Abstract
Objective:
The low sensitivity of the real-time reverse transcription-polymerase chain reaction (rRT-PCR) test on the nasopharyngeal swab for SARS-CoV2 virus could cause infections among health care professionals and could be a source of viral spread. The aim of this study was to verify whether lung ultrasonography (LUS) had a negative predictive value (NPV) high enough to rule out coronavirus disease 2019 (COVID-19) in a cohort of health care employees, working in emergency departments (EDs).
Materials and Methods:
A multicenter prospective observational study was conducted in two EDs in Northeast Italy. An adjudication committee established the definitive diagnosis of COVID-19.
Results:
A cohort was enrolled of 155 possible patient cases (two true positives). Twenty-one health care employees presented with suggestive symptoms for COVID-19. The rRT-PCR test was positive in one of the two patients. LUS was suggestive for COVID-19 pneumonia in four patients. The diagnostic accuracy of LUS was 98.7% (95% confidence interval [CI] = 95.4%–99.8%). The NPV was 100% (95% CI = 100%–100%).
Conclusion:
LUS has a high enough NPV for excluding a COVID-19-related pneumonia in a cohort of health care employees who were exposed to the SARS-CoV2 virus.
Since the first reports of coronavirus disease 2019 (COVID-19) in Italy, health authorities have implemented specific protocols to triage the SARS-CoV2-infected patients in the emergency department (ED). 1 These protocols, acknowledging variations across health care systems, provided for early isolation of highly suspected patients. It also allowed for the execution of a real-time reverse transcription-polymerase chain reaction (rRT-PCR) test on samples of body secretions (i.e., in the ED, a nasopharyngeal swab) and further examinations to establish the severity of the COVID-19 disease (i.e., arterial blood gas analysis, chest radiography, lung ultrasonography [LUS]). The health personnel assigned to identify positive SARS-CoV2-infected patients, as well as wearing personal protective equipment (PPE) (e.g., gloves, visor, N95 mask, gown), were subjected to surveillance checks, such as a nasopharyngeal swab. 2 The importance of guaranteeing health care workers’ safety lies in protecting the health of those workers 3 and preventing them from being a source of coronavirus spread. 4 Health care professionals have an increased risk of exposure and are potential carriers of subsequent transmission. More than 3300 healthcare workers were infected in China (4% of 81 285 reported infections) in 2020. 5 In Spain, on March 25, 2020, nearly 6500 medical staff members were affected (13.6%) of the country’s 47 600 total cases, 1% of the health care workforce. 6 Therefore, it is essential to identify health care professionals infected with the SARS-CoV2 virus compared to healthy staff, who may present with COVID-19 symptoms but are not very specific. During this epidemic, the use of LUS became central to both identifying infected patients and monitoring them once they were hospitalized.7,8
This study was designed to verify whether LUS had a negative predictive value sufficient to be used to rule out COVID-19-related pneumonia, with a cohort of health workers working in two EDs.
Materials and Methods
Setting and Participants
From March 30, 2020, to April 22, 2020, every health care employees from two rural EDs in the Northeast of Italy (Hospital of Latisana serves 25 000 patients/year and Hospital of Tolmezzo serves 15 000 patients/year) were asked to participate voluntarily in an LUS screening procedure. This was done to establish the presence or absence of diagnostic findings compatible with pulmonary COVID-19 involvement. The following health care employees were considered—42 physicians, 72 nurses, 21 support personnel, and 25 ambulance drivers—stationed in the EDs. During this time, the two EDs were dealing with a prevalence of about 20% incidence of COVID-19, among the patients who entered.
Data Collection, Adjudication, and Follow-up
The clinical data were collected prospectively. An adjudication committee established the definitive diagnosis of a COVID-19-related pneumonia. The final diagnosis was, based on clinical data, rRT-PCR test on the nasopharyngeal swab (as well as any subsequent swab test performed during hospitalization), chest radiograph, and computed tomography (CT), for those who needed it. The adjudication committee was composed of two experienced emergency physicians blinded to the LUS findings. At the end of the review of each case, the adjudication committee expressed a dichotomous opinion: “positive” or “negative” to COVID-19-related pneumonia. An experienced radiologist and another qualified emergency physician reviewed the chest radiographs or CT images. These diagnostic results were categorized in a final report as “suggestive for COVID-19 pneumonia” or “non-suggestive for COVID-19 pneumonia.” These diagnostic steps were completed before the adjudication committee viewed these images. The rRT-PCR tests used in this study were Roche Cobas SARS-CoV-2 essay (F. Hoffmann-La Roche AG, Basel, Switzerland) and Liaison MDX DiaSorin (DiaSorin S.p.A., Saluggia VC, Italy).
The health care employees were re-evaluated weekly for up to 1 month or until the SARS-CoV2 infection was confirmed via nasopharyngeal molecular swab, if symptoms appeared earlier than 1 month.
LUS Diagnosis
The LUS examinations were performed before the rRT-PCR result, for SARS-CoV-2. Therefore, the two sonographers were blinded to the report of this examination, but not the possible presence of potentially suggestive symptoms. The LUS examinations were performed by dividing the chest into 12 areas: 4 anterior right areas, 4 anterior left areas, 2 right posterior areas, and 2 left posterior areas. The images were evaluated for the presence of the following sonographic findings: bilateral B lines, consolidations, subpleural consolidations, thickening and irregularities of the pleural line, and pleural effusion. For early identification and maximum LUS sensitivity, the examination was considered positive if even one of area of the lung demonstrated at least one of the previously mentioned suggestive signs of pulmonary involvement, from COVID-19 (see Figure 1). The undetermined cases were re-discussed by the two sonographers together to categorize them into “positive” or “negative” for COVID-19 pneumonia, according to the International Proposed LUS protocol. 7 The two sonographers were two emergency physicians with more than 15 years of experience in LUS. The two ultrasound equipment systems used were MyLab Alpha (Esaote S.p.A., Genova, Italy), with convex and linear transducers.
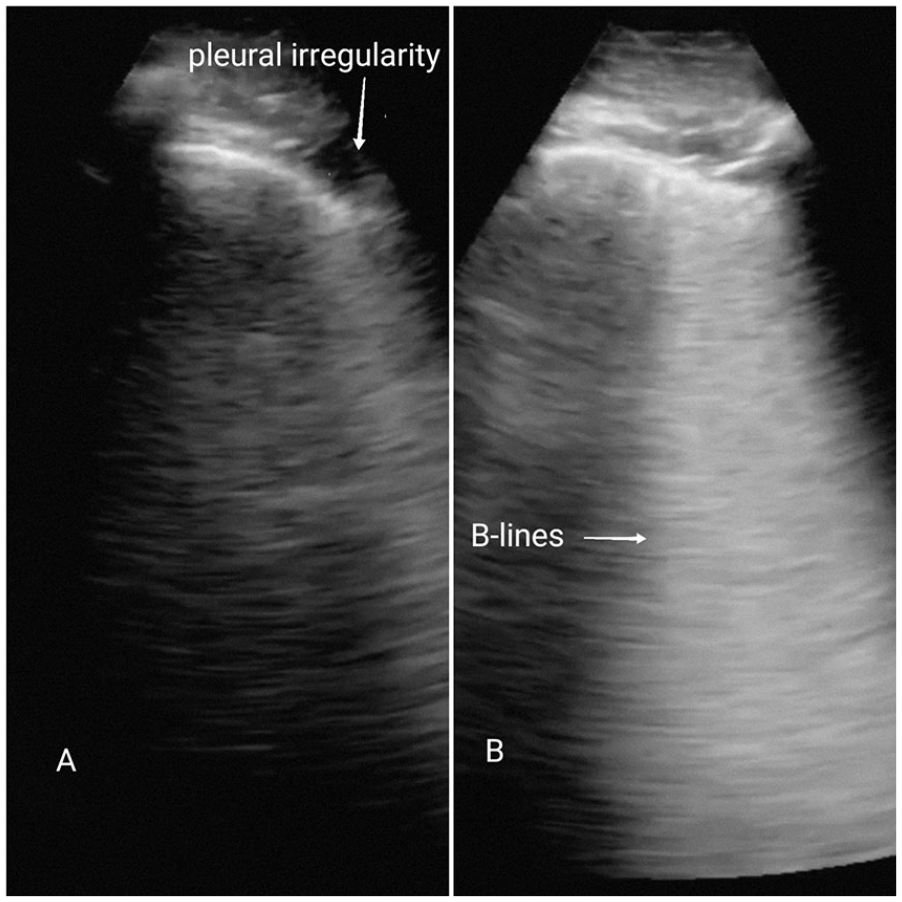
Typical LUS diagnostic findings with COVID-19 pneumonia. (A) Pleural irregularities as anechoic spots that interrupt the pleural line; (B) focal B lines, scattered, sometimes confluent but not homogeneously distributed.
Sample Size
A convenience sample of 150 suitable health care employees was considered. In calculating the confidence interval’s (CI) magnitude, it was hypothesized that at least 140 employees, who agreed to participate in the study, would be negative for COVID-19-related pneumonia on the LUS examination. Estimating an approximately 98% negative predictive value for LUS meant a calculated 95% CI would range from 96% to 100%. The final sample was 153 healthcare employees examined with LUS for a disease prevalence of 1.3% (95%CI = 0.2%–4.6%). The study had a sufficiently precise CI to detect a reliable negative predictive value.
Statistical Analysis
Descriptive statistics were reported based on the clinical and demographic characteristics of this cohort: sex, age, the presence of symptoms suggestive for COVID-19, the LUS results, and the CT results.
The categorical variables were expressed in absolute value (and respective percentage). Statistical significance between the two groups was calculated using Pearson’s χ2 test (or exact Fisher’s test, if appropriate).
Due to their nonparametric distribution, the continuous variables were shown with the median value and interquartile range (first and third interquartile). Statistical significance between the two groups was calculated using the Kruskal–Wallis test.
Statistical significance was considered for an α error value less than or equal to 0.05 (i.e., a two-tailed
Based on a contingency table (considering the frequencies of the True Positives, False Positives, True Negatives, and False Negatives), sensitivity, specificity, positive and negative likelihood ratios, positive and negative predictive values, and diagnostic accuracy of the LUS were calculated.
All statistical analyses were performed using the open-source R-CRAN project Version 4.0.0. statistical software. The followed packages were implemented: “irr,” “caret,” and “compareGroups.”
Results
During the period, 155 health care employees were enrolled. The median age of this cohort was 43 years. The characteristics of the enrolled cohort are provided in Table 1.
A comparison between positive and negative patient clinical data for COVID-19-related pneumonia.
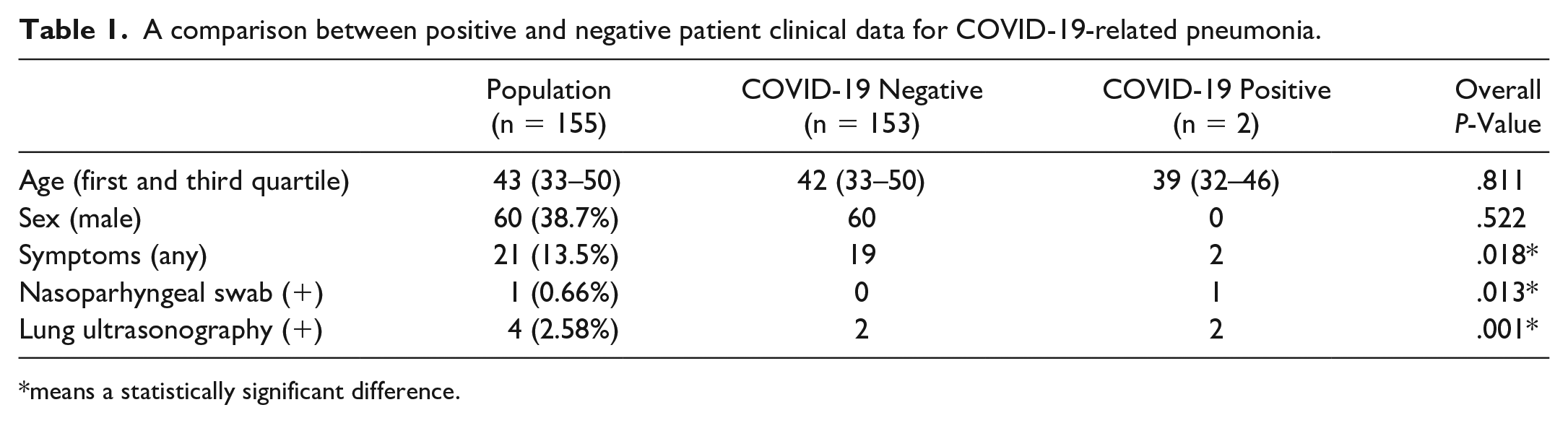
means a statistically significant difference.
The adjudication committee established two true-positive diagnoses for COVID-19. The remaining 153 cases were true negative (see Figure 2). By dividing the cohort based on the diagnosis of COVID-19-related pneumonia, the presence of compatible symptoms and the positivity of the nasopharyngeal swab are the two variables that were statistically significantly different between the two groups (
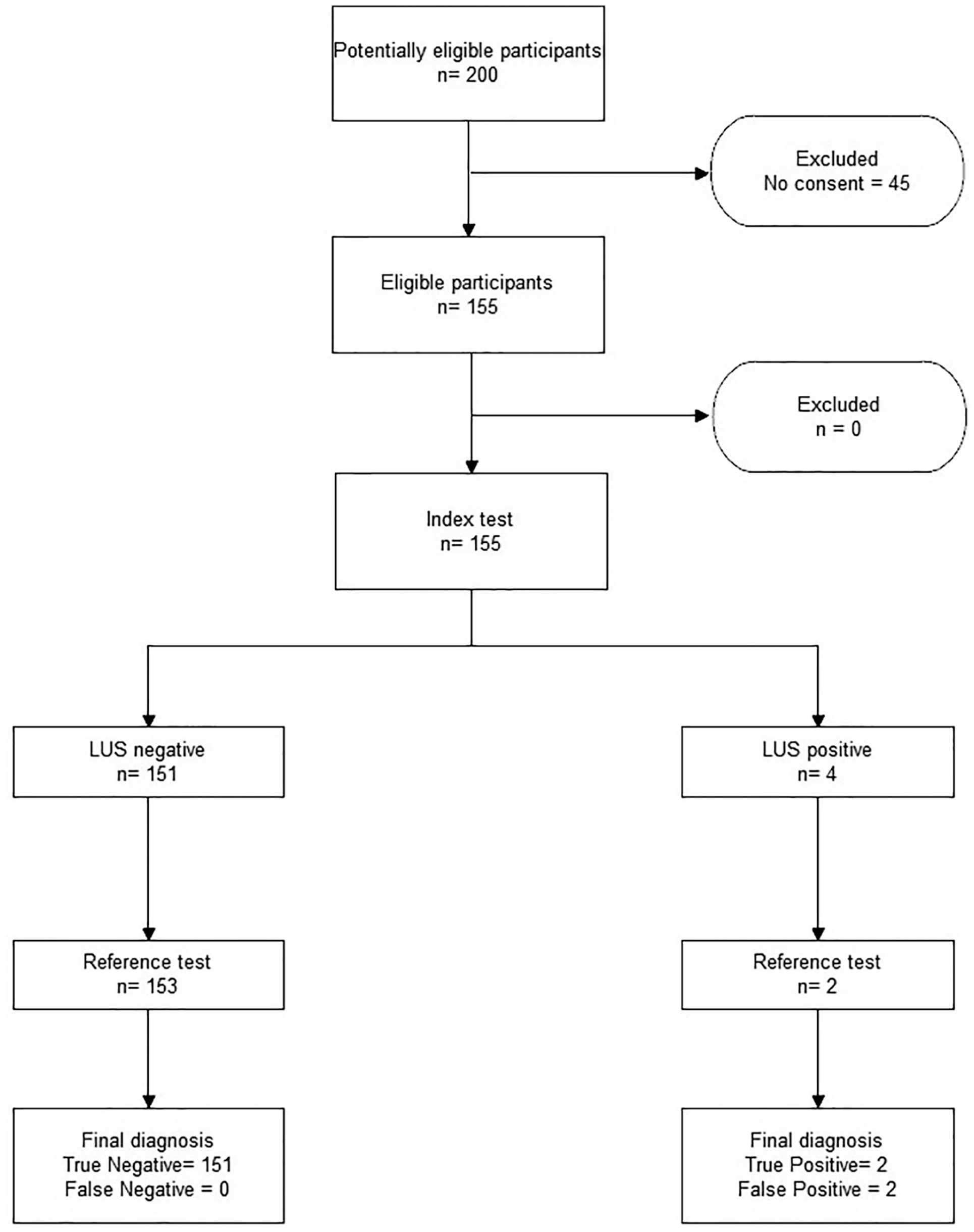
A patient enrollment flow chart. Lung ultrasonography was suggestive for COVID-19 pneumonia in four patient cases (two false-positive patient cases).
There were 21 health care employees who presented with suggestive symptoms (2 true positives and 19 false positives). A cough was present in six patients, cold/rhinitis in ten patients, fever in three patients, sore throat in two patients, myalgia/arthralgia in two patients, and, finally, one case had exertional dyspnea. The rRT-PCR test on the nasopharyngeal swab was positive for one of the two true-positive COVID-19 cases. Neither of the two patients who were rated as “false positives” developed symptoms suspicious of COVID-19, during the month-long follow-up period.
LUS was suggestive for COVID-19 pneumonia in four patients: two patients were true positives, whereas two other patients were false positives. The sensitivity and specificity of LUS in this cohort were 100% (95% CI = 15.8%–100%) and 98.7% (95% CI = 95.4%–99.8%), respectively. Based on these values, the diagnostic accuracy was 98.7% (95% CI = 95.4%–99.8%). The positive and negative likelihood values were 76.5 (95% CI = 19.3–303.1) and 0, respectively. Therefore, the positive predictive value was 50% (95% CI = 20.2%–79.8%) and the negative predictive value was 100% (95% CI = 100%–100%) (see Figure 3).
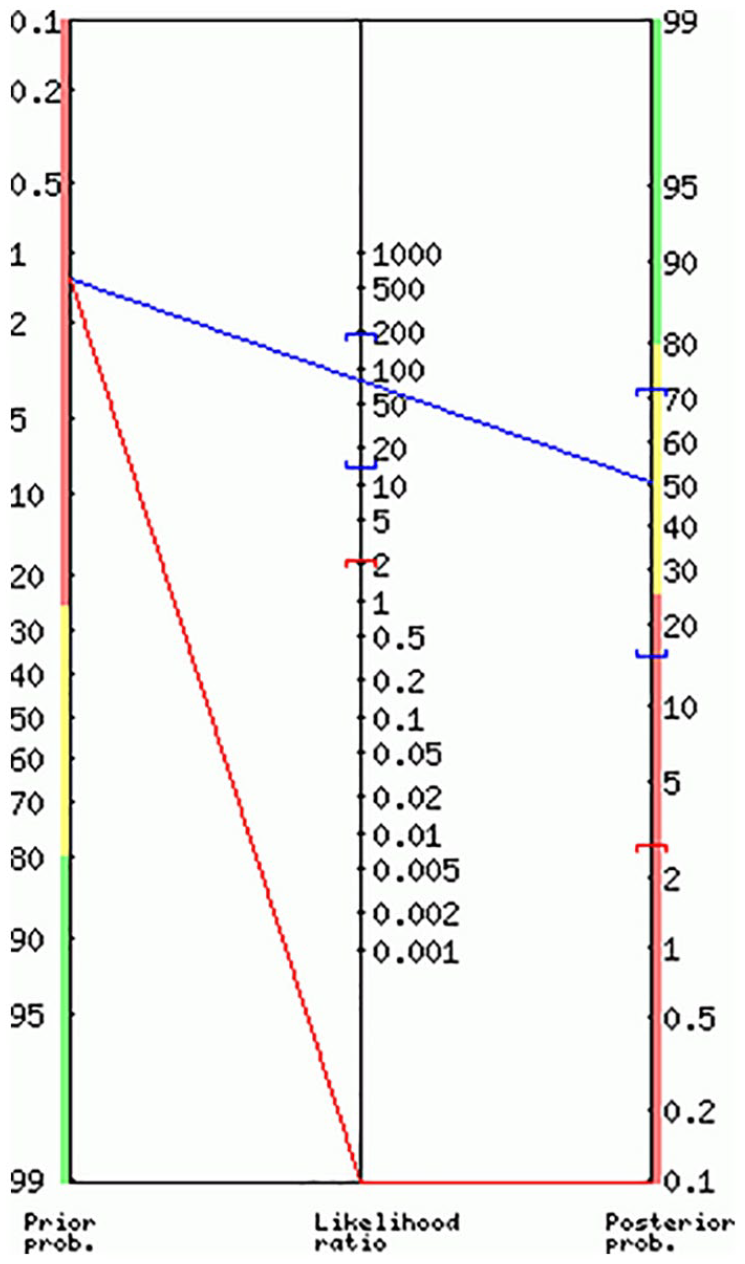
The likelihood ratio nomogram for lung ultrasonography in diagnosing COVID-19 pneumonia. The positive likelihood ratio was 76.50 (19.31–303.12); the negative likelihood ratio was 0.
Discussion
This cohort study demonstrated that LUS was a useful diagnostic test for ruling out COVID-19 involvement in a low prevalence group of patients. Furthermore, the cohort studied was very comparable to the patient population, within countries affected by the COVID-19 pandemic, regarding the reduction in peak infection. This result was twofold; primarily, LUS was useful for quickly verifying the effectiveness of the internal protocols provided on the isolation pathways of COVID-19-infected patients and PPE efficiency.
The community of health care employees represented a cohort at high risk of exposure, but with a low probability, in part due to PPE.9,10 These health care employees were within a group of works at a low prevalence for COVID-19 disease. Therefore, it may be reflective of the general population when the SARS-CoV-2 infection peak was low. 11 It becomes essential to identify infected patients and exclude uninfected patients with an adequate margin of safety, in this situation.
The literature shows that asymptomatic cases can constitute up to 60% of COVID-19 patient cases. 12 Given the high infectivity of patients, it is essential to exclude with reasonable certainty the cases that, even with symptoms suggestive of COVID-19 (given their non-specificity), have not been infected.
The role of asymptomatic patients is increasingly emerging in the spread of infection.13,14 However, there is still no adequate diagnostic screening method. The rRT-PCR tests have had a variable sensitivity (37%–71%) and long processing times,15–17 as well as a chest radiograph that has insufficient sensitivity (69%).
18
Although a high sensitivity (98%)
19
is associated with a CT examination, it exposes a non-negligible ionizing risk.
20
Subjects positive for SARS-CoV2 but asymptomatic may have changes in the lung parenchyma. This was recently demonstrated with a radiographic tomography examination conducted on cruise ship passengers.
21
More precisely, lung disease pattern was present in 54%, ground glass in 80%, and consolidations in 20%.
22
In a retrospective study of 29 COVID-19 patients, Yang et al compared LUS with CT.
23
They found that LUS was more sensitive than CT in the diagnosis of regional alveolar-interstitial pattern (60% vs. 38.5%,
The clinical experiences reported thus far in the literature indicate a low specificity for sonographic diagnostic findings.24,25 Changes in the pleural line and the presence of multiple B lines are sonographic findings common to other forms of interstitial lung disease, such as fibrosis, emphysema, or early pulmonary edema. LUS is probably ideal as a diagnostic tool to exclude rather than confirm a COVID-19-related pneumonia. 26 These results, conducted on a cohort of health care employees, confirmed this hypothesis.
The containment strategies, adopted by various countries, consisted mostly of the limitation of and reduction in physical contact between people and in the use of masks and gloves. These measures have probably allowed for the “flattening” of the curve, of the epidemic. 27 However, the reignition of infectious foci can occur in places where cases of residual infection can concentrate, such as hospitals.28,29 The use of a bedside imaging examination that is non-invasive and easily repeatable allows for expanding the potential diagnostic assessment. Since the hypothesis of mass screening seems to be one of the few plausible strategies, LUS has the potential to be an adequate tool, in terms of negative predictive value.
Limitations
This cohort study was conducted in two EDs with a relatively low prevalence of COVID-19, among hospitalized patients. There were not enough data to determine how these results could be applied in another context. In addition, the sonographic diagnosis of COVID-19-related pneumonia was established by physicians, with a long experience using LUS. In other health care settings, LUS could have a clinical performance that is not comparable to this cohort.
As was reported in this study, the rRT-PCR test with a nasopharyngeal swab did not demonstrate optimal sensitivity. Furthermore, the main limitation is that there is no reference diagnostic examination that can be considered as the clinical gold standard. This inherent difficulty was overcome by creating an adjudication committee to evaluate all the clinical documentation and tests available.
Furthermore, due to the nature of the COVID-19, the two sonographers could not be blinded to the presence of any suggestive symptoms.
Conclusion
In this cohort study, LUS had a good enough negative predictive value for excluding pulmonary disease, due to SARS-CoV2, among health care employees exposed to the virus but with a low prevalence of the disease.
Footnotes
Acknowledgements
We would like to thank Dr Marcella Montico for her statistical advice.
Authors’ Contributions
R.C. designed the study, reviewed the final diagnosis, and supervised the final draft; G.A. reviewed the radiographic images; C.G. collected the data; D.O. performed the statistical analysis, drafted the first draft, and supervised the final draft; S.C. designed the study, collected the data, drafted the first draft, and supervised the final draft; P.P.P. reviewed the final diagnosis; C.R. collected the data; D.C. collected the data; A.G.D. collected the data; and S.M. reviewed the radiographic images.
Ethical Approval
No formal ethical approval was required given the observational and non-invasive nature of the study.
Informed Consent
Informed consent was requested and acquired for each of the study participants.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because Guidelines for humane animal treatment did not apply to the present study because no animals were used to conduct this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Trial Registration
Not applicable.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent for Publication
Each patient included in the study signed informed consent for participation in the study, collection of personal data, and consent to the publication of data obtained from the analysis of clinical data, according to the Declaration of Helsinki.
Availability of Data and Material
Data are available following a reasoned request.
