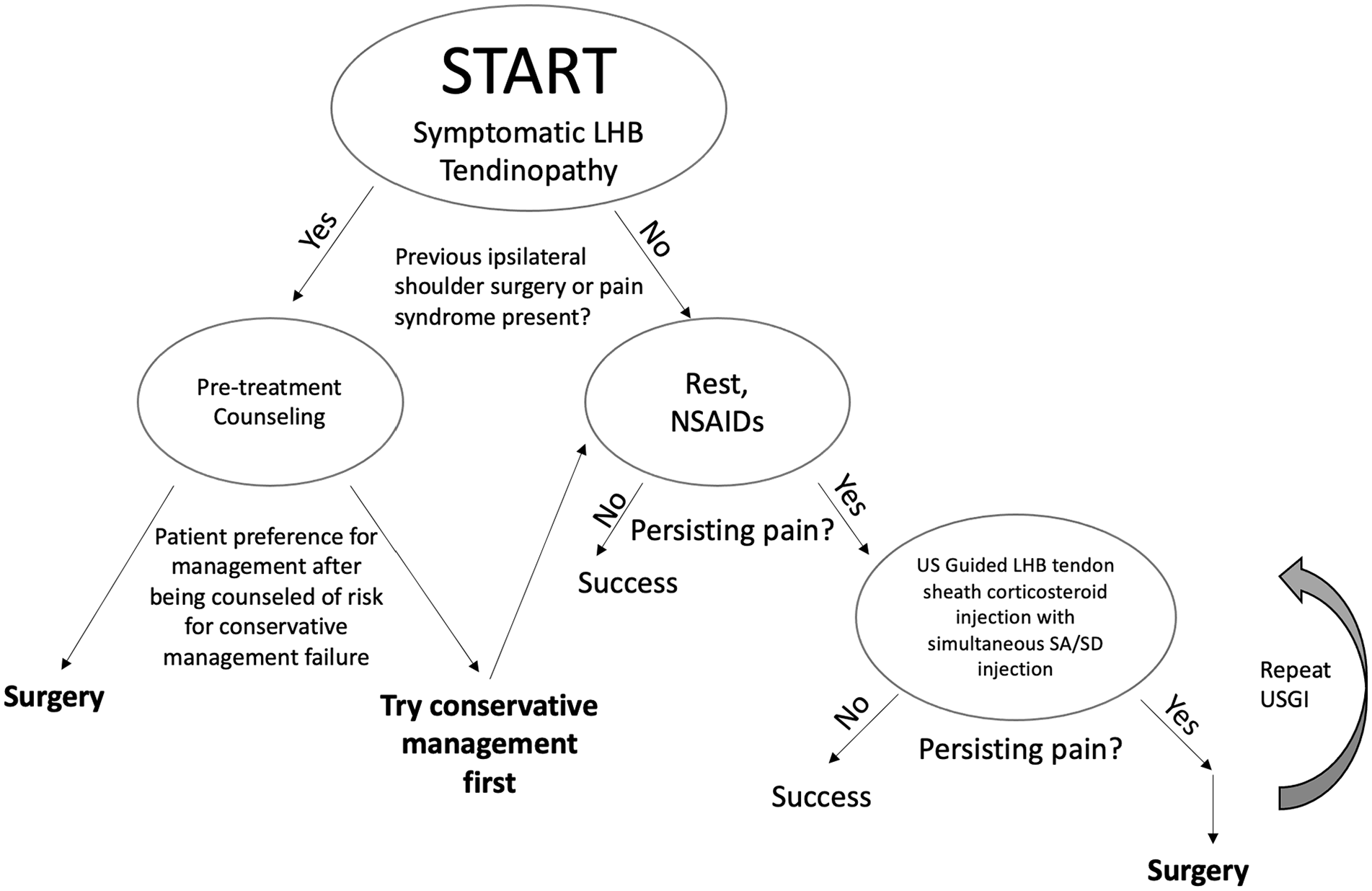
The proposed treatment algorithm. LHB, long head of biceps; NSAID, non-steroidal anti-inflammatory drug; SA/SD, subacromial/subdeltoid; USGI, ultrasound-guided injection.
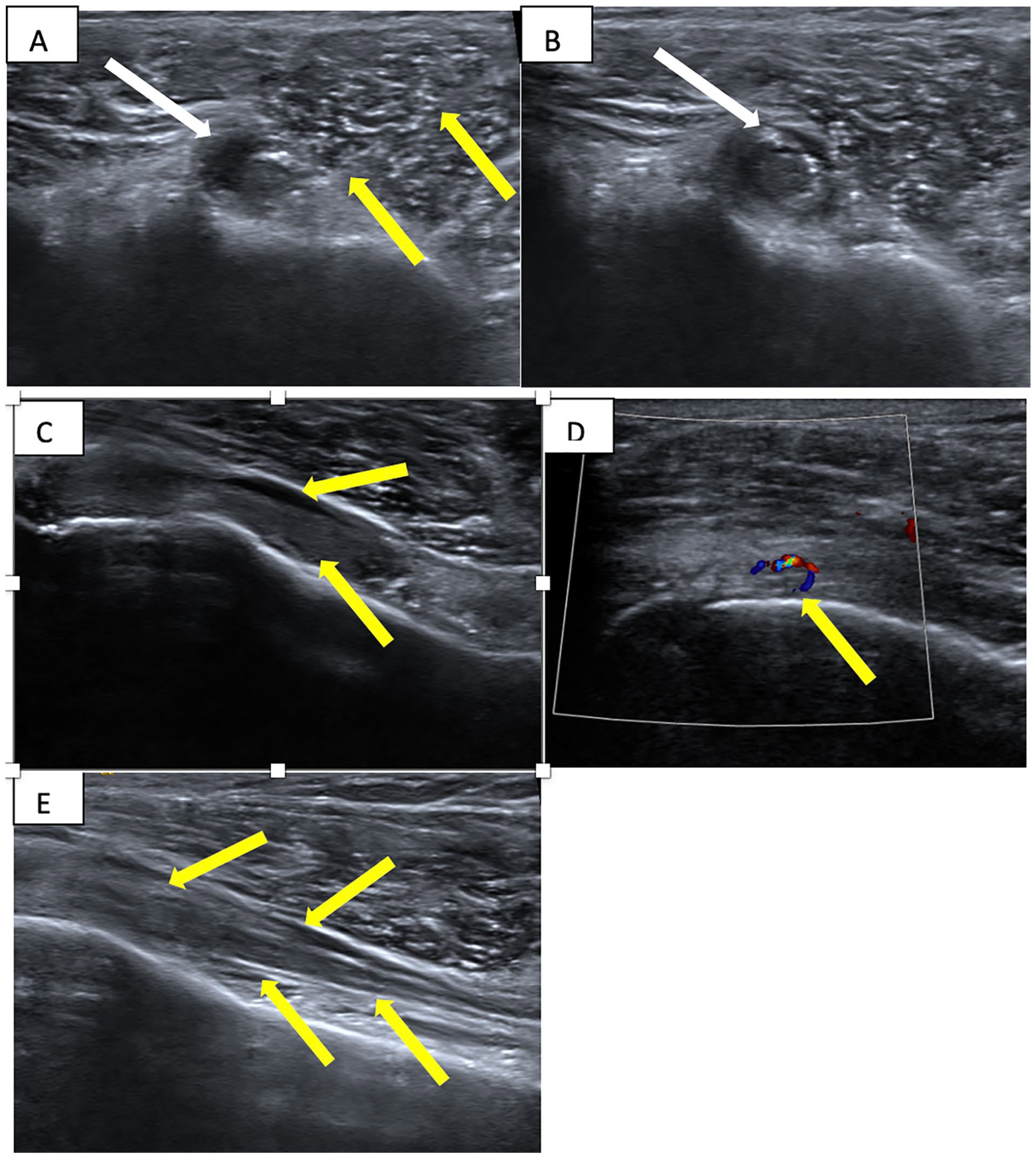
(A) Short-axis sonogram at level of bicipital groove demonstrates biceps tenosynovitis and enlarged tendon (white arrow) as the needle approaches the sheath (yellow arrows). The needle is seen in the tendon sheath housing the enlarged heterogeneous tendon with its tip deep to the tendon. (B) Ultrasound-guided injection of the long head of biceps (LHB) tendon sheath. Post-injection short-axis sonogram demonstrates steroid and anesthetic mixture surrounding the LHB tendon within the biceps tendon sheath (white arrow). (C) Longitudinal view of the LHB tendon in anatomic position within the bicipital groove. Note that the tendon is flush against the bone. There is tendinosis and tenosynovitis (yellow arrow). (D) Longitudinal view of the LHB tendon in the bicipital groove with power Doppler showing tendon hyperemia in this patient with tenderness over the bicipital groove with light pressure by the ultrasound probe (yellow arrow). (E) Longitudinal view of the LHB tendon after injection. Echogenic material is within the sheath and displaces the tendon from its normal position against the bone. The yellow arrows denote the injectate surrounding the tendon.
Guidelines for post-injection follow-up are institutionally defined as a 6-week post-injection visit or phone call with the patient’s referring sports medicine physician to discuss response to therapy. A total of 47 injections had no follow-up information available; these patients were excluded from the study. To identify underlying systematic bias, age, sex, and BMI for this population was recorded and compared with the remaining participants. Participants were divided into one of three groups. The first cohort comprised of those who returned for follow-up after injection and reported near-complete or complete symptom resolution, designated complete relief (CR). The second cohort included patients who returned to clinic and reported some improvement, but continued symptoms and were designated partial relief (PR). The last cohort included patients who returned to clinic and had little to no response following LHB injection and were designated no relief (NR). Demographic, anatomic, clinical, and pathologic structural variables were subsequently used for statistical analysis during the chart review process. The status of anatomic structures was determined by available data from either imaging studies (including sonography or magnetic resonance imaging [MRI]) or from arthroscopy. When these data were not available, individuals were excluded from sub-analyses.
Injection Method
All injections were performed by a fellowship-trained musculoskeletal radiologist. Using a high-frequency linear-array transducer, the patient’s LHB tendon sheath was injected using an in-plane approach with direct visualization (Figure 2A and B). The needle was guided into the sheath on the side of the tendon until it hit the bone behind the bicipital groove. Once on bone, the needle was slightly retracted and a small test injection of 1% lidocaine without epinephrine was injected. Injectate was composed of 0.5% ropivacaine and triamcinolone acetonide. The dose of triamcinolone acetonide injections was either 40 or 80 mg, with a total final injectate volume of 3 mL. A subset of patients with sonographic and clinical findings of subacromial/subdeltoid (SA/SD) bursitis had concurrent injection of local anesthetic and corticosteroid into the SA/SD bursae. Patients with sonographic evidence of severe LHB tendinopathy (markedly enlarged, hypoechoic, or hyperemic tendon with or without intra-tendon fissuring), but no evidence of SA/SD bursitis, underwent 80 mg triamcinolone acetonide injection into the LHB sheath injection. Intra-tendon sheath injection was evident sonographically by free flow of the injected material around the biceps tendon (Figure 2C–E). Biceps tendon sheath distention without fluid extravasation was the determining factor for a successful injection.
Participant Data Extraction
Information on physician identified clinical features was collected from orthopedic specialist encounter note. It was important to specify the search to indications of any form of Ipsilateral Shoulder Surgery including arthroplasty and non-arthroplasty procedures. It was also important to include a prior diagnosis of Adhesive Capsulitis by screening for International Classification of Diseases (ICD) code (M75. 00). An MSK Musculoskeletal radiologist reviewed prior radiographic images for glenoid fraying or tears, labral tears, and biceps brachii tears. Complications were documented from patient note in orthopedic post-injection follow-up visit.
Differences in age and sex among the three response groups (CR, PR, and NR) were compared using analysis of variance (ANOVA) and post hoc Tukey honest significant difference (HSD) analysis. The remaining data are presented as dichotomous variables and are compared across the three separate response groups using a χ2 test.
These data included clinical features, structural pathologies, treatment variables, procedural complications, and patient demographic factors. Differences were considered significant at P < .05. A secondary analysis was conducted wherein individuals from CR and PR were grouped into a single population. This created a (response) group and a non-response group. A χ2 analysis for each variable was conducted. A Bonferroni adjustment for multiple χ2 comparisons was applied and our cutoff P value became .038. The statistical analysis was performed using Stata 11.1 (StataCorp, College Station, TX, USA).
Results
Demographics
There were 65 female patients and 50 male patients, with the mean age of 48.4 years and a standard deviation of 14.8 years (range, 18–78). The sex distribution across the three response groups—CR, PR, NR—was comparable. However, the age distribution across the three response groups was significant (P = .01). A post hoc analysis using Tukey HSD with Kramer adjustment for size was used to identify significant differences between the pairs. It revealed significant differences between PR versus NR (P = .01). Twenty-four patients with a history of chronic pain were deemed “pain syndrome” patients. These patients showed a higher rate of partial or no response to treatment, when compared with patients without chronic pain (P < .001).
Other features of the patient’s history, including whether the patient engaged in repetitive overhead throwing motion, was not associated with pain resolution (P = .136).
Pre-Intervention Treatment
In all, 88 individuals performed pre-intervention physical therapy. Forty-six individuals demonstrated CR, 26 individuals demonstrated PR, and 16 demonstrated NR. Across the groups, there were no differences (P = .700).
Lost to Follow-Up
The 47 individuals who were lost to follow-up were predominantly women (54%) with a mean age comparable with our analyzed population (P > .05). Four individuals received bilateral shoulder injections. Three individuals received 80-mg triamcinolone injection. There were 11 subacromial/sub-bursal injection. Additional information was not collected from the population.
Clinical Features
A history of ipsilateral shoulder surgery was associated with treatment response (P = .0114). A prior diagnosis of adhesive capsulitis showed no significant associations with treatment response (P = .366). An important caveat, only 99 of the 115 individuals had adequate pre-intervention imaging that permitted identification of glenoid, labral, and biceps tear or fraying. Regarding treatment outcomes, patients designated as having a normal rotator cuff without a partial- or full-thickness tendon tear demonstrated no significant differences compared with patients having an abnormal cuff with either a partial- or full-thickness tear (P = .75). The glenoid labral status was not associated with treatment outcome when patients were designated as having a normal labrum without fraying or tear versus an abnormal labrum with either fraying or tear (P = .595). The status of the LHB tendon was also not associated with treatment outcome when patients were designated as either having tendinosis, a partial tear, or a complete tear (P = .3327). There was no statistical difference in therapeutic response whether radiographically detectable osteoarthritis was present or not (P = .700).
Treatment Variables
Injection into the SA/SD bursa following LHB sheath injection was associated with a statistically significant chance of treatment success (P = .033). However, injection steroid dose (40 vs 80 mg) was not associated with treatment outcomes (P = .660).
Complications
Following USGI, there were two patients with LHB rupture. Both patients had a history of chronic LHB pathology. One was a 63-year-old man with a rupture during a tennis game and the other was a 74-year-old man with a history of prior arthroscopic rotator cuff tear repair and subacromial decompression. This composed a 1.7% incidence of tendon rupture.
All descriptive characteristics including clinical, demographic, and procedure associated complications between participants belonging to complete response, partial response, and no response group are summarized in Table 1.
Univariate Analysis of Patient Demographic, Radiographic, and Procedural Factors.
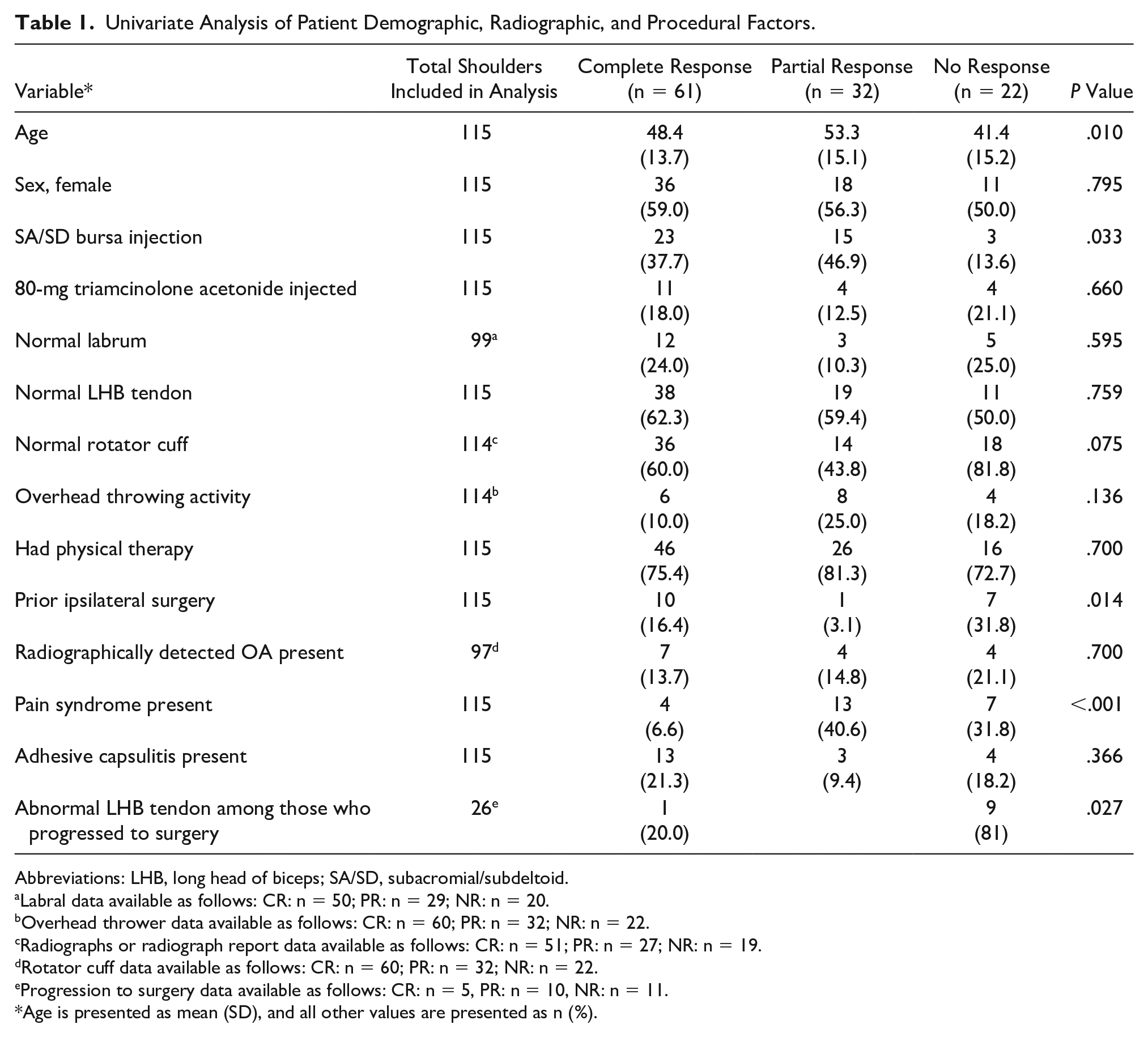
Abbreviations: LHB, long head of biceps; SA/SD, subacromial/subdeltoid.
Labral data available as follows: CR: n = 50; PR: n = 29; NR: n = 20.
Overhead thrower data available as follows: CR: n = 60; PR: n = 32; NR: n = 22.
Radiographs or radiograph report data available as follows: CR: n = 51; PR: n = 27; NR: n = 19.
Rotator cuff data available as follows: CR: n = 60; PR: n = 32; NR: n = 22.
Progression to surgery data available as follows: CR: n = 5, PR: n = 10, NR: n = 11.
Age is presented as mean (SD), and all other values are presented as n (%).
Logistic Regression
An ordered logistic regression was performed using those variables that reached statistical significance in the univariate analysis of differences among response groups. A random effects model was incorporated as no individual analyzed provided within-subject variability. These variables included age, pain syndrome, ipsilateral shoulder surgery, and concurrent SA/SD bursa injection. In this model, only pain syndrome (odds ratio [OR] = 5.7, P < .001, 95% CI, 2.2–15.0) was a significant predictor of outcome. A summary of all variables used in the logistic regression along with the individual significance are tabulated in Table 2.
Multivariate Analysis of Statistically Significant Patient Demographic, Radiographic, and Procedural Factors.
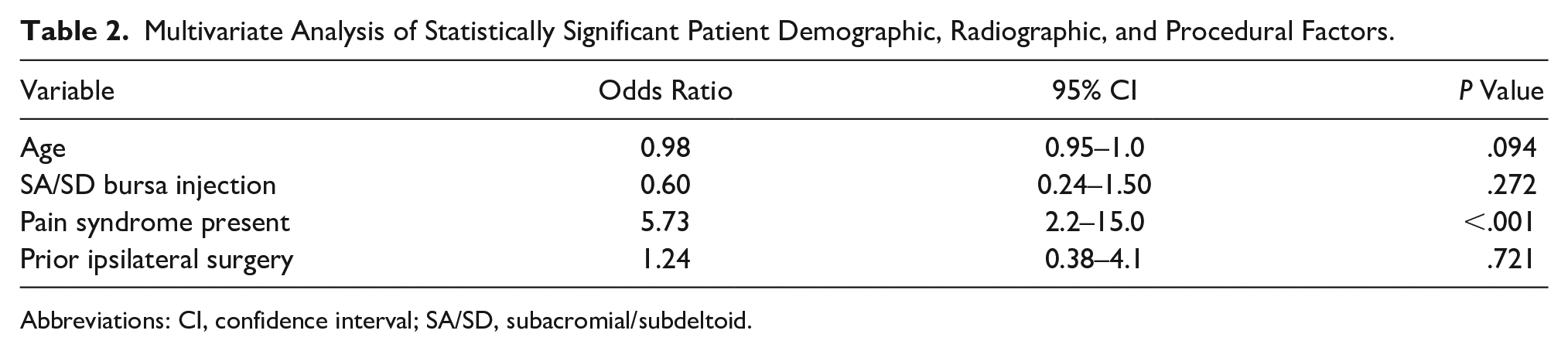
Abbreviations: CI, confidence interval; SA/SD, subacromial/subdeltoid.
Grouped Analysis
Grouping of CR and PR was done to create a response and no response comparison. These results (Table 3) were comparable with those conducted for the three separate groups except for analysis of individuals with pain syndrome and rotator cuff pathology. After grouping, there were no significant differences between individuals with pain syndrome and the sample population (P = .16). In addition, abnormal rotator cuff was associated with improved outcome compared with those with a normal rotator cuff (P = .018).
Grouped Analysis of Patient Demographic, Radiographic, and Procedural Factor.
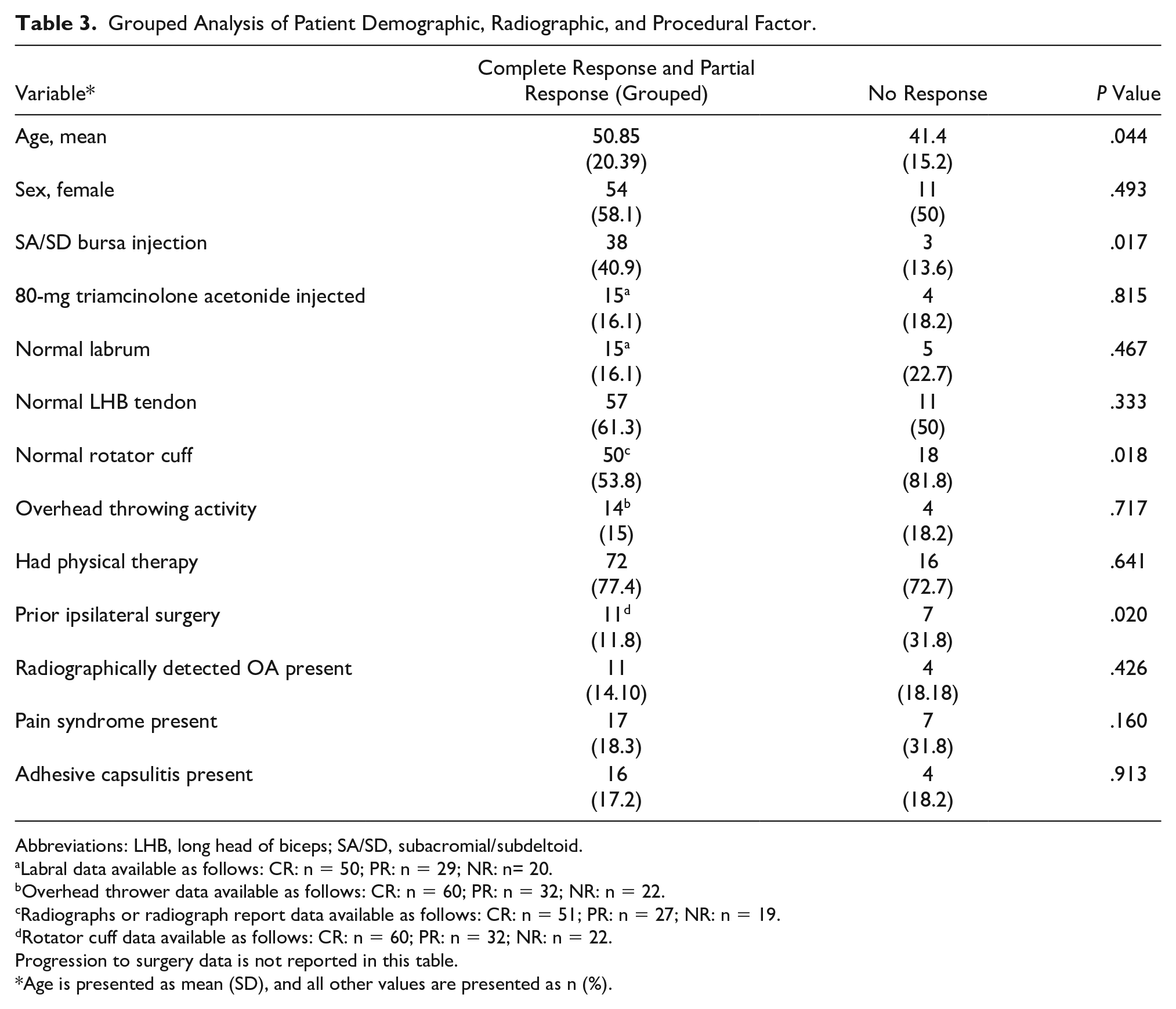
Abbreviations: LHB, long head of biceps; SA/SD, subacromial/subdeltoid.
Labral data available as follows: CR: n = 50; PR: n = 29; NR: n= 20.
Overhead thrower data available as follows: CR: n = 60; PR: n = 32; NR: n = 22.
Radiographs or radiograph report data available as follows: CR: n = 51; PR: n = 27; NR: n = 19.
Rotator cuff data available as follows: CR: n = 60; PR: n = 32; NR: n = 22.
Progression to surgery data is not reported in this table.
Age is presented as mean (SD), and all other values are presented as n (%).
Discussion
The data in the present study support existing publications for LHB tendon sheath injection as a possible treatment modality within the realm of non-surgical management for LHB tendon pathology, even in patients with known labral pathology, and especially with rotator cuff pathology. It is important to note that the presence of osteoarthritic findings should not serve as a deterrent for corticosteroid injection. Moreover, simultaneous SA/SD bursa injection should be considered in patients who undergo USGI if there is clinical and/or sonographic suspicion of concomitant bursitis or clinical evidence of subacromial impingement.
One of the best predictors of USGI failure is the presence of a “pain syndrome” as designated in this study. However, when assessing any response would be offered by steroid injection, individuals with pain syndrome are no different than those without pain syndrome. Therefore, when considering any clinical improvement within 6 weeks, having a past medical history of an inflammatory arthropathy or pain syndrome should not preclude this treatment. Furthermore, the exact reason as to why these patients exhibited less than expected complete response is not clear, possibilities include a lower pain threshold due to alterations in the afferent pain pathway (at the receptor level, peripheral nerve level, and/or within the central nervous system) or unrecognized concomitant pathologies (such as radiculopathy or plexopathy).
Based on this study cohort, younger patients had worse outcomes compared with older patients. Although speculative in nature, the reason for this may be because younger patients might be more active and may thus be less likely to comply with activity modification recommendations (such as limiting aggravating activity for a time period), especially if they participate in competitive or recreational sporting activities. Based on our results, not only did most patients have good relief or PR outcomes, but because USGI is less invasive, there are theoretically fewer associated risks, complications, and overall cost compared with surgery. As previously mentioned, certain subsets of patients require pre-treatment counseling to set expectations and establish behavior modification strategies that will increase the probability of USGI therapeutic success. In addition, patients with clinical examination or sonographic findings suggestive of subacromial impingement may benefit from simultaneous SA/SD bursal USGI.
Limitations
There are several limitations to this study, primarily related to the retrospective nature of the data collection. First, a standardized numeric scale would have been ideal to grade therapeutic response, but such detail was not recorded prospectively. Second, not all patients had complete data sets available which may lead to our statistics being underpowered; however, we believe that a sufficient number of participants were included for each variable studied to render meaningful clinical data. Third, some variables were assessed with different modalities and by different physicians. For example, the accuracy in evaluating the LHB tendon is likely different using ultrasound, MRI, or arthroscopy. Fourth, only adults were included in this study preventing examination of this intervention in children and adolescents with identical pathology. Fifth, we systematically lost 44 individuals to follow-up, the composition of this population was representative of our study population and therefore we presume bias from this was minimal. Last, because of the retrospective nature of this study, randomization and blinding was not possible, indicating the highest rigor to identify causality was not attainable.
Conclusion
USGI can present as a safe and efficacious way to treat acute LHB tendinopathy in most patients. Patients suffering from pain syndromes are predisposed to incomplete response. In addition, given the benefit of a simultaneous SA/SD bursa injection, especially in patients with clinical or sonographic evidence of subacromial impingement, this should be considered along with USGI of the LHB. Nevertheless, the authors are now planning on using these data to build a predictive model for when it is most efficacious to offer corticosteroid injection for LHB tendinopathy.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
