Abstract
Due to the rarity of the disease, there are no guidelines or consensus for the management of solid pseudopapillary neoplasm of the pancreas with liver metastasis. We describe a patient with solid pseudopapillary neoplasm of the pancreas with multiple liver metastases who was successfully managed with staged resection. A previously healthy 50-year-old female presented to the clinic with dull epigastric pain. The contrast-enhanced computed tomography showed a well-defined cystic lesion at the distal body and tail of the pancreas with multiple liver deposits. A core needle liver biopsy was suspicious of a hepatic epithelioid hemangioendothelioma. A distal pancreatectomy with splenectomy and a left hemi-hepatectomy were performed with a subsequent staged right posterior sectionectomy to address the inadequate future liver remnant. The final histology with immunohistochemistry revealed a pancreatic solid pseudopapillary neoplasm. The patient had satisfactory surgical and oncological outcomes with no recurrence. As these are slow growing tumours, appropriately spaced staged liver resection was feasible to allow hypertrophy of the liver remnant and physiological recovery of the patient. The discrepancy in the histology and cross-sectional imaging led to a pre-operative diagnostic dilemma, and the final diagnosis was reached after surgical resection. Thus, resectable metastatic solid pseudopapillary neoplasm of the pancreas may be offered a primary curative radical surgery before considering other treatment modalities.
Introduction
Solid pseudopapillary neoplasm (SPN) of the pancreas accounts for 1%–2% of exocrine pancreatic neoplasms. 1 It is more prevalent among young females with the mean age of presentation being around 20–35 years.1,2 These are slow growing, non-functioning tumours usually arising from the body and tail regions of the pancreas. 2 Therefore, SPNs are usually associated with non-specific symptoms and are often diagnosed incidentally with cross-sectional imaging. Abdominal discomfort or pain is the commonest symptom.1,2 SPN is considered to have a low malignant potential, and it rarely metastasises to the liver, omentum and mesenteric lymph nodes. 3 Because of the rarity and unusual presentation, the diagnosis may become challenging. Surgical excision is considered the treatment of choice due to the favourable long-term survival and poor response to chemotherapy and radiotherapy. 3
Due to the rarity of the disease, there are no guidelines for the management of the SPN with liver metastases. However, recent reports have suggested surgical resection as a feasible and effective option for multiple liver metastases.3–5 We describe a patient with SPN of the pancreas with multiple liver metastases who was successfully managed with staged resection while discussing the challenges encountered.
Case presentation
A previously healthy 50-year-old female self-presented to the clinic with dull epigastric ache associated with dyspeptic symptoms for 7 years. There was no significant weight loss or loss of appetite. She did not have a family history of malignancy. She had no significant drug or allergy history or psychosocial history. Her past surgical and medical histories were unremarkable.
She had a palpable epigastric intra-abdominal mass that moved with respiration. Abdominal ultrasonography showed multiple heterogeneous cystic lesions in the body and tail of the pancreas. Contrast-enhanced computed tomography (CECT) of the abdomen showed a 11.6 cm × 9.4 cm well-defined cystic lesion with a thin rim of peripheral calcification at the distal body and tail of the pancreas. There were multiple liver metastases in both lobes of a non-cirrhotic liver (Figure 1). There was no para-aortic or mesenteric lymph node enlargement. Core needle biopsy of the liver was suspicious of a hepatic epithelioid hemangioendothelioma (HEHE). At this stage, the differential diagnoses were synchronous pancreatic SPN and HEHE or a metastatic SPN.
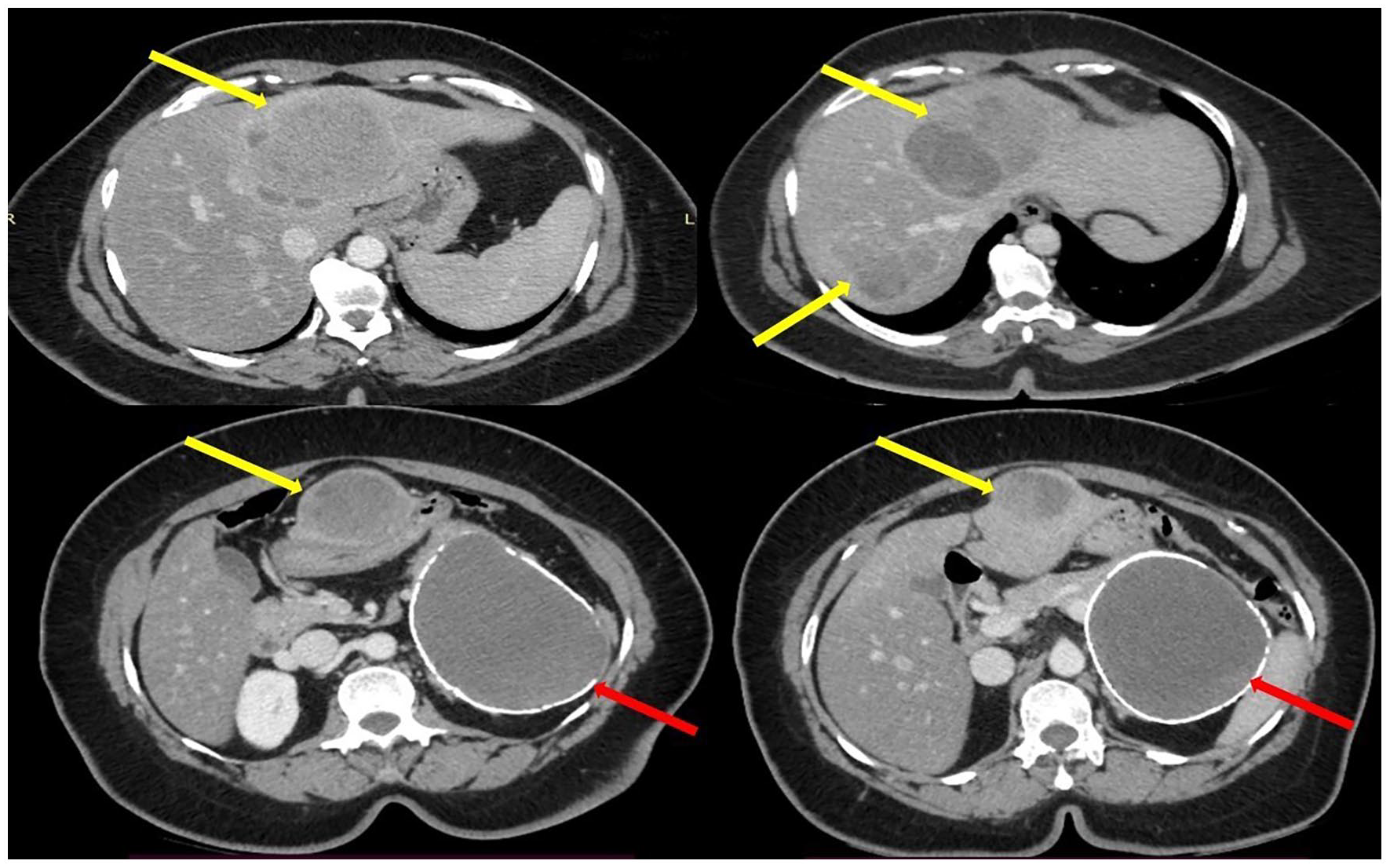
CECT showing multiple deposits on the left liver lobe and the right posterior sector (yellow arrow) and a well-defined cystic lesion with a thin rim of peripheral calcification at the distal body and the tail of the pancreas (red arrow).
The patient was discussed at the hepatopancreatobiliary multi-disciplinary team (MDT) meeting which included hepatopancreatobiliary surgeons, pathologist, radiologist and oncologist. It was concluded that the most probable diagnosis was an HEHE of the liver with a benign pancreatic cyst. As the main stay of treatment for HEHE was surgery, a decision was taken to perform a two-stage resection of liver nodules with a distal pancreatectomy.
After routine pre-operative optimization, a distal pancreatectomy with splenectomy and a left hemi-hepatectomy were performed via open approach (Figure 2). Metastatic deposits of the right hemi liver were not addressed at the primary surgery due to concerns regarding inadequate future liver remnant. Ligation or arterial embolization was not performed prior to intervention as compensatory right lobe hypertrophy following a left hepatectomy was expected. Ligation of the right posterior sectoral blood supply was not performed during the first surgery due to the high risk of liver failure as only the two remaining segments would be functioning. The patient’s initial recovery was uneventful. However, on Day 6, a low volume bile leak was detected and she was discharged with the abdominal drain on Day 10. Subsequently, she was managed with endoscopic retrograde cholangiopancreatogram (ERCP) and biliary stenting as the low volume bile leak was persistent (Clavien–Dindo IIIA).

(a) Left hemi-hepatectomy, (b) distal pancreatico-splenectomy and (c) right posterior sectionectomy.
Histology revealed an SPN of the pancreas which was well circumscribed and encapsulated in most parts but showed focal infiltration of the surrounding pancreatic tissue and peripancreatic adipose tissue. The solid areas were composed of poorly cohesive monomorphic cells with eosinophilic or clear vacuolated cytoplasm. Pseudopapillae were present with aggregation of cells around fibrovascular stalks. The liver deposits were composed of loosely arranged similar cells with presence of pseudopapillae (Figure 3). The findings were further validated by immunohistochemistry (diffusely positive for vimentin, focally positive for with CD10, negative for neuron-specific enolase (NSE) and chromogranin A). Re-imaging with CECT after 2 months showed compensatory hypertrophy of the liver with adequate future liver remnant. The previously noted focal lesions in segments VI and VII were identified without any interval change in size. The new histological diagnosis was discussed at the second MDT. A decision was reached to perform a posterior sectionectomy. A volumetry was performed to assess the future liver remnant which was deemed satisfactory.

(a) H&E stains ×10 showing poorly cohesive monomorphic cells with eosinophilic or clear vacuolated cytoplasm. Pseudopapillae were present with aggregation of cells around fibrovascular stalks. (b) The liver deposit was composed of loosely arranged similar cells with presence of pseudopapillae.
She underwent an uneventful surgery and was discharged on post-op Day 6 (Figure 2). Histology revealed well-defined metastatic deposits from SPN resected with adequate margins. Subsequently, the patient was followed with serial CECT scans with no evidence of recurrence at 18 months of follow-up.
Discussion
SPNs of the pancreas are infrequently seen epithelial tumours with a low malignant potential. The main morphological differential diagnosis is pancreatic neuroendocrine tumour. Therefore, immunohistochemical exclusion of neuroendocrine tumours using neuroendocrine markers is considered important in the diagnosis of SPN. However, SPNs can be infrequently positive for neuroendocrine markers such as chromogranin A. 6 In the present case, diffuse positivity for vimentin, focal positivity for with CD10 and negative stains for NSE and chromogranin A validated the diagnosis. Aberrant nuclear expression of beta-catenin is characteristic in most cases of SPN. 7 However, assessment of beta-catenin was not done as rest of the panel was suggestive of SPN, as immunohistochemical facilities are limited and expensive in our setting. 8 Interestingly, the initial biopsy was suggestive of hepatic epithelioid HEHE which are rare vascular tumours of endothelial origin. The pancreatic neoplasm was initially considered an incidental benign finding. Due to the rarity and overlapping microscopic features, HEHE are usually misdiagnosed with histological examination. 9 This highlights the importance of immunohistochemical examination for accurate pathological diagnosis as it is essential to differentiate synchronous versus metastatic tumours. 10 Furthermore, CECT demonstrated a well-defined cystic lesion with peripheral calcification. Other differential diagnosis for calcified cystic lesions of the pancreas includes mucinous cystic neoplasms, serous cystic neoplasms and intraductal papillary mucinous neoplasm. 11 Fortunately, the patient received the appropriate treatment as both tumours (HEHE and SPN) are usually managed with surgical excision as the first line of treatment. Review of the initial biopsy with the final histology could have given further clues regarding the discrepancy. However, this could not be achieved as the first biopsy was performed at a different centre.
The biological behaviours of SPN of the pancreas are unpredictable. The clinical or histological characteristics have been inconsistent in classifying the tumour biology and aggressiveness. Local invasion of adjacent blood vessels, peritoneal cavity and adjacent viscera are macroscopic features that may suggest an aggressive tumour biology. Furthermore, cellular pleomorphism and increased mitosis are microscopic characteristics implicated in metastasis and recurrence. However, these features are highly unreliable in predicting the aggressiveness of the tumour. Furthermore, absence of these features does not exclude malignant behaviour. Our patient had metastatic disease without significant malignant histological characteristics. However, there was focal infiltration into the pancreatic and peripancreatic tissues.
SPNs of the pancreas are often identified in the advanced stage due to the indolent and asymptomatic nature of the disease. However, due to its low malignant potential, the prognosis is usually favourable. Our patient was diagnosed of SPN at the age of 50 years which is well above the mean age of presentation which is around 20–35 years. Usually, pancreatic neoplasms such as mucinous cystic neoplasms and intraductal papillary mucinous neoplasms are diagnosed during the fifth decade of life.12,13 A case series of 14 patients by Patil et al. 14 described that the age range was 13–45 years with a median age of 20 years. However, another series of 10 patients from Turkey described an age range of 18–71 years with a median age of 39 years, with two patients above the age of 50 years. 15 This may suggest regional variation of age at presentation due to the genetic factors and health seeking behaviour. In females, role of oestrogen in the pathogenesis and growth rate of SPN have been postulated. 16 It is likely that the patient developed the disease at a young age and presented late due to the slow growing nature of the tumour. Despite the late presentation and metastatic disease, the tumour was resectable with favourable prognosis. Therefore, radical excision should be offered even for older patients with metastatic disease due to the favourable prognosis following complete resection.
In general, the best choice of treatment is surgery even in advanced disease. This is because, the adjuvant systemic therapy show inferior response probably due to the low malignant potential of the tumour. The evidence for adjuvant systemic therapy is only restricted to a few anecdotal reports and has shown conflicting results. 4 Various modalities of systemic therapy regimes have been attempted which include cisplatin and 5-fluorouracil and Gemcitabine based therapy. 4 Furthermore, adjuvant chemotherapy with IPE regimen (ifosfamide, cisplatin, VePesid) has been used for multiple peritoneal metastases in the lower abdomen, liver and spleen following initial surgery. 17 The place of neoadjuvant in SPN is controversial and not recommended. It is unclear whether adjuvant systemic chemotherapy should be given routinely to address the potential micrometastasis as few anecdotal reports have shown benefits in non-resectable cases. However, a decision was taken not to give adjuvant chemotherapy as the surgical margins were clear and to date, and the patient has no evidence of recurrence.
Trans-arterial tumour embolization and transcatheter arterial chemo embolization (TACE) have been seldom used for metastatic lesions but were associated with poor outcome.4,18 Radiofrequency ablation (RFA) is considered a safe and effective modality for multiple unresectable liver metastases. 19 However, dedifferentiation and epithelial–mesenchymal transition of the tumour may occur when the RFA is incomplete. 4 Therefore, the utility of RFA should be limited to unresectable tumours. Combined irradiation and chemotherapy for locally advanced pancreatic tumour was found to reduce the tumour size but was ineffective in preventing the advancement of liver metastasis. 20 The reports on survival of metastatic and aggressive SPN and their risk factors are limited. A meta-analysis showed a disease-specific survival of 150 months for aggressive SPN with systemic involvement. 21 Furthermore, the 5- and 10-year survival rates were approximately 70% and 65%, respectively. Patients with metastasis at diagnosis had poorer mean recurrence free survival (22 vs 48 months). 21 However, it was not statistically significant. 21
With regard to surgical treatment, organ sparing surgery such as local resection and enucleation may be performed in small tumours. However, in larger lesions of the tail and body of the pancreas, distal pancreatectomy is required which is often combined with splenectomy. Large lesions in the head of the pancreas would require a pancreaticoduodenectomy. To address the multiple liver metastases in our patient, we performed a radical excision of the primary tumour coupled with staged resection of the liver metastasis due to the concerns related to inadequate future liver remnant. As these are slow growing tumours, the staged surgical resections could be appropriately spaced to allow hypertrophy of the liver remnant and physiological recovery. Thus, staged resection was feasible and effective in our patient with multiple resectable liver metastasis with acceptable morbidity to the patient. The patient developed post-op bile leak; however, it was successfully managed with ERCP and stenting.
Conclusion
SPN of the pancreas with liver metastasis is a rare phenomenon, and there are no guidelines or consensus for management. We described a patient with metastatic SPN of the pancreas who was successfully managed with staged resection. The discrepancy in the histology and cross-sectional imaging led to a pre-operative diagnostic dilemma, and the final diagnosis was reached only after surgical resection. Therefore, clinicians should be aware of possible discrepancies that could be encountered. As these are slow growing tumours, staged liver resection with appropriate spacing was feasible to allow hypertrophy of the liver remnant and physiological recovery. Thus, primary curative radical surgery may be offered for resectable metastatic SPN of the pancreas before considering other treatment modalities. Multi-organ surgical treatment is possible in these patients and could improve overall survival. The survival of patients with and without metastatic disease and their risk factors should be further evaluated.
Footnotes
Author contributions
K.R., M.N., U.d.S. and S.D. contributed to collection of information and writing of the manuscript. U.J., A.P. and C.d.S. contributed to writing of the manuscript and final approval. All authors read and approved the final version for publication.
Availability of data and materials
All data used to support the findings of this study are included within the article.
Consent for publication
Written informed consent was obtained from the patient for anonymized information and accompanying images to be published in this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval and consent to participate
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
