Abstract
Objective:
To assess the feasibility and reliability of incorporating quantitative shear wave elastography (SWE) data as an additional means of characterizing focal atheromata during routine carotid sonographic examinations when compared with conventional gray-scale Gray-Weale (GW) methods.
Methods:
Inpatients and outpatients undergoing routine carotid examinations at a medical center were included in the pilot study. Patients with prior history of endarterectomy, interventional therapeutic procedures, or absence of plaque formation were excluded. All examinations were performed by registered vascular technologist using a Mindray Resona 7 ultrasound equipment system with an L9-3U transducer in a carotid examination mode. The images were later reviewed by two independent, blinded observers and correlated to GW characterization.
Results:
There were 41 atheromatous lesions evaluated during the pilot study. The range of absolute (low to high) average and median Young’s modulus (YM) kPa values vary widely across each plaque type; they nonetheless still exhibit evidence of increasing stiffness as the gray-scale sonographic appearance becomes denser. The same observation is true of the average kPa and median kPa values as increasing YM values indicate increasing stiffness of interrogated tissues.
Conclusion:
This pilot study affirms the feasibility and reliability of incorporating quantitative SWE data as an additional means of characterizing focal atheromata during routine carotid sonographic examinations; further large-scale studies are needed to determine the sensitivity and specificity of individual kPa values associated with specific plaque types.
Keywords
Atherosclerosis is a dynamic pathologic condition affecting the arterial walls. The inflammatory and biochemical processes associated with this condition culminate in the development of atherosclerotic plaques that can obstruct blood flow in the affected vessel. Stratifying risk associated with atherosclerotic plaques in the carotid arteries is important in predicting complications that could ultimately result in cerebrovascular events, that is, transient ischemic attacks (TIAs) and stroke. Assessment of plaque echogenicity and integrity of the fibrous cap using gray-scale sonography has demonstrated efficacy in assisting with patient management and surveillance. 1
Gray-scale sonography assessment using the Gray-Weale (GW) classification method has, in some protocols, been incorporated into the diagnostic process to stage plaque. While this technique is relatively reliable, it is a subjective visual appraisal and is prone to inter-observer variability. 2 Prior studies have demonstrated that grading of sonographic gray-scale images is not consistent among observers and ranges from 66% to 75% agreement using this modality alone. 2 Moreover, GW classification does not provide any quantitative data regarding the elasticity, or stiffness, of the focal atheromatous lesions being interrogated, data that can be instrumental in guiding appropriate patient care. The GW classification criteria are as follows 3 :
Type I: predominantly sonolucent plaque with a thin echogenic cap
Type II: substantially sonolucent lesions with small areas of echogenicity
Type III: predominantly echogenic lesions with small areas of sonolucency
Type IV: uniformly echogenic lesions (equivalent to homogeneous).
Ultrasound shear wave elastography (SWE) offers an additional tool for the assessment of carotid plaque. 4 In general, SWE is a noninvasive method of evaluating the elasticity, or stiffness, of human soft tissue. Quantification is achieved by using onboard software algorithms that calculate Young’s modulus (YM) which is, simply described, the elastic property of a substance, that is, its deformation in response to an applied stress. Using an acoustic “push pulse,” the interrogated tissue sample generates transverse sound waves (shear waves). The velocity values of these shear waves is extracted from the returning acoustic data set and is used in calculating YM values using the following formula:
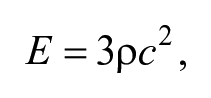
where
The 2dSWE uses multiple data points acquired simultaneously from an operator-defined region of interest (ROI). The size, shape, and location of the ROI can vary based on the operator’s judgment of best placement. Multiple push pulses are transmitted into the ROI, and the received data points are used to create both a color-coded map displayed on the monitor and a readout of mean quantitative YM values. Prior studies have demonstrated 2dSWE estimates of YM correlated well with GW classification. 4 Incorporating 2dSWE into carotid artery assessment is relatively easy but carries the potential for significant inter-operator variability related to selection of size, shape, and location of the ROI.
The pSWE method assesses plaque stiffness at a single point within the interrogated lesion using a single, fixed-dimension ROI size. Applying pSWE to the area of interest within a plaque may be a more reliable and reproducible method of assessing stiffness during routine carotid duplex examination in the vascular laboratory setting. Real-time pSWE also offers the potential to overcome data acquisition challenges associated with vessel motion and inter-operator variability, as well as providing improved penetration of heavily calcified plaques. While both 2dSWE and pSWE have demonstrated good sensitivity and specificity in liver fibrosis evaluation, there is scant data regarding the use of either method in atherosclerosis assessment. 7
There are technical limitations of applying SWE methods to focal arterial plaques that should be noted. The YM values are calculated under the assumption that the tissue being sampled is isotropic with homogeneous composition. In fact, atheromata and the underlying arterial walls are anisotropic in nature and the shear wave methods used to assess other tissues may not be optimal for vascular imaging. 6 In addition, there are inherent difficulties accurately applying both 2dSWE and pSWE methods to pulsatile arteries as the exact position of the sampled area within the sonographic ROI changes over the cardiac cycle. As the primary aim of this study was to assess the reliability and feasibility of incorporating the use of quantitative SWE in routine carotid duplex imaging, pSWE was chosen as the method for acquiring data to compare the GW assessment.
Materials and Methods
Participants
Patients undergoing routine carotid artery sonographic examinations were assessed in the typical fashion. All examinations were performed at a medical center’s vascular laboratory in Baltimore, Maryland, and consisted of inpatients and outpatients. The single inclusion criterion was the presence of one or more focal carotid atheromata identified during the performance of the diagnostic duplex examination using gray-scale imaging alone. Exclusions to participation in the study included the absence of plaque, history of carotid endarterectomy, or other interventional therapeutic procedures. No additional inclusion or exclusion criteria were applied. Determination of inclusion or exclusion to the study was made by the registered vascular technologist performing the initial diagnostic examination. No additional patient demographic data were included in this study, that is, age, sex, vascular history, or other comorbidities.
Data Collection
A Mindray Resona 7 ultrasound equipment system was used with an L9-3U linear transducer using a standardized carotid preset mode. All pSWE data points were calculated using an onboard software algorithm. Examinations were performed by registered vascular technologists under the supervision of a vascular surgeon. When focal atheromatous lesions were identified in the distal common carotid artery, carotid bifurcation, and/or proximal internal carotid artery, standard and enlarged (zoomed) gray-scale images were obtained. Both longitudinal and transverse planes of the appropriate segment were recorded and labeled accordingly. Data were obtained from right and/or left sides when the presence of plaque was noted. The pSWE acquisitions were obtained in the transverse plane in each plaque both to assure that the largest dimension of the lesion was being sampled and to overcome inaccuracies associated with potential off-axis imaging. These images were later reviewed by two independent, blinded observers, one registered vascular technologist working as a manager on staff and one registered vascular technologist working as a consultant and not associated with the hospital. The GW type was determined based solely on gray-scale plaque appearance. Five pSWE data points were gathered in each plaque and YM values were obtained and recorded.
In addition to average and median YM values obtained for each sampled plaque, onboard analysis software also calculates the interquartile range (IQR) for each data set as a quantitative measure of accuracy. Data sets with an IQR value of >30% were excluded from this study per the standard industry recommendation. The software package also calculated the average values of
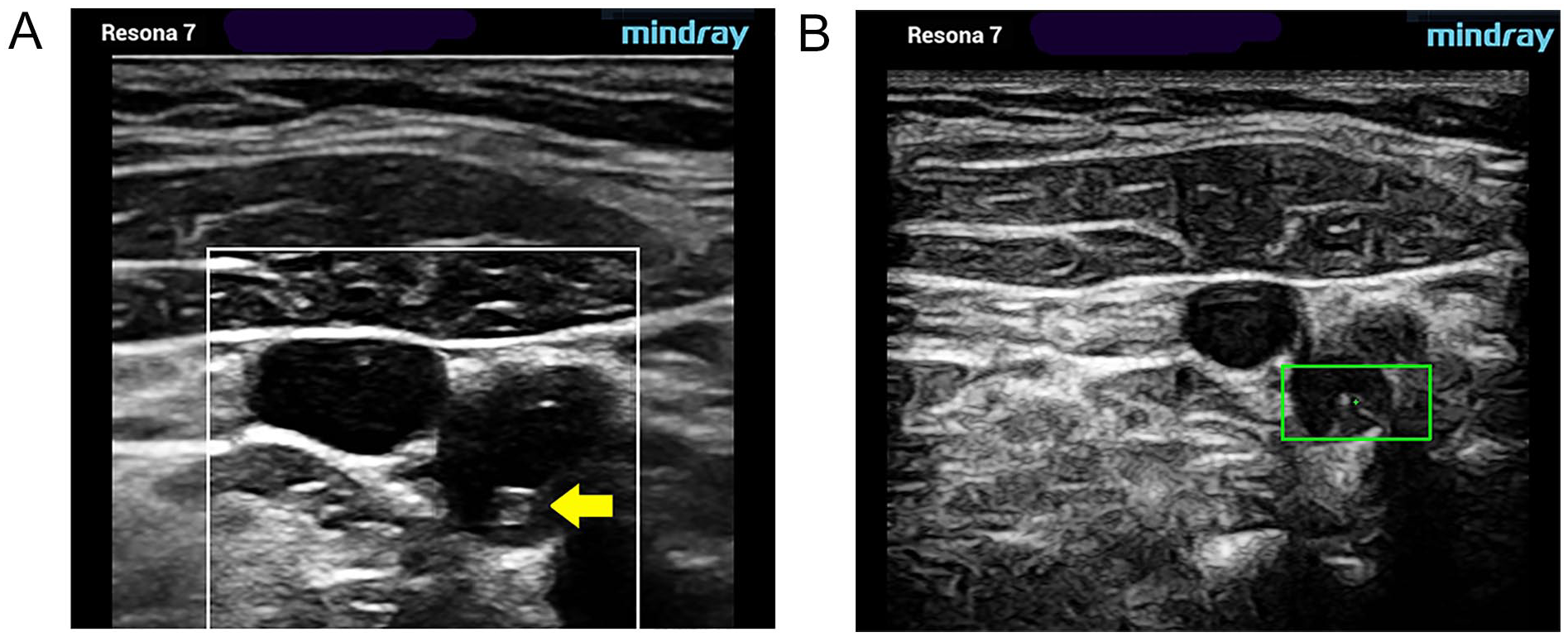
Type II. Substantially sonolucent lesion with small areas of echogenicity (yellow arrow). Gray-scale (A) and pSWE acquisition images (B). pSWE, point shear wave elastography.
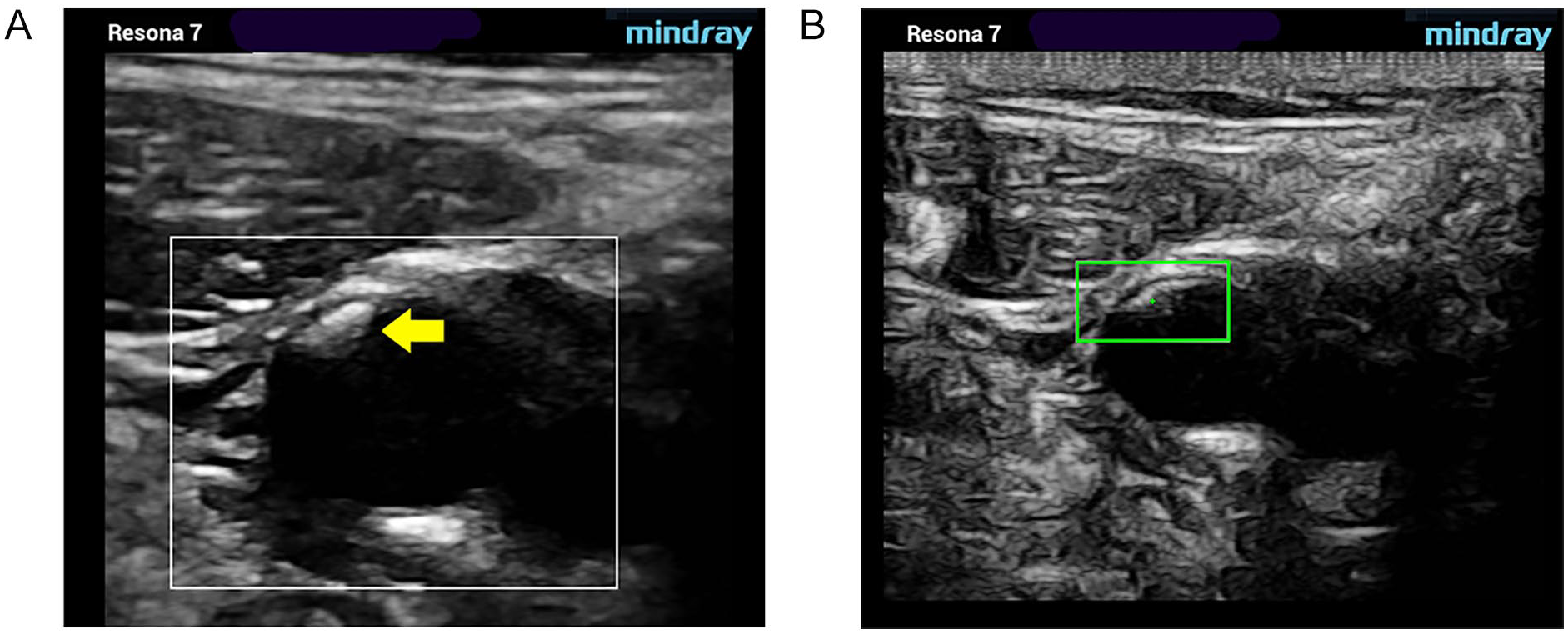
Type III. Predominantly echogenic lesion with small areas of sonolucency. Gray-scale (A) and pSWE acquisition images (B). pSWE, point shear wave elastography.
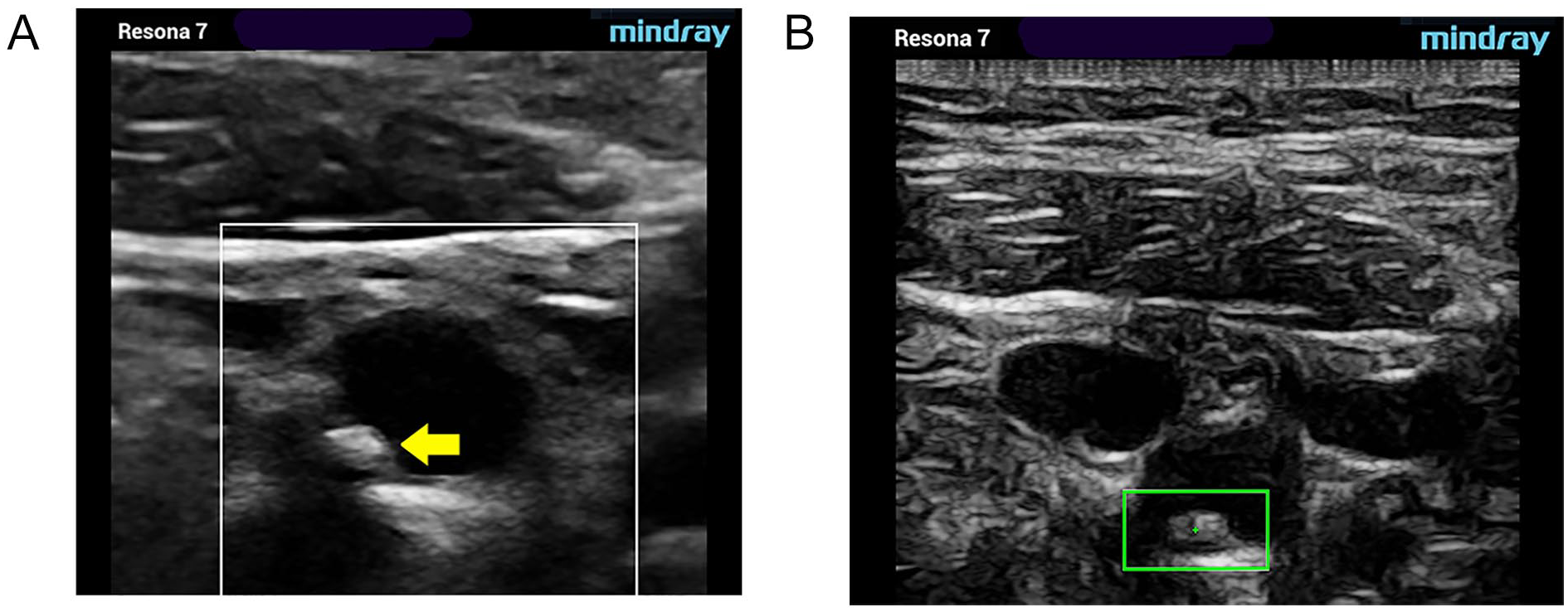
Type IV. Uniformly echogenic lesion (equivalent to homogeneous). Gray-scale (A) and pSWE acquisition images (B). pSWE, point shear wave elastography.
In the modified sonographic examination protocol, acquisition of pSWE images and data were grouped and performed sequentially after routine carotid diagnostic assessment. Time stamps are automatically displayed in the meta-data on each stored image. Calculating the delta between beginning and ending times of the series provides a metric for the additional amount of time required to incorporate SWE assessment into routine carotid ultrasound examinations.
Results
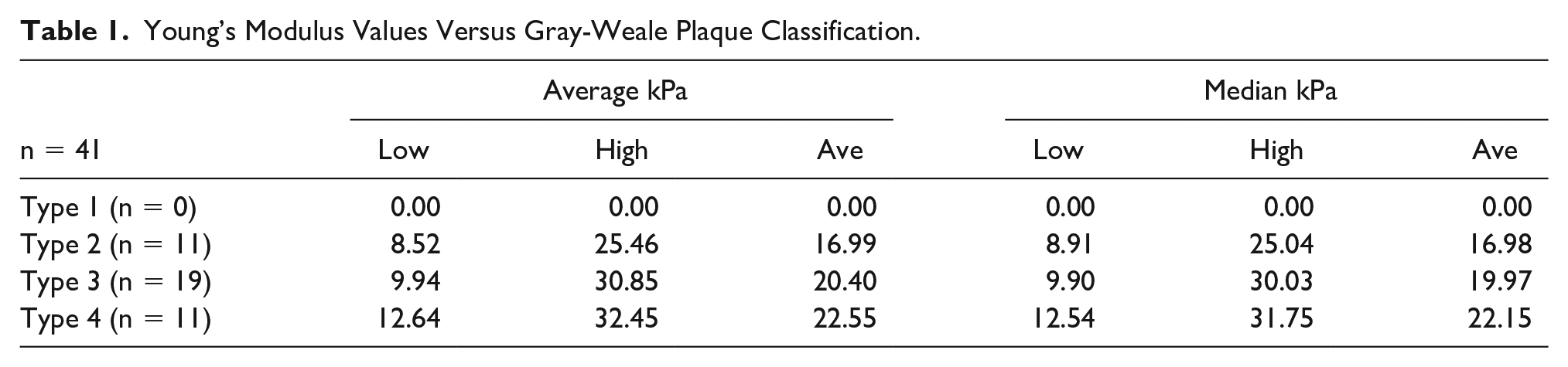
A total of 41 atheromatous lesions were evaluated. Unfortunately, no Type 1 lesions were encountered within the timeframe of this study and, therefore, cannot be included. Table 1 shows the distribution of the 41 lesions grouped by GW classification and the associated YM (average and median kPa) values. While the range of absolute (low to high) average and median YM (kPa) values vary widely across each plaque type, they nonetheless still exhibit evidence of increasing stiffness as the gray-scale sonographic appearance becomes denser. The same observation is true of the average (Ave kPa and median kPa) values as increasing YM values indicate increasing stiffness of interrogated tissues. The correlation between quantitative assessment of plaque stiffness and gray-scale appearance using the GW plaque classification method is consistent with other published studies and is discussed below. After discussion with a biostatistician, it was determined that there was insufficient quantity and variety of data to apply meaningful statistical correlation methods.
Young’s Modulus Values Versus Gray-Weale Plaque Classification.
Average and median acquisition time for the additional pSWE data points obtained in each study were recorded and are presented in Table 2.
Time Requirements for Additional SWE Data Acquisition.

Abbreviation: SWE, shear wave elastography.
Discussion
Carotid artery atherosclerosis represents a current health problem as it accounts for approximately 20% to 25% of all cerebral ischemic events (CIEs) in the United States. 8 The role of triplex sonography in the assessment of the extracranial carotid arteries is well established and has become, in most cases, the initial evaluatory modality for patients presenting with known or suspected carotid artery disease.9,10
Added to conventional subjective gray-scale assessment of individual atherosclerotic lesions, the introduction of quantitative methods, such as SWE, has provided the potential for further characterizing these lesions (plaques) as vulnerable or not. Vulnerable lesions are those which, based on their histologic and morphologic features, predispose a patient to an increased risk of a CIE due to plaque or thrombus embolization. Composition of these lesions varies, and each may contain an amalgamation of lipids, inflammatory cells, cellular debris, calcium, fibrous connective tissue, and thrombus. 11 The histologic content ultimately determines the gross structural morphology and overall stiffness of these lesions which ultimately determines their sonographic appearance. There is ample evidence in the medical literature that particular structural types of plaque are predictive of an increased risk of both cerebral and cardiac ischemic sequelae.12,13
SWE has demonstrated efficacy in providing additional information about the compositional features of carotid plaques, which can be particularly useful in identifying vulnerable or symptomatic plaques. In a study by Li et al, vulnerable plaques yielded a significantly lower YM value than those of stable plaques. The presence of plaque hemorrhage, thrombus, and other factors contributing to plaque vulnerability was also associated with a significantly lower YM. 14 In a study by Lou et al, which evaluated YM values in plaques present in patients presenting with cerebrovascular events, significant correlations were found between GW classification and mean YM values. The YM values of plaque stiffness were lower in lesions present in the carotid arteries of symptomatic patients than those present in the asymptomatic group. 6 The results obtained in this study are consistent with those obtained in other published studies.
The addition of slightly more than 2 minutes to routine carotid sonographic examination time makes the incorporation of quantitative SWE data quite practical in most clinical scenarios. It should be noted that in assessing the feasibility of incorporating these data points into routine vascular laboratory examinations, the additional amount of time only applies to instances were appreciable focal plaquing is identified. The overall impact on patient throughput when incorporating this additional criterion into routine carotid ultrasound testing should be negligible.
Conclusion
The current study demonstrates the feasibility and reliability of incorporating quantitative plaque assessment methods into routine carotid sonographic examinations. Quantitative SWE data correlated well with observer plaque scoring using GW assessment, and incorporation was relatively simple to apply. The use of SWE methods increases total examination time by slightly over 2 minutes in patients with appreciable focal carotid bifurcation plaquing. However, it provides demonstrated and advantageous information on plaque morphology, useful in both risk assessment and management decisions in patients with carotid artery disease.
While this pilot study affirms the feasibility and reliability of incorporating quantitative SWE data as an additional means of characterizing focal atheromata during routine carotid sonographic examinations, further large-scale studies are needed to determine the sensitivity and specificity of individual kPa values associated with specific plaque types. Additional studies to correlate pSWE data obtained in individual plaques with either surgical or histopathologic determination of lesion vulnerability would also be needed to add the predictability of this modality to the diagnostic armamentarium.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
