Abstract
Objective:
The development of ciliochoroidal effusions and secondary acute angle-closure crisis (AACC) is an uncommon side effect of topiramate, a common antiepileptic now FDA-approved for migraine prophylaxis. The mechanisms that underlie the development of ciliochoroidal effusions after topiramate use remain unclear.
Materials and Methods:
Ultrasound biomicroscopy (UBM) was also performed in all participants after stopping topiramate.
Results:
Six patient cases are presented with medication-induced AACC following the initiation or escalation of topiramate. Ciliochoroidal effusions were confirmed by gray-scale sonography in all patients at presentation. The images revealed either plateau iris configuration or atypical plateau iris configuration. Plateau iris configuration is defined by presence of an anteriorly rotated ciliary body processes and an absent posterior sulcus. Atypical plateau iris configuration refers to when the iris inserts directly into the ciliary body face.
This case series, of medication-induced angle-closure crisis, suggests that plateau iris configuration is a shared anatomical feature in the development of topiramate-induced ciliochoroidal effusions.
Topiramate (Topamax™) is a sulfa-derivative monosaccharide that was first approved for use as an antiepileptic medication in 1996. 1 It has been gaining popularity for its use in nonepileptic conditions, including bipolar personality disorder,2,3 alcohol dependence, 4 smoking cessation, 5 and migraine prophylaxis. 6 In 2012, evidence-based guidelines on episodic migraine prevention were updated with topiramate indicated as a first-tier treatment option.7,8 With over 8.5 million prescriptions a year per the Agency for Healthcare Research and Quality Survey, topiramate was reported as the 89th most prescribed drug as of 2018. 9
As the range of indications for topiramate’s use has continued to expand, the prevalence of topiramate-induced adverse effects has concomitantly increased. Although rare, with only 23 reported cases out of approximately 825,000 users in a post-marketing surveillance report,10-12 complications of myopic shift and acute angle-closure glaucoma secondary to ciliochoroidal effusions are a known side effect with potentially devastating consequences to vision. 13 Ciliochoroidal effusions have been associated with other sulfa derivatives as well, including sulfamethiozole, acetazolamide, and sulfapyridine. 14 In this study, the intent was to demonstrate that ultrasound biomicroscopy (UBM) was an important test. Furthermore, it was important to demonstrate that six cases of topiramate-induced ciliochoroidal effusions were all found to have underlying typical plateau iris (PIC) or atypical plateau iris configuration (APIC), after resolution of the ciliochoroidal effusions.
Materials and Methods
This was a retrospective review of patients seen at the W.K. Kellogg Eye Center, University of Michigan with topiramate-induced ciliochoroidal effusion between May 2005 and November 2017. Approval from the University of Michigan Institutional Review Board was obtained, and the study followed the tenets set forth by the Declaration of Helsinki. Participants were included in the study if they met the following criteria: clinically significant ciliochoroidal effusions after using topiramate (Topamax™), and had ultrasound imaging during their clinical course. Exclusion criteria included ocular disease or condition that had the potential to affect angle anatomy, including prior penetrating trauma, prior anterior segment surgery (excluding cataract surgery), nanophthalmos (<21.0 mm axial length), or uveitis.
The primary outcome measure of this study was angle morphology on UBM after resolution of the effusions. Secondary outcome findings included duration of topiramate usage before symptom onset, demographic information including sex and age, and symptoms on presentation. The ultrasound equipment system with transducers to image the eye is shown in Figure 1. Representative structures of the anterior segment of a normal eye, forms of PIC (ie, typical and atypical), and plateau iris syndrome with angle closure are shown in Figure 2.
This figure shows a typical ultrasound instrument with ultrasound transducers (Quantel Absolu, Bozeman, MT) used to image ocular structures. On the left side of the monitor, there is a 20 mHz transducer used for posterior segment imaging. On the right side of the monitor, there is a 15 mHz transduer (left) also used for posterior segment imaging and a 50 mHz transducer with a green sleeved cover filled with solution used for anterior segment imaging (right).
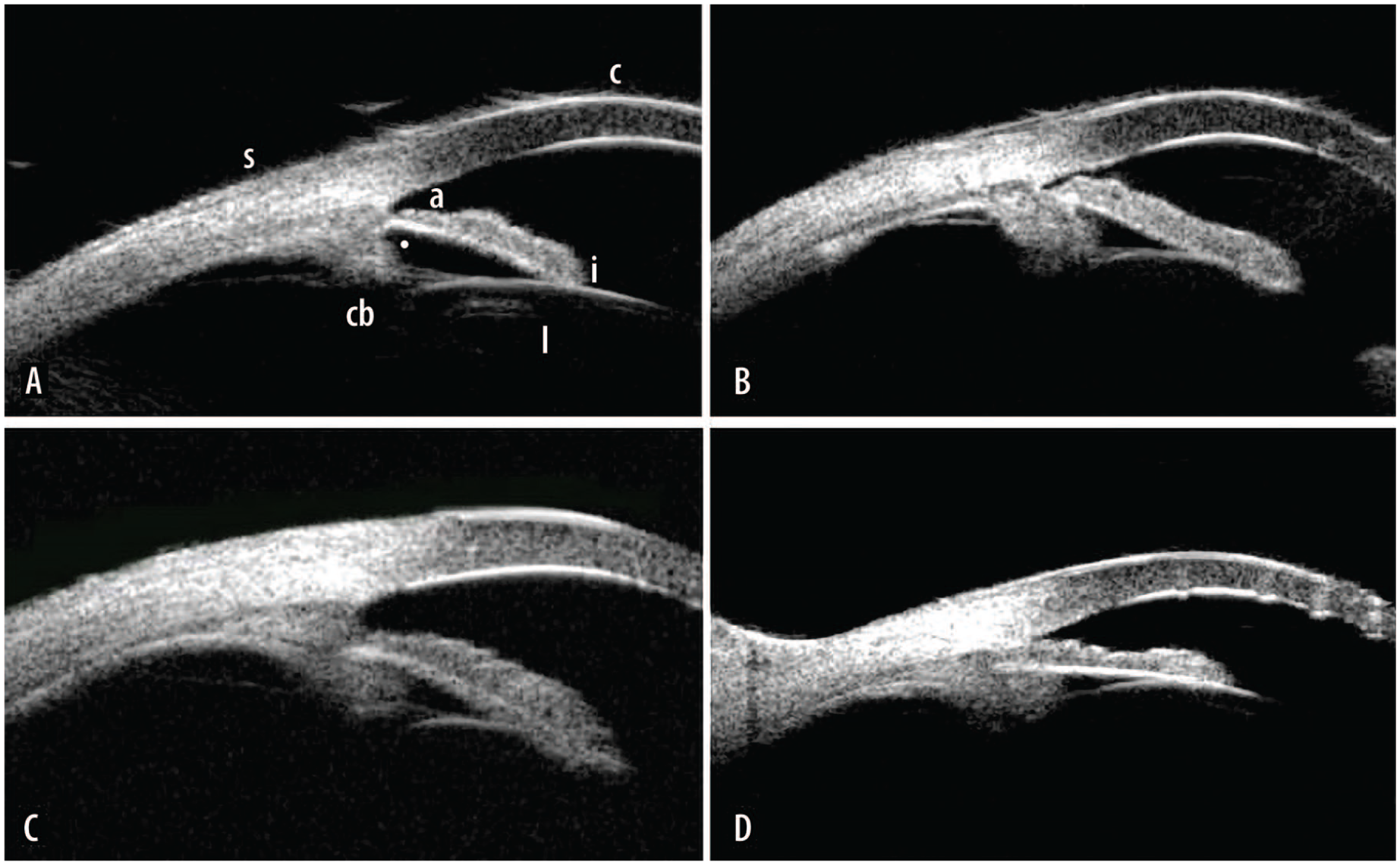
Ultrasound biomicroscopy images of anterior segment angle. Image A shows normal anatomy showing cornea [c], open angle [a], iris [i], portion of crystalline lens [l], ciliary sulcus [•], ciliary body [cb] process, and sclera [s]. Image B shows typical plateau iris configuration with narrow angle compared with A, absent ciliary sulcus compared with A, and anterior rotation of the ciliary body process. Image C shows atypical plateau iris configuration with open angle, iris root inserted on ciliary body, absent ciliary sulcus, and anterior rotation of the ciliary body process. Image D shows plateau iris syndrome with closed angle, absent ciliary sulcus, and anterior rotation of the ciliary body process.
Results
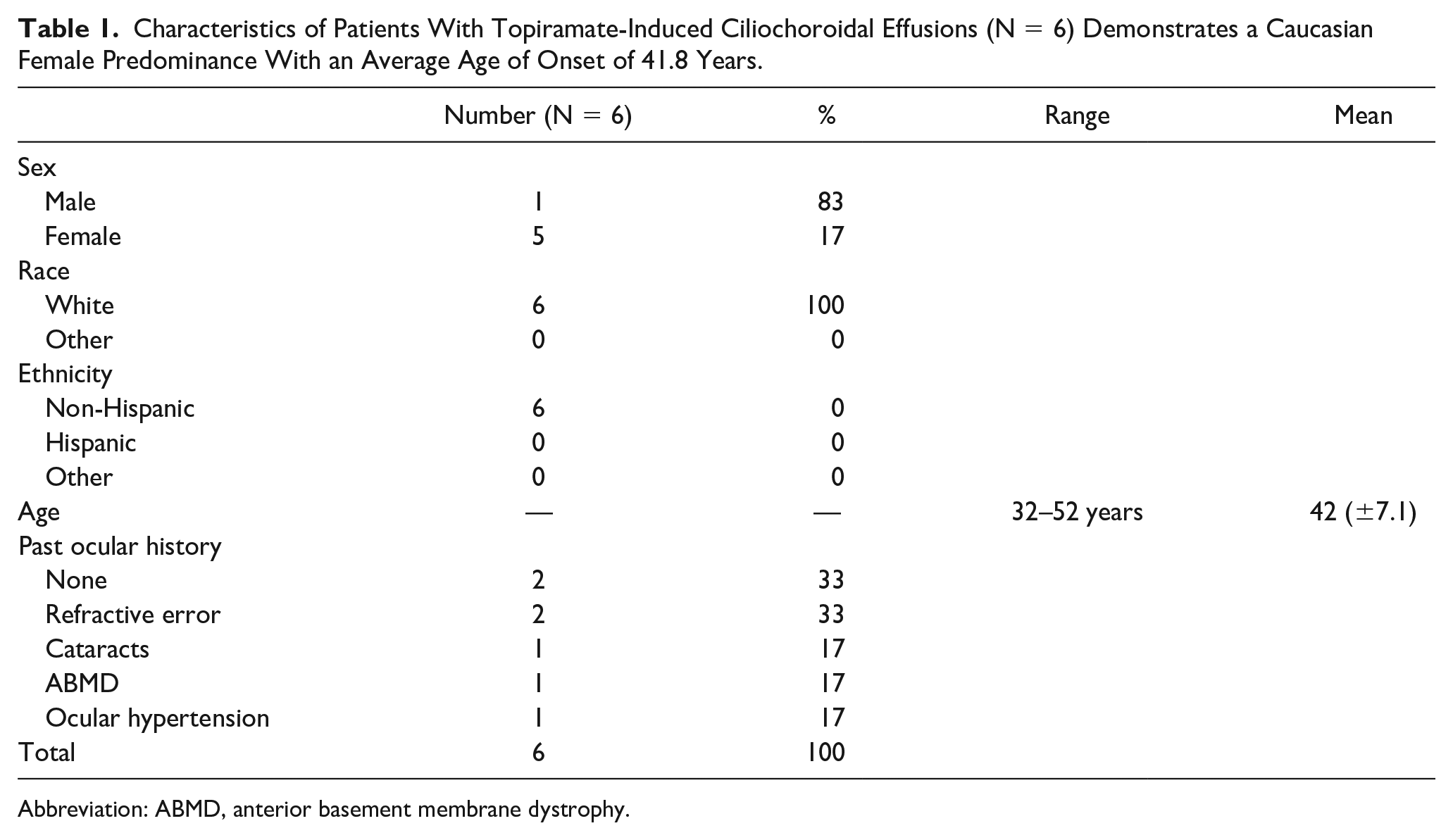
Eight patients presented with topiramate-induced choroidal effusions, but two patients were excluded from the review due to prior diagnoses uveitis and nanophthalmos. The average age of onset among the remaining six patients was 42 ± 7.9 years (range = 32–52). The majority (5/6 cases, 83%) were female, and all identified as non-Hispanic white (Table 1). The most common reason for using topiramate was for migraine prophylaxis (4/6 cases, 67%) (Table 2). The average time between medication start or dosage increase and symptom onset was 13 ± 2.7 days (range = 8–14).
Characteristics of Patients With Topiramate-Induced Ciliochoroidal Effusions (N = 6) Demonstrates a Caucasian Female Predominance With an Average Age of Onset of 41.8 Years.
Abbreviation: ABMD, anterior basement membrane dystrophy.
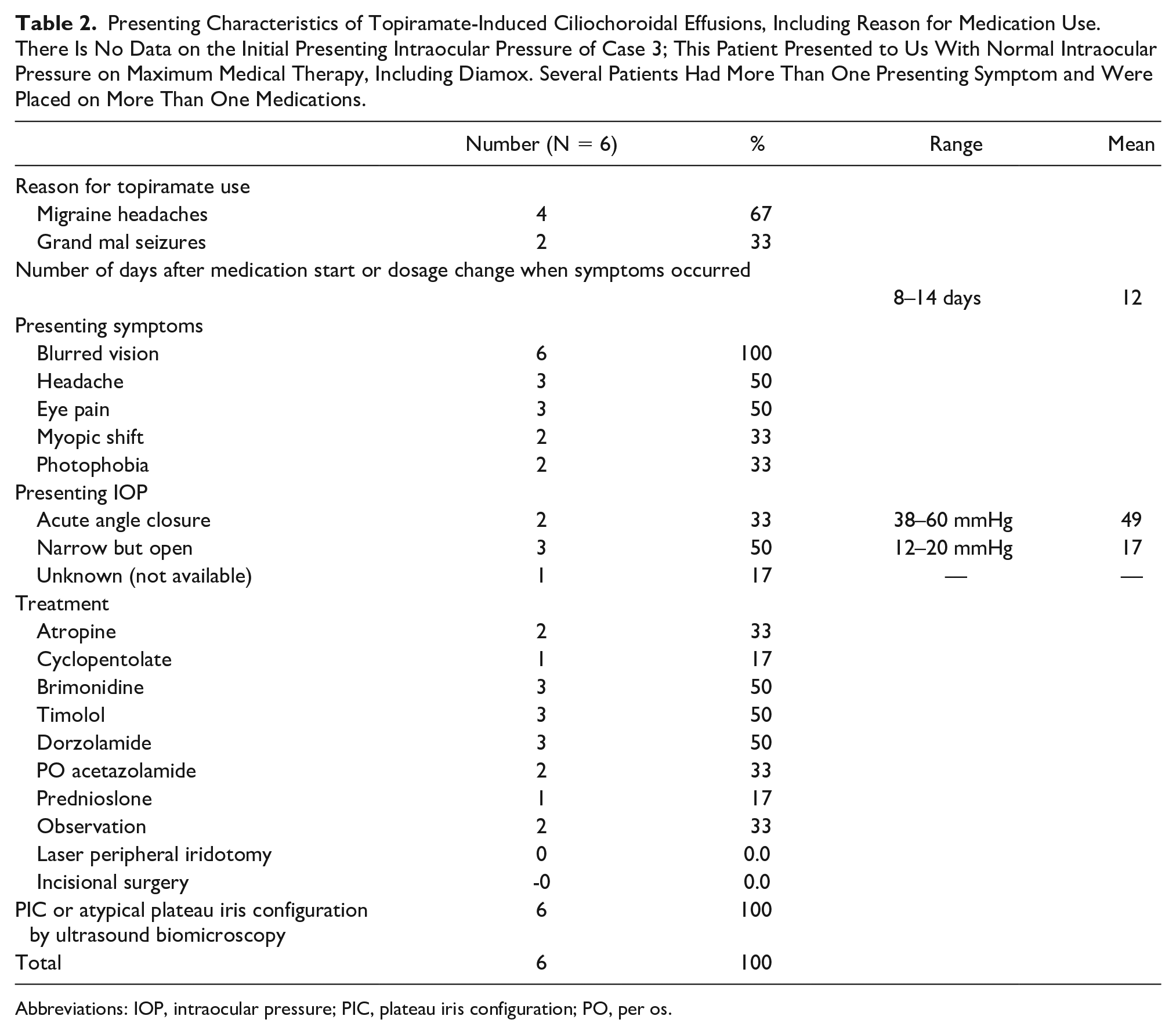
Presenting Characteristics of Topiramate-Induced Ciliochoroidal Effusions, Including Reason for Medication Use. There Is No Data on the Initial Presenting Intraocular Pressure of Case 3; This Patient Presented to Us With Normal Intraocular Pressure on Maximum Medical Therapy, Including Diamox. Several Patients Had More Than One Presenting Symptom and Were Placed on More Than One Medications.
Abbreviations: IOP, intraocular pressure; PIC, plateau iris configuration; PO, per os.
The most common presenting symptom was blurred vision (6/6 cases, 100%) (Table 2). Cases 2, 3, and 4 (3/6 cases, 50%) presented in angle-closure crisis of both eyes. We were unable to obtain the records from Case 3’s presenting emergency room visit; the IOP was within normal limits on maximum medical therapy upon evaluation by our clinic. The other two cases had an average presenting IOP of 49 (range = 38–60), which was medically managed until resolution of the effusions. All three cases received a topical cycloplegic (atropine 1% or cyclopentolate 1%), a topical carbonic anhydrase inhibitor (dorzolamide 2% or brinzolamide 1%), timolol 0.5%, and brimonidine 0.2%. Two cases were also prescribed 500 mg extended release oral acetazolamide twice per day. Case 3 had bilateral laser peripheral iridotomies prior to presentation, which had no effect on the IOP. No incisional surgeries were performed.
Cases 1, 5, and 6 had normal IOP on presentation (mean = 17, range = 10–20), but were all documented to have narrow angles and a shallow peripheral anterior chamber on examination. Case 5 had an isolated and painless acute 5-diopter myopic shift.
Five cases had a gray-scale imaging at the time of diagnosis demonstrating active ciliochoroidal effusions, and four had a UBM demonstrating the anterior ciliochoroidal effusions (Figure 3). Case 3 was initially diagnosed and treated at an outside facility, with resolution at the time of referral. Case 5 was seen over the weekend when UBM was not available, and had complete resolution at the time of the Monday follow-up appointment. All cases were seen in follow-up after resolution of symptoms, and all had documented ciliochoroidal effusion resolution by sonographic exam. Later recall of these cases showed typical or atypical PIC by UBM (Figure 3).
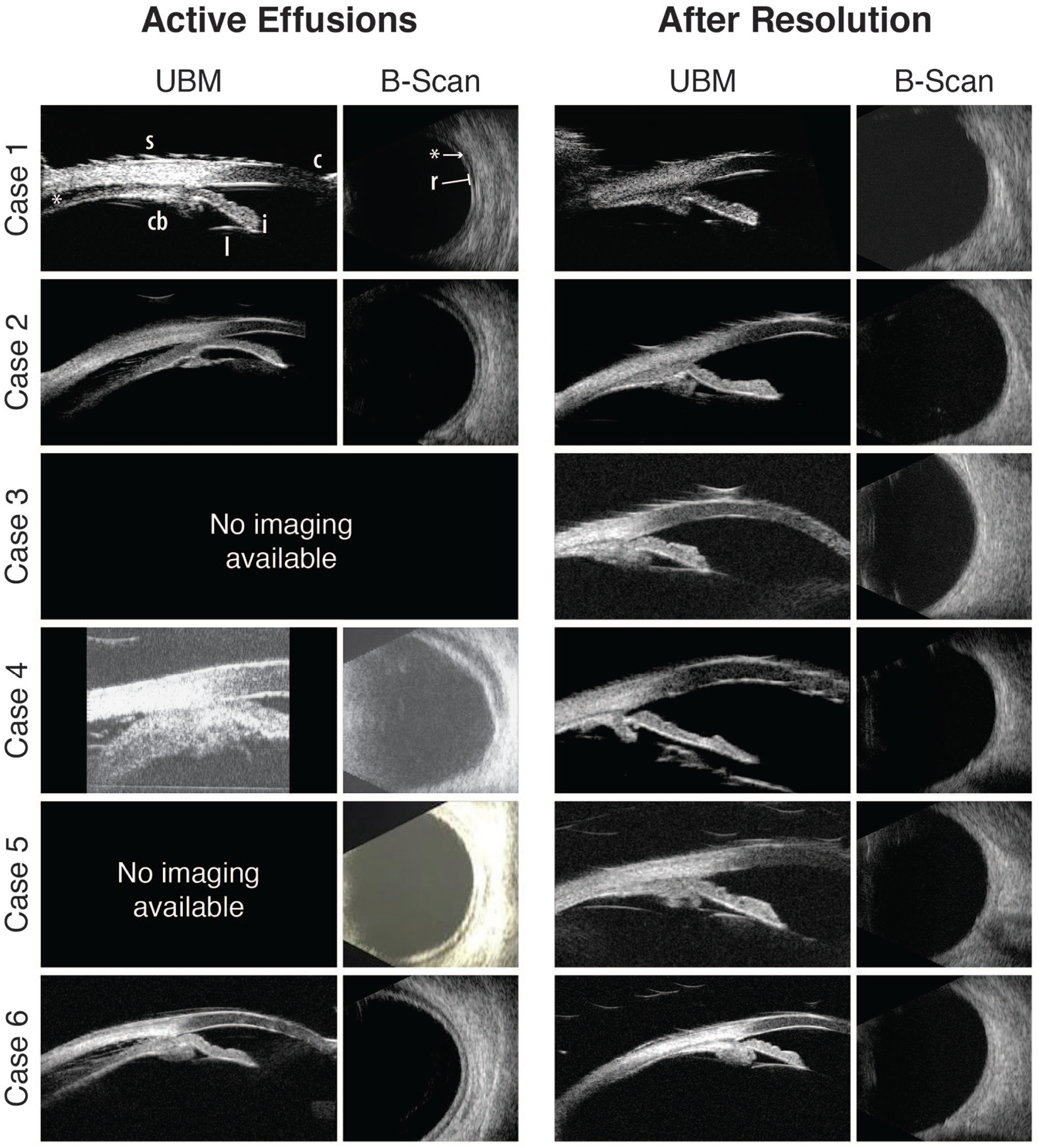
Ultrasound imaging of cases during active ciliochoroidal effusions and after resolution showing the anterior segment by UBM and the posterior segment with gray-scale imaging. Case 1, active effusions have the UBM image showing the cornea [c], iris [i], portion of crystalline lens [l], absent ciliary sulcus [not shown], ciliary body [cb] process, sclera [s], and [*] shows the effusion which looks like a “dark line.” Case 1, active effusions, gray-scale image showing the thin retina [r] and [*] shows the effusion which looks like a “dark line.” Case 1, after resolution shows the same structures and no effusion in both the UBM and gray-scale images. In addition, all of the after resolution UBM images show plateau iris.
Discussion
In this study, 100% of cases seen at the Kellogg Eye Center had evidence of typical PIC or atypical PIC on UBM testing following topiramate cessation and resolution of the ciliochoroidal effusions. There have been three separate case reports of medication-induced ciliochoroidal effusions that occurred in patients with typical or atypical PIC.15-17 The most recent case reported by Agarwal showed UBM images during the topiramate-induced ciliochoroidal effusion and after the resolution of the ciliochoroidal effusion. While it is difficult to estimate the frequency of such idiosyncratic drug reactions, a recent health services research project reported that angle-closure glaucoma among individuals using topiramate was estimated at one out of every 2505 new topiramate users. 18 In this health services data set based upon billing codes, there were no clinical gonioscopy data or ultrasound testing to determine the presence of typical or atypical PIC. While this small number of cases is inadequate to suggest an association between this angle anatomy and idiosyncratic drug reaction with ciliochoroidal effusions, it is certainly curious that these cumulative nine cases of drug-associated ciliochoroidal effusions share a common ultrasound imaging finding of typical or atypical PIC.
The term PIC is defined as the presence of anteriorly positioned ciliary processes that results in a narrow or absent ciliary sulcus. The term atypical PIC was proposed by Man et al. to describe a similar configuration of anteriorly rotated ciliary processes with iris root insertion into the top of the ciliary processes, rather than into the angle. 16 Using UBM in conjunction with the examination, PIC was found to have a prevalence up to 37.0% in patients with chronic primary angle-closure glaucoma and 19.2% in patients with open-angle glaucoma. 19
The reasons for why typical or atypical PIC may be associated with such idiosyncratic drug-induced ciliochoroidal effusions is not understood but may lie in the fluid balance of the uveal tissues in the eye. Quigley proposed that the fluid balance of the iris may play a pivotal role in the risk for acute angle-closure crises in subjects with narrow angles. 20 Although initially thought to be the result of an osmotic disturbance with the lens resulting in swelling and shallowing of the anterior chamber, Craig et al. reported that lens swelling only accounted for a small portion of shallowing (9%–16%) and instead, was predominantly due to ciliochoroidal effusion. 21 Another study by Hesami et al. further supported these findings as significant increases in myopia were reported in patients following a month of topiramate use while reporting no significant changes in anterior chamber angle, depth, or volume. 22 While the relationship of this typical or atypical PIC and intraocular fluid balance is not known, the potential role of this anatomical configuration in these idiosyncratic drug reactions with ciliochoroidal effusions is of interest going forward by using ultrasound imaging to evaluate this anatomical configuration after resolution of these medication-associated ciliochoroidal effusions. With this additional examination of these uncommon idiosyncratic reactions, it may be possible to model the relative risk for such medication-induced ciliochoroidal effusions.
There are several limitations of this study that include a small number of cases of topiramate-induced ciliochoroidal effusions. While it was considered a comprehensive medical record search to identify more cases, this approach was challenging as there are no ICD-9 or ICD-10 codes specific for ciliochoroidal effusions and adverse drug reaction. Although the included list is meant to be comprehensive, it is possible that the six cases do not include every case seen at the University of Michigan during this time period described in the methods. As the case series is retrospective and encompasses patients seen by multiple providers in different subspecialties and clinics, dynamic gonioscopy was not always performed to determine the presence of plateau iris. Due to this limitation, UBM imaging was used to determine the anatomical configuration of typical or atypical PIC after resolution of the ciliochoroidal effusions.
Conclusion
In summary, UBM imaging after resolution of topiramate-induced ciliochoroidal effusions demonstrated that all six cases had typical or atypical PIC. To the best of our knowledge, this is the largest case series to identify a shared structural anatomy of typical or atypical PIC in this idiosyncratic drug reaction of topiramate-induced ciliochoroidal effusions. Agarwal’s case study on topiramate-induced ciliochoroidal effusions also demonstrated PIC by UBM imaging. 17 While these combined cases showed typical or atypical PIC by UBM after resolution of the drug-induced ciliochoroidal effusions, follow-up imaging after resolution is needed to generate additional data on this observation. While this concordance between typical or atypical PIC and medication-induced ciliochoroidal effusions is limited to these new six cases and one recent case report, 17 if the clinician is knowledgeable of typical or atypical PIC in a patient, who has medical conditions treated with topiramate, then it is reasonable to advise such patients of this potential relationship. As these cases report reversal of this idiosyncratic reaction after stopping topiramate, informing the signs and symptoms to these patients, who have known typical or atypical PIC, is prudent. At the very least, it is our responsibility to educate patients of the possibility of this uncommon idiosyncratic condition, especially in patients who may already be noted to have this underlying angle anatomy of typical or atypical PIC.
Footnotes
Acknowledgements
A special thanks to Sarah E. Alward, MLA, who assisted with figure composition and editing. The University of Michigan echographers (Dr Bernadete M. Ayres, Tanya McClendon, and Elizabeth A. Parrish) provided the initial scans of these cases.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Sayoko Moroi has received funding for clinical trials in the past 3 years form Allergan, Aerie Pharmaceuticals, Icare USA, and NEI/NIH (R01EY022124). Dr Sayoko Moroi received royalties for her contribution on Shields Textbook of Glaucoma, 6th and 7th editions from Wolters Kluwer.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Heed Ophthalmic Foundation (EAB).
