Abstract
This case presentation describes a solid pseudopapillary neoplasm found in a young 41-year-old female. This mass was initially detected with sonography and later confirmed with an ultrasound-guided biopsy. The neoplasm was scheduled to be surgically resected, as a cure, which has a favorable long-term prognosis. This case provides information on the imaging characteristics and natural history of this benign neoplasm.
Solid pseudopapillary neoplasms (SPN), of the pancreas, are rare. They account for 1%–2% of exocrine pancreatic tumors. 1 They were first described by Frantz in 1959 and have had many interchangeable titles, including Gruber-Frantz tumor and solid and papillary epithelial. In 1996, The World Health Organization (WHO) renamed this lesion as a solid pseudopapillary neoplasm and classified it as borderline malignant neoplasm, of the exocrine pancreas.1-3
SPN is mainly seen in young females of Asian and African American descent. 4 It usually affects females in their third decade of life, with a mean age of presentation being 23 years. The male-to-female ratio is 1:10. Even though SPNs can be found anywhere along the pancreas, the majority of them are found in the body and tail of the organ. 2
On sonographic imaging, these lesions may consist of both solid and cystic elements, while surrounded by a well-defined capsule.1,2 Most patients present with abdominal pain, but asymptomatic cases can be detected incidentally on imaging studies. 5 Complete resection is a curative treatment in most cases. 4
Case Report
A 41-year-old Caucasian female presented to the emergency department with epigastric and chest discomfort that radiated to her back. She had both dizziness and nausea.
A right upper quadrant diagnostic medical sonogram (DMS) was performed using a 6-MHz curvilinear transducer and a Logiq E9 ultrasound equipment system (GE Healthcare, Sacramento, CA, USA). The radiologist reported that a 2.0-cm hypoechogenicity was noted and was measured along the pancreatic body. This lesion was well circumscribed and did not demonstrate any acoustic enhancement or posterior shadowing (see Figures 1 and 2). Doppler analysis did not demonstrate any significant vascularity, associated with this lesion (see Figure 3).
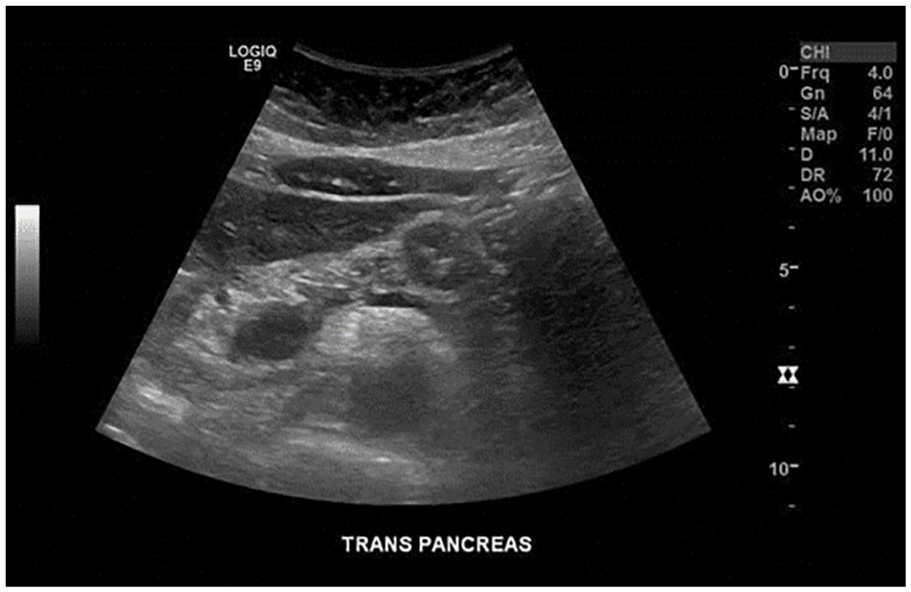
A transverse sonographic view of the abdomen shows a hypoechoic mass within the pancreatic body.
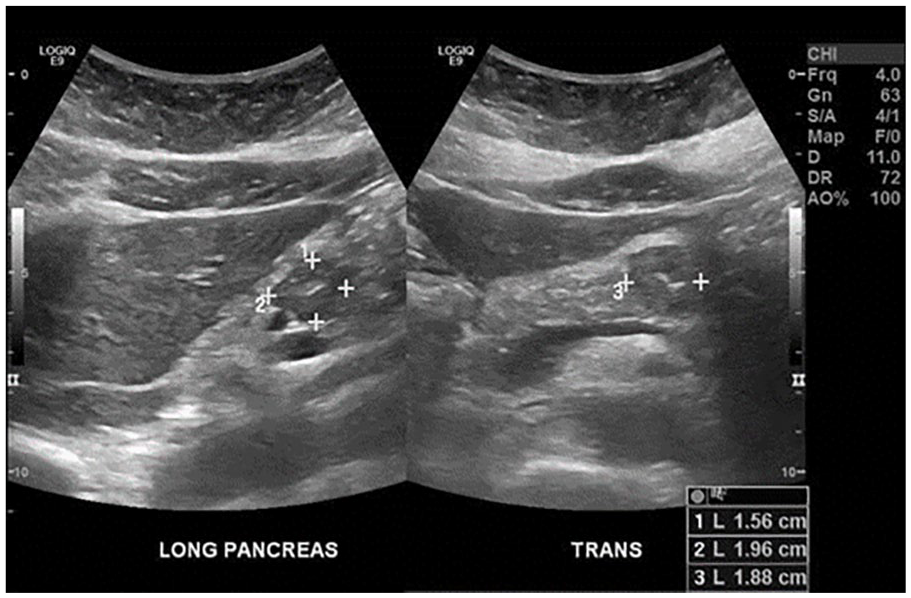
A series of measurements of the pancreatic mass, which was seen in both the longitudinal and transverse sonographic planes.
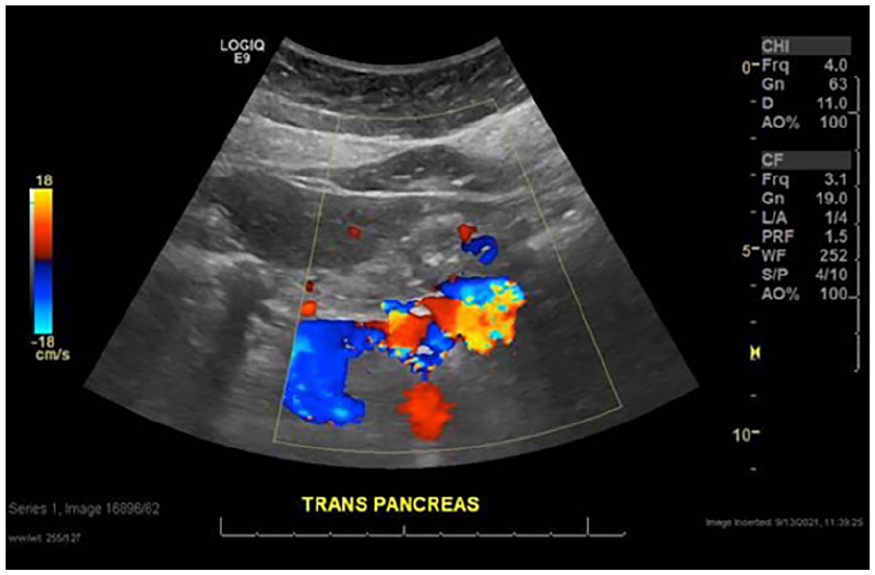
A transverse sonographic view of the abdomen shows no significant vascularity within the pancreatic mass, itself.
Computed tomographic (CT) images of the abdomen and pelvis were acquired with 145 mL Omnipaque 300 intravenous contrast, and this study confirmed the DMS findings. The CT report described a 2-cm exophytic, hypodense, soft tissue mass in the superior aspect of the pancreatic body (see Figure 4). These diagnostic findings were highly suspicious for a pancreatic neoplasm, but there was no associated pancreatic ductal dilation.
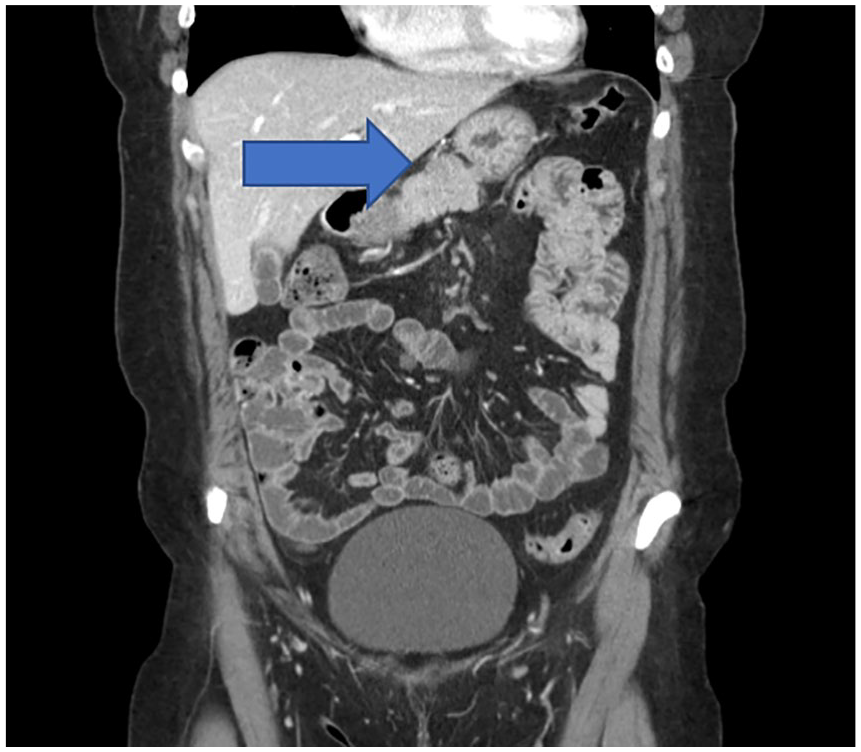
A coronal computed tomographic image demonstrates a soft tissue mass seen within the pancreatic body (indicated by the blue arrow).
In the meantime, the patient’s epigastric and chest pain improved with medication. Her lab results, including standard chemistries, complete blood count, liver function tests, and lipase, were within normal ranges. The patient was subsequently discharged home with an outpatient follow-up appointment to be evaluated by gastroenterology (GI).
Within 2 days of discharge, GI performed an endoscopic ultrasound with fine needle aspiration (EU-FNA). It showed nonhoneycomb lobularity in the pancreatic parenchyma and stranding in the head, uncinate body, and tail of the pancreas. A hypoechoic and slightly heterogeneous solid mass, with central calcification, but without any microcysts, were seen in the body of the pancreas. This mass measured 2.2 × 1.5 cm with no vascular invasion (see Figures 5 and 6). Using a trans-gastric approach, under Doppler guidance, to avoid vascular injury, the mass was biopsied with multiple passes. The biopsy was completed using a BS Acquire 22-gauge fine needle biopsy needle (see Figure 7).
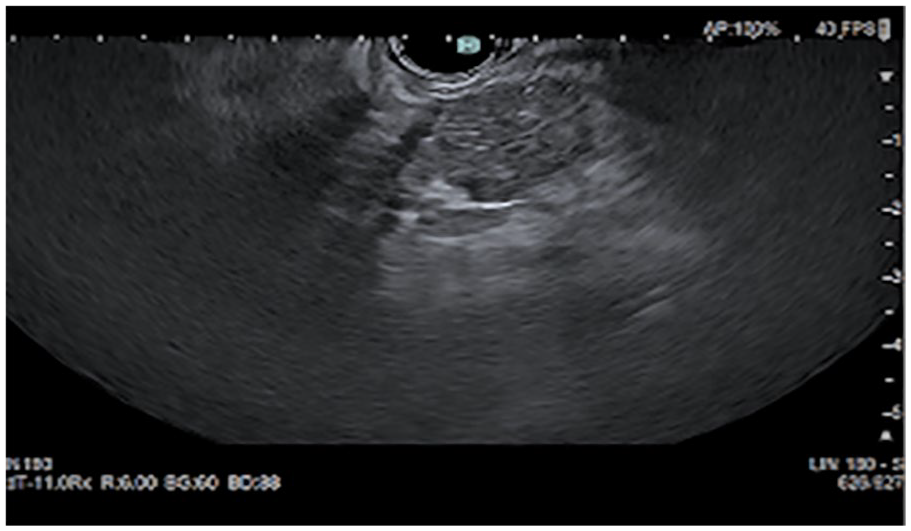
An endoscopic sonographic image of the hypoechoic pancreatic mass.
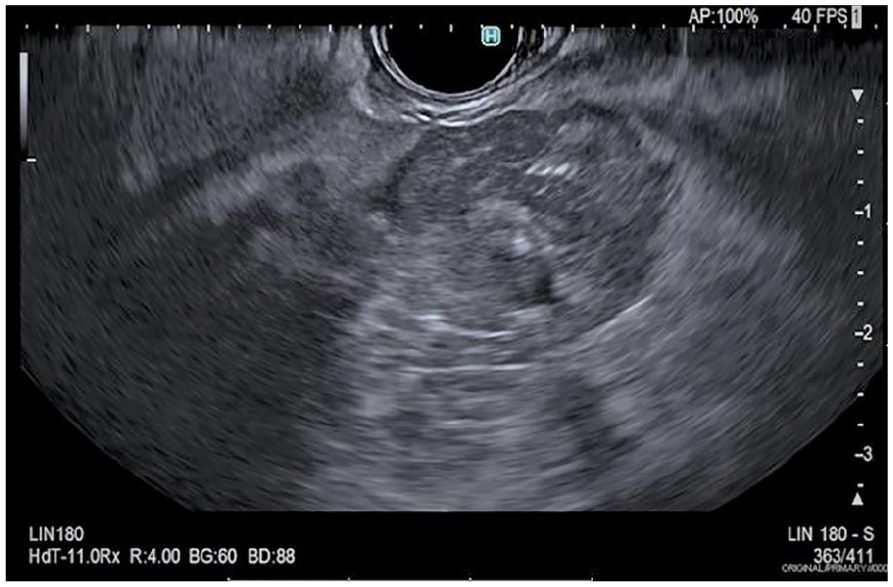
An additional endoscopic sonographic image if hypoechoic pancreatic mass.
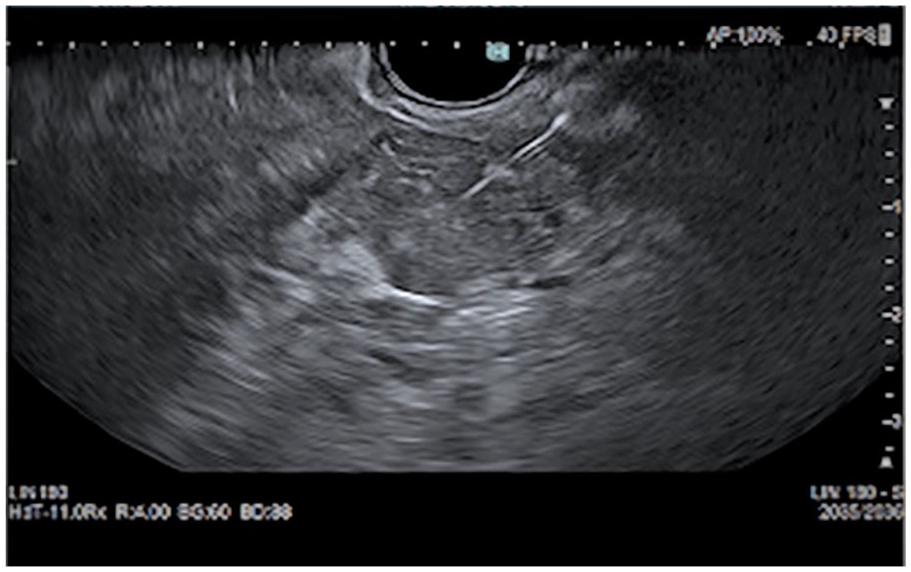
An endoscopic sonographic image of hypoechoic pancreatic mass, with a linear echogenic biopsy needle inserted.
An immunohistochemical analysis revealed that these mass cells were positive for beta-catenin, CD56, CD10, and AE1/AE3, while chromogranin and e-cadherin values were negative. These diagnostic findings were consistent with a solid pseudopapillary neoplasm. Based on these pathological findings, the patient was scheduled for surgery to extract the entire mass.
Discussion
Etiology
The etiology of SPN is unclear. Most researchers suspect that SPNs originate from multipotent primordial cells, whereas some suggest an extra-pancreatic origin.1,2,6
Presentation
SPNs present with metastases in approximately 15% of patient cases. The most common metastatic sites are adjacent structures, such as the liver and omentum. 2 There have also been cases of splenic invasion and these are mostly seen with large SPNs located, in the tail of the pancreas. 1 If the tumor grows large enough to cause obstruction, jaundice may occur.4,5 Blood lab results are generally normal, such as normal serum amylase levels and normal pancreatic cancer markers (like CA19-9, carcinoembryonic antigen, and α-fetoprotein.) Because patients present with nonspecific symptoms and normal blood labs, the diagnosis is commonly made incidentally with imaging, ultrasound and/or CT. 7
Imaging
Studies have shown that the tumor size varies widely, anywhere from 3.0 to 15.0 cm. Tumor sizes greater than 5.0 cm are associated with a greater risk of high-grade malignancy. 8 An additional report found that when the tumor is greater than 6 cm, the possibility of vascular involvement and capsular invasion increases significantly. 3
On DMS, an SPN can appear well-defined and predominantly cystic, solid, or a complex lesion with both cystic and solid components. This lesion may or may not have calcifications.5,8 Beak et al. demonstrated that imaging features of small SPNs, less than 3.0 cm in size, were different than the imaging features of larger SPNs. This study showed that small SPNs generally appear purely solid with sharp margins, as seen in the present case study, while SPN greater than 3.0 cm appear as well-defined cystic and solid masses. 9 It is believed that the larger masses outgrow their blood supply and undergo cystic degeneration, necrosis, and hemorrhage. The degeneration of the mass can also acquire dystrophic calcifications. Calcification can occur centrally or peripherally. 1
SPNs do not exhibit any unique features that help distinguish them from other pancreatic masses. Of tumors that have cystic and solid components, the differential diagnosis includes serous microcystic adenoma and mucinous cystic neoplasm. 7 Microcystic adenoma are benign, consist of small cyst (less than 2.0 cm), and may have calcifications. They are frequently seen at the head of the pancreas. Microcystic adenomas may also present with a central stellate fibrotic scar (seen in approximately 50% of the tumors). Mucinous cystic adenomas are predominantly malignant and consist of larger cyst (greater than 2.0 cm) and may have a calcified rim. They occur in the tail 60% of the time. 10 Even though both of these tumors are mostly found in females, they rarely occur in young females with the typical age of presentation being between the fifth and seventh decade of life.8,11
The differential diagnosis for solid appearing masses includes pancreatoblastoma and pancreatic adenocarcinoma. Pancreatoblastomas are seen in young children and often present with liver metastases. These lesions are more aggressive than SPNs. Pancreatic adenocarcinoma is the most common pancreatic malignancy, accounting for 95% of all pancreatic masses. 10 These mainly arise at the head of the pancreas. About 70% of these tumors are found in older patients. Their typical sonographic appearance is hypoechoic, with virtually no calcifications or cystic degenerations.10,11 Other clues to help differentiate some of these masses from SPNs include the absence of lymph node enlargement that might be present in a metastatic process. Moreover, SPNs do not show the infiltrative changes in the surrounding pancreatic parenchyma that may be seen in more aggressive masses.10,11 Elevated carcinoembryonic antigen (CEA) level, serum amylase, and bilirubin may be seen in other pancreatic masses and are not typically seen with SPNs. 8
DMS is a great imaging technique for capturing the entire pancreas, for diagnostic evaluation. Unfortunately, despite technology advancements, using optimal scanning techniques, such as placing the patient in the upright position, filling the stomach and duodenum with water, the entire pancreas is not visualized in most patients.
CT, with intravenous contrast, is the most frequently used imaging modality to evaluate and characterize these pancreatic masses. Magnetic resonance imaging (MRI) can provide a more accurate diagnosis as it can better define hypervascularity, shape, and the cystic and hemorrhagic features of these tumors.6,3A significant disadvantage is that MRI is more expensive and time consuming than a CT study.
Although clinical and diagnostic imaging features can be helpful, a fine needle aspiration is the most reliable way to confirm the diagnosis. SPN cells are characteristically positive for α1-antitryspin, CD56, CD10, and vimentin and negative for endocrine markers such as CD68, lysosome, chromogranin, and bombesin.4,5 These diagnostic results were also found in the present case study.
Treatment
The definitive treatment of SPN is complete resection. Even if the resected tumor is malignant, the prognosis is favorable after surgery. The 5-year survival is as high as 97%. 4 The patient, in the current case study, underwent a pancreatic resection of the distal pancreas, a non-Whipple procedure, with no complications reported.
Conclusion
Solid papillary neoplasms are rare pancreatic tumors seen mainly in young females with a good prognosis. Although CT and MRI studies can optimally characterize SPNs, DMS also can play an important diagnostic role. DMS can help describe the mass, assess vascularity, and help guide fine needle aspirations of these tumors.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
