Abstract
Lung ultrasonography (LUS) has been used as a clinical diagnostic tool for the location of pleural fluid and marking patients for a thoracentesis, for decades, but has not been deemed as appropriate for other diagnostic uses. In the context of the COVID-19 pandemic, the necessity of a portable, low-cost, and non-ionizing diagnostic choice is needed to assess patient lungs. LUS has become a strong candidate to fill this diagnostic gap. With the use of Lichtenstein’s bedside LUS in emergency (BLUE) protocol, LUS may have potential to diagnose lung disease and assist with treatment decisions. While evidence of LUS as a COVID-19 diagnostic tool is not conclusive, early diagnostic results are promising. Further research on the use of LUS and the clinical implementation of the technique have a true potential to improve patient outcomes.
Lung ultrasonography (LUS) has been universally employed as a means of locating pleural fluid and marking the patient for a thoracentesis (also known as a lung tap, needle thoracostomy, or needle decompression of the pleural space). These sonographic-assisted therapeutic procedures are most often demonstrated using a current chest radiograph, to provide a panoramic view of the thorax and the suspected location of the pleural fluid. This use of sonography to provide guidance is often performed by a sonographer but could be performed by a pulmonologist or radiologist. Novice sonographers are sometimes intimidated to participate in these guided procedures due to their level of academic training in physics and lack of exposure to this examination.
An understanding of ultrasound physics principles is helpful when imaging the lung. A stated premise is that sound attenuation is higher in the lung, compared to other soft tissue interfaces and this is due to the presence of air. 1 The propagation of the speed of sound is also known to be much lower in lung tissue. 1 This discrepancy has not been considered important because ultrasound does not penetrate lung tissue and many consider it adequate for diagnostic imaging. 1 The pleural interface acts as a specular reflector and as a result, the lung tissue appears rather unstructured, with superimposed artifactual reverberations. It has been stated, for all the physics concepts provided, that the lungs should be avoided during scanning and a major reason is due to the large amount of reflection occuring at multiple interfaces, within the tissue. 2 Given this educational context and general lack of experience with imaging the lungs, it isn’t surprising that many sonographers hesitate to interrogate the lungs.
An additional challenge to implementing LUS has been the lack of empirical evidence related to sonographic assessment of the lungs. The American College of Radiology’s (ACR) Appropriateness Criteria, for specific clinical concerns, provides best practice guidance for providers and is backed by meta-analyses, but the criteria for using LUS for specific patient conditions lacks endorsement compared to other imaging options. 3 This 2018 document provides clinicians with a warning that the effectiveness for detecting central lung infections is limited, due to the aerated lung tissue that exists between the transducer and the pathology. 3 In most of the pathologic variants provided for diagnosis, LUS was either rated as “may be appropriate” or “not appropriate,” 3 which likely dissuades the widespread use of LUS in providing a non-ionizing diagnostic tool for lung pathology. One set of exceptions does exist as the 2019 ACR appropriateness criteria for the utilization of LUS, to detect pediatric lung pathology, has been more highly rated. 4 In most of the subvariations, provided for diagnosis, LUS was either rated as “appropriate” or “may be appropriate.” The 2019 evidence-based guidelines are more promising due to the concern for ionizing radiation in this young population of patients. This acknowledgement of reducing ionizing radiation for examining the lungs is more promising than what was provided for adults in 2009, as the ACR guidelines for the detection of pneumonia only included options for a radiograph or computed tomography (CT). 5
One benefit from the COVID-19 pandemic has been the need to find a portable non-ionizing way to monitor patients with lung disease. This has forced the provider community to reassess the role of LUS, in detecting pneumonia and COVID. The ACR continues to rely on infrequent radiographs and CT of the chest. 6 In a set of 20202 guidelines, for the diagnosis of COVID-19 infiltrates, the ACR guideline reaffirms that there is no diagnostic role for LUS. 7 As scientific evidence lags behind the potential for use of LUS in detecting lung pathology and specifically, COVID-19 residual disease, it is important to continue to update and share our experiences. This will help clinicians to move beyond using sonography to mark a lung for a pleural fluid tap and include LUS for diagnostic surveillance. The Centers for Disease Control and Prevention (CDC) state that post-COVID conditions are a wide range of new, returning, or ongoing health problems people can experience 4 or more weeks after initial infection with the SARS-CoV-2 virus that causes COVID-19. 8 The significant number of patients living with the wide spectrum of COVID-19 residual disease could have improved outcomes if an effective a diagnostic tool was provided. We need to continue our use of LUS for these patients as well as gathering empirical evidence, to upgrade current practice guidelines.
Methodology for Conducting LUS—Reliance on Lichtenstein’s Blue Protocol
The POC sonographic application of evaluating the thorax for a possible pneumothorax is also part of the extended focused assessment with sonography in trauma (eFAST) exam. 9 The technique described to assess the thorax for determining a pneumothorax is to image the parietal and visceral pleura with sonography (see Figure 1). Additionally, it is important to watch for dynamic movement of the underlying lung tissue. 9 An additional sonographic sign in evaluating the lung for a potential pneumothorax is the documentation of a comet tail artifact. This is commonly referred to as B-lines that run perpendicular to the pleural line (see Figures 2 and 3). An additional method for documenting the movement or lack of sliding of lung tissue is the use of M-mode. Using M-mode is affirmative for a pneumothorax based on recording a series of parallel horizontal lines, on the tracing (see Figure 4). 9 This is compared to the expected normal movement of the lung that creates a disrupted tracing that has been compared to the pattern of a sandy beach intersecting with the rolling waves of the ocean. 9 These aforementioned imaging techniques have been reported to have a 66% sensitivity and 100% specificity.10-12
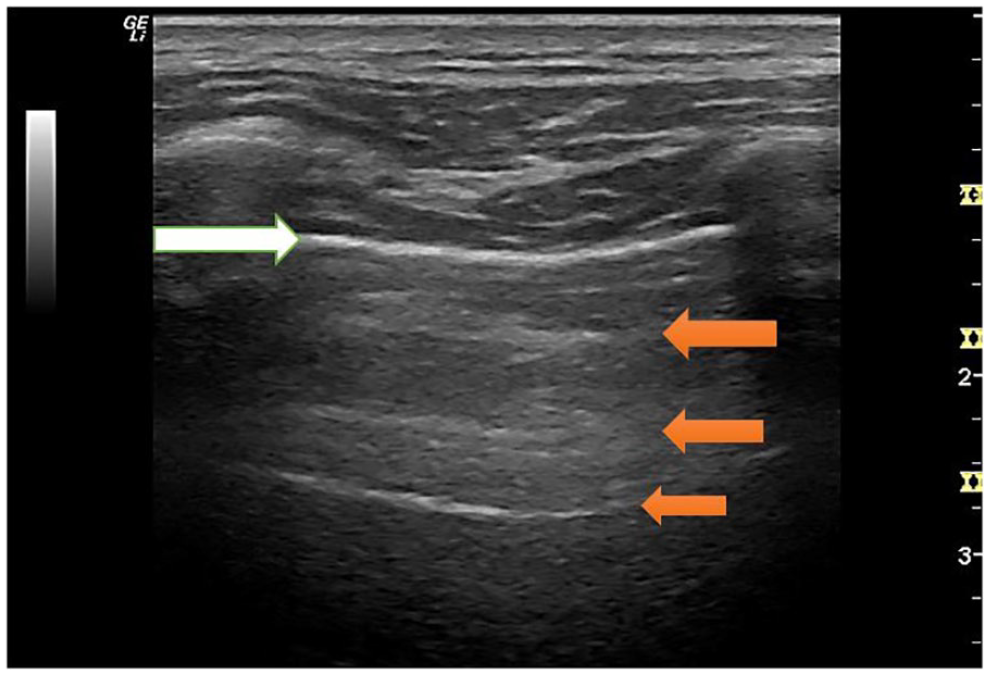
This is an upper BLUE point, intercostal sonographic image of the adult lung. The white arrow points to the well-defined pleural line interface of the lung tissue. The orange arrows indicated the A-line sonographic artifact, associated with a well maintained pleural interface. The rib shadows are seen at the right and left of the pleural interface and this view has been called the “bat-wing sign.” BLUE, bedside lung ultrasound in emergency.
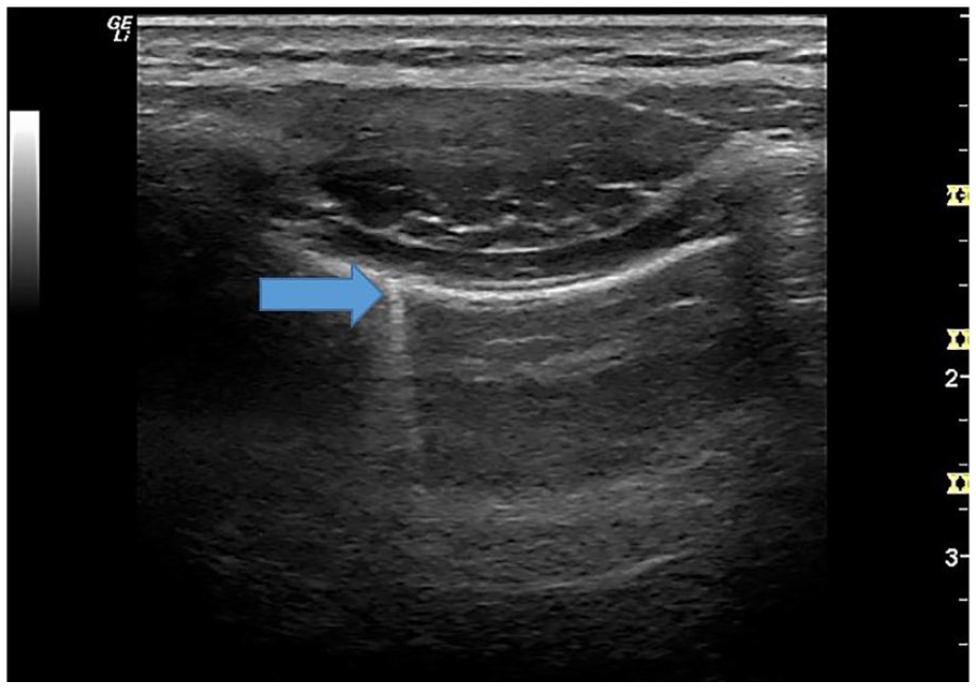
The small break captured on this upper BLUE point, intercostal sonogram of the adult lung demonstrated a resulting B-line artifact. The blue arrow points to the resulting B-line artifact from the break in the pleural line. These breaks in the pleural can be noted but several of these breaks in the pleura have been associated with COVID-19 infection. BLUE, bedside lung ultrasound in emergency.
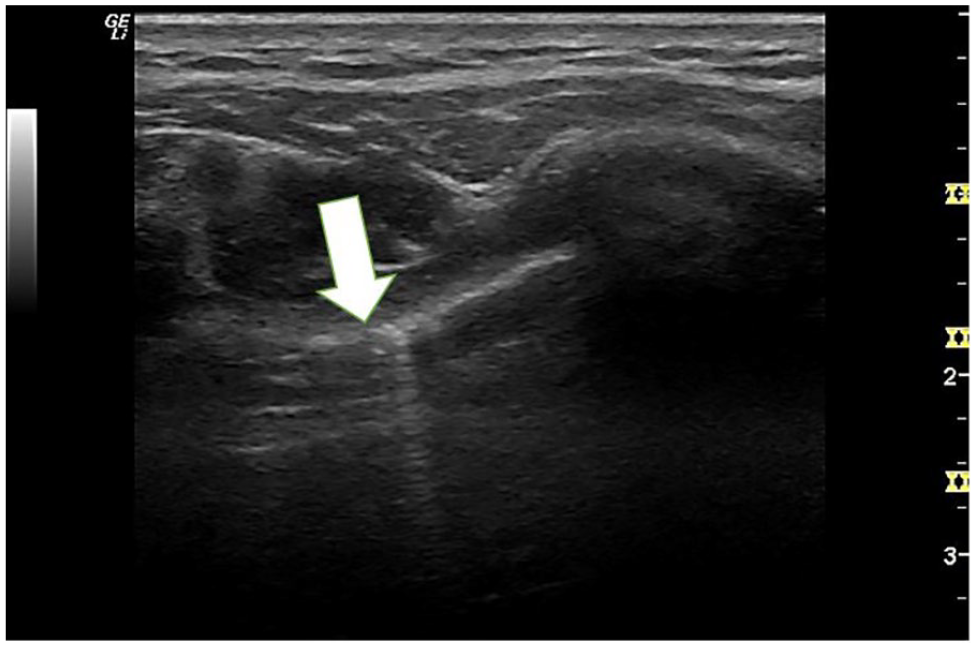
This intercostal view of the pleural interface appears to have tiny disruptions in the echogenic line, which has been seen with loss of aeriation (as well as COVID). The white arrow points to the pleural interface and note the irregularities along the line and B-line artifacts.
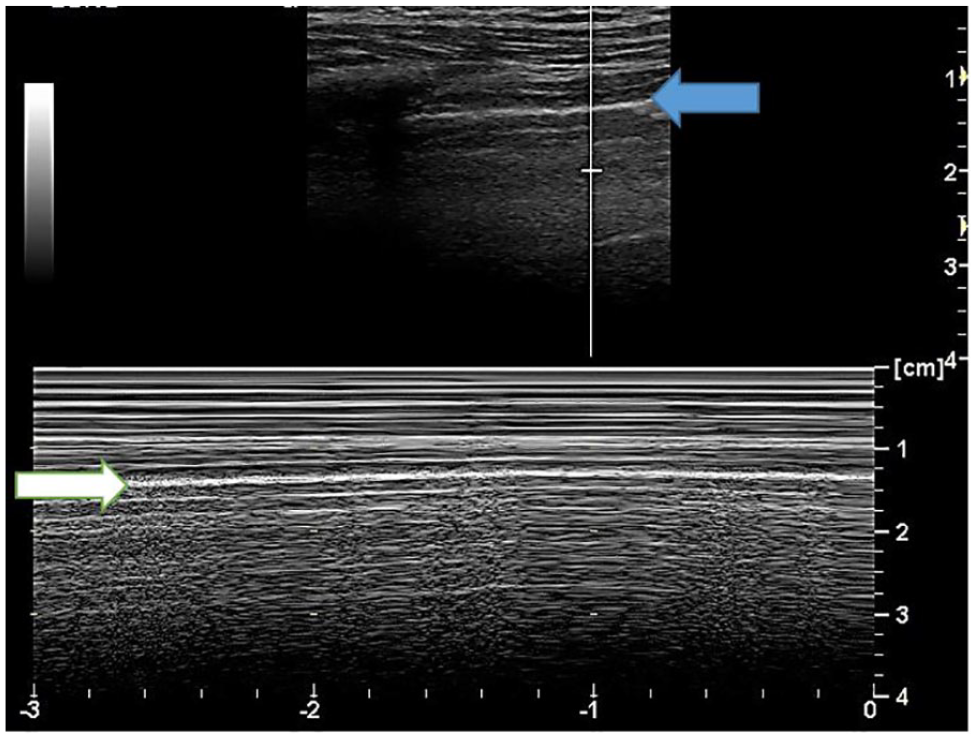
The M-mode tracing that allowed for documentation of the adult lung sliding (aeriation of lung tissue). The movement provided on the time line creates the pattern known as the “seashore sign.” The white arrow indicates the pleural line and the movement captured by M-mode. The blue arrow indicate the gray scale pleural interface.
Beyond just using LUS as a diagnostic tool to triage emergency department patients, Dr. Lichtenstein is considered the “father of LUS,” based on his international practice and seminal publication on the technique. Lichtenstein’s imaging technique is referred to as the bedside lung ultrasound in emergency (BLUE) protocol for LUS.13,14 To get the maximum diagnostic benefit from a LUS, it is highly advocated to follow the BLUE protocol and use all the scanning points to assess the underlying lung tissue. (
Utilizing LUS to Reduce Absorbed Radiation Dose in Patients With Lingering COVID-19 Symptoms
SARS-CoV-2 is a viral infection that causes COVID-19. This novel coronavirus causes a wide-ranging disease course, some infected individuals require hospitalization and supplemental oxygen, while others have few or no symptoms. Age and co-morbidity including cardiovascular disease, obesity, and diabetes have been identified as risk factors for severe reactions. 15 Young healthy individuals have also suffered from becoming critically ill, particularly when comorbidities are present. A significant proportion of individuals with COVID-19 experience damage to their lungs, heart, or kidneys,16,17 even if their disease course is not severe. Global diagnostic data taken from hospitalized patients has helped to unravel some of the mysteries of this disease. What remains elusive is the degree to which residual disease and chronic pathologic changes have developed in those adults who were infected but not hospitalized. “Long COVID” is a new term18-20 that has been developed to describe those patients, reporting to primary healthcare offices with the lingering effects of the disease.
Potential Use of LUS for Evaluating Patients With COVID-19
There is evolving evidence that point of care lung ultrasound (POCUS) is the ideal imaging modality to diagnose and monitor patients with respiratory illness due to SARS-CoV-2 and other pulmonary diseases such as pneumonia, pleural diseases, and interstitial lung disease (ILD).21,22Although a reverse transcription-polymerase chain reactive (RT-PCR) test is required to confirm COVID-19 diagnosis; this test has low sensitivity. For that, researchers have recommended using chest CT as a diagnostic tool to confirm pneumonia and COVID-19 since it has better sensitivity than the nasopharyngeal swab. 23 However, there are significant concerns with adopting the use of CT, especially for children and pregnant women. Using CT or chest radiography regularly in pediatrics should be carefully evaluated, as increased use of these modalities could increase potential radiation exposure. 24 Additionally, COVID-19 is a global pandemic and the inaccessibility of CT equipment in rural areas with low to medium income make it inconvenient to use it as a diagnostic tool. Consequently, physicians sometimes use a chest radiograph as the diagnostic tool to replace CT; however, there is concern that the chest radiograph doesn’t have enough sensitivity and specificity to confirm disease. 25 Moreover, while the chest CT has better accuracy compared to LUS and the chest radiograph, a chest CT of children has disadvantages, such as the possible need for sedation and high cost. 24 LUS findings in patients with COVID-19 and pneumonia can help facilitate the triage of patients and isolate them from spreading disease, without the need for further investigation. Considering LUS as a first line diagnostic tool could help rapidly identify the infection and start pharmacological therapy for those who present with severe respiratory symptoms. 25 LUS can also be used to monitor the dynamic changes of the pulmonary mechanics of the ventilated patients by using the BLUE protocol and this has the potential to aid in recruitment maneuvers. LUS could further aid physicians in deciding to place patients in a prone position given the amount of consolidation imaged in the posterior segment of the lungs. Turning patients in the prone position while on a ventilator has been found to be beneficial for COVID-19 patients. 26
Bedside LUS should be done following a systematic approach to evaluate the whole thorax. Following the BLUE protocol, each hemithorax is divided by the axillary lines into anterior, posterior, and lateral. 14 Then each area is further subdivided into two halves to produce 5 lung zones per hemithorax for a total of 10 zones for both lungs. 27 Some researcher advocates 14 lung zones where the posterior side is divided into 3 zones. 28 (see Figure 5). A curvilinear transducer is the preferred choice to evaluate both the pleura and the lungs tissues. Linear transducers and low-frequency transducers could be used to highlight the peripheral lung parenchyma or the pleural pathologies. Positioning the U.S. transducer between the intercostal space, perpendicular to the ribs would maximize the surface area in 1 single scan. Another transducer position is parallel to the ribs or in a transverse orientation. Using both orientations would allow for a thorough lung and pleura examination. Scanning the lung according to the BLUE protocol as described, will help score the LUS images and eventually will benefit in diagnosing and determining the severity of COVID-19.
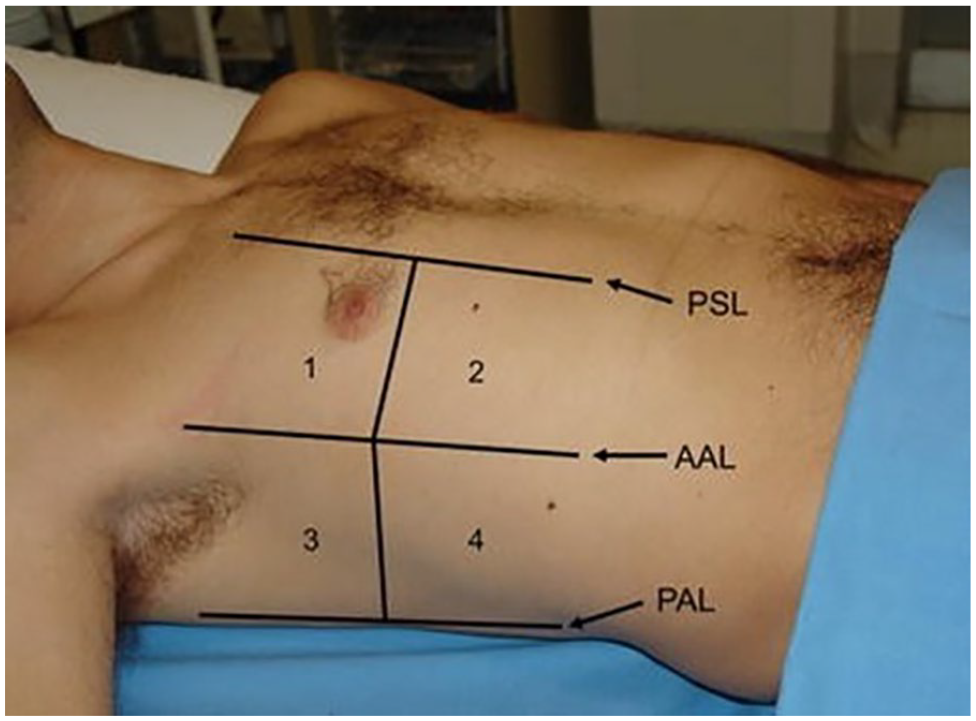
A diagram of the BLUE protocol zones on the anterior portion of the chest. A posterior view provides a fifth zone (superior and inferior) and provides 12 zones for a full chest examination. BLUE, bedside lung ultrasound in emergency. This image is reprinted with permission.
How LUS Can Be Applied to Monitor Disease in Pediatric Patients
There are few published articles that can provide evidenced based guidance for applying LUS to the diagnosis and surveillance of pediatric patients with COVID-19. One seminal meta-analysis that has recently published describes the most common imaging appearances seen in children infected with the virus. 29 Based on 131 quality publications, Hoang et al noted that diffuse bilateral ground-glass opacities were the most common appearance noted on the chest CT for children. 28 These chest CT results were coupled with the disappointing normal appearing chest radiograph for these pediatric patients. 29 It was also noteworthy that the analysis of all the pediatric patients across all the published studies found that 72 of the patients had co-infections attributed to influenza, 29 which only complicates the diagnostic assessment. This highest level of evidence did lack the inclusion of LUS, given its infrequent use and lack of published studies. Nevertheless, the role for LUS seems very important given the lack of evidence on a chest radiograph and the need to monitor resolution of the chest CT findings. Another important point raised in the Hoang et al meta-analysis was that the average age of the patients was between 8 and 9 years of age and they were largely asymptomatic upon initial assessment. 29 This would underscore the need for an alternative non-invasive, non-ionizing imaging technique for screening these young patients with suspected exposure to the COVID-19 virus.
Besides the need for acute screening of children is the growing evidence that children are living with long lasting conditions attributed to COVID-19. In an Italian study of children that contracted COVID-19, a majority of the cohort reported at least one persistent complaint even after 120 days post viral infection. 30 In this cohort, the most common symptoms were fatigue, muscle and joint pain, headache, insomnia, breathing issues and heart palpitations. 30 These sorts of clinical presentations would also make the use of LUS important for the purpose of surveillance and clinical management of these patients. Name et al have published a textbook on their clinical use of LUS. They gathered their expert advice through the use of LUS at the bedside of critical ill pediatric patients. They provide this lower level of evidence based on their experience. The most common LUS findings among hospitalized pediatric patients with COVID-19 are as follows: pleural line irregularities, B-lines, patchy areas of white lung, subpleural consolidations, occasionally air bronchograms, pleural effusions. 31 Based on Buonsenso et al’s clinical experience, they have found that among pediatric patients, diffuse sonographic short and long artifacts, as well as isolated small subpleural consolidations are the most commonly encountered. 31 Given these varied levels of evidence, the need continues to implement the use of LUS and report clinical findings so that pediatric patients are provided with both an expedited diagnosis as well as surveillance for “long COVID” symptoms. The protocol for imaging children in both the inpatient and outpatient environment is the same as adults. Buonsenso et al’s recommendations for scanning are that LUS should be performed in the longitudinal and transverse imaging planes. 31 When examining young patients, especially if they are uncooperative, sonographic imaging should be completed intercostally, to insure that the widest surface of the lung is covered. 31 (See Figures 6 and 7)
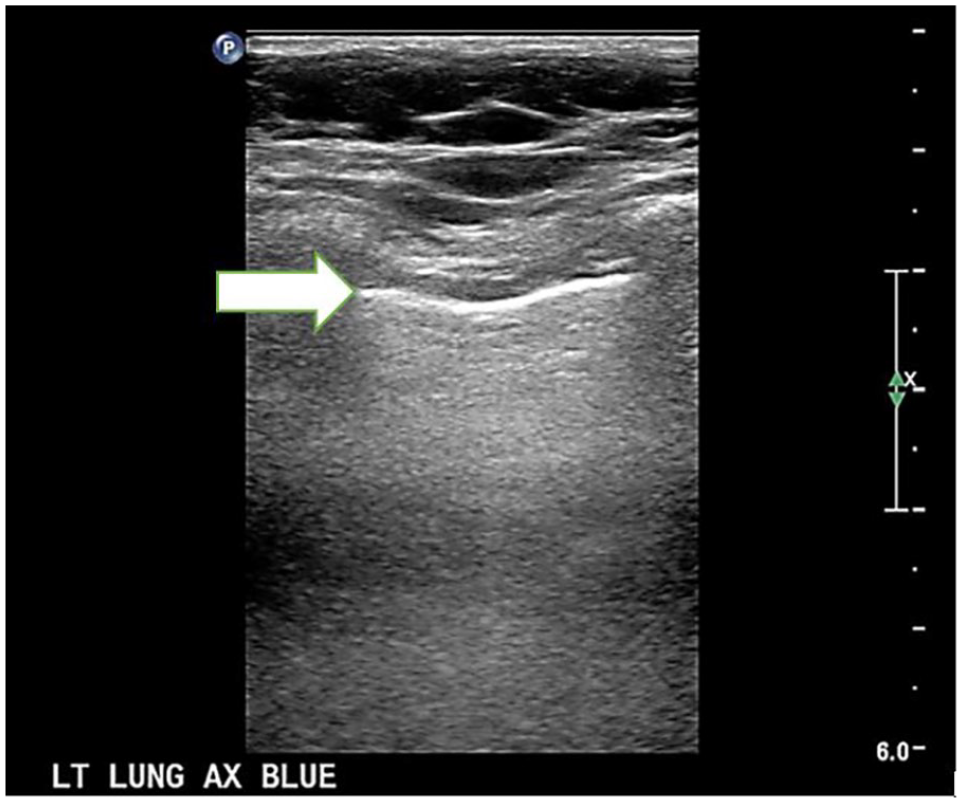
An 11-year-old COVID survivor provided an upper axillary, intercostal sonographic image of the lung tissue. The white arrow indicates the intact pleural line and the small A-line artifacts that project below it.
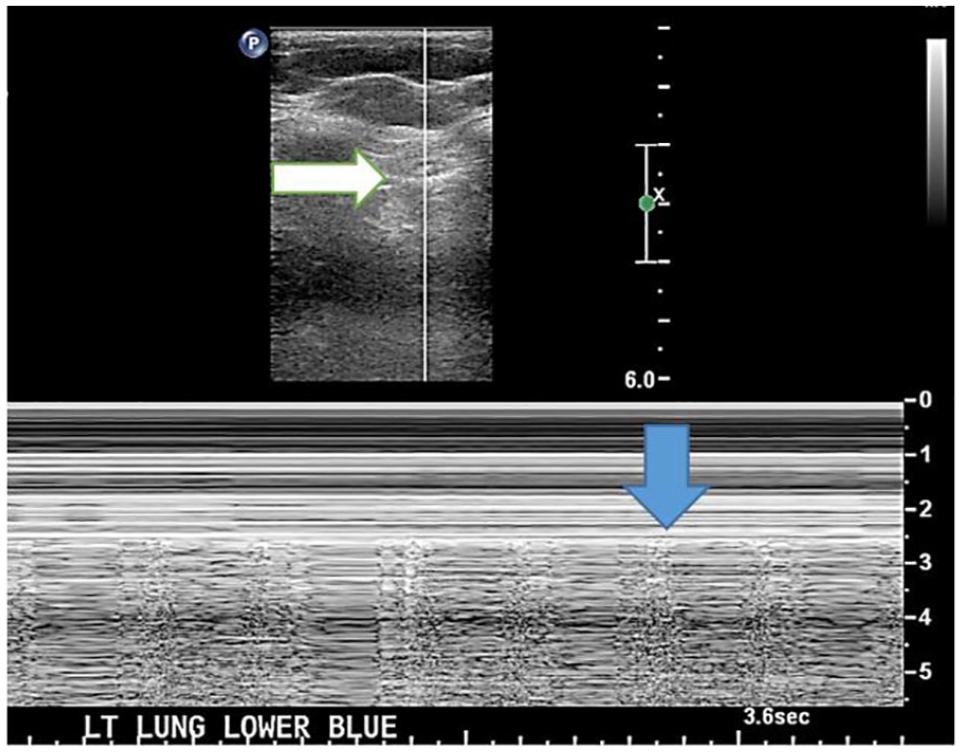
An 11-year-old COVID survivor provided this M-mode tracing to document the aeriation of the underlying lung tissue and the “seashore sign” artifact. The blue arrow in the gray scale image points to the pleural line and the blue arrow points to the M-mode tracing of that same interface.
Potential for Scoring the LUS Diagnostic Findings
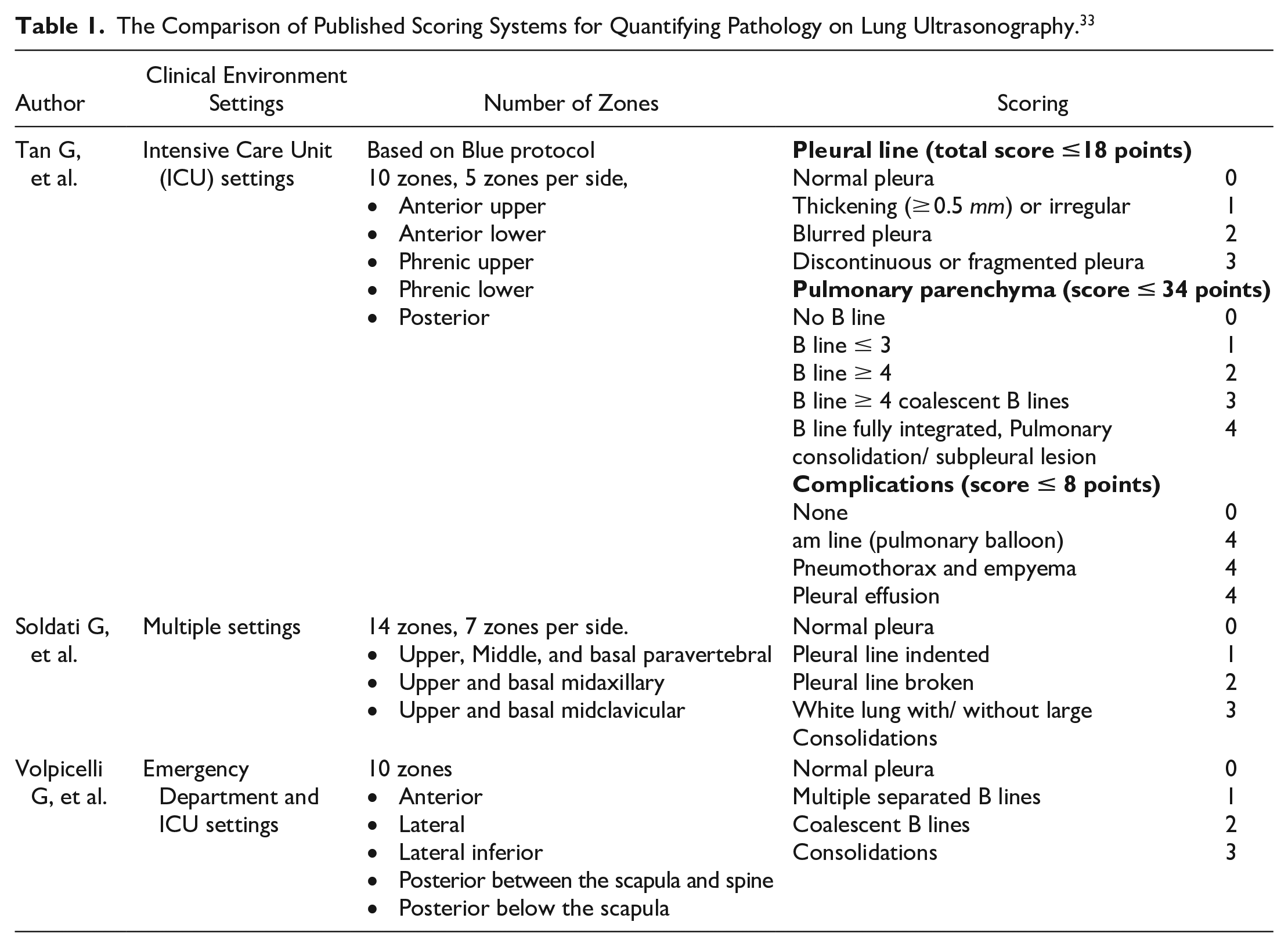
COVID-19 may cause inflammation and pulmonary congestion; consequently, lung tissue degradation, fibrotic changes, and interstitial thickening of lung tissue may develop. These rapidly progressive pulmonary changes might appear as ground-glass opacities or lung consolidation on a chest radiograph or CT. In LUS, the typical findings are pleural line thickening, fragmentation, irregularities, sub-pleural consolidations, and more than 3 coalescent B-lines. 32 (See Figure 8) These findings were identical in adults as well as children, however, these findings were less common in children than adults. 32 LUS findings correlate with the extent of the disease severity and parenchymal lung injuries. LUS scoring system is a semiquantitative method to evaluate the pathological changes of the lungs and the pleura. This scoring system was used before? COVID in the intensive care unit (ICU) to evaluate patients with acute respiratory distress syndrome (ARDS). 33 With the COVID-19 surge, ER and ICU physicians tried to find a measure to assess the changes of the pulmonary system, monitor the prognosis of the disease, evaluate the pulmonary mechanics, the effectiveness of the treatments, and eventually ease the burden on the medical health system34-36 (See Table 1).
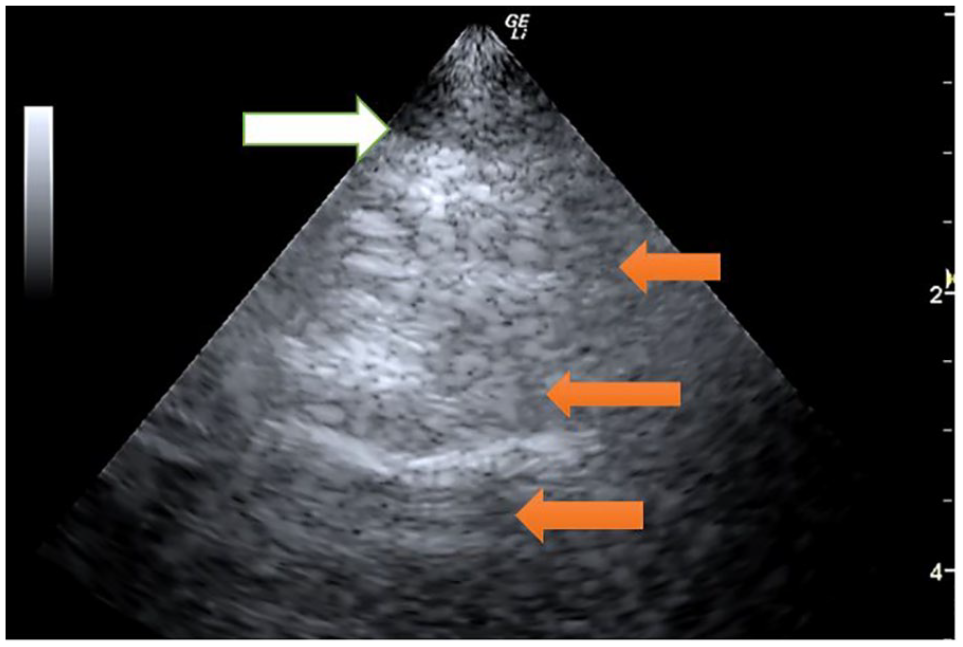
A 8-year-old COVID survivor who still complains of shortness of breath and some times of unexplained fatigue. Note the white arrow pointing to the pleural line which is difficult to see clearly maintained. This lung image was created with a phased array transducer to better fit into the smaller intercostal spaces. The orange arrows indicate areas that
The Comparison of Published Scoring Systems for Quantifying Pathology on Lung Ultrasonography. 33
Summary and Next Steps
More evidence is needed regarding the use of LUS to provide a compatible diagnosis of COVID-19 pathology, given the use of either a chest radiography or CT. The publication of evidence of the LUS imaging artifacts, attributed to disease, and the scoring of these images will be highly influential for the revision of clinical guidelines. Certainly there is an important diagnostic role for LUS in the treatment and surveillance of adults and children suffering from lingering COVID-19 symptoms (termed “long COVID”). Sonography continues to be an important non-invasive, non-ionizing diagnostic choice but needs additional evidence as to the efficacy of detecting and monitoring COVID-19 pathology.
Peer Reviewer Guarantee Statement
The Editor/Associate Editor of JDMS is an author of this article; therefore, the peer review process was managed by alternative members of the Board and the submitting Editor/Associate Editor had no involvement in the decision-making process.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
