Abstract
Objective:
The aim of this study was to assess the diagnostic accuracy of lung ultrasonography (LUS) and a local clinical triage protocol complicated by varied clinical severities of COVID-19, in pregnant patients who had computed tomography (CT) and reverse transcription polymerase chain reaction (RT-PCR) results.
Materials and Methods:
This retrospective cohort study was conducted in a tertiary maternity unit, during the pandemic, between March 2020 and November 2021. The pregnant patients included in the study were diagnostically categorized as asymptomatic, mild, moderate, severe, and critical, upon hospital admission and their presenting disease severity. All of the patients, as well as those with mild and asymptomatic severity, LUS were evaluated accuracy based on their RT-PCR results.
Results:
In total, 1531 pregnant patients underwent a primary emergency department clinical evaluation, in the pandemic clinic. The sensitivity, specificity, positive predictive value, and negative predictive values for LUS were 70.5%, 72.45%, 88.1%, and 45.8%, respectively.
Conclusion:
In this large cohort of patients, LUS was shown to be quick, safe, effective, and reproducible diagnostic tool for the screening of COVID-19 pneumonia, in this obstetric practice. This clinical evidence also provided a new set of triage strategies, during the pandemic, for pregnant patients.
Over 764 million people have been diagnosed with COVID-19 worldwide as of January 7, 2024, and over 7 million have died as a result. 1 In the early days of the SARS-CoV-2 pandemic, clinicians developed rapid clinical algorithms from their experiences with previous coronavirus outbreaks. 2 For the time being, the SARS-CoV-2 pandemic has subsided, but the mutated virus continues to circulate.
Reverse transcription polymerase chain reaction (RT-PCR) testing is considered the diagnostic gold standard for detecting the COVID-19 infection, and the symptomatology of patients is also important factor in making the ultimate diagnosis. 3 The RT-PCR test is time-consuming, and it has high false negative rate. 4 The symptoms related to SARS-CoV-2 infection and the complaints related to pregnancy can be confusing for patients and clinicians. 5 The diagnosis of COVID-19, during pregnancy, might be considered controversial, due to the physiological changes occurring in pregnant patients and the potential for adverse pregnancy events. 3 The diagnosis can be established based on a clinical evaluation, with chest imaging (e.g. radiography, computed tomography [CT], and sonography), which aids in the identification or exclusion of pulmonary complications.3,6 Regarding the use of CT in pregnancy, the American College of Radiology (ACR) does not recommend it as a primary imaging method in the diagnosis and follow-up of COVID-19. 7 Related professional associations, such as the American College of Obstetricians and Gynecologists, the Royal College of Obstetricians and Gynecologists, and the Society for Maternal-Fetal Medicine have recommended that the use of chest CT should not be avoided, based on clinical necessity. 7 SARS-CoV-2 pneumonia develops at a later stage of the disease, therefore, choosing CT for the initial diagnostic tool, to detect suspected COVID-19, in pregnant patients, is a challenging decision for physicians and patients.
Lung ultrasonography (LUS) has been found suitable for the evaluation of SARS-CoV-2 pneumonia, as pulmonary changes most likely occur in the subpleural localization 8 and obstetricians have familiarity with the use of sonography.9,10 In clinical practice, obstetricians can examine the patient’s lungs immediately following an obstetric sonogram. This would enable these physicians to identify the presence of abnormalities and specific diagnostic patterns, as well as determining whether additional multidisciplinary treatments are needed. 11 In this study, the aim was to assess the diagnostic accuracy of LUS and a local clinical triage protocol complicated by varied clinical severities of COVID-19, in pregnant patients who had CT and RT-PCR results.
Materials and Methods
This retrospective study was conducted in a tertiary maternity unit, which was accepted as a pandemic clinic, between March 2020 and November 2021. The pregnant patients admitted to the clinic or referred to the clinic’s emergency department, due to suspected COVID-19, were recruited. The patients who were not pregnant and who could not be evaluated with LUS were excluded.
The hospital’s electronic information repository for the patients’ archived medical records were reviewed and analyzed. Data were procured on the demographic and clinical characteristics of the patients, as recorded in the electronic medical records. The gestational trimesters were categorized as follows: first trimester 0 to 13+6 weeks, second trimester 14 to 27+6 weeks, and third trimester ≥28 weeks.
This study was approved by the local ethics committee and the Ministry of Health’s COVID-19 Scientific Research Evaluation Commission (IRB# 2020/287).
Clinical Management
The diagnosis and treatment of pregnant patients, infected with SARS-CoV-2, was guided by the local clinic’s protocol, based on the current guidelines of the World Health Organization (WHO) and the Turkish Ministry of Health, at the time of their clinical management. An obstetric sonogram, LUS, and nasopharyngeal swab RT-PCR test were performed in the emergency department, 12 and a chest CT was recommended, as part of the pandemic conditions.
The pregnant women included in the study were diagnostically classified as asymptomatic, mild, moderate, severe, and critical, upon their admission to the maternity unit’s emergency department, which was based on their clinical evaluation and disease severity. 3 The clinical management patient groups are presented in Table 1.
Clinical Management Groups of the SARS-CoV-2 Infection.

Lung Ultrasonography
Lung ultrasonography was performed by evaluating the lungs with a EA 720 ultrasound equipment system (Esaote SpA, Genoa, Italy) and a convex transducer with a bandwidth frequency of 1 to 8 MHz, utilizing the equipment’s obstetrical preset. All obstetrics residents and specialists attended a one-day didactic session led by a radiologist, with more than six years of expertise, and a colleague (M.Y.), with experience using LUS to assess pregnant patients. 10 Each patient’s lung involvement was scored between 0 and 3, based on the LUS diagnostic results. 13 The LUS examination followed the routine obstetric sonogram, at admission to the emergency department or within 24 hours by obstetrics residents or specialists, based on pandemic conditions. The 14 anatomic locations covered using LUS were 3 posterior, 2 lateral, and 2 anterior in the hemithorax and intercostal spaces in supine, right lateral, and left lateral positions, for at least 10 seconds. Following the LUS, the highest diagnostic score was recorded for each area, such as anatomic landmark #1 = score 0, anatomic landmark #2 = score 1, as an example. 10 An LUS score of 0 was deemed normal, a score of 1 was mild, 2 was considered moderate, and 3 was considered severe lung involvement, based on the work of Soldati et al. 13 After the overall LUS evaluation, the patient’s highest lung score was used as part of the clinical triage protocol. Definitions of the LUS scores are summarized in Table 2, and images related to LUS scores are presented in Figures 1 to 4.
The Definition and Scoring Used for Assessment of the Lung Sonograms.

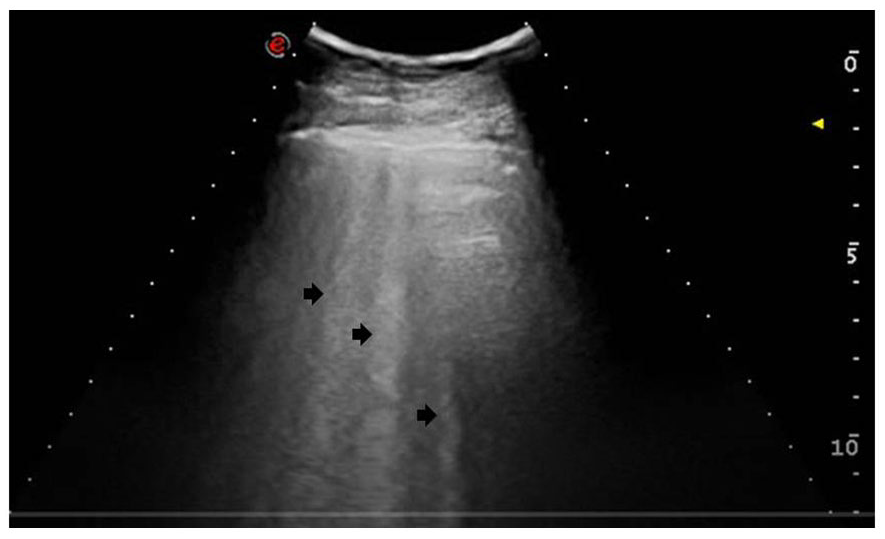
The lung sonogram provided demonstrates a diagnostic score of 0. This is based on the physiological A-line artifacts that are marked with arrows. A normal pleural line is also noted.
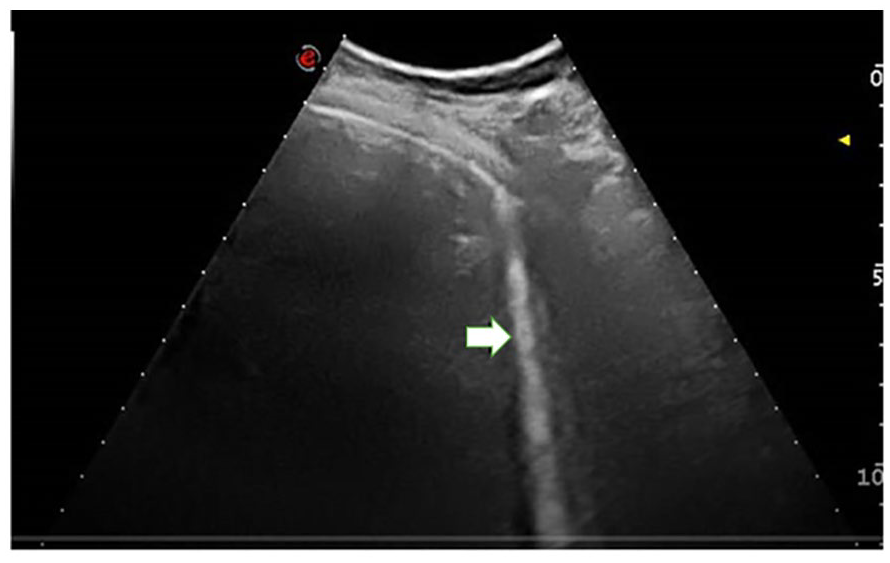
The lung sonogram provided demonstrates a diagnostic score of 1. This is based on the appearance of a thick, irregular pleura and the B-line artifacts, which are marked with an arrow.
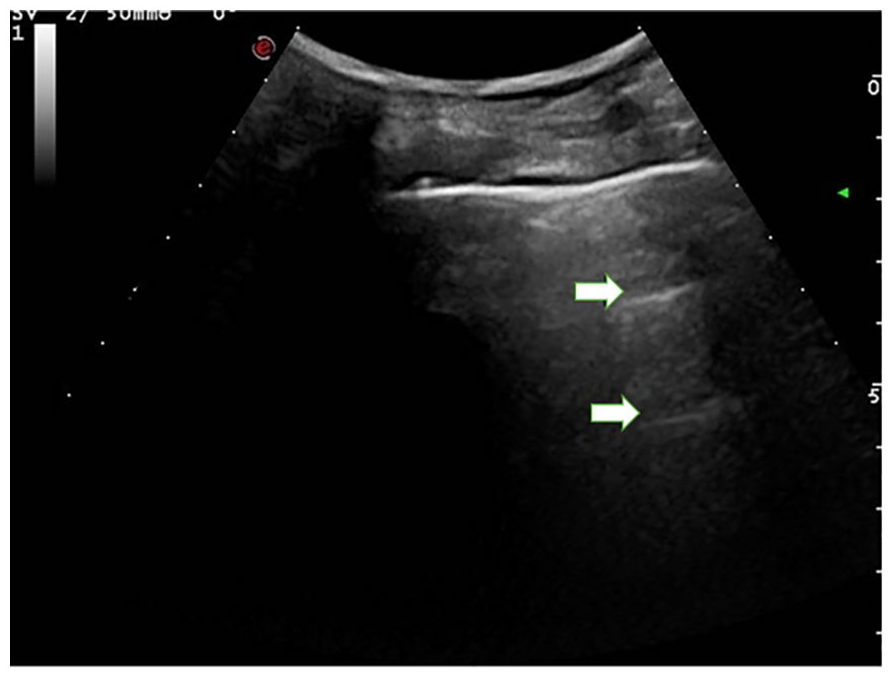
The lung sonogram provided demonstrates a diagnostic score of 2. This is based on the broken and thickened pleural line and the B-lines artifacts, which are marked with arrows.
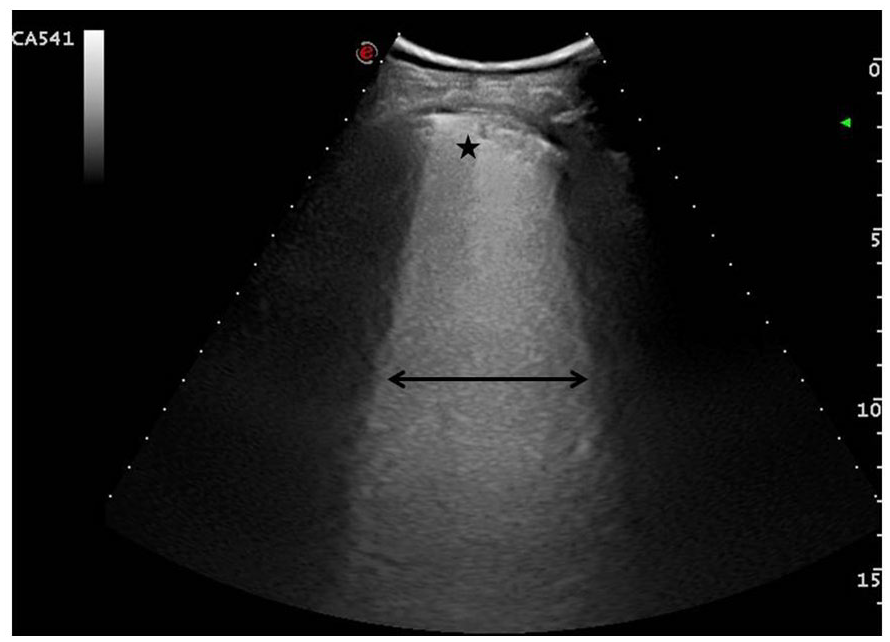
The lung sonogram provided demonstrates a diagnostic score of 3. This is based on the sonographic appearance of a “white lung” pattern and is indicated by a double-headed arrow. A star marker is placed to indicate the broken portion of the pleural line and a small area of subpleural consolidation.
Pregnant patients evaluated with LUS were grouped as LUS “positive” (LUS scores of 1, 2, and 3) and LUS “negative” (LUS scores of 0), as a standard evaluation for statistical analysis. However, asymptomatic patients with an LUS score of 1 were accepted as LUS “negative” in the clinic’s triage protocol, and this analysis was made separately. Distribution of patients, based on the clinic’s triage protocol, is presented in Figure 5. In addition, patients with mild and asymptomatic clinical severity were analyzed for LUS accuracy based on their RT-PCR results. The other clinical severity groups were excluded due to reasons provided in the study results.
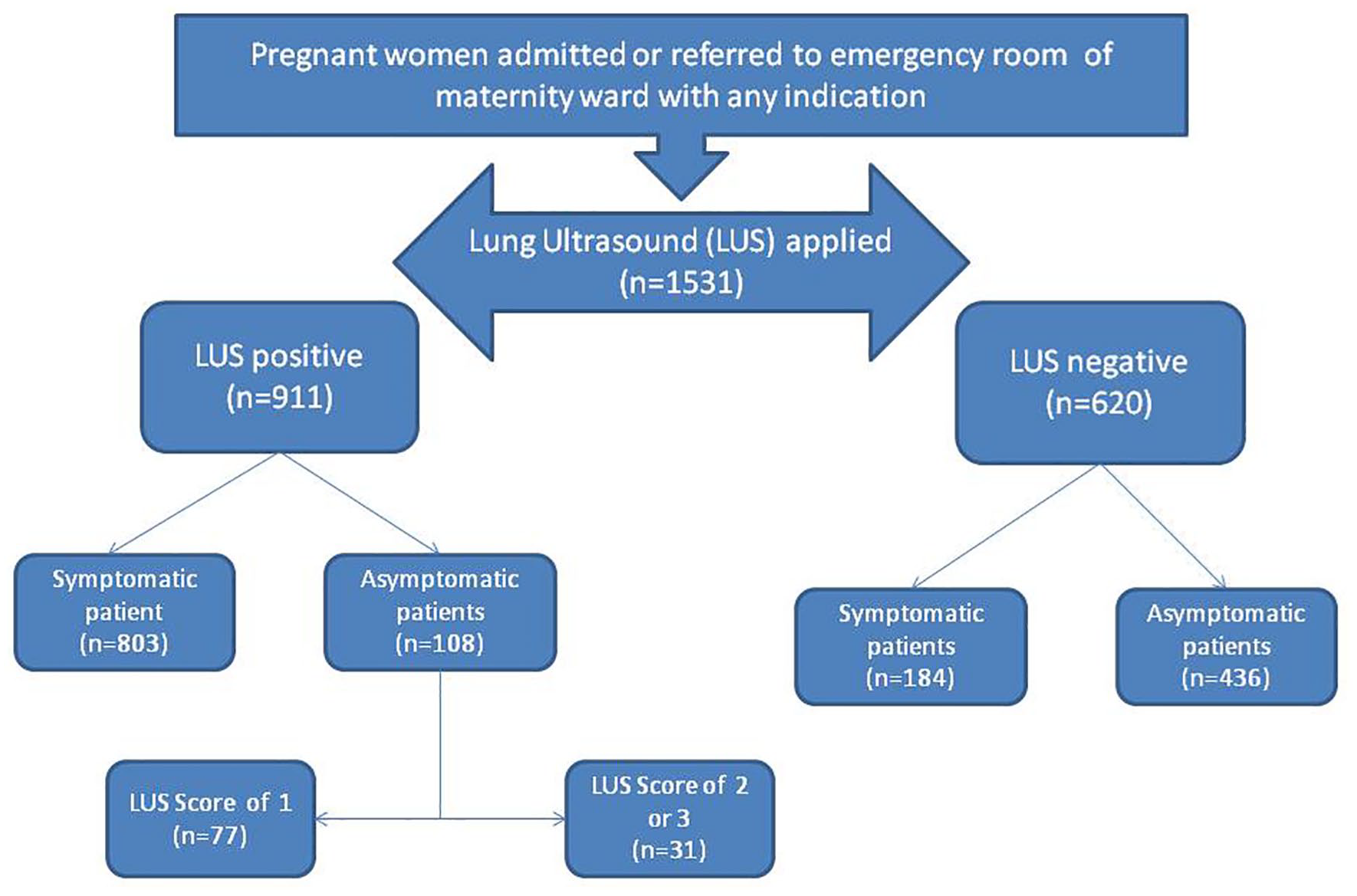
The distribution of the pregnant patients enrolled in the study based on their lung sonogram and the clinical triage protocol.
Computed Tomography
A chest CT was recommended for the diagnosis of SARS-CoV-2 pneumonia, for all pregnant patients, admitted or referred to the emergency department, under pandemic conditions.
Computed tomography images were acquired with the patient in the supine position, utilizing full inspiration and without the use of contrast agents. The diagnostic evaluation was performed by a radiologist. Pregnant patients were examined on an Optima 520 or Ingenuity Core 128 CT equipment system (Optima-GE Healthcare, Waukesha, Wisconsin or Ingenuity-Philips Healthcare, Koninklijke Philips N.V., Amsterdam, The Netherlands) and the imaging parameters included an 80 kV tube voltage, 50 mAs automatic tube current modulation, 5 mm slice thickness, 5 mm slice interval, 16, 36.0 display field of view (DFOV) and 512 × 512 matrix, a helical acquisition was used (See Figure 6 for CT and LUS images). Personal protective lead shields were employed for the thyroid, abdomen, and pelvis. The dose-length product (DLP) ranged from 25 to 100 mGy. Patients exhibiting SARS-CoV-2 pneumonia-related findings on their CT examination were categorized as CT positive, whereas those without such findings were deemed as CT negative for statistical analysis.
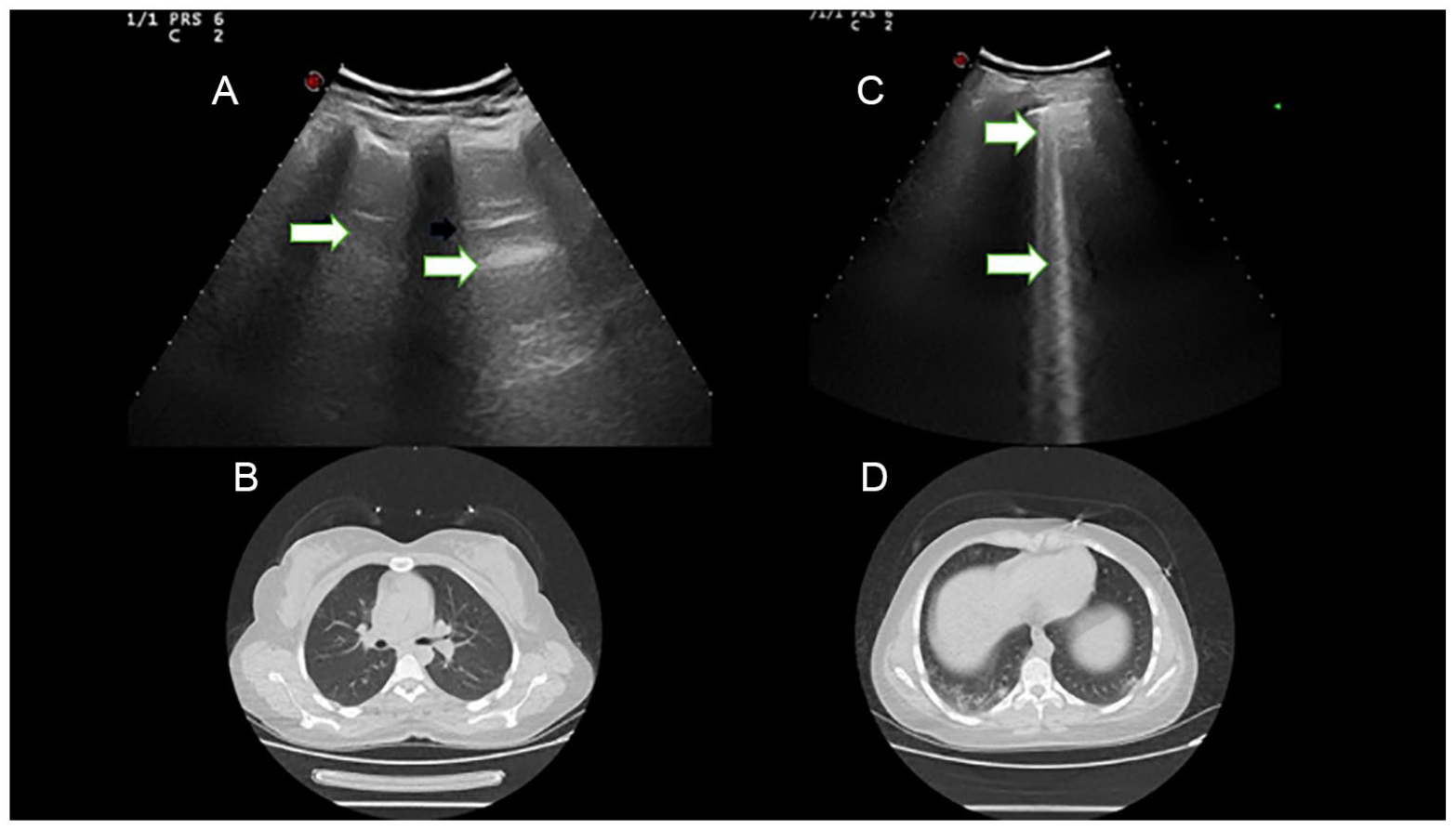
A selection of images from a study patient are provided for review. (A) The lung sonogram is taken of the right upper lung segment. The image demonstrates physiological A-line artifacts that are marked with arrows. This image was given a diagnostic score of 0. (B) A chest computed tomogram (CT) is provided without findings in the upper segmental lobes, of the lungs. (C) An additional lung sonogram taken on the same patient, at the right lower lung segment. This image demonstrates a thickened pleural line and B-line artifacts marked with arrows. This image was given a diagnostic score of 2. (D) Another CT image slice, depicting the lower segment lobes, of the lungs. This image demonstrated characteristics of viral pneumonia, showing peripheral “ground glass” opacities and patchy infiltrations, particularly in the right basal lobe.
Statistical Analysis
IBM SPSS Statistics version 25.0 (IBM Corporation, Armonk, New York) was used for statistical analysis. The continuous variables were presented as mean ± standard deviation, and the categorical variables were presented as frequencies and means. Categorical variables were analyzed with chi-squared and Fisher’s exact tests according to the assumption of normality. Diagnostic accuracy measures (sensitivity, specificity, predictive values, and likelihood ratios) with corresponding 95% confidence intervals (CIs) were calculated using 2 × 2 contingency tables. A
Results
In total, 1531 pregnant women underwent a primary evaluation in the emergency department, of the pandemic clinic. The demographic and obstetric characteristics of the pregnant patients are presented in Table 3. Clinical characteristics of these patients are summarized in Table 4.
The Demographic and Obstetric Characteristics of the Pregnant Patient Cohort.
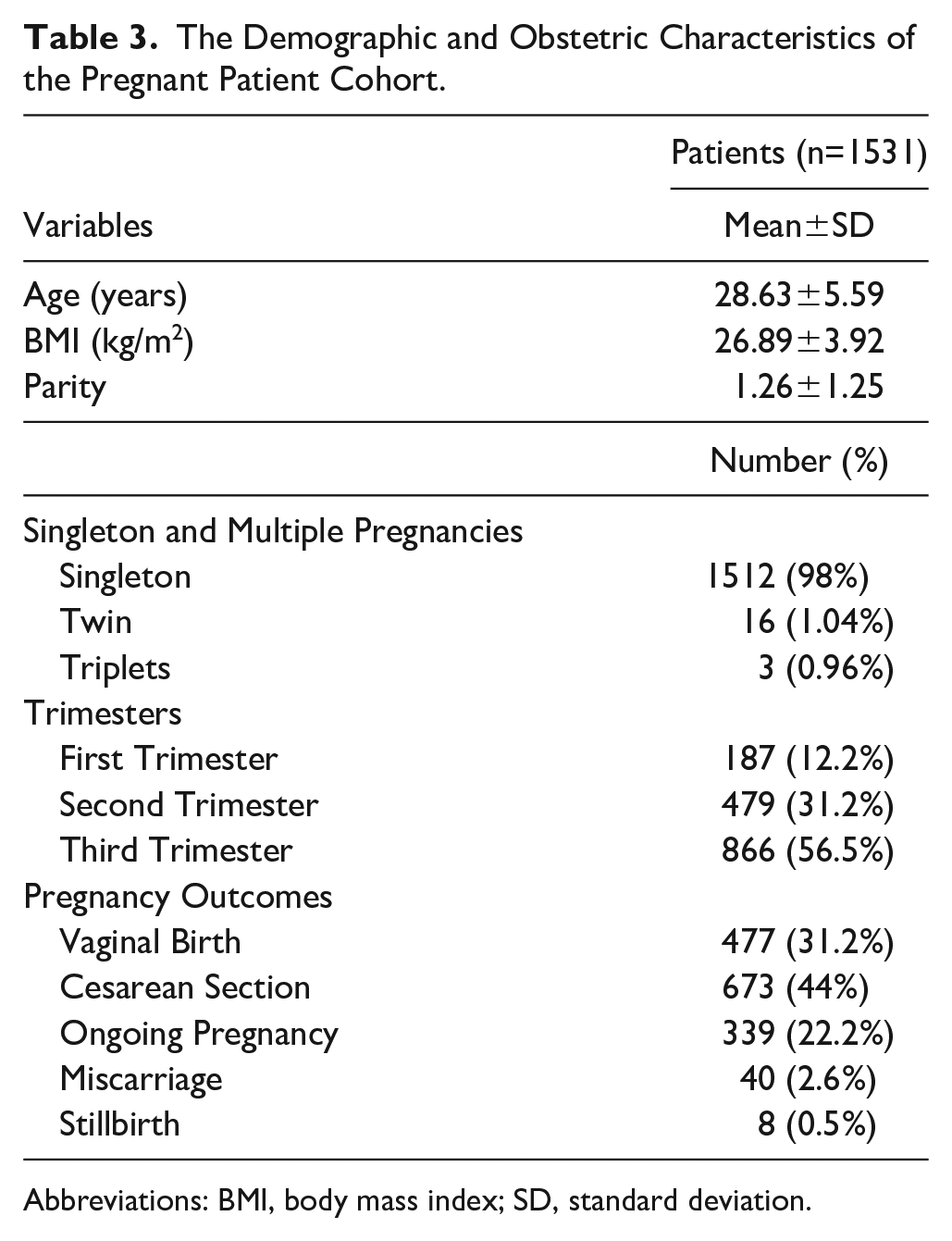
Abbreviations: BMI, body mass index; SD, standard deviation.
The Study Cohort’s Clinical Characteristics and Diagnostic Assessments.
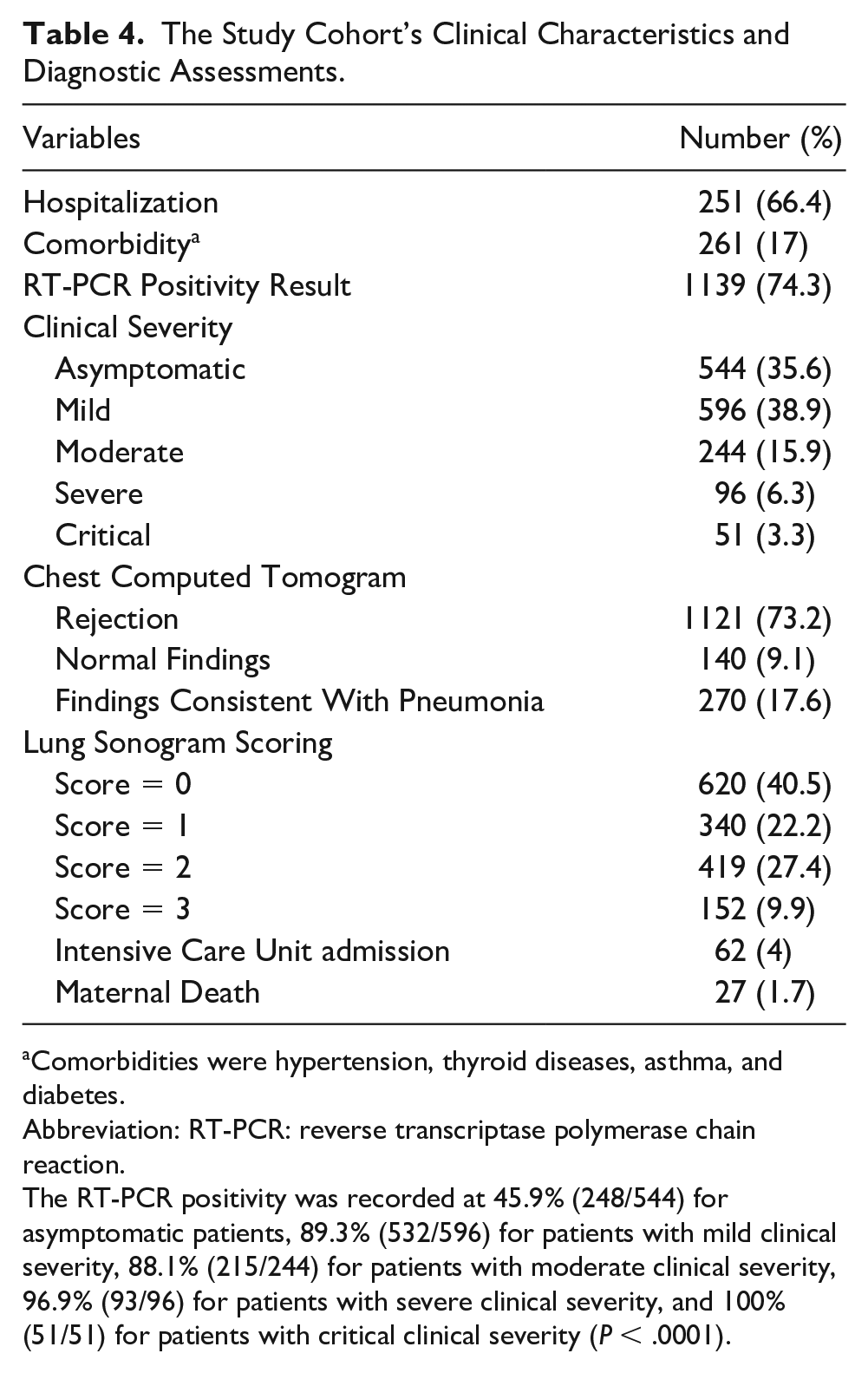
Comorbidities were hypertension, thyroid diseases, asthma, and diabetes.
Abbreviation: RT-PCR: reverse transcriptase polymerase chain reaction.
The RT-PCR positivity was recorded at 45.9% (248/544) for asymptomatic patients, 89.3% (532/596) for patients with mild clinical severity, 88.1% (215/244) for patients with moderate clinical severity, 96.9% (93/96) for patients with severe clinical severity, and 100% (51/51) for patients with critical clinical severity (
Distribution of LUS scores according to clinical severity is presented in Table 5 (
The Distribution of the Study Cohort’s Lung Ultrasonogram (LUS) Scores and the Diagnostic Findings from Their Chest Computed Tomograms (CT), Based on Disease Severity.
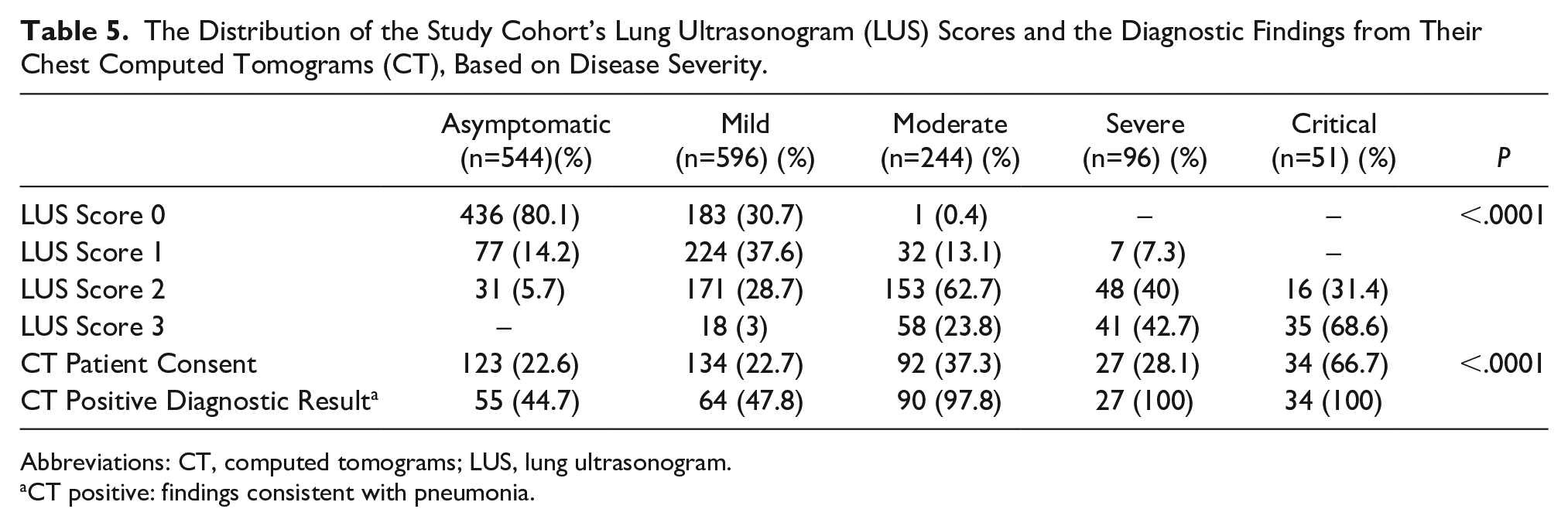
Abbreviations: CT, computed tomograms; LUS, lung ultrasonogram.
CT positive: findings consistent with pneumonia.
Four hundred and ten (26.8%) patients agreed to a chest CT. The diagnostic chest CT results are presented in Table 5 (
The accuracy of LUS and chest CT combined with using the RT-PCR test, as a diagnostic gold standard, are presented in Table 6.
The Diagnostic Accuracy of Lung Ultrasonography (LUS) and Chest Computed Tomography (CT) Based on the Diagnostic Gold Standard of the RT-PCR Test Results.
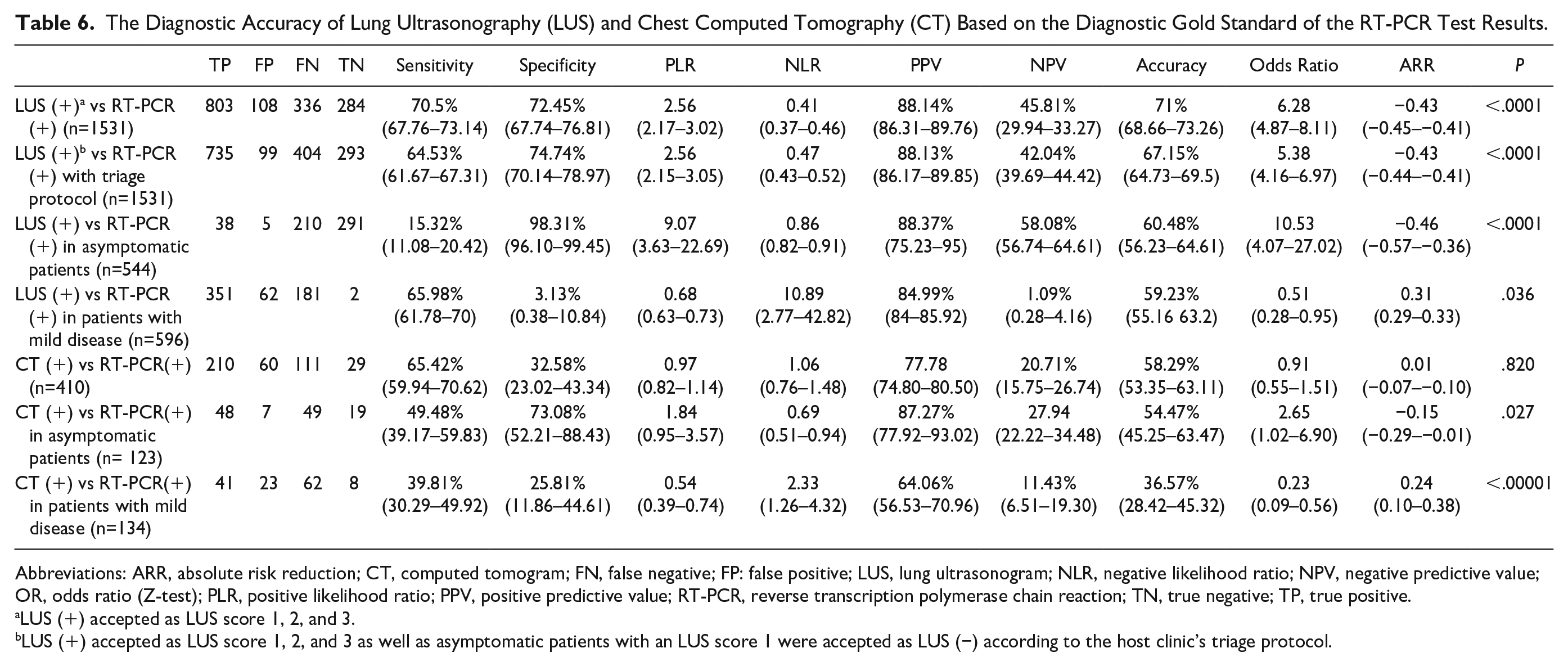
Abbreviations: ARR, absolute risk reduction; CT, computed tomogram; FN, false negative; FP: false positive; LUS, lung ultrasonogram; NLR, negative likelihood ratio; NPV, negative predictive value; OR, odds ratio (Z-test); PLR, positive likelihood ratio; PPV, positive predictive value; RT-PCR, reverse transcription polymerase chain reaction; TN, true negative; TP, true positive.
LUS (+) accepted as LUS score 1, 2, and 3.
LUS (+) accepted as LUS score 1, 2, and 3 as well as asymptomatic patients with an LUS score 1 were accepted as LUS (−) according to the host clinic’s triage protocol.
Discussion
In this large cohort of pregnant patients, LUS was found to have good diagnostic detection, when used for screening, during the COVID-19 pandemic. When patients were classified according to their clinical severity of their SARS-CoV-2 infection, a high specificity for LUS was noted among asymptomatic patients and moderate sensitivity in patients with mild severity. The diagnostic value for LUS was 100% for patients who were deemed clinical outlook was severe and or critical.
Lung ultrasonography has showed superior diagnostic performance over CT, particularly in asymptomatic patients. In this study, previously introduced parameters used in study clinic were used to calculate the diagnostic performance of the LUS.12,14 In previous research, these clinical parameters offered a very good triage performance, particularly during the pandemic’s peak periods and RT-PCR tests are not available. Continued use of this screening procedure proved to a safe, effective, and fast screening method for every pregnant patient admitted to the delivery room. 14 The specificity of LUS increased from 72.45% to 74.74%, and the positive predictive value was unchanged in this study, regardless of recruiting local clinical triaged patients (See Table 6).
In the meta-analysis by Jari et al., early in the pandemic, they emphasized that LUS was suitable and useful for detecting patients at risk, particularly in the initial evaluation. They reported that the sensitivity of LUS was comparable to the moderate sensitivity of RT-PCR, but its specificity was low. 6 In the review by Gil-Rodríguez et al., 15 they stated that the use of LUS could be restricted, as COVID-19 prevalence declines, as the positive predictive value would decline. They felt that it may be appropriate to use LUS in cases that resulted in a negative PCR, but with clinical suspicion, in the context of the pandemic. 15
In a meta-analysis, that included 15 studies where the RT-PCR test was used as a diagnostic standard, they reported that the sensitivity of LUS ranged between 68% and 96% and its specificity varied between 21% and 91%. 6 In a multi-centered study, based on 1462 patients, the predictability of LUS was examined by creating mild-severe and mixed patient groupings, which was based on clinical severity. 16 In that study, the sensitivity of LUS was found to be 32% and specificity 94%, for patients with mild clinical severity. 16 In comparison to this study, asymptomatic patients were not included, as this cohort came from the general population, and their LUS scoring system, used in that analysis, was different. 16 In general, it is important to note that there are different LUS scoring systems, as part of many published studies and hampering the standardization of LUS data. This variability in LUS scoring has been mentioned as a major limitation in determining the diagnostic accuracy of LUS.6,15,17 The accuracy of LUS has shown considerable variation across a variety of published studies.
This study was carried out in the tertiary center, which had a maternity unit with the highest capacity, during the pandemic. The size of the study cohort (n=1531), the homogeneity of the cohort, and the application of clinical managements with pragmatic approaches are positive attributes of this study. Generalizability of the study’s results is also limited due to the high percentage of patients who rejected a CT examination. At the time of this study, there were different viral variants, as described by World Health Organization (e.g. Delta variant, etc.). The diagnostic power of diagnostic imaging techniques could differ based on specific SARS-CoV-2 variants and the greater or lesser lung disease involvement. Nevertheless, this study appears to have been only marginally influenced by viral variant differences, as the diagnostic accuracy of LUS consistently reached 100% in cases with moderate, severe, and critical clinical severity. This might underscore the efficacy of LUS as a reliable diagnostic screening tool even for mild or asymptomatic patients. Perhaps due to the high patient rejection rate of CT, its diagnostic performance showed significantly lower sensitivity compared with LUS. Particularly in asymptomatic patients, LUS outperformed CT.
During the initial phases of the pandemic, LUS was predominantly assessed in conjunction with RT-PCR or COVID-19 symptoms, as studies involving pregnant patients, aimed to diagnose or monitor the disease.12,18 Additional patient case studies further explored the comparison between LUS and CT. Beyond these patient cases, the evaluation of LUS and CT has primarily focused on symptomatic COVID-19 individuals, with a recent limited utilization of CT, for investigations of pregnant patients.19,20
The rejection rate of CT in the current cohort was 77.2%. High CT rejection rates may have occurred during the period of this study due to high rate of hospital admission, suboptimal sterilization in tomography rooms, and the theoretical possibility of ionizing radiation exposure to the fetus. These rates may have decreased over time with the ongoing waves of the pandemic, the level of societal knowledge increased, and the pandemic infection rate improved. However, due to this significant bias, the demonstrated diagnostic performance of CT, in the study cohort, is a limitation.
Although the results of this study have lost its priority due to the pandemic abating, it could still be used in underdeveloped countries where the ongoing SARS-CoV-2 infection rates continue unabated. In addition, the diagnostic performance of imaging methods in the corona virus-based outbreaks should continue to be investigated. In future studies, there may be a role for hand-held or laptop ultrasound systems to perform LUS, in a pandemic to gauge the presence of viral pneumonia. This portability of equipment has a role to provide bedside imaging of patients for surveillance in the intensive care units. It also may be a shorter learning curve for obstetricians to provide LUS, given their sonographic experience and can contribute these patients struggling through a pandemic.
Limitations
The major limitation of this study was the design, which has internal and external threats to validity. It also utilized a convenient sample of patients, and therefore, these results are unique to them. In addition, this study was based on a retrospective review of data curated from the patient’s electronic records, which have impacted the accuracy of reported information. This study primarily focused on a specific demographic group of pregnant patients and the evaluations of the LUS were provided by obstetricians. This diagnostic data collection was very specific and also further hampers the generalizability of the results to diverse populations. Moreover, the low acceptance rate of chest CT among pregnant women posed a challenge, preventing a comprehensive analysis of the diagnostic performance of CT.
Conclusion
During the SARS-CoV-2 pandemic, the diagnostic performance of LUS was found to be adequate for diagnostic detection compared with the RT-PCR test, in pregnant patients admitted to the hospital with suspicion of having contracted COVID-19. The high rate of pregnant patient’s rejecting a CT examination (77.2%) limited the impact of the study results. Nevertheless, in this cohort of patients, LUS was found to be quick, safe, effective, and reproducible particularly for screening for COVID-19 and combined with new triage strategies, in an obstetric practice, during the recent pandemic.
Footnotes
Acknowledgements
The authors specially thank Fatih Hakan Koklu PhD, whose expertise and commitment were instrumental in overcoming various challenges.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval for this study was obtained from the local ethics committee and the Ministry of Health COVID-19 Scientific Research Evaluation Commission (Decision No.: 2020/287).
Informed Consent
Informed consent was not sought for this study because all case data were de-identified and/or aggregated and followed ethics committee or IRB guidelines (also referred to as the Honest Broker System).
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because no animals were used during the study.
Trial Registration
Not applicable.
