Abstract
Gallbladder carcinoma is rare and difficult to detect in the early stages of the disease process, due to lack of symptoms. Sonography is typically the first modality of choice for assessing gallbladder pathology due to its high sensitivity, portability, real-time imaging capability, and non-ionizing technique. Conventional gray-scale and color Doppler sonographic imaging may be ambiguous for diagnosing solid tumors, such as gallbladder carcinoma. In this case, gallbladder carcinoma was definitively diagnosed utilizing contrast-enhanced ultrasound, allowing for quick patient treatment options and an optimal surgical outcome.
Keywords
Adenocarcinoma of the gallbladder (GB), also known as gallbladder carcinoma (GBC), is the most common biliary tract malignancy,1,2 and the fifth most common malignancy, in the gastrointestinal tract.,3,4 GBC accounts for 80% to 95% of biliary tract cancers.,5,6 The outcome of GBC is considered poor, with the overall 5-year survival rate less than 5%.7,8 Worldwide, GBC affects over 140,000 patients and results in greater than 100,000 deaths each year. 9 Diagnosis of GBC is challenging in the early stages due to nonspecific or lack of patient symptoms and the highly invasive nature of the disease. 10 The only potential curative option is surgical resection; however, the curative resection rate remains low at 22% to 38%, due to locally advanced disease at time of resection.11,12 If an early stage GBC diagnosis is made, a 5-year survival rate of 75% can be achieved if adjuvant therapy is performed. 8
GBC is rare in the Western world including the United States, United Kingdom, Canada, Australia, and New Zealand. The peak incidence is in the sixth to seventh decades of life and is three to five times more prevalent in the female population. 13 Risk factors for GBC include obesity, metabolic syndrome, diabetes mellitus, gallstone disease, history of chronic cholecystitis, choledochal cysts, anomalous pancreaticobiliary duct junctions, and polyps greater than 1 cm in size.13,14 An estimated 22% of patients with a “porcelain GB,” will develop GBC. 13 Patients with gallstones measuring larger than 3 cm are associated with a greater than tenfold increased risk of developing cancer, compared to patients with smaller gallstones.15,16 The following case report exhibits the limitations of conventional gray-scale and color Doppler sonography in the diagnosis of GBC, and presents an innovative method for making the diagnosis using contrast-enhanced ultrasound (CEUS), when gray-scale and color Doppler sonographic imaging is uncertain.
Case Report
A 91-year-old Caucasian female was referred to the palliative care homebound program for medically complex history and a recent fall. Past medical history was significant for diabetes mellitus type 2, chronic right heart failure, and chronic kidney disease stage 4. The patient developed decreased appetite and increased fatigue in recent months. On physical exam, the abdomen appeared slightly rounded and demonstrated no rebound tenderness or guarding pain. Initial laboratory data showed an elevated alkaline phosphatase (ALP) of 354 U/L (normal range 35–104 U/L), alanine aminotransferase (ALT) of 232 U/L (normal range 7–45 U/L), and aspartate aminotransferase (AST) of 71 U/L (normal range 8–43 U/L). Bilirubin remained within normal limits of 0.8 mg/dL (normal <= 1.2 mg/dL). The medical care team ordered an outpatient abdominal sonogram, due to the elevation of liver enzymes and to possibly detect cholangitis or hepatic steatosis.
Gray-scale sonography (LOGIQ E9, GE Health Care) using a 6 MHz curvilinear transducer demonstrated GB cholelithiasis, including a 3.5 cm gallstone, and bulky tumefactive sludge, throughout the lumen (Figures 1 and 2). A subtle retraction of the GB wall adjacent to a 4 cm component of presumed sludge was noted, where an underlying neoplasm could not be excluded. Scattered color Doppler was visualized throughout but was felt to be artifact (Figure 3). Given the patient’s medical history of renal insufficiency, CEUS of the GB was recommended. Intravenous (IV) iodinated contrast agents used commonly in computed tomography (CT) examinations are contraindicated, when a patient has significant renal insufficiency. The contrast agents may further impair renal function in patients already suffering from underlying renal insufficiency. CEUS can be used safely as an alternative to iodinated contrast in these patients. When the patient returned for the advanced sonographic imaging, additional gray-scale images were obtained of the GB (Figure 4). On this examination, the sonographer demonstrated arterialized blood flow within the suspicious area using spectral Doppler technique (Figure 5). For better demarcation and to confirm the suspected gray-scale finding of solid neoplasm, CEUS was performed to assess for a focal, enhancing GB tumor. 2.4 mL of sulfur hexafluoride lipid-type A microspheres (Lumason, Bracco Diagnostics Inc.) were injected, as off label use. Prior to the administration of contrast, the sonographer located the optimal position for visualization of previously presumed GB sludge versus a solid neoplasm. The patient was pre-screened for any contraindications for contrast, namely a prior allergic reaction. A 20-gauge IV catheter was accessed in the left antecubital fossa in the examination room, which was previously placed by the radiology IV technologist. A 20-22-gauge needle is preferred as a smaller gauge potentially destroys the microbubbles. This IV access location is optimal as the patient’s right arm is inaccessible due to sonographer scanning technique and patient position. An additional sonographer intravenously injected 2.4 mL of contrast slowly over 2 seconds followed by a 10 mL saline flush, which clears the IV tubing outside of the patient and ensures delivery of the contrast agent into the IV system. It is important injections are not performed too rapidly to prevent microbubble rupture. At this institution, the scope of practice for sonographers includes IV contrast agent and saline injections.
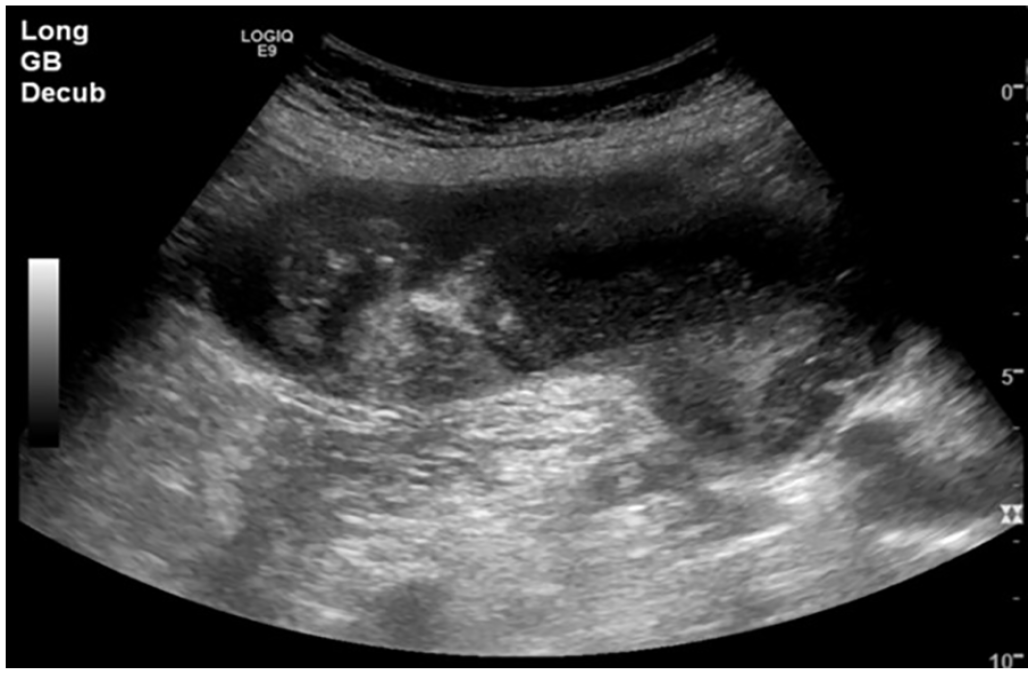
Longitudinal gray-scale sonographic image of the gallbladder demonstrates tumefactive sludge throughout the lumen.
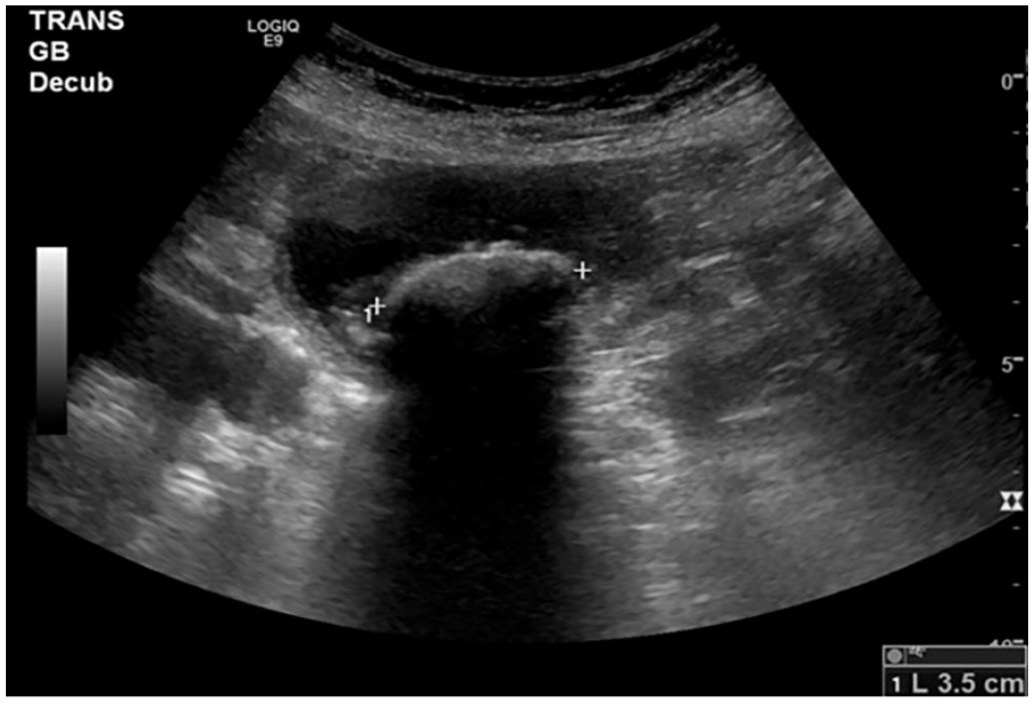
Transverse gray-scale sonographic image of the gallbladder demonstrates a large gallstone measuring 3.5 cm.
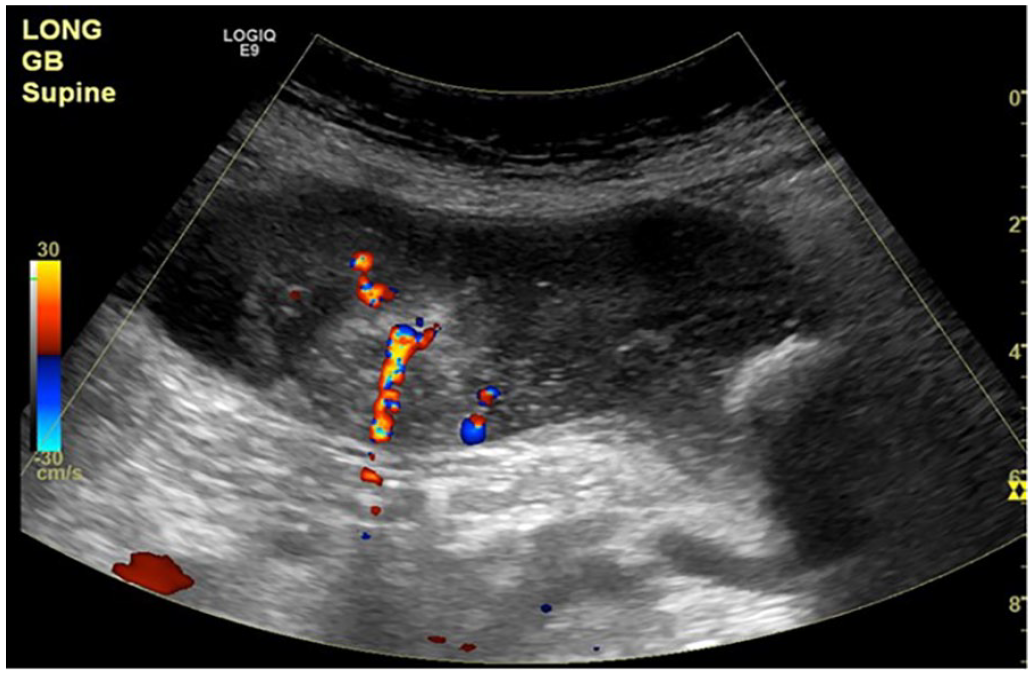
Longitudinal sonographic image identifies scattered color Doppler flow appearing as artifact.
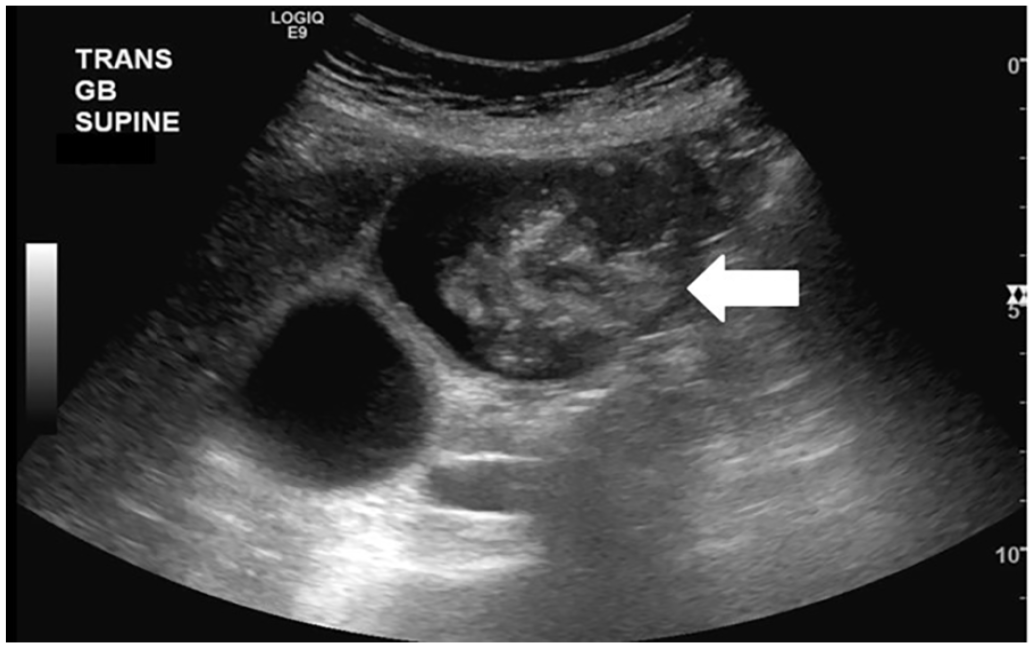
Transverse gray-scale sonographic image of the gallbladder detects the suspicious area of concern (white arrow).
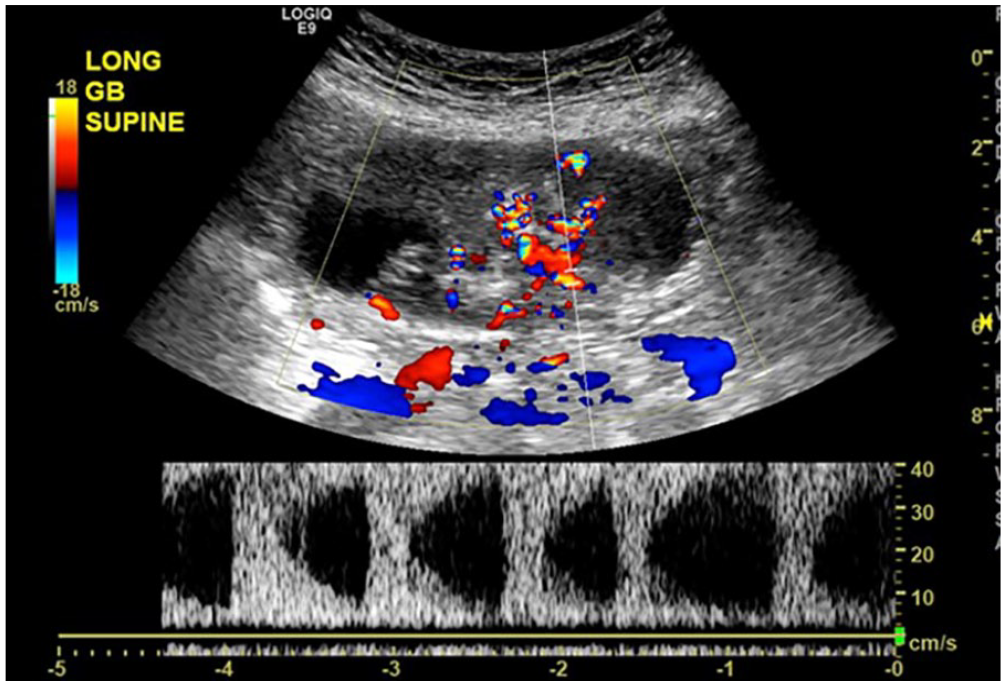
Longitudinal spectral Doppler sonographic image of the gallbladder demonstrates arterialized blood flow within the suspicious area of concern.
Shortly following the injection, the sonographers and the radiologist observed the enhancement pattern of the GB, dynamically. CEUS images demonstrated rapid enhancement of the GB sludge-like area confirming the gray-scale suspicion of GB neoplasm concerning for primary GB adenocarcinoma. (Figures 6–8) This intraluminal polypoid mass measured 5.0 cm x 3.0 cm. There was no definite extension of the neoplasm beyond the GB wall. The sonographer removed and assessed the IV site and catheter that was placed and completed all the necessary documentation within the patient’s medical record.
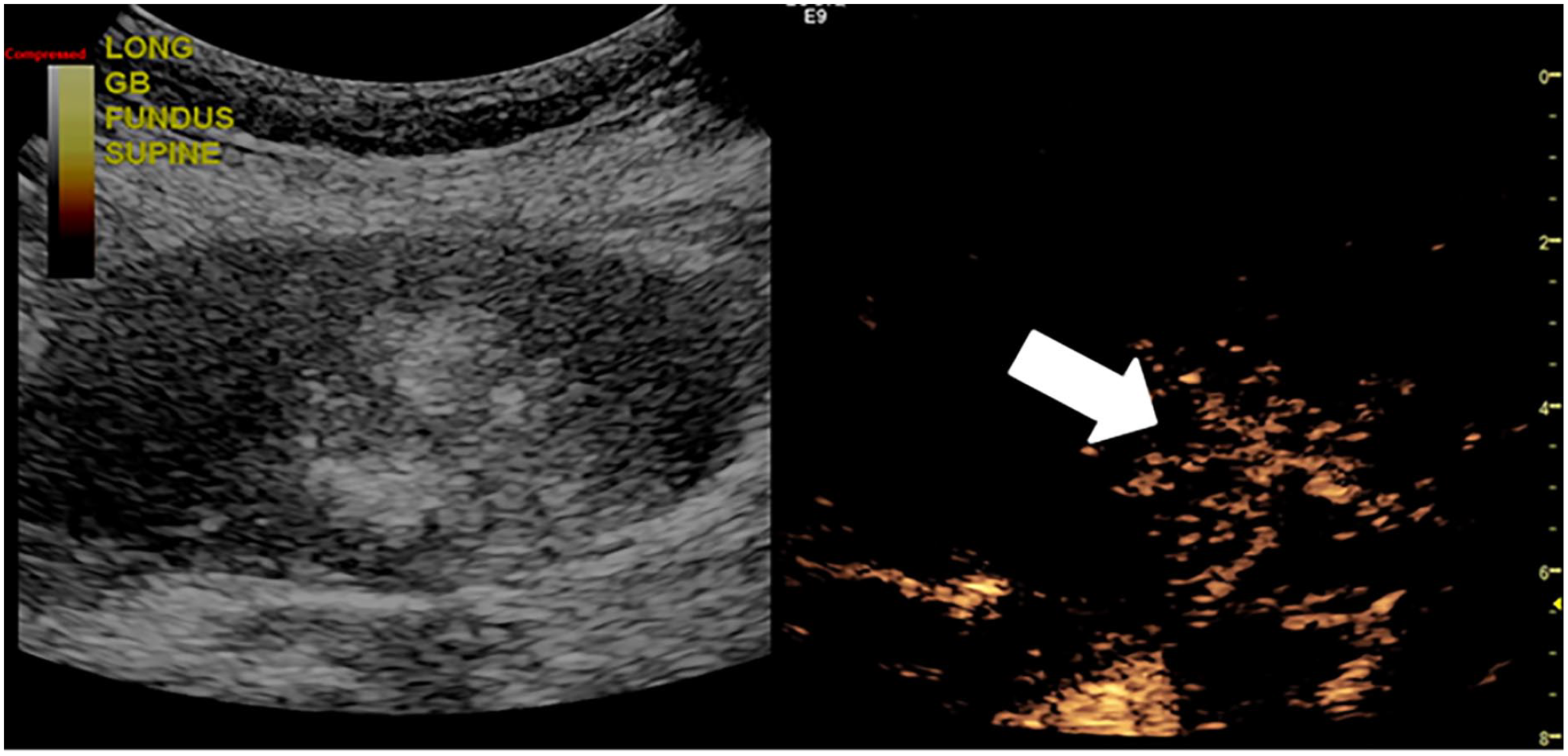
Contrast-enhanced ultrasound dual image demonstrated rapid enhancement within the gallbladder at 17 seconds following the injection. Note the branched intralesional vessels (white arrow).
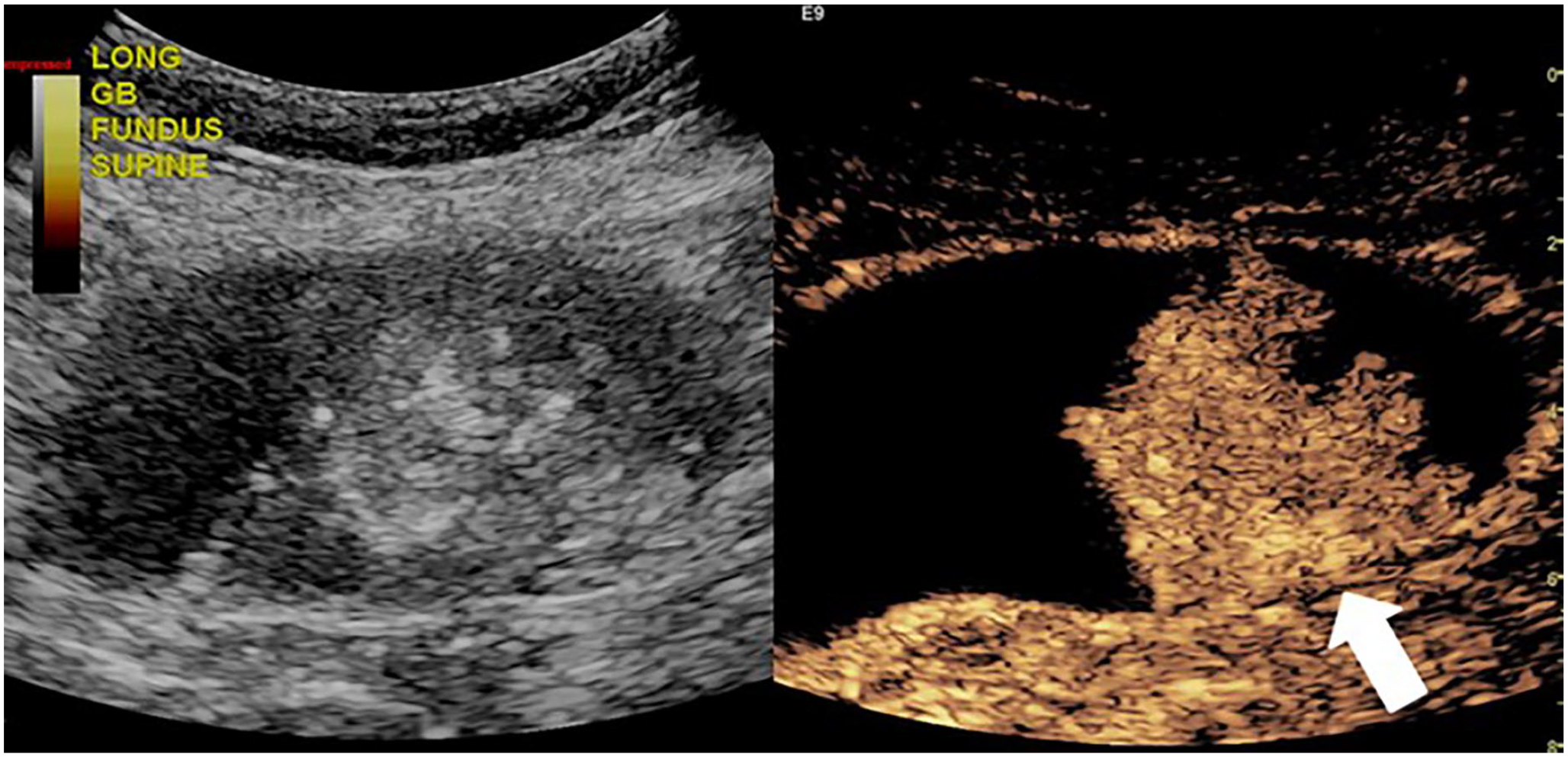
Contrast-enhanced ultrasound dual image confirmed a solid hyperenhancing mass at 31 seconds following the injection (white arrow).
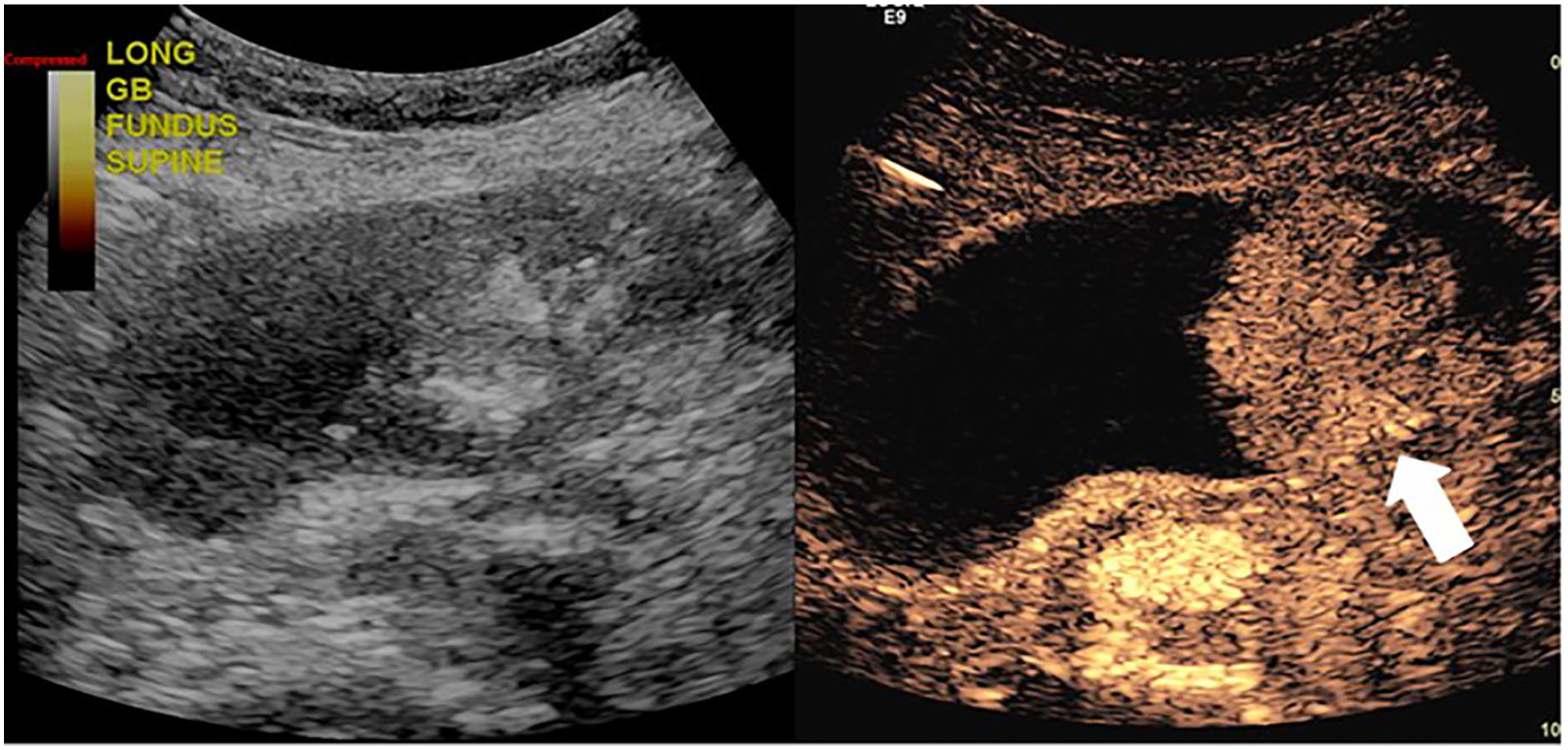
Contrast-enhanced ultrasound dual image of the solid mass remained hyperenhanced at 1 minute and 41 seconds following the injection (white arrow). Note it did not demonstrate rapid washout as expected.
After the patient care team discussed the results of the sonogram with the patient, they requested further workup including a surgical consultation to review all management options. A magnetic resonance imaging (MRI) examination was ordered and performed approximately 1 week following the CEUS for hepatobiliary cancer staging. There were two polypoid enhancing GB masses identified with the largest measuring 5.2 cm and the smallest measuring at 2.3 cm. (Figure 9) The larger mass near the fundus extended through the GB wall without invasion of adjacent structures. There were no findings to suggest gross hepatic extension or hepatic metastases. Also noted was a large gallstone and fundal GB adenomyomatosis (ADM). There were several prominent porta hepatis lymph nodes measuring 9 to 10 mm. A chest CT was also ordered and performed, without evidence for lung metastases. The calcified fundal GB mass was noted (Figure 10). Further laboratory blood work showed elevated carbohydrate antigen 19-9 (CA19-9) of 474 U/mL (normal <35 U/mL) and increased values of ALP at 442 U/L 1 month following the initial laboratory data. After all the information was presented to the patient, they elected to undergo a cholecystectomy to help prolong life.
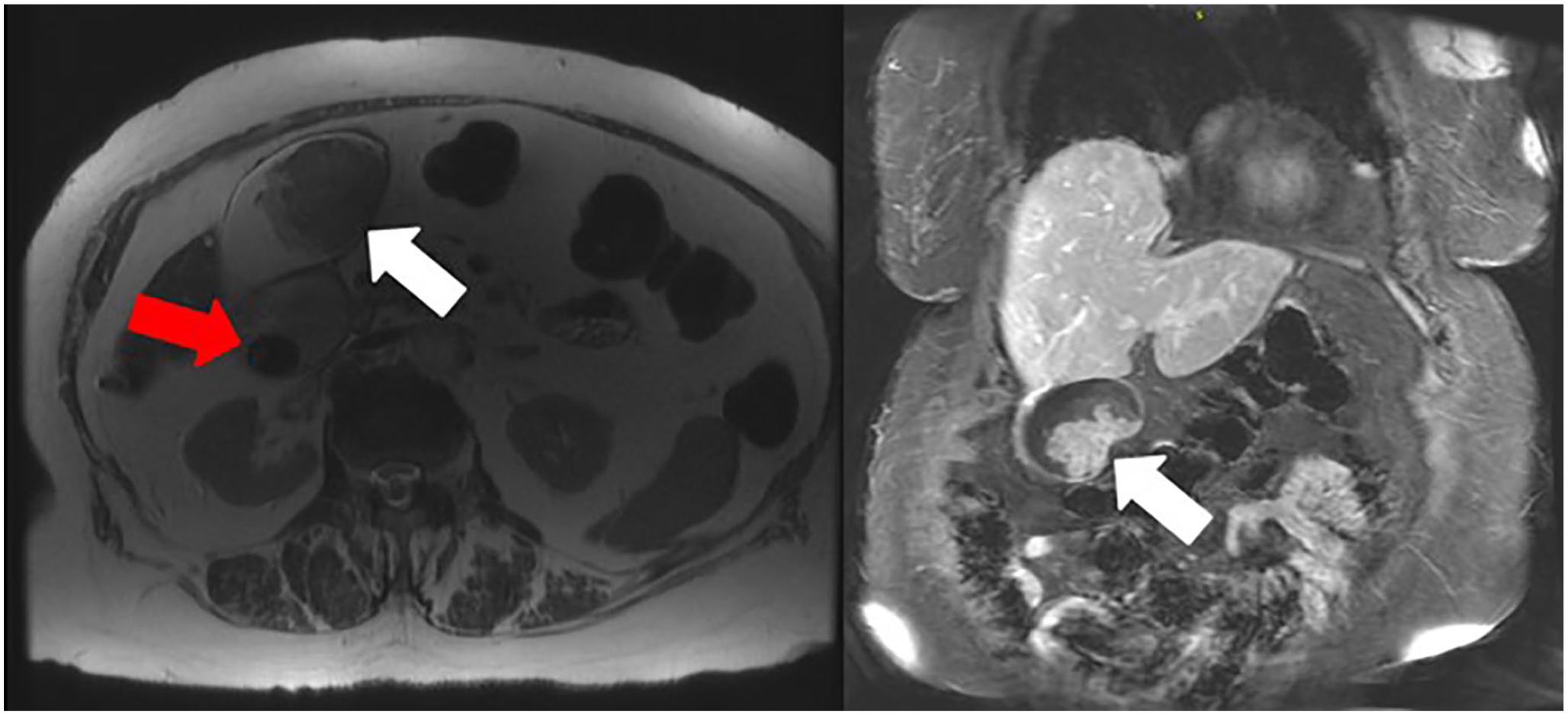
Magnetic resonance imaging examination demonstrated the larger 5.2 cm solid GB mass (white arrows). The axial T2 weighted non-fat saturation image is shown on the left and the gallstone is also visualized (red arrow). The right image depicted the mass in the coronal plane post Gadolinium contrast administration.
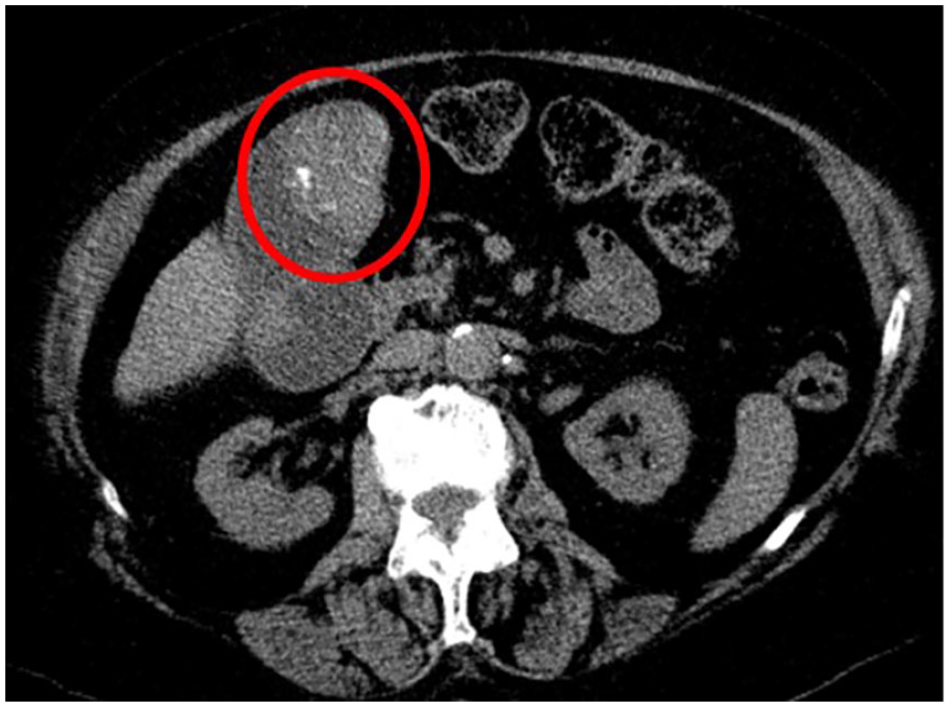
Computed tomography examination demonstrated a vague calcified mass in the fundus of the gallbladder on this axial image (red circle).
Within 1 month of the initial sonographic findings, the patient underwent a laparoscopic cholecystectomy (Figure 11). The surgical procedure was substantially more difficult due to the large tumor size. The final pathology report revealed grade 2 invasive adenocarcinoma, mixed intestinal, oncocytic, and biliary type. The largest tumor invaded the perimuscular connective tissue without serosal involvement. Lymph nodes, which were resected, were negative for tumor. Chronic cholecystitis and cholelithiasis were also noted on the pathology specimen examination.
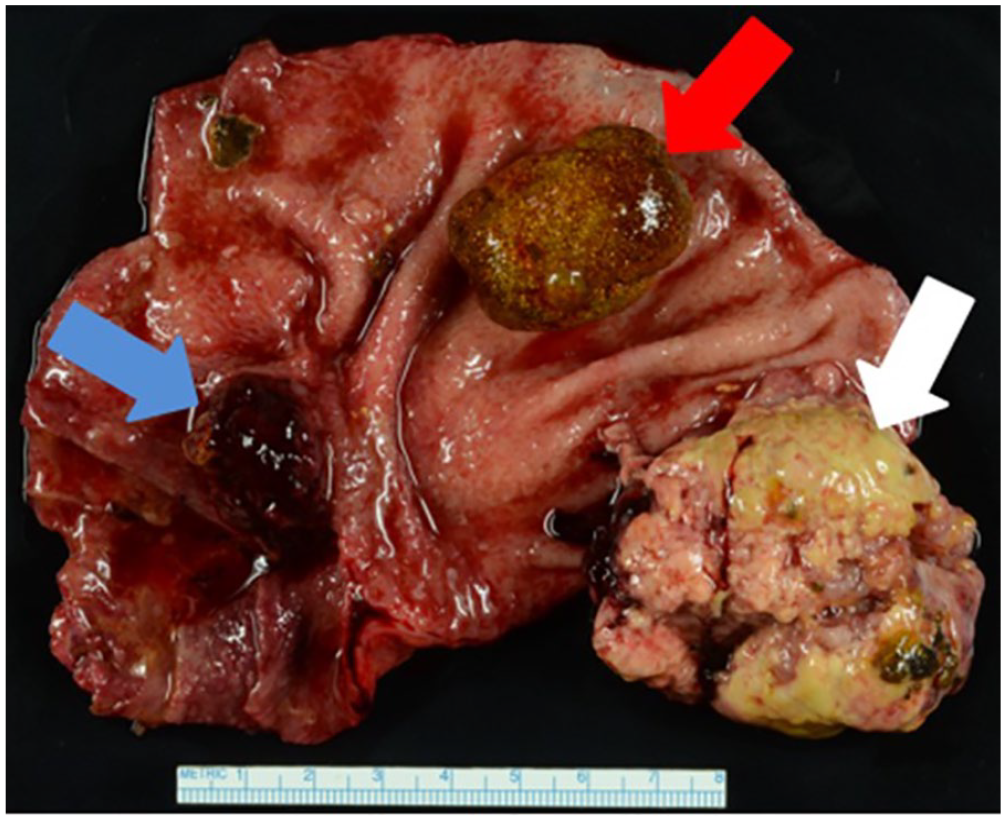
Pathology gross specimen photograph of the surgically open gallbladder. The large mass noted (white arrow), large gallstone (red arrow), and small mass (blue arrow).
Discussion
In 1777, Maximilian de Stoll first described GB carcinomas. Studies have shown adenocarcinoma is the most common histological type accounting for 80% to 97%.17,18 Some other less common malignancies include squamous-cell carcinoma, adenosquamous-cell carcinoma, and papillary adenocarcinoma. Fong et al described these in 3% to 20% of cases. 17 GBC is considered rare and difficult to diagnose within early stages due to lack of patient symptoms in majority of cases. A GBC is only preoperatively suspected in 30% of all patients with 70% of cases being incidental findings during cholecystectomies. 4 Only 15% to 47% of patients with preoperatively suspected GBC are suitable for resection. 19 Autopsy results have revealed GBC in up to 3.4% of patients over 60 years of age with cholelithiasis. 20 Cross-sectional imaging is essential and may be helpful aiding in diagnosis when symptoms or suspicious clinical findings are present.
GB ADM is an epithelial proliferation and hypertrophy of the muscularis layer with outpouching of the mucosa into the thickened muscular layer. ADM is typically asymptomatic and usually detected in 2% to 8% of cholecystectomy cases.21,22 Several reports have documented ADM associated with GBC, although not typical.21,22 In 1988, Katoh et al. were the first to report noninvasive GBC in localized ADM. 23 Morikawa et al. reported only 3.2% (3 of 93) of ADM cases identified GBC. 24 Kai et al. reported approximately 40% of patients with ADM were found to be in advanced stages of GBC. 25
The presence of a large GB mass that completely fills the lumen and often invades the surrounding liver parenchyma is highly suggestive of GBC visualized in about 40% to 65% of cases using sonography, CT, or MRI 13 (Figure 12). Sonographically, the mass appears heterogenous and predominantly hypoechoic filling most of the GB lumen. 13 Gallstones, tumor calcifications or “porcelain GB” may be also present. 26 On unenhanced CT, GBC is typically hypodense with up to 40% of tumors demonstrating hypervascular foci of enhancement equal to or greater than that of the liver following contrast administration. 27 GBC usually appears hypo-to isointense signal on T1 weighted sequences and demonstrates moderately hyperintense signal on T2 weighted sequences on MRI examinations. 28
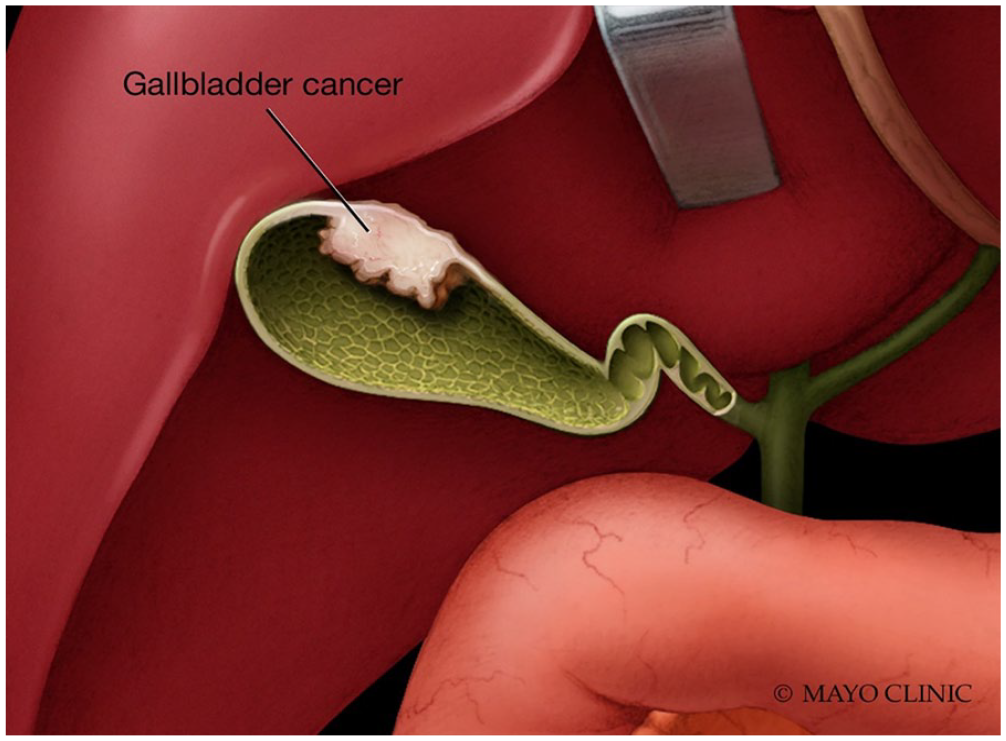
Diagram illustrates gallbladder carcinoma invading the fundal wall. Used with permission of Mayo Foundation for Medical Education and Research. All rights reserved.
Sonography is typically the first modality of choice assessing for GB pathology due to its high sensitivity and real-time imaging. (Figure 13) The normal sonographic appearance of the GB is a completely anechoic fluid filled lumen with a thin smooth wall measuring less than 3 mm. Folds may be present and are considered normal. The patient should fast at least 4 to 6 hours prior to exam to prevent a contracted GB, which can obscure pathology. The sonographer should utilize multiple scanning windows including supine and left lateral decubitus patient positioning. A 2-6 MHz curvilinear transducer is typically used.
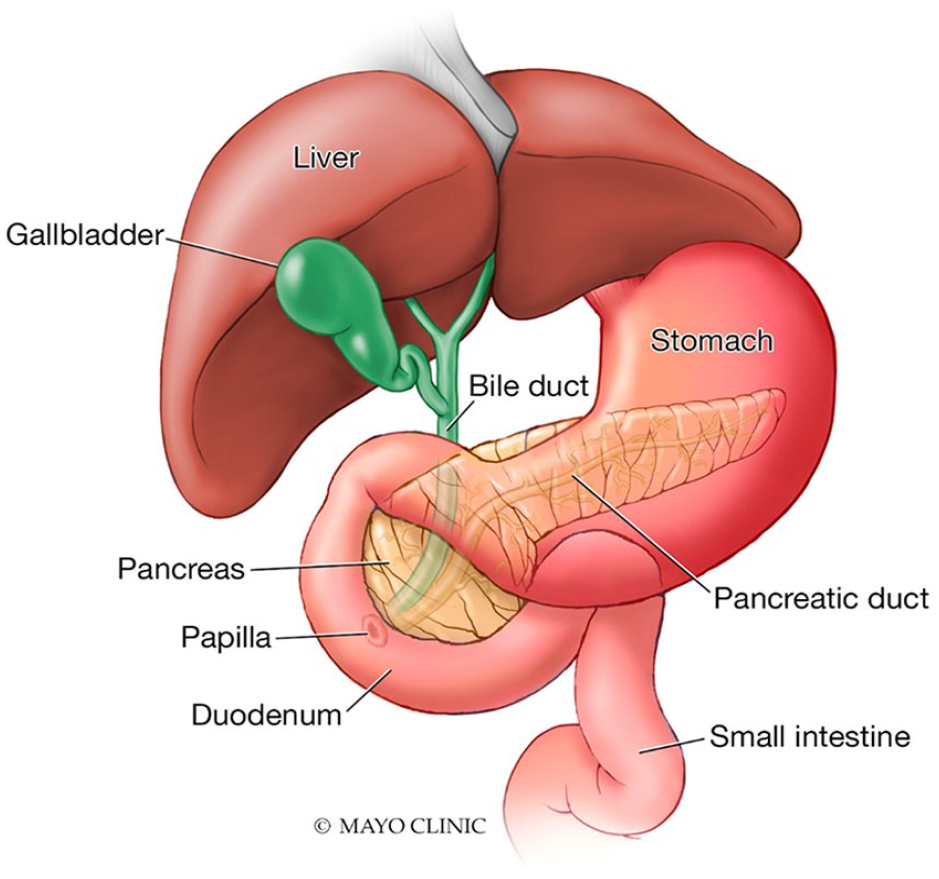
Diagram illustrates the normal gallbladder and biliary tract anatomy. Used with permission of Mayo Foundation for Medical Education and Research. All rights reserved.
Limitations of conventional sonographic imaging may occur when multiple types of GB masses are present especially when filling the GB lumen. Motionless sludge may mimic GBC and distinguishing the two oftentimes can be difficult. These pseudotumors may lead to unnecessary surgical interventions. 29 It is difficult to visualize invasion of the GB wall, adjacent liver infiltration and nodal and peritoneal metastases using sonography alone. 29 Malignancy is usually associated with larger GB polyps and sonographic Doppler techniques have shown a sensitivity and specificity of around 80% diagnosing GBC. 30 The spectral Doppler cut-off value of 20 to 30 cm/s is proposed for characterizing a malignant versus benign disease process. 30 Bach et al. reported a poor sensitivity in which only 37% of advanced disease cases were identified on conventional sonography. 31
This case report demonstrates that CEUS can be helpful to differentiate between motionless sludge and solid tumor. CEUS is a relatively new, cost-effective, noninvasive technique improving resolution limitations. Its application is now recognized as a useful tool in addition to conventional sonography and other cross-sectional imaging in assessment and diagnosis of GB disease.32-39 Ultrasound contrast agents are gas-filled microbubbles injected intravenously lasting 3 to 4 minutes for high-quality diagnostic imaging. As the contrast degrades due to bursting bubbles, the gas is expelled through the lungs. Currently,
CEUS has been successfully used while imaging the liver, kidney, and pancreas. 38 CEUS has now been prompted for GBC cases due to the low specificity on conventional sonography. 39 Numata et al. reported high diagnostic rates identifying GBC using CEUS with 75% sensitivity, 100% specificity, and 91% accuracy. 32 Xie et al. concentrated on GB wall destruction on CEUS, and results showed a high sensitivity of 84.8% and high specificity of 100%. 40 Zhang et al. studied the diagnostic efficiency of conventional sonography compared to CEUS between malignant and benign GB lesions. They reported the sensitivity and specificity rates of conventional sonography as 82.4 and 89.8%, respectively. In comparison, CEUS demonstrated 94.1% sensitivity and 95.5% specificity rates. 41 CEUS can be advantageous over conventional sonography as it can distinguish sludge, show early liver invasion and localize small tumors that may be missed on CT. 37 Normal GB CEUS findings include uniform enhancement of the GB wall, homogeneous enhancement of the adjacent liver, and an anechoic GB lumen without enhancement. 37 Sludge within the GB is typically mobile and appears echogenic; however, it may remain motionless and adhere to the GB wall imitating GBC or adenoma. 42 CEUS can confirm sludge as complete nonenhancement during the arterial and venous phases due to the lack of blood supply within.37,41 Hattori et al. reported the effectiveness of CEUS using the contrast agent, Levovist (Nippon Schering, Japan), for diagnosis of GB polypoid lesions. 43 The most reliable imaging characteristic for GBC is GB wall destruction, but other features include hyperenhancement and rapid washout of the contrast agent within 35 seconds. 40 Other classic CEUS findings indicative of GBC are described as having linear or branched intralesional vessels, or a tortuous-type tumor vessel, which enhances in the arterial phase and washes out quickly in the late phase.29,32,44 Liu et al. achieved a multi-institutional CEUS study of 152 patients with GBC and reported a combination of GB wall destruction and linear or branched intralesional vessels were indicative of GBC. 29
CA19-9, a mucinous protein, may be elevated in patients with bile duct, gastric, and pancreatic cancers.44,45 Carcinoembryonic antigen (CEA), a broad-spectrum tumor marker, is a protein polysaccharide complex, which may be found in gastrointestinal cancer as well as in the normal embryonic gut, pancreas and biliary tree. CA19-9 and CEA are used as predictive markers for gastric and pancreatic cancer. 46 The most measured tumor markers evaluated preoperatively for GBC diagnosis and prognosis are CEA and CA19-9.47-49 Hatzaras et al. reported that CA19-9 appeared to be the only predictor of survival for those patients undergoing resection. 45 It has been discovered that CA19-9 is a prognostic marker in GBC. 48 It was recommended that CA19-9 values be routinely monitored prior to surgical resection. 45 The sensitivity and specificity of CA19-9 are 77.5 and 68.7%, respectively for GBC diagnosis. 49 Surgical resection is the only curative treatment for GBC, but only 10% of patients are good surgical candidates, which lead to the overall poor survival rates. 50 In more than 75% of cases, the GB is surgically removed laparoscopically.8,51 It has been found with elevated CEA and CA19-9 markers, the median survival and 3-year survival rates were 6.2 months and 2.2%, respectively. 46 GBC is not sensitive to chemotherapy or radiation therapy treatments. 13 However, a retrospective study completed by Gold et al reported statistically superior survival rates with adjuvant chemoradiation. 52 Adjuvant therapy is recommended following surgical resection in patients with stage II GBC or higher. 53 CA19-9 may guide personalized neoadjuvant therapy in patients with GBC. 2
Conclusion
Sonography remains a cost-effective, accessible, non-nephrotoxic and non-ionizing tool for diagnosis of GB disease and is the initial imaging modality of choice. GBC is an often fatal disease, with optimal outcomes requiring a timely imaging diagnosis. Conventional gray-scale and color Doppler imaging may be equivocal for detection of a solid mass. Therefore, further evaluation with CEUS may be helpful. The key CEUS finding of GBC is a focal area of rapid enhancement and quick washout within the GB lumen. As illustrated in this case, CEUS differentiated between benign non-mobile sludge and viable tumor. A rapid diagnosis of GBC in this patient helped with surgical planning, preoperative counseling regarding survival and risk, as well as potentially improving long-term outcome.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
