Abstract
Solid pseudopapillary neoplasm (SPN) is a rare epithelial neoplasm of the pancreas, with low malignant potential, that usually occurs in young women. With advancing diagnostic imaging techniques and technology, the number of documented SPN cases has increased. Due to a varying morphologic appearance, SPNs often fail to show typical imaging findings, making it difficult to establish a definitive diagnosis. A case is reported of a SPN discovered on a routine abdominal sonogram, further characterized with magnetic resonance imaging, and confirmed with surgical pathology analysis. Sonographers should be aware of this rare pathology and should consider SPN in the differential diagnosis when encountering an indeterminate pancreatic neoplasm, especially in a young female patient.
Introduction
Solid pseudopapillary neoplasm (SPN) of the pancreas is a rare cystic neoplasm that has been reported to account for between 0.17% and 2.7% of all nonendocrine tumors of the pancreas.1,2 Distinct characteristics of a SPN include prevalence in young females, uncertain histogenesis, generally low malignant potential, and an excellent long-term prognosis, despite its aggressive appearance at presentation. 3 Accurate preoperative diagnosis aids surgical decision making and can require multimodality imaging management. This article presents a case of SPN in which diagnosis was suspected on imaging and confirmed by pathology, enabling appropriate surgical management.
Case Report
A 14-year-old female presented for an abdominal sonogram to evaluate generalized upper abdominal pain. There was no history of nausea, vomiting, weight loss, or appetite changes. The patient had no known history of pancreatitis, alcoholism, diabetes, or biliary disease.
The sonographic examination was performed using a GE Logic 9 (GE Healthcare Ultrasound, Waukesha, Wisconsin, USA) ultrasound system using a standard 5-MHz curved-array transducer. Imaging of the pancreas demonstrated a well-defined, round, hypoechoic 15-mm lesion within the head and body of the pancreas (Figure 1). The lesion did not demonstrate internal or peripheral vascularity on color Doppler analysis. The main pancreatic duct proximal to the lesion was dilated, measuring 4 mm in maximum anterior-posterior dimension (Figure 2). The remainder of the abdominal sonogram yielded no additional pathology.
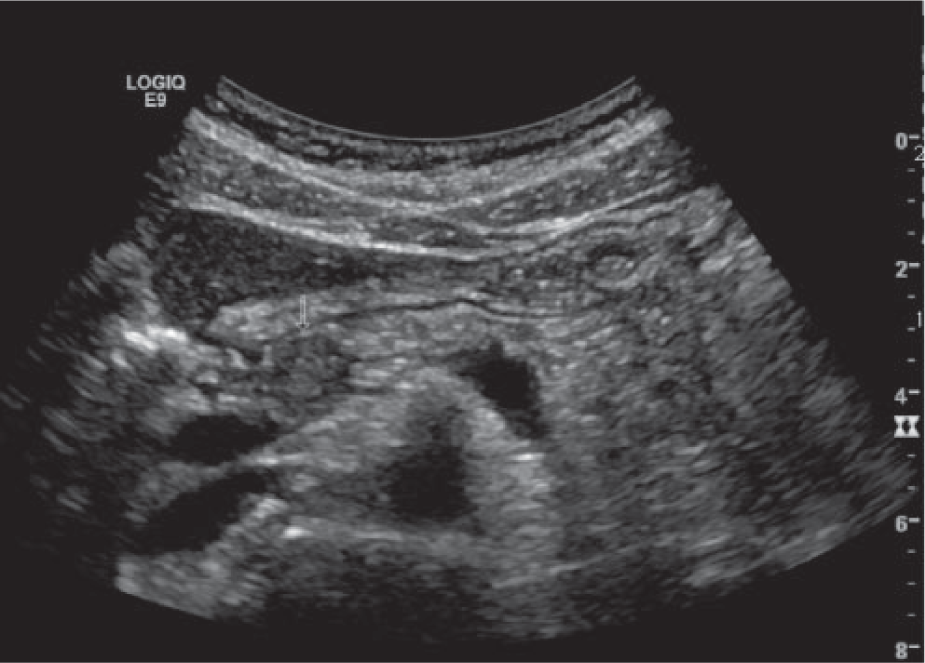
Transverse gray-scale image showing a well-circumscribed, 14-mm hypoechoic lesion within the pancreatic head (arrow).
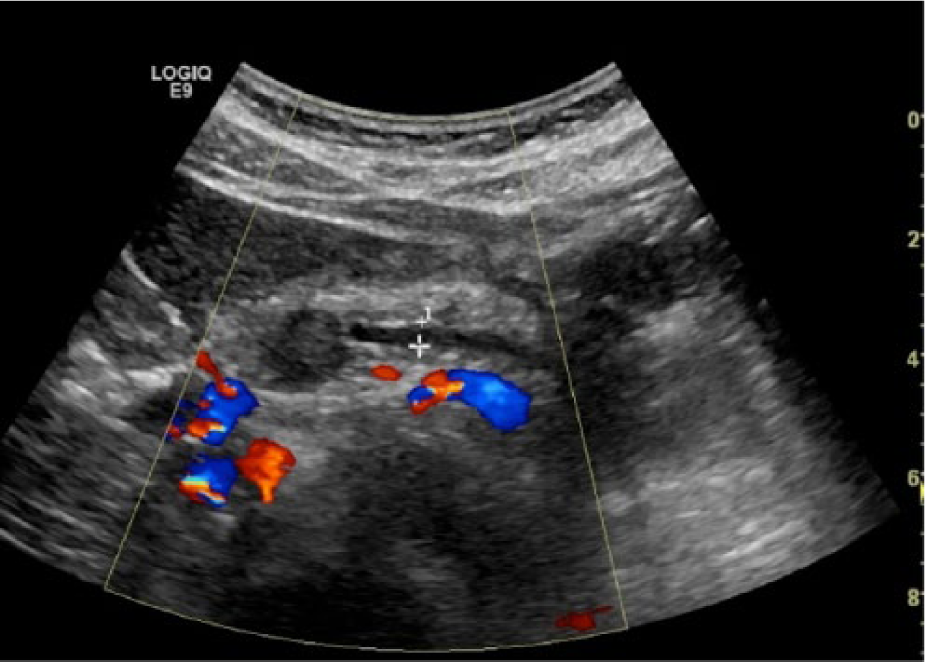
Transverse image with color Doppler imaging showing main pancreatic ductal dilation proximal to the avascular, hypoechoic lesion residing in the pancreatic head.
Follow-up magnetic resonance imaging (MRI) was performed using a GE Optima 450W 1.5 Tesla (GE Healthcare, Waukesha, Wisconsin, USA). The MRI detected a 12-mm round mass within the pancreatic body corresponding to the sonographic findings. The mass demonstrated low T1 signal and less contrast enhancement than the adjacent pancreatic tissue, and an intermediate high T2 signal without cystic component (Figures 3 and 4). The pancreatic body and proximal tail demonstrated diminished T1 signal with delayed contrast enhancement, consistent with chronic pancreatitis and fibrosis (Figure 3). Similar to the sonographic examination, the main pancreatic duct proximal to the lesion was dilated, measuring up to 4 mm in maximum dimension (Figures 3 and 4). Differential diagnoses based on the sonogram and MRI examinations were pancreatic solid pseudopapillary tumor, adenocarcinoma, and pancreaticoblastoma.
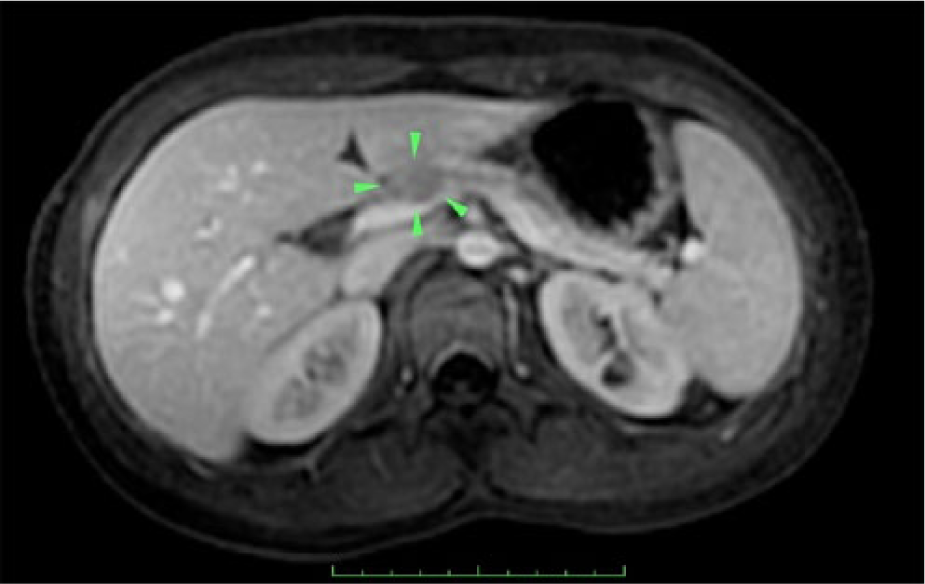
T1 weighted axial, contrast-enhanced, fat saturated magnetic resonance image showing the low signal intensity pancreatic mass (arrowheads) with proximal pancreatic ductal dilation and proximal pancreatic atrophy. The mass can be seen to be less enhancing than the adjacent pancreatic parenchyma.
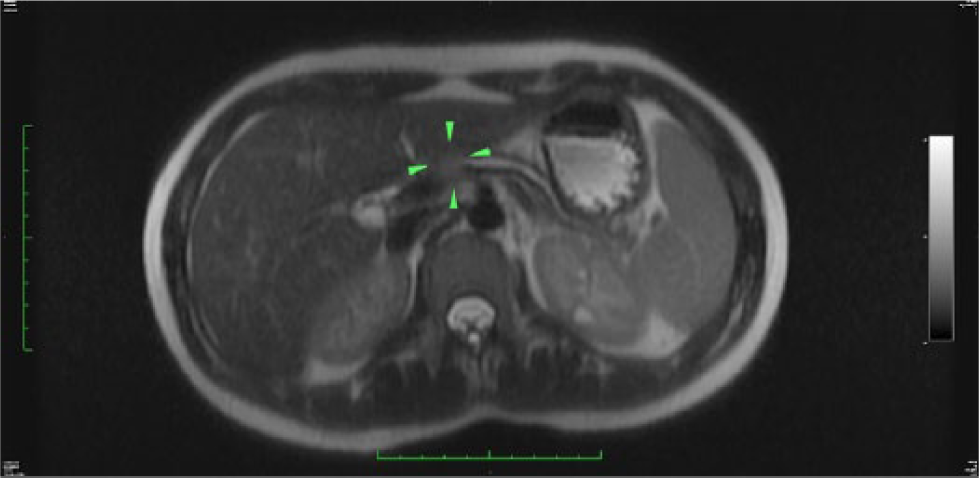
T2 weighted axial magnetic resonance image showing a mildly hyper-intense mass in the pancreatic head mass (arrowheads) with proximal pancreatic ductal dilatation and pancreatic atrophy.
The patient underwent surgical resection of the central pancreatic tumor along with a distal pancreatectomy and splenectomy. Gross examination of the resected tumor revealed a well-circumscribed, solid, yellow tan mass without hemorrhage or necrosis (Figure 5). Microscopically, the tumor was composed of monomorphic epithelioid cells with round nuclei and uniform chromatin surrounded by a delicate fibrovascular network (Figure 6A). Some nests showed necrosis of tumor cells within the center of the nests and some cells showed clear cytoplasm with numerous eosinophilic hyaline globules (Figure 6B). Immunohistochemical stains showed the tumor cells to be nuclear immunoreactive toward beta-catenin and nonreactive toward chromogranin and trypsin, thus confirming a solid pseudopapillary tumor (Figure 6C). Additional pathology analysis confirmed that there was no regional metastatic lymph node involvement. The patient tolerated the procedure well, and at 8-week follow-up, abdominal computed tomography yielded no evidence of residual tumor or metastatic disease.
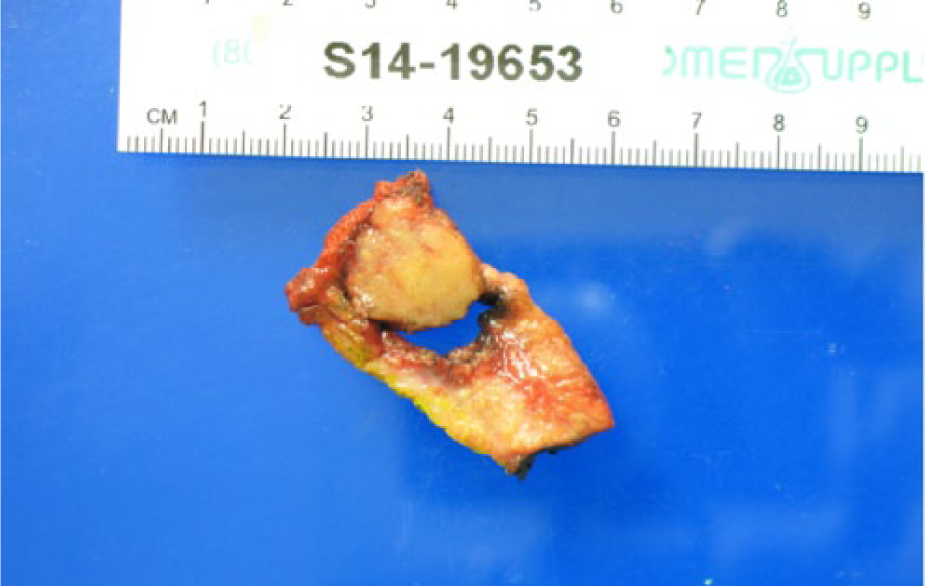
Photograph of the surgically resected specimen showing a 17-mm solid mass within a cystic cavity surrounded by pancreatic tissue.
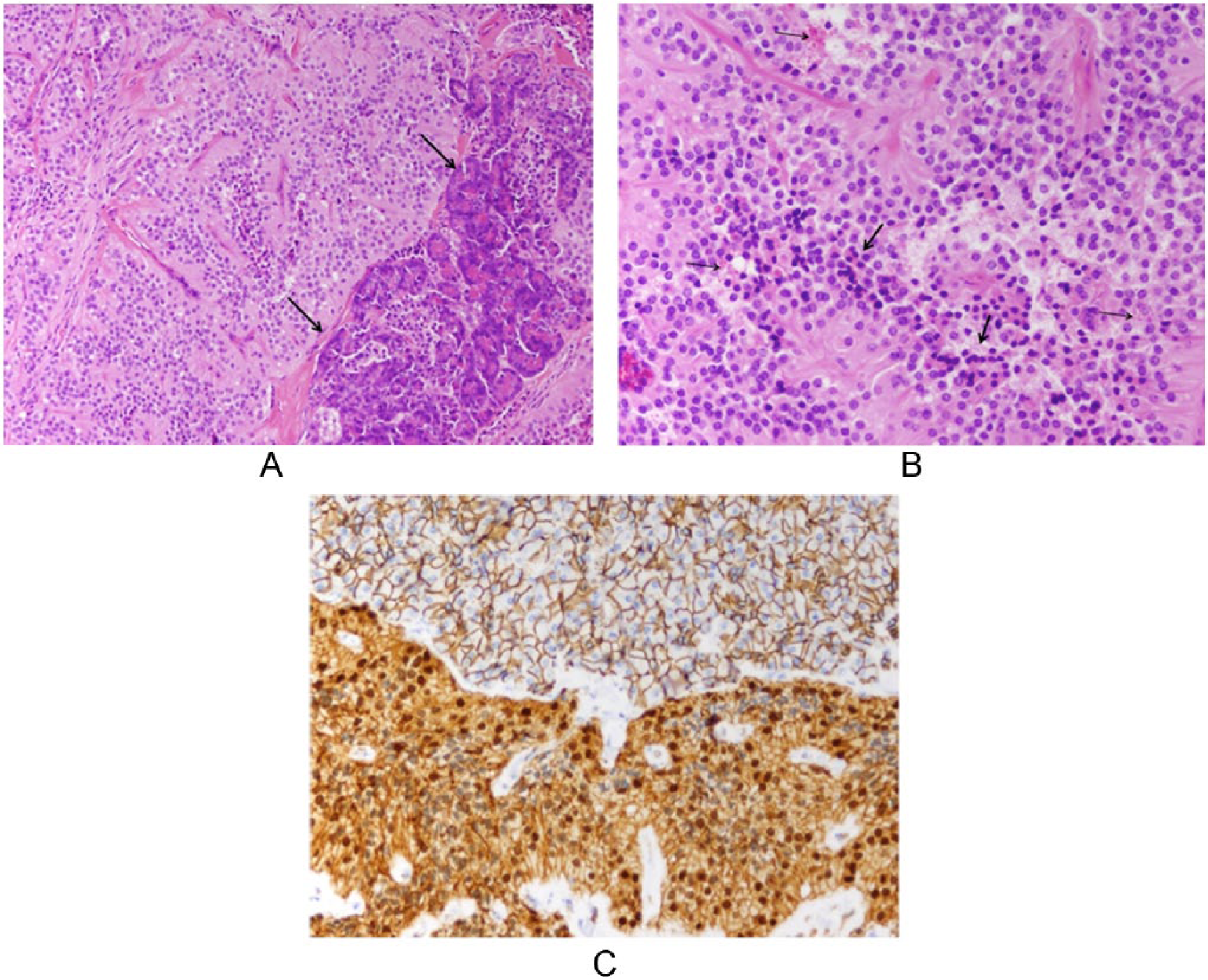
Histopathologic evaluation of the specimen: (A) A nest of monomorphic round tumor cells entrap an island of normal pancreatic parenchyma (arrows) at its edge (hematoxylin and eosin stain; 100× magnification). (B) May cells showed clear cytoplasm filled with hyaline globules (thin arrows). Cellular necrosis was present in the center of the nests, farthest from the blood supply (thick arrows) (hematoxylin and eosin stain; 200× magnification). (C) Beta-catenin nuclear immunoreactivity of tumor cells (bottom of image) as compared to the normal membranous pattern seen in the normal pancreas (top of image) (200× magnification).
Discussion
Pathology
Solid pseudopapillary neoplasm of the pancreas is a rare entity, constituting about 1% of all pancreatic neoplasms. 1 The tumor is exocrine in origin and typically found in young women, between the second and third decades of life. 2 Clinically, patients are commonly asymptomatic; those with symptoms usually report generalized or vague abdominal discomfort. In addition, pancreatic laboratory values are typically normal, making incidental finding a common means of detection for SPNs. 3 If the SPN grows large enough, obstruction of the main pancreatic duct or biliary tree would manifest obstructive and pancreatitis symptoms. Solid pseudopapillary neoplasms classically occur in the head or tail of the pancreas but can be found throughout the pancreas. 2
Solid pseudopapillary neoplasms have a varying morphological appearance, ranging from solid to cystic with degeneration. 4 Gross examination of the neoplasm will show a well-encapsulated mass, with internal areas of necrosis and fibrosis. 5 Solid pseudopapillary neoplasms have a characteristic cytologic profile positive for antitrypsin, vimentin, neural cell adhesion molecule (CD56), and common acute lymphocytic leukemia antigen (CALLA or CD10). 6 Most SPNs are benign in nature with malignancy occurring rarely. 7 Of note, a single literature review found that 66 of the 452 cases evaluated demonstrated malignancy with regional metastasis. 8 Given the chance of malignancy, surgical resection of the tumor along with accompanying pancreatectomy, splenectomy, or pancreatico-duodenectomy is the course of treatment for SPNs. Studies have shown that 95% of patients with total tumor resections have full survival 9 ; a local recurrence rate of 6.2% is reported for radical resections. 5
Imaging Features
Because small tumors are frequently asymptomatic, SPNs often are large at diagnosis. They are well-encapsulated tumors that contain varying amounts of hemorrhage, necrosis, and cystic changes. These are characteristic findings that allow for differentiation from other solid pancreatic neoplasms on cross-sectional imaging. 10 Sonography and MRI are modalities used in the detection, diagnosis, and management of SPNs.
Sonography is commonly used as the initial imaging tool when evaluating generalized abdominal pain, and as such, SPNs could present on routine examination. Solid pseudopapillary neoplasms generally present on sonogram as a well-defined, nonspecific mixed solid and cystic pancreatic mass; however, as in the case presented, the mass can appear purely solid without cystic areas. 11 Typically, smaller tumors appear hypoechoic without cystic components, with larger tumors containing internal areas of hemorrhage or cystic degeneration. 11 When the tumor is located within the pancreatic head, ductal dilatation can be seen in the main pancreatic duct and common bile duct. Definitive diagnosis of pancreatic neoplasms, including SPNs, cannot be achieved with sonography alone, and they require additional imaging with an advanced modality such as MRI.
Because of its high-contrast resolution between focal pancreatic disease and normal pancreatic parenchyma, MRI is regularly used for differentiation and characterization of pancreatic tumors that have been documented initially with sonography. Magnetic resonance imaging better depicts fat, hemorrhage, cystic changes, and pancreatic ductal dilatation, all of which are suggestive of SPN. 10 Magnetic resonance imaging of a SPN shows a well-circumscribed mass, with low signal intensity on T1 weighted imaging, and a high to intermediate signal intensity on T2 weighted imaging. 12 For predominately cystic lesions, signal intensity resembles characteristics closer to that of fluid on T2, whereas smaller SPNs have a more homogeneous appearance. 10 Serous microcystic adenoma, mucinous cystic neoplasm, pancreaticoblastoma, and cystic islet cell tumor could be considered as differentials on MRI. However, SPN should be considered more likely when a well-encapsulated, large, solid, and cystic mass with areas of hemorrhagic degeneration presents within a woman younger than 30 years. 13
Conclusion
A case is presented of a rare pancreatic tumor, solid pseudopapillary neoplasm, initially discovered with sonography and later characterized with MRI. Surgical resection and subsequent histology confirmed a diagnosis of SPN. With the increasing use of sonography on initial investigation of generalized abdominal pain, sonographers need to have knowledge of the wide presentation of pancreatic pathology. Solid pseudopapillary neoplasm should be considered in the differential diagnosis when encountering a solid or solid and cystic pancreatic neoplasm with sonography, especially in women younger than 30 years. Although definitive diagnosis of SPN cannot be determined with sonography alone, when used in conjunction with MRI and biopsy, sonography can play a decisive role in the detection, treatment, and surveillance of this rare pathology.
Footnotes
Acknowledgements
The authors would like to thank Ruzena Jelinek, RDMS, RVT, for providing the sonographic images.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
