Abstract
Gallbladder carcinoma (GBC) is a lethal and rare disease despite it being the most common malignancy of the biliary tract. Most cases of GBC are also associated with cholelithiasis and inflammation. The poor prognosis of GBC is mainly due to a lack of symptoms in the early stages of the disease. Early detection and cholecystectomy can lead to a higher survival rate when the cancer is confined to the gallbladder mucosa. This case study describes a man in his late 50s who presented with elevated liver function tests and was diagnosed incidentally with GBC with sonography.
Gallbladder carcinoma (GBC) is the most common malignancy of the biliary tract, with a reported incidence of 2.2 per 100 000 per year worldwide. 1 Early diagnosis is uncommon because of nonspecific symptoms until the disease has progressed into adjacent tissues. The median survival of diagnosed GBC cases is low, as reported at only 6 months. 1 The most common risk factor for GBC is the presence of gallstones with a reported incidence of 74% to 92% in affected patients. 2 Studies have shown other risk factors such as female sex, age, cigarette smoking, ethnic origin, and increased body mass.2,3 Many of these risk factors are linked to the prevalence of cholesterol gallstone disease. Multiparous females with elevated body mass have a predisposition to cholesterol gallstone formation. The proposed reason for this phenomenon is that female sex hormones increase secretion of cholesterol into the bile, thereby increasing gallstone production. The chronic irritation due to gallstones is thought to lead to dysplasia of the epithelium, which can lead to a carcinoma. 3 While the prevalence of GBC, in the United States, is relatively low, other geographic areas show a high rate of disease. Chile has one of the highest mortality rates in the world from GBC and it is reported as the second most frequent cause of death in women. 1 The normal gallbladder comprises four layers, with the surface layer being composed of a single layer of epithelium that has no submucosa. The GBCs are of epithelial origin and as high as 90% are histologically classified as adenocarcinomas. 2 This single muscular layer and lack of submucosa of the gallbladder are factors that contribute to the high rate of metastatic disease and tumor extension, beyond the gallbladder wall.2,3 Tumor extension beyond the gallbladder results in a poor prognosis with a 5-year survival rate, with advanced disease, of less than 5%. 3 Early diagnosis is usually incidental but leads to better patient outcomes. A carcinoma, confined to the gallbladder, can be treated with cholecystectomy and adjuvant therapy which results in a good prognosis, as well as an overall survival rate of approximately 88%. 1
Case Report
A 57-year-old male patient was referred to hepatology with reported acute hepatitis and continued elevated Liver Function Tests (LFTs), trending upward. Specifically, there was an elevation in aspartate aminotransferase (AST) at 115 U/L and alanine aminotransferase (ALT) at 88 U/L. Review of the hepatitis profile revealed that he did not have acute hepatitis, as his hepatitis A and hepatitis B antibodies were reactive. These antibodies were reported to be secondary to previous exposure or prior vaccination.
The patient had no family history of liver disease and was not an alcohol abuser. He was a patient with type II diabetes with hyperlipidemia and obesity (body mass index [BMI] 44). He had been prescribed atorvastatin, for his high cholesterol, but stopping taking it for 3 months, due to elevated LFTs. Despite this change in medication, the LFTs were still elevated and it was suggested he most likely had non-alcoholic fatty liver disease (NAFLD). Based on these findings, the patient was referred for a fibroscan (liver elastography) to determine the incidence and degree of hepatic steatosis. This fibroscan was deferred due to the coronavirus pandemic. The patient was ultimately referred for a routine abdominal sonogram, after the monthly lab report demonstrated no decrease in LFT results.
The patient reported to the ultrasound department approximately 1 month later, for an abdominal sonogram. The right upper quadrant of the abdomen was imaged in multiple longitudinal and transverse planes. Sonographic imaging of the liver demonstrated a heterogeneous echo pattern of the liver parenchyma, without evidence of a distinct mass. The liver also appeared hyperechoic with poor definition of the periportal fat planes and with an increased liver/kidney echogenicity ratio, compatible with hepatocellular disease. Normal flow was documented in the portal vein. The pancreas, common bile duct, and right kidney had a grossly normal sonographic appearance.
Focused interrogation of the gallbladder revealed an intraluminal, heterogeneous solid mass with internal vascularity, as demonstrated by color Doppler (see Figures 1 and 2). The solid lesion was identified in the fundus of the gallbladder and measured 3 cm × 4 cm × 2.5 cm (see Figure 3). Also noted was a hypoechoic area adjacent to the gallbladder, in the liver, suggesting fatty sparing or possible tumor extension into the liver (see Figure 4). The mass was described as highly suspicious for a GBC and a magnetic resonance imaging (MRI) of the abdomen, with contrast, was recommended for diagnostic confirmation.
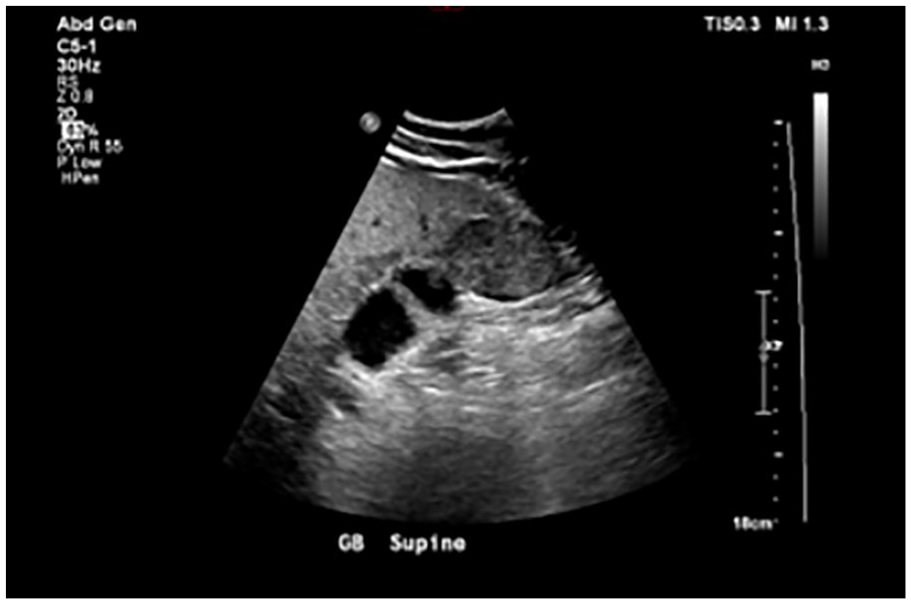
A longitudinal view of the gallbladder, demonstrating a heterogeneous solid mass in the fundus.
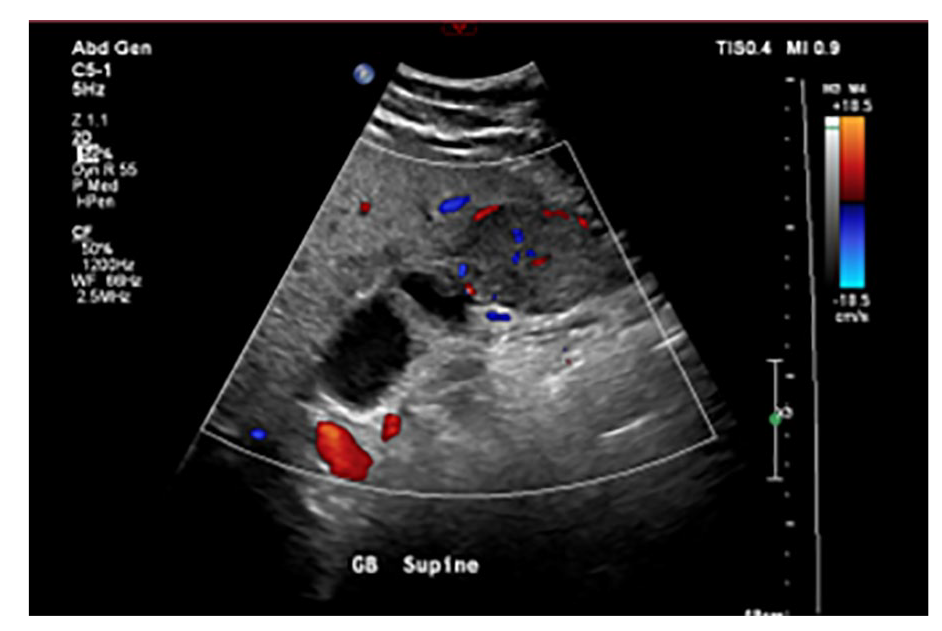
A longitudinal view of the gallbladder, with color Doppler, showing internal vascularity of the mass.
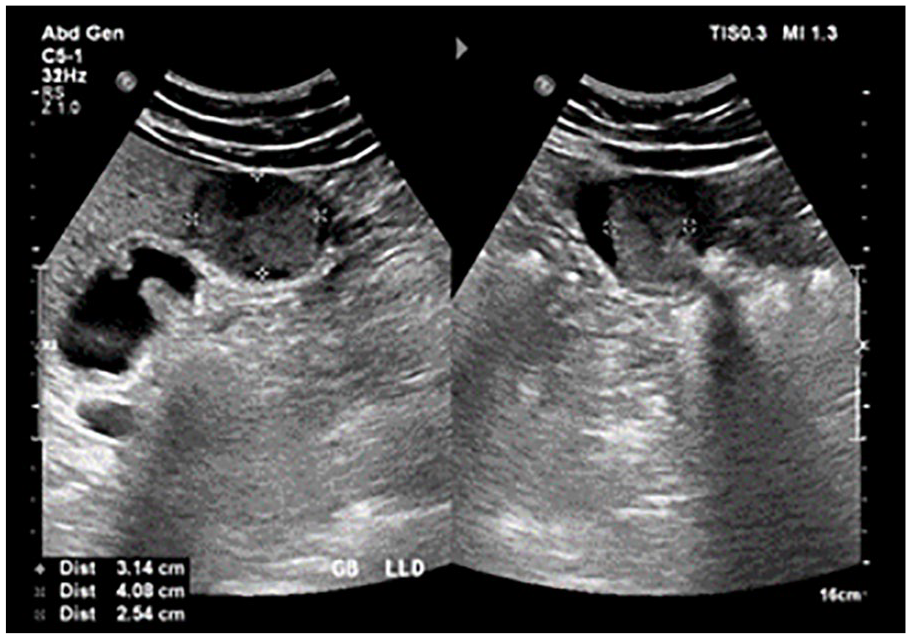
A longitudinal and transverse sonographic view of the gallbladder (GB) showing measurements of the suspicious mass.
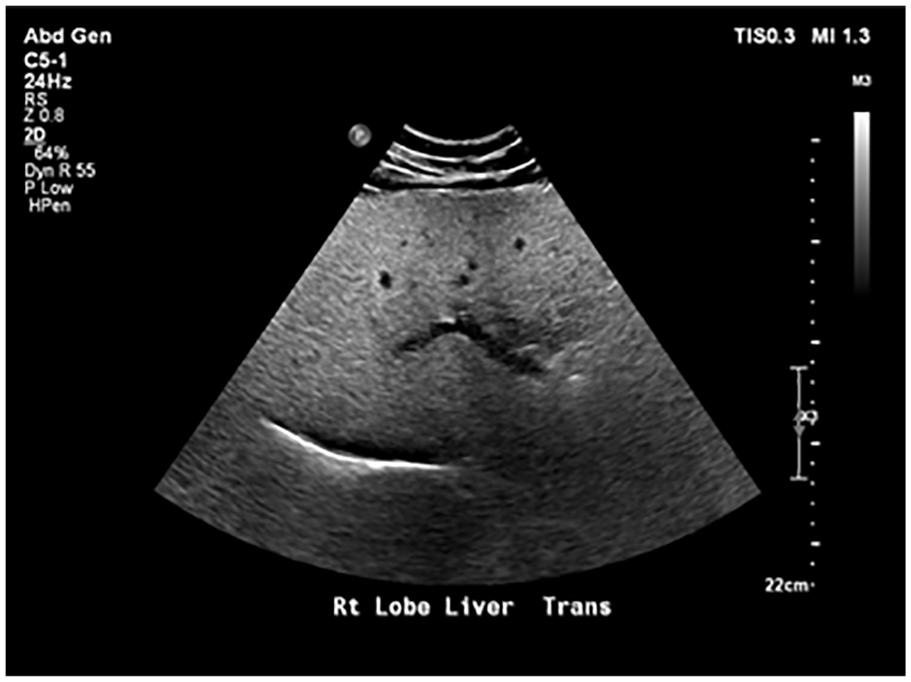
A transverse sonographic view of the liver showing hypoechoic area of possible tumor invasion.
The MRI, of the abdomen, confirmed a hyper-vascular multifocal gallbladder mass (see Figures 5 and 6) with local invasion, of the liver segments 4B and 5 (see Figure 7). The mass now measured 6.4 cm × 5.9 cm × 4.7 cm. No biliary ductal dilatation or enlarged lymph nodes were seen, as part of the MRI. Based on these imaging findings, the patient was referred for a surgical consultation.
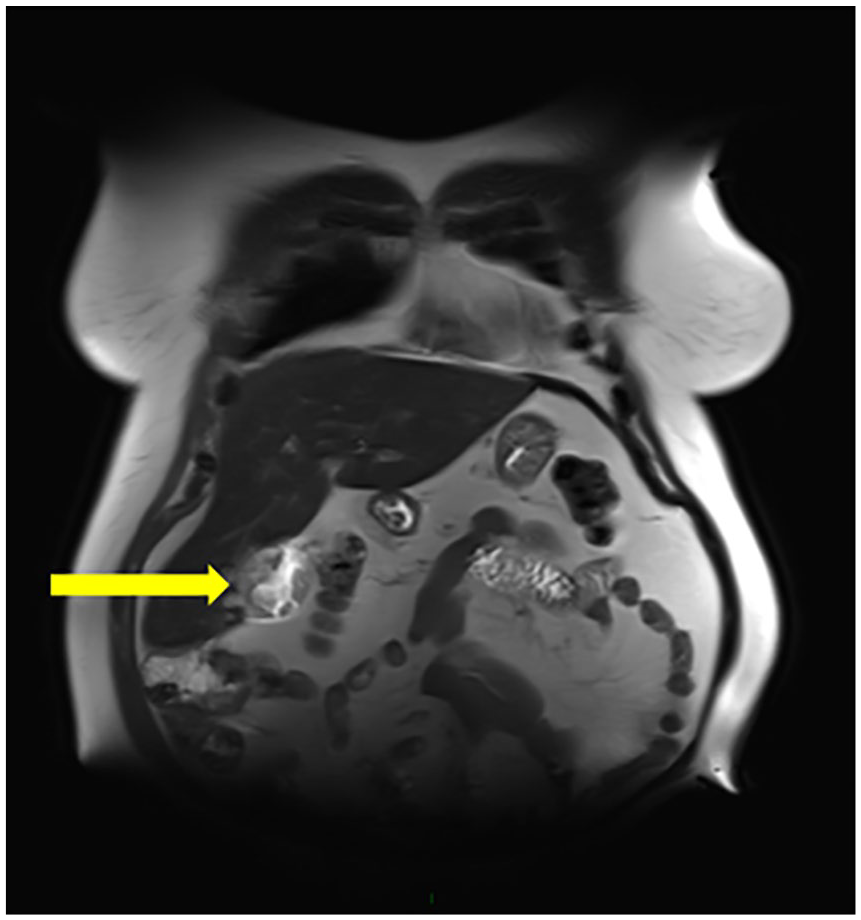
A magnetic resonance image of the abdomen with a coronal view that demonstrates a solid, vascular lesion in the gallbladder (GB) (denoted with a yellow arrow).
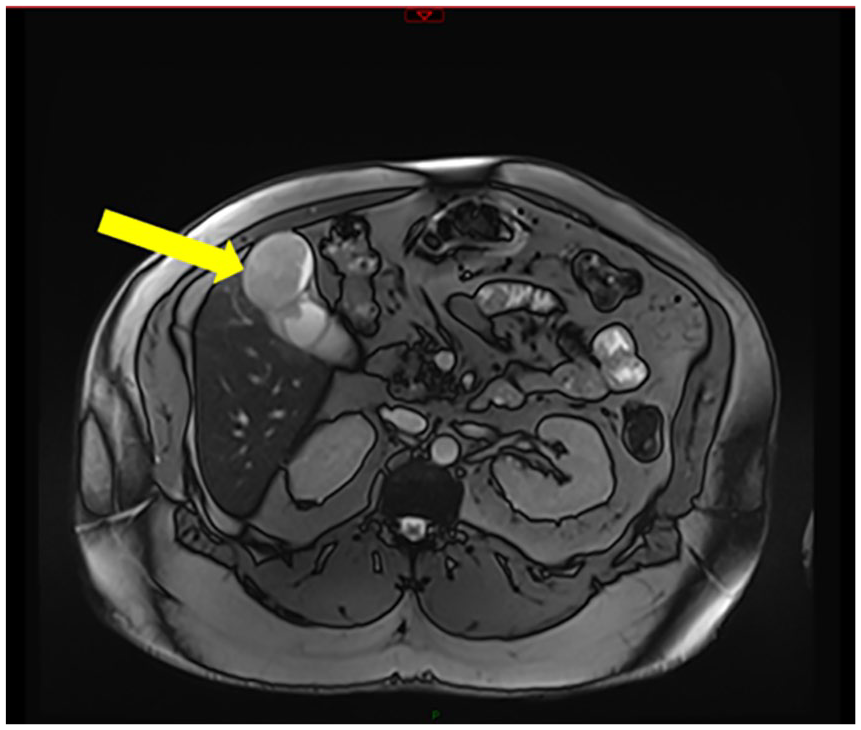
A magnetic resonance image of the abdomen with an axial view that shows a solid, vascular lesion in the gallbladder (GB) (denoted with a yellow arrow).
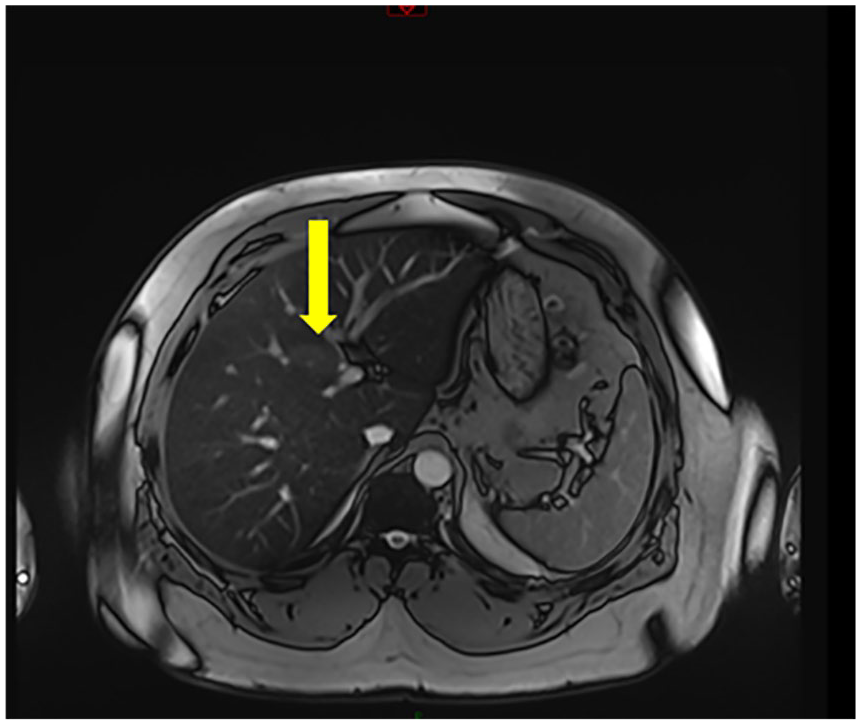
A magnetic resonance image of the abdomen with an axial view that demonstrates tumor extension into the 4B and 5 liver segments (denoted with a yellow arrow).
One month after the MRI, the patient underwent open radical cholecystectomy, with partial liver resection and portal lymphadenectomy. The clinical surgical assessment confirmed no evidence of nodal involvement and no evidence of distant metastasis. The patient remained hospitalized 3 days post-procedure but was able to ambulate the next day with well-controlled pain. He presented to the clinic 10 days later for postoperative staple removal and reported feeling great. He reported a good appetite and bowel function with no episodes of pain, nausea, or vomiting. The follow-up telehealth visit, 20 days post-procedure, also documented a self-report of feeling “fantastic” and no further complaints. The patient was then referred to oncology for consultation and was treated for 6 months of chemotherapy with capecitabine.
The patient returned for a follow-up computed tomogram (CT) at 15 months after surgery. Imaging identified subtle soft tissue thickening in segment 5, of the liver, along with enlarged porta hepatis and portacaval lymph nodes. The patient reported no physical symptoms despite the CT showing disease recurrence. The diagnosis of metastatic disease was confirmed by nodal biopsy, and the course of oncological treatment was changed to palliative chemotherapy with cisplatin and gemcitabine, until progression.
Discussion
There are five major disease stages of GBC. Stage 0 describes cancer in situ, which has not spread beyond the site. Stage I is denoted by a cancer that is confined to the inner layers. In stage II, cancer has spread to the outer layers. Stage III is gauged by a cancer that has spread to one or more organs, possibly lymph nodes. Stage IV is the most advanced stage and has spread to distant parts of the body, which is likely unresectable. The only curative treatment for GBC is cholecystectomy and is the preferred treatment for stage I tumors. Stage II and III tumors should be treated with removal of the gallbladder along with a possible limited hepatic resection and portal lymphadenectomy, depending on the area infiltrated. 4 Adjuvant chemotherapy and/or radiation is an option for high-risk patients. Palliative chemotherapy improves survival for patients with unresectable or metastatic disease. 1
The biggest factor in increasing the survival rate of patients with GBC is early detection. It is reported that only 24% of patients with early disease are treated. 4 Sonography is the preferred imaging modality for the gallbladder and is therefore the best option for diagnosing early-stage GBC. Most of the tumors (60%) originate in the fundus of the gallbladder, with another 30% of tumors found elsewhere. 2 Sonographic findings are most commonly a solid mass, wall thickening, or intraluminal polypoid masses. Carcinomas that present as solid tumors typically have irregular margins and a heterogeneous appearance as well as feeding vessels identified by color Doppler. 5 Stones are usually present, which appear as echogenic foci with acoustic shadowing. Shadowing from stones can reduce visibility of the gallbladder wall and can lead to a missed diagnosis. Moving the patient into alternate positions can ensure that all of the gallbladder has been visualized and imaged thoroughly. Wall thickening is a more challenging indicator of disease, as it may be subtle or nonspecific. It may present as diffuse or focal but retrospective studies have shown that 65% of patients had some form of wall thickening, in those confirmed cases. 4 Differentiating neoplastic polyps from non-neoplastic polyps is also not definitive by sonography, although size greater than 1 cm has proven to be a reliable predictor for malignancy. 5 The CT, with contrast enhancement, has proven to be valuable in confirming and staging the diagnosis, by identifying tissue invasion of the liver as well as lymph node involvement and distant metastasis. 2
Advances in contrast-enhanced ultrasonography (CEUS) are aiding in the differentiation of malignant and benign lesions in the gallbladder. Researchers have found that CEUS can help distinguish if the wall of the gallbladder is intact or discontinuous, which is a high predictor of malignancy and not easily seen with conventional sonography. Zhang et al 6 reported that these findings result in a high sensitivity rate of 0.82 and specificity rate of 0.93. Invasion of the surrounding tissues was more easily identified with the use of contrast as well as correctly differentiating between chronic cholecystitis and wall thickening due to GBC. A significant imaging characteristic in the setting of malignant wall thickening was shown as a discontinuous layer of the wall and was more evident with the use of CEUS. 6 A research study by Xie et al 7 also reported significant findings regarding the integrity of the gallbladder wall. Their research with CEUS correctly identified destruction of wall intactness in the cases of malignancy in all of the patients in their study. This study also reported on the washout time of contrast in lesions of the gallbladder. Contrast washout, within 35 seconds of administration, proved a high predictor of malignancy and was reported in 91% of GBC in that study. The hypo-enhancement of liver lesions in late phase is consistent with malignancy but this same pattern cannot be applied to GBC. According to Xie et al, 7 the malignant lesions as well as the benign lesions showed hypo-enhancement in the late phase. While all of these studies show promising results, more research is needed to determine the true significance of CEUS in accurately diagnosing GBC.
Conclusion
This case study provides an example of how early detection of GBC can lead to improved overall survival rates for patients. Although the current case study patient did have disease recurrence, his survival thus far has been more than 18 months. This is three times the reported median survival of 6 months. He has remained asymptomatic and continues to report a good quality of life.
When GBC is found before extensive tissue invasion, it becomes a more treatable disease. Sonography plays a vital front-line role in gallbladder imaging and can ultimately improve patient outcomes. Increased awareness of and careful attention to the imaging characteristics of GBC can assist the sonographer and the interpreting radiologist achieve an early and accurate diagnosis.
Footnotes
Acknowledgements
The authors would like to thank the sonographers from the Central Arkansas Veterans Healthcare System for providing the images and case information for this paper.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
