Abstract
Emphysematous cholecystitis (EC) is a rare but deadly variant of acute cholecystitis noted by the presence of gas in the gallbladder lumen or wall. This gas, emitted by bacteria, is thought to result from gallbladder ischemia caused by vascular compromise with subsequent bacterial invasion. The two most common bacteria responsible for EC are Clostridia perfringens and Escherichia coli. Symptoms of EC are similar, although often less overt, compared to those of acute cholecystitis (AC). Sonography and radiography are common methods of diagnosing EC, but computed tomography (CT) carries the highest diagnostic accuracy rate. Laparoscopic cholecystectomy is the most common method of treating EC, although conservative therapy and hyperbaric oxygen therapy have also been effective in some cases.
Emphysematous cholecystitis (EC) is an uncommon yet potentially fatal variant of acute cholecystitis, characterized sonographically by the presence of gas in the gallbladder wall.1,2 The first mentions of EC in the literature were in 1896 by Welch and Flexner. 3 Five years later, Stolz further described the presence of gas in the gallbladder after performing autopsies on three patients.3,4 In the early 1900s, Hegner described the radiographic features of EC, while Pene postulated that perhaps instead of being a distinct pathology, EC was possibly a variant of acute cholecystitis (AC).1,3
Etiology
Emphysematous cholecystitis, also referred to as acute gaseous cholecystitis, aerocholecystitis, clostridial cholecystitis, pneumocholecystitis, or gas gangrene of the gallbladder, has been suggested to be an uncommon outcome of acute cholecystitis, occurring in only 1% to 4% of cases.1,3 It has been postulated that following an initial bout of acute cholecystitis, the disease progresses in a manner that results in gallbladder ischemia, with the subsequent invasion of gas-producing bacteria within the gallbladder wall.1,5 Ischemia is thought to result from primary vascular compromise to the gallbladder, yielding necrosis and an environment where bacteria can proliferate. 6 Once housed within the gallbladder wall, the bacteria produce gas as they develop and replicate. However, other means of gas or air accumulation in the gallbladder may occur, including an incompetent sphincter of Oddi, tissue infarction, drugs, or an enterobiliary fistula secondary to abdominal trauma. 7 For this reason, an understanding of the possible mechanisms of the disease is important for the imaging professional.
Compromised or inadequate vascular supply to the gallbladder is often secondary to other conditions such as atherosclerosis, arterial embolism, vasculitis, or systemic hypotension/hypoperfusion. 8 Anatomic pathology studies of EC show a high incidence of endarteritis obliterans affecting the cystic artery, with the inflammation of the intimal layer of the vessel causing a reduction of the lumen with ultimate vascular occlusion.3,9 With the occlusion of the cystic artery, ischemic damage occurs to the gallbladder, bacteria opportunistically invade, and subsequent replication of gas-producing organisms begins to accumulate within the gallbladder wall. 3 Systemic hypoperfusion and transhepatic arterial embolization that result in embolization of the cystic artery have also been shown in several cases of EC following emergency cardiopulmonary resuscitation. 3
EC has also been reported after endoscopic retrograde cholangiopancreatography (ERCP), even in the absence of stones. 10 An ERCP is a radiographic imaging examination used to evaluate the liver, biliary tree, and pancreas that requires insertion of the endoscope via the patient’s esophagus. The endoscope, under video guidance, is maneuvered through the stomach and into the proximal portion of the duodenum, where the sphincter of Oddi is identified. Radiographic contrast is allowed to perfuse the biliary tree for diagnostic analysis, making possible the instillation of air and perhaps bacteria within the biliary tree. Others have reported the onset of EC after angiographic procedures. 6
Bacterial Invasion
The two most common microorganisms cultured in EC are Clostridium perfringens and Escherichia coli, although others such as Proteus vulgaris, Aerobacter aerogenes, Staphylococcus, Streptococcus, Klebsiella, and Bacteroides fragilis have been implicated.11,12 All of these organisms have been shown to be capable of releasing gas during the process of decomposing glucose under anaerobic conditions. 4
C. perfringens, which proliferates in places of low oxygen saturation, is a Gram-positive, anaerobic bacterium responsible for histotoxic and gastrointestinal diseases in both humans and animals.12,13 Ranging from types A through E based on their lethal toxin-producing ability, C. perfringens type A has been identified to cause food poisoning and non-food-borne gastrointestinal diseases (NFBGIDs). 13 C. perfringens is also responsible for 50% of biliary tract infections. 14 Decreased pH levels from acidic tissue ischemia, combined with inflammation from an occluded or compromised cystic artery, create a favorable environment for bacteria to migrate from the intestinal tract to the biliary tract and grow in bile. 7 It has been demonstrated that C. perfringens can migrate from the duodenum to the biliary tree and thus be found in bile cultures during pathologic analyses. 12 Other sources of C. perfringens include the lungs, pelvis, and decubitus ulcers. 15 Risk factors associated with clostridial infections include diabetic foot, uremia, and intestinal disease. 14 Therefore, besides EC, C. perfringens is often suspected as the primary causative agent of tubo-ovarian and liver abscesses, necrotizing enterocolitis, and discitis. 15
Another organism commonly found at the site of EC is E. coli, a gram-negative bacteria. 16 This bacterium is divided into two categories: intestinal pathogens, which cause diarrhea, and extraintestinal E. coli, which causes a variety of infections, including urinary tract infections, meningitis, and septicemia. 17 E. coli has been cited as the most common causative agent for acute renal disease as well. 16 It is commonly found in the gastrointestinal tract and, with optimal conditions, can also migrate into the biliary tract. 12
Clinical Findings
Clinical findings of EC may be indistinguishable from those of AC in some patients. 10 Patients with AC often present with right upper quadrant pain, fever and vomiting, notable tenderness over the gallbladder, and abdominal pain that may increase after a fatty meal. 18 Focal tenderness over the gallbladder with transducer pressure, the sonographic Murphy’s sign, may also be present in patients with AC.
Similarly, patients with EC can present with right upper quadrant pain, fever, nausea, and vomiting. 19 The literature does not comment on the presence or absence of Murphy’s sign for EC. Both diseases cause an elevation in white blood cell counts and bilirubin, although AC also causes elevated aminotransferase, alkaline phosphate, and amylase.7,18 EC has been noted to be associated with an increase in serum glucose. 7 Although EC has been mentioned as a variant of AC, some clinical findings and statistics do differ from the latter (Table 1).1,5,18–23 One sequela of EC that is of primary concern is gallbladder perforation, which occurs much more often in EC patients compared with AC patients and thus yields a higher concern for immediate medical or surgical intervention. 20 Gallbladder perforation from undifferentiated EC carries a significant mortality and morbidity rate, resulting from nonspecific clinical and imaging findings that can result in a late diagnosis. 20
There are several known predisposing conditions for EC. Recurrent gallstones and cholecystitis have been shown to increase the risk of developing EC. 24 Patients with uncontrolled diabetes mellitus present a much higher risk of developing EC 25 ; 50% of patients with EC have a history of diabetes mellitus compared with only 10%-15% of patients with AC. 26 This is secondary to the ischemic environments and increased possibility of infection related to the disease. 3 Diabetic neuropathy, which can cause insensitivity to lower limb trauma, incomplete urination, and fecal incontinence, creates multiple entryways for infectious micro-organisms. 3 Decreased ability to destroy microorganisms, altered immunity, and impaired antioxidant systems involved in bacterial activity all contribute to the increased risk of a diabetic patient developing EC.27,28 Hyperglycemia also increases the risk of developing EC because of its direct correlation to microvascular angiopathy, which is predictive of soft tissue and organ infections in diabetic patients. 29 Diabetic arteriopathy leads to distal tissue and organ parenchymal ischemia, thus increasing the possibility of bacterial proliferation. 3
Sonographic and Other Imaging Findings
EC is a disease that has begun to be diagnosed more frequently, primarily because of the increased use of sonography to diagnose biliary disorders. 10 The frequency of discovery has revealed a broad spectrum of EC, ranging from mild to severe. 10 Abdominal sonography and radiography have up to 95% sensitivity for diagnosing EC 2 ; however, diagnosis of EC with sonography is dependent on the amount and location of gas in the gallbladder. 12
While AC and EC share many similarities, the sonographic appearance of the two does vary, making a vigilant sonographer the likely first line of discovery. Sonographic findings of AC include a combination of wall thickening, pericholecystic fluid, enlarged cystic artery, and hyperemia noted with color Doppler imaging analysis. 21 Sonographic diagnosis of EC is made by observing gas in the gallbladder wall, lumen, or pericholecystic fluid. 7 EC is divided into three stages, based on the amount of air distributed within the biliary tract (Table 2). 11
Stages of Emphysematous Cholecystitis.

Gas bubbles can be detected with imaging 72 hours after the onset of symptoms, and bile cultures will yield positive growth of bacteria. 1 The diagnosis of EC with sonography is often dependent on the amount and location of the gas. 12 A smaller amount of gas is depicted as echogenic foci with reverberation artifact, while a greater amount of gas produces a wide band of acoustic shadowing within the gallbladder fossa (Figures 1 and 2). 11 Multiple echogenic foci, or gas bubbles, can also be visualized rising in the gallbladder lumen from the dependent portion to the independent portion. 4 This is termed the “effervescent” or “champagne” sign, since the bubbles rise similarly to those in a glass of champagne.7,10
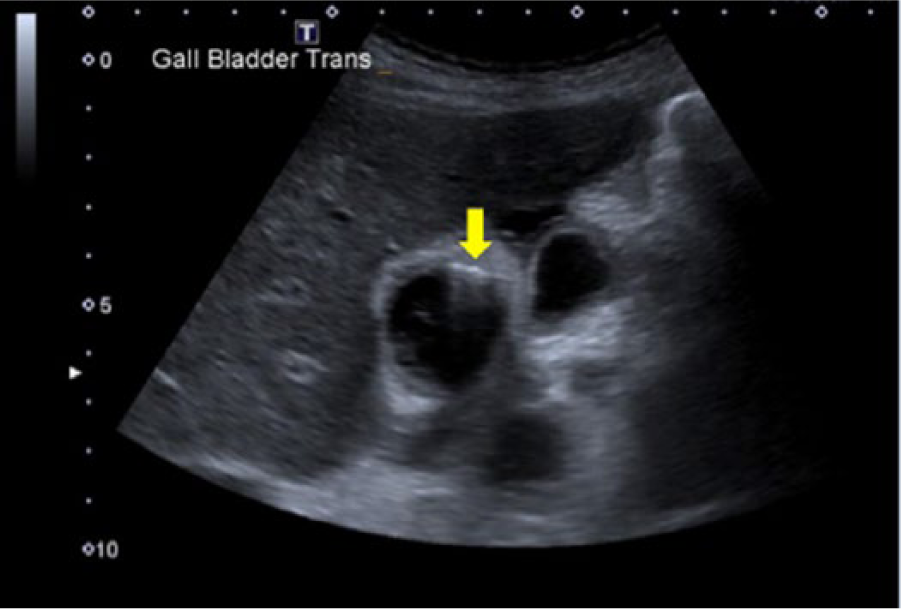
Transverse gray-scale image of the gallbladder with a small amount of air that produces a reverberation artifact (arrow). Some pericholecystic fluid can also be noted. (Reprinted with permission from Hacking C. Emphysematous cholecystitis. Radiopaedia.org. http://radiopaedia.org/cases/emphysematous-cholecystitis-2.)
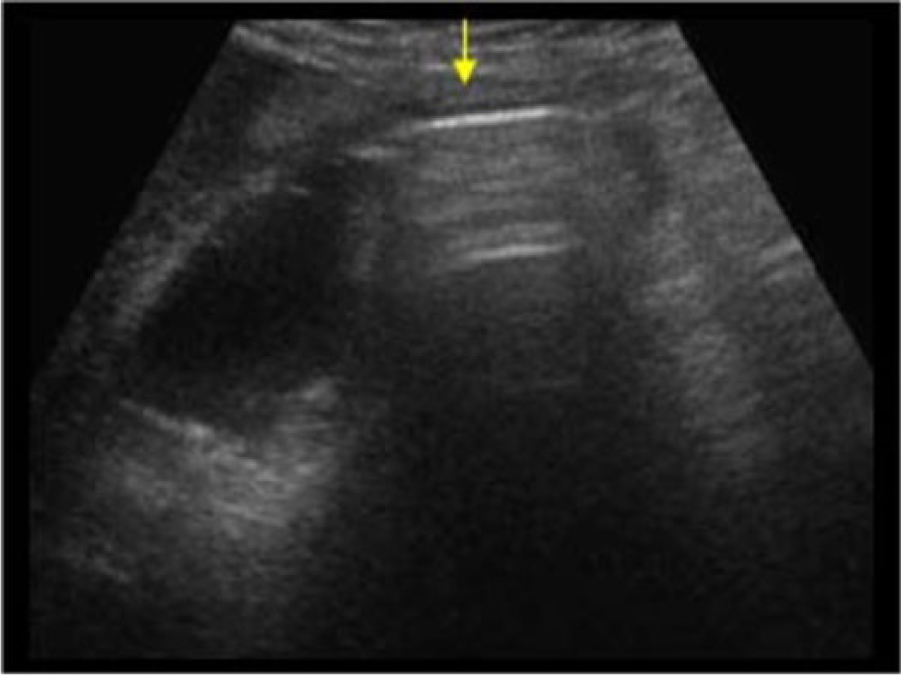
Longitudinal gray-scale image of the gallbladder showing a wide band of acoustic shadowing/reverberation in the lumen (arrow). (Reprinted with permission from Geeertsma TSA. Emphysematous cholecystitis. Ultrasoundcases.info. http://ultrasoundcases.info/Slide-View.aspx?cat=164&case=4139.)
Two types of fluid collections can be present with EC. Type 1 fluid collections, which are not usually indicative of gallbladder perforation, present as a thin, anechoic crescent-shaped fluid collection adjacent to the gallbladder wall and are often visualized alongside the “champagne” sign. 22 Type 2 fluid collections, which always present with gallbladder wall perforation and abscess formation, are large, round, or irregularly shaped and complex. 22 These fluid collections may also be seen without imaging evidence of a discernible gallbladder. 22
Various gallbladder conditions can obscure the gallbladder or give faulty diagnoses during imaging. False negatives with sonography can result from air in the gallbladder or pericholecystic air obscuring the gallbladder by mimicking bowel loops. This can be prevented by scanning through the intercostal spaces and imaging using multiple changes of patient position.11,25 Multiple gallstones in the lumen or a porcelain gallbladder can make visualization of the gallbladder problematic. However, M-mode has been demonstrated to improve detection of the subtle movement of air pockets with probe compression. 11 Adenomyomatosis, which produces reverberation artifact within the gallbladder wall, can also mimic gas from EC, but adenomyomatosis tends to produce a specific V-shaped comet tail reverberation artifact, while EC often presents as a linear-shaped dirty shadow with mobility (Figures 3 and 4).11,30
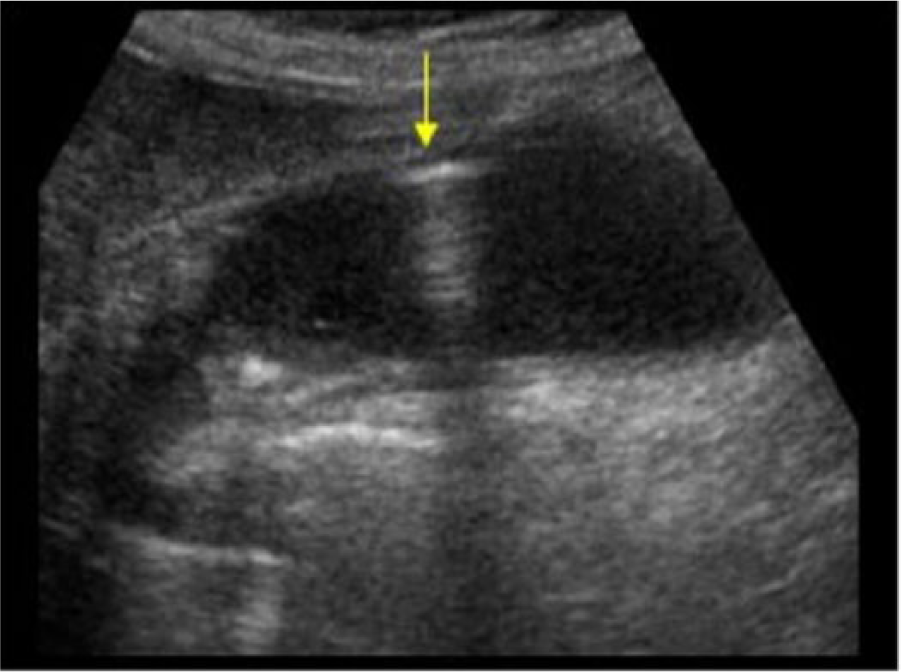
Longitudinal gray-scale image of the gallbladder showing air in the gallbladder lumen (arrow). Reverberation and acoustic shadowing can be seen posterior to the echogenic linear structure. A stone obstructing the gallbladder neck can also be noted. (Reprinted with permission from Geertsma TSA. Emphysematous cholecystitis. Ultrasoundcases.info. http://ultrasoundcases.info/Slide-View.aspx?cat=164&case=4139.)
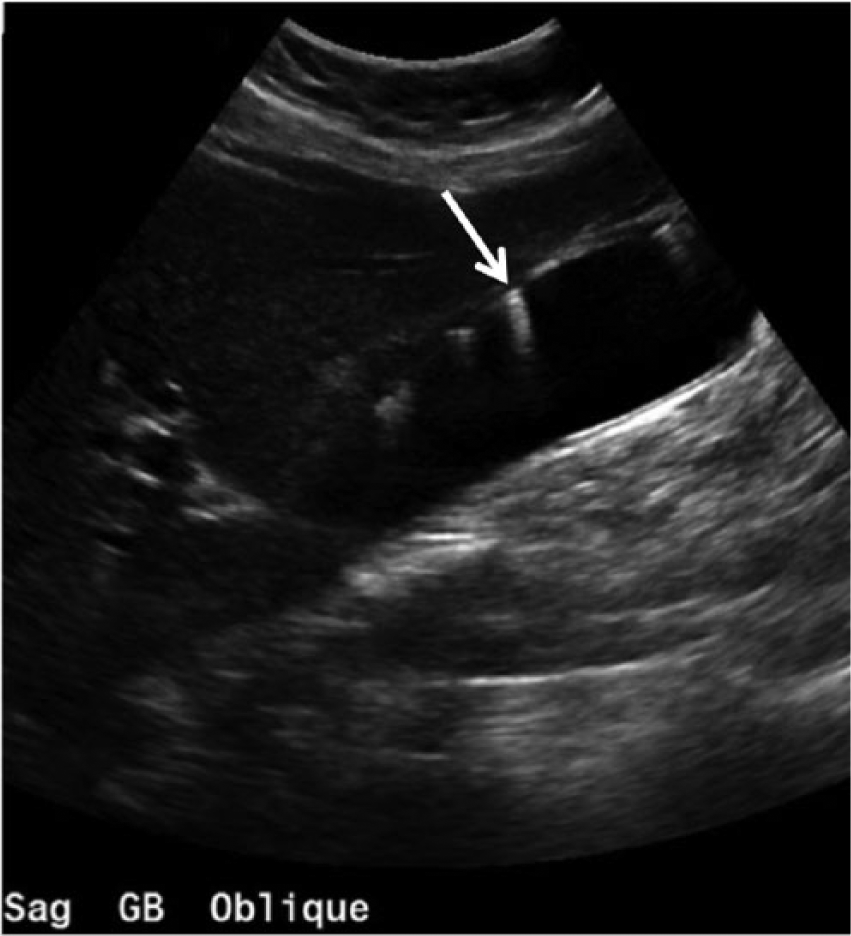
Longitudinal gray-scale image of the gallbladder with adenomyomatosis (cholesterol crystals) noted on the anterior gallbladder wall (arrow). (Reprinted with permission from Gaillard F. Adenomyomatosis. Radiopaedia.org. http://radiopaedia.org/cases/adenomyomatosis-4.)
Computed tomography (CT), with nearly 100% sensitivity, is precise for detecting EC due to its ability to detect the exact location of gas in the gallbladder wall, gallbladder lumen, or biliary tract with high specificity (Figure 5).4,5,12 Furthermore, complications of EC such as pericholecystic inflammatory changes, abscess formation, perfusion alterations, and miniscule amounts of intraperitoneal free air are readily detected with CT. 4 CT also has the ability to differentiate between EC, cholecystoenteric fistula, gallbladder lipomatosis, and duodenal perforation. 4 However, because of the general ambiguity regarding EC symptoms, CT is not typically ordered for patients presenting with abdominal pain, leaving sonography as the most common imaging modality employed for biliary tree analysis. 12 For this reason, sonographers should have a thorough understanding of both the clinical and sonographic findings of EC.
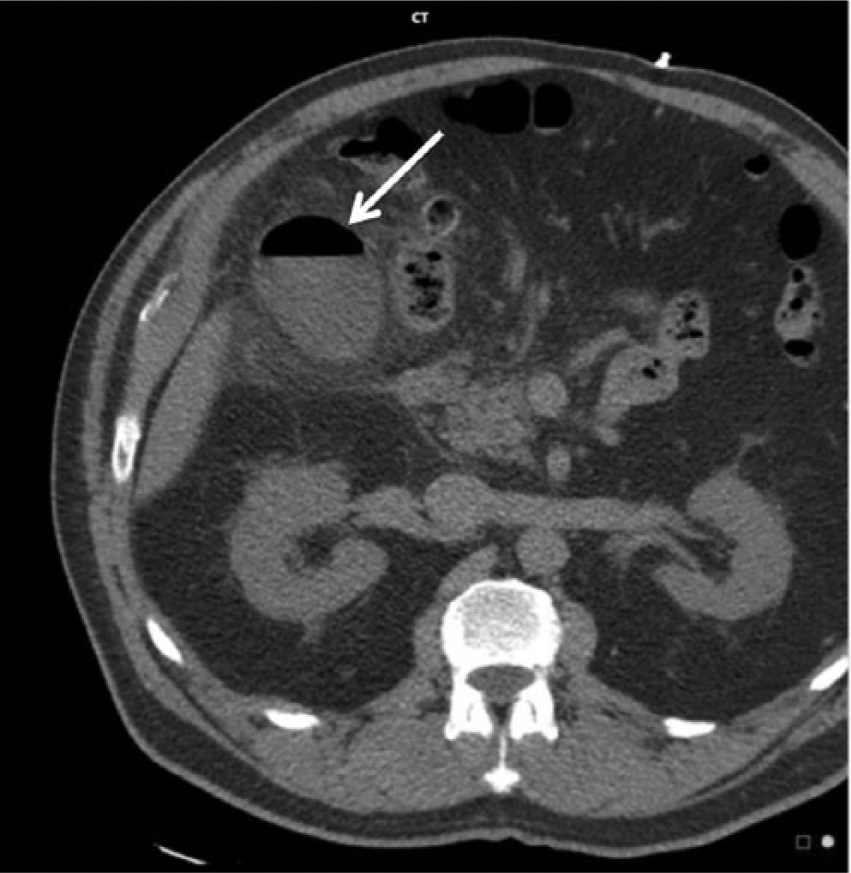
Computed tomography axial noncontrast view of the abdomen. Gas is noted within the wall of the gallbladder with an air/fluid level in the lumen (arrow). (Reprinted with permission from O’Donnell C. Emphysematous cholecystitis with local perforation. Radiopaedia.org. http://radiopaedia.org/cases/emphysematous-cholecystitis-with-local-perforation.)
A plain abdominal radiograph, which carries a 95% accuracy rate, can also be used to diagnose EC.2,31 A supine, erect, or decubitus radiograph of the abdomen will demonstrate one or more round bubbles or a pear-shaped lucency in the right upper quadrant (Figure 6). 32 Abdominal radiography can depict a gaseous halo around the gallbladder and gas-fluid level in the gallbladder, both of which are suspicious findings indicating the diagnosis of EC. 31
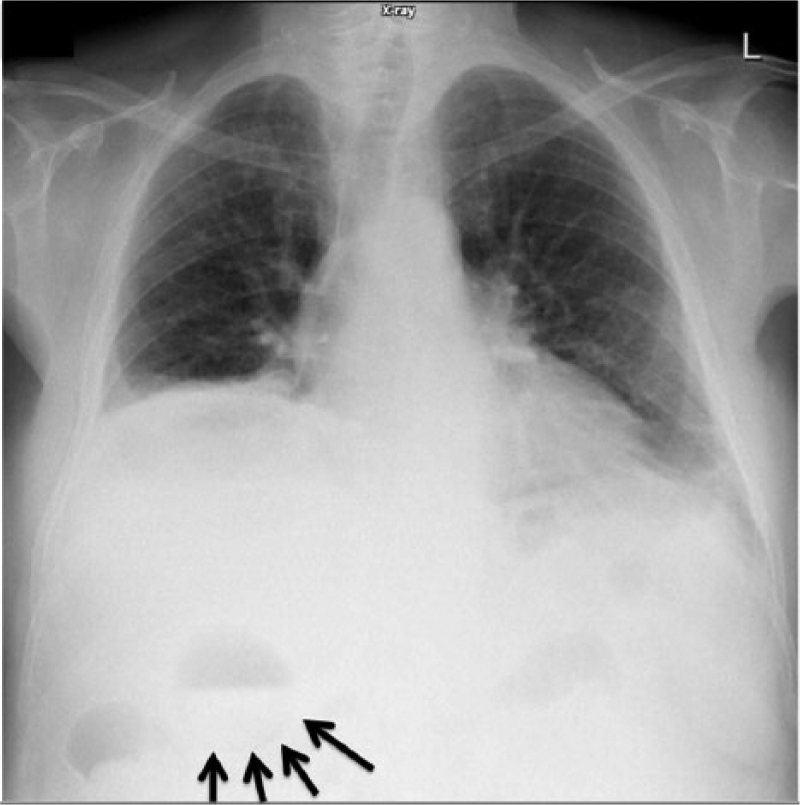
Upright chest radiograph showing, in the right upper quadrant, an air-fluid level with a curvilinear lucency visible around the fluid component (arrows). (Reprinted with permission from Gerstenmaier JF. Emphysematous cholecystitis. Radiopaedia.org. http://radiopaedia.org/cases/emphysematous-cholecystitis.)
Magnetic resonance imaging (MRI) also is accurate in the diagnosis of EC. MRI can differentiate EC from adenomyomatosis and provide detail regarding intraluminal gas and intramural necrosis.11,32 The appearance of gas in the gallbladder lumen and wall is that of signal void areas. 33 More specifically, intraluminal gas comprises various floating signal void bubbles in the upper dependent portion of the gallbladder lumen and/or extrahepatic bile duct. 33 Gallstones, when present, usually rest in the lower dependent portion of the gallbladder on MRI. 33
Prognosis and Treatment
A prompt diagnosis is essential to minimize the morbidity and mortality rates associated with EC. 34 Although the symptoms are nonspecific, a patient’s condition can worsen significantly from complications of perforation or gangrene. 7 Since EC has a high risk of perforation and gangrene, systemic sepsis is more likely in EC than in a nonemphysematous variants of cholecystitis. 6
Treatment for EC includes laparoscopic or open cholecystectomy, percutaneous cholecystostomy, conservative therapy, or hyperbaric oxygen therapy.2,7,12 Laparoscopic cholecystectomy is considered a safe emergency procedure for EC, but it is usually performed only when the patient is hemodynamically stable. 2 Advanced research has allowed for a minimally invasive approach for cholecystectomy while adhering to fundamental surgical principles used in open surgery. 6 However, some researchers claim that laparoscopic cholecystectomy is not preferable due to the instability of the gallbladder wall, and thus it should only be executed when other intervention is futile.1,10 A critically ill, unstable, or very elderly patient with EC can undergo percutaneous drainage of the gallbladder if gallstones are not present.1,7 Cholecystostomy can also be attempted to temporarily control sepsis. 6
Milder EC may be treated with other nonsurgical options. 10 Conservative therapy provides the patient with intravenous fluids, antibiotics, and analgesia to combat the disease and has proven successful in some cases. 12 The antibiotics, however, must be sensitive to anaerobes and enteric Gram-negative and Gram-positive organisms. 7 If antimicrobial therapy does not provide results within 72 to 96 hours, cholecystectomy is often suggested. 34
Conclusion
EC is an uncommon variant of AC, found more often in diabetic patients, that has the potential to be fatal. Because symptoms of EC are similar to those of AC, patients are often referred to the sonography department for initial diagnosis, thus placing the sonographer on the frontlines of diagnosis. For this reason, the sonographer must be capable of acquiring a thorough clinical history and correlating that information with sonographic findings. An incorrect diagnosis can give way to delayed and/or suboptimal treatment, with a consequential degradation of patient health. The suspicion of EC with sonography will allow further imaging and testing to take place, thus prospectively leading to a quicker diagnosis and subsequent immediate life-saving intervention.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.

