Abstract
Systemic lupus erythematosus (SLE) is a chronic idiopathic autoimmune disease. SLE can involve almost any part of the eyes. However, bilateral angle-closure glaucoma due to lupus choroidopathy that is accompanied by polyserositis and nephropathy is rare. We report a 21-year-old woman whose clinical manifestations were diagnosed as bilateral angle-closure glaucoma caused by ciliochoroidal effusion. Subsequently, SLE and lupus nephritis were diagnosed on the basis of malar rash, photosensitivity, proteinuria, positive anti-Smith and anti-DNA antibodies, and a renal histopathological biopsy. After 1 month of treatment with steroids and immunosuppressive drugs, the patient’s intraocular pressure returned to normal, visual acuity improved, and lupus nephritis was effectively controlled. Bilateral secondary acute angle closure caused by SLE choroidal disease can be an ocular manifestation of SLE, and is usually accompanied by polyserositis and nephropathy. High-dose steroids and immunosuppressive therapy should be immediately and actively provided for this condition.
Keywords
Introduction
Systemic lupus erythematosus (SLE) is a chronic idiopathic autoimmune disease caused by the combination of autoantibodies and abnormally activated immune complexes. 1 SLE can involve almost any part of the eyes. Ocular symptoms of SLE include blepharitis, keratoconjunctivitis sicca, scleritis, retinal vascular disease, choroidopathy, and optic neuropathy. However, bilateral angle-closure glaucoma due to lupus choroidopathy that is accompanied by polyserositis and nephropathy is rare. We report a patient who developed SLE with angle-closure glaucoma as the ocular symptom, and discuss the possible pathogenesis and treatment of this disease.
Case report
A 21-year-old Chinese woman presented with a 2-day history of decreased bilateral visual acuity, accompanied by bilateral eyelid edema. There was no ocular history or family history. Uncorrected visual acuity was 20/400 in both eyes. The best corrected visual acuity of both eyes was 20/100, with −6.00 spherical equivalent in both eyes. The intraocular pressure was 35 mmHg in the right eye and 30 mmHg in the left eye, as measured using a non-contact tonometer. There was eyelid edema and severe conjunctival chemosis in both eyes. Slit-lamp biomicroscopy showed a clear cornea, shallow anterior chamber, and narrow anterior chamber angle in both eyes. The pupil size and light reflection of both eyes were normal. A fundus examination showed that the optic nerve had no edema and the retina of the posterior pole was wrinkled (Figure 1a). Ultrasound biomicroscopy showed a shallow anterior chamber, 360° angle closure, and edema and pronation of the ciliary body (Figure 1b). B-scan ultrasonography showed choroidal thickening with effusion (Figure 1c). Using optical coherence tomography, serous retinal detachment was detected in both eyes (Figure 1d). The patient was initially diagnosed with acute angle-closure glaucoma and provided medication (brinzolamide and compound tropicamide eye drops twice a day, and 200 mL of intravenous 20% mannitol per day) to reduce intraocular pressure. To identify the cause of the patient’s acute angle-closure glaucoma, she underwent a further systemic examination. The results of all laboratory tests were as follows: proteinuria was 2090.83 mg/24 hours, the plasma albumin level was 27.1 g/L, complement levels were low (complement 3 level: 0.308 g/L; complement 4 level: <0.0712 g/L), and the immunoglobulin level was increased (19.4 g/L). The titer for antinuclear antibody was positive (1: 100), anti-dsDNA antibody was positive at 531.81 RU/mL (positivity >100), anticardiolipin antibody was positive at 13.1 RU/mL (positivity >10), and anti-nucleosome, anti-Smith, ribonucleoprotein/Smith, and anti-histone antibodies were positive. Chest computed tomography showed bilateral pleural effusion and atelectasis. Cardiac sonography showed pericardial effusion, and abdominal and pelvic computed tomography showed diffuse exudative changes in the abdominal cavity and mesenteric soft tissue, pelvic effusion, and a small amount of pleural effusion on both sides. A renal biopsy (Figure 2a) showed slight proliferation and segmental endothelial cell proliferation in the glomerular mesangial region and compound red substance deposition in periodic Schiff–methenamine silver–Masson staining in the mesangial area. Furthermore, there was mild edema in the proximal tubule, a small amount of protein casts and cellular casts in the distal tubule, and fluorescence in the mesangial area as shown by immunofluorescence. These findings were consistent with lupus nephritis (type II, activity index: 2). Electron microscopy (Figure 2b) showed glomeruli in kidney tissue. The arrangement of the capillary loops was irregular, and most of the vascular lumens were narrow or occluded. The basement membrane of the capillaries in some segments was irregularly thickened, and scattered electron dense deposits were observed on the epithelial side and under the endothelium. Podocytes were swollen, some vacuoles were degenerated, and the foot processes were diffusely fused. Mesangial cells and the mesangial matrix were severely proliferated, and there were massive electron dense deposits in the mesangial area. Focal inflammatory cell infiltration was observed in part of the mesangial area. These findings suggested the diagnosis of SLE with multiple serositis and lupus nephritis. The patient was administered methylprednisolone sodium succinate 500 mg pulse therapy for 3 days, with a gradual reduction in dose, combined with oral mycophenolate mofetil (0.25 g three times per day).

(a) A fundus examination shows that the posterior pole is wrinkled; (b) Ultrasound biomicroscopy shows 360° angle closure, and edema and pronation of the ciliary body; (c) B-scan ultrasonography shows choroidal thickening with effusion; (d) Optical coherence tomography shows serous retinal detachment.
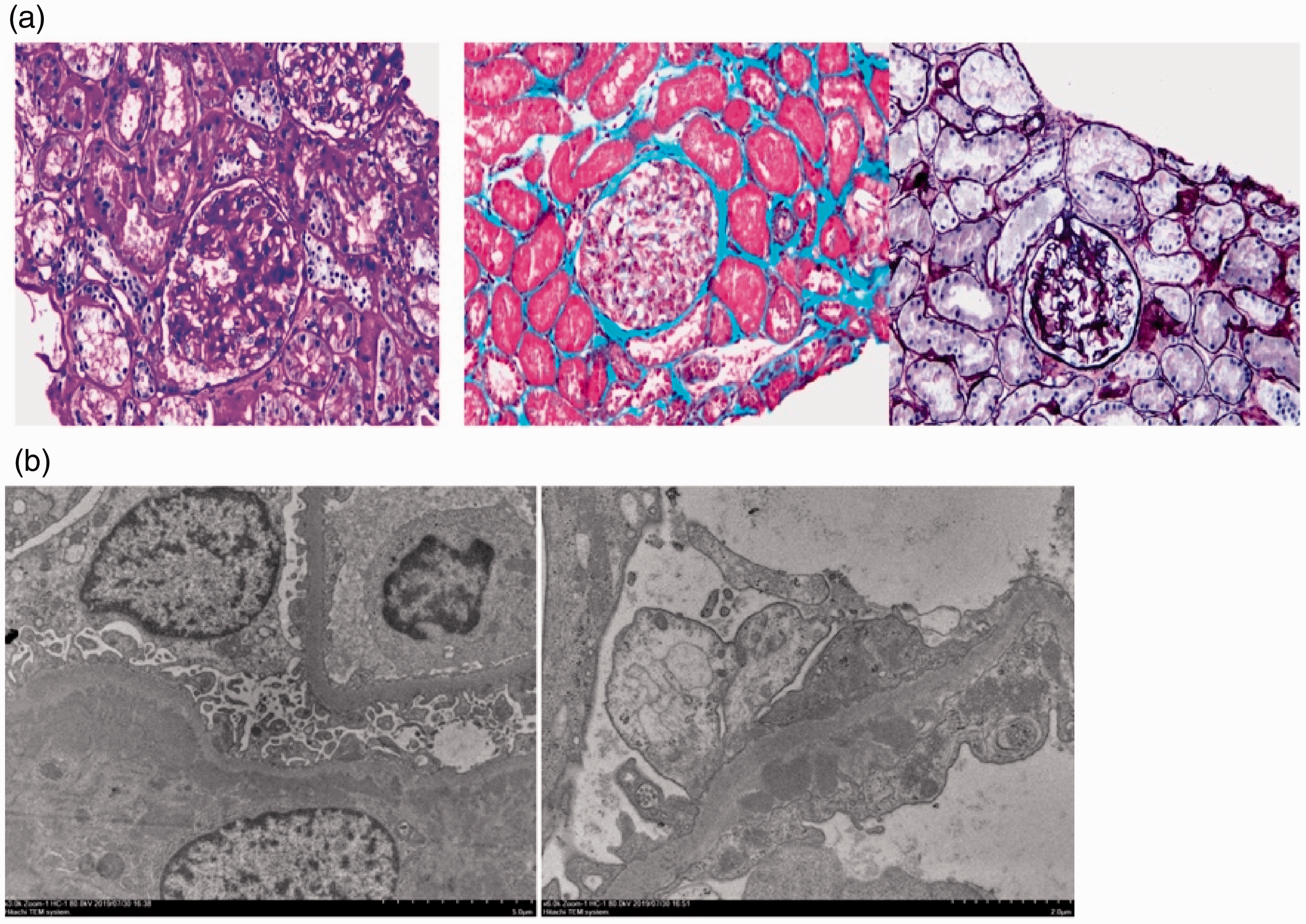
(a) Pathology of a renal biopsy; (b) Electron microscopic examination.
After 20 days of treatment, the patient’s best corrected visual acuity was 20/32 in the right eye, with −2.50 spherical equivalent, and 20/25 in the left eye, with −1.50 spherical equivalent. The intraocular pressure was decreased to 15 mmHg in the right eye and 16 mmHg in the left eye. Images of the fundus showed no obvious subretinal effusion (Figure 3a). Bilateral B-ultrasound showed slight choroidal thickening (Figure 3b). Optical coherence tomography showed that there was still a small amount of residual fluid under the retina, but the original normal structure was basically restored (Figure 3c). Laboratory studies showed that the albumin level was 40.7 g/L and protein was negative in the urine. During the subsequent 6 months, with steroids and immunosuppressive drug therapy, secondary acute angle closure did not recur and the nephritis remained under control.

(a) A fundus examination shows no obvious subretinal effusion; (b) B-ultrasound shows slight choroidal thickening; (c) Optical coherence tomography shows that the original normal structure has basically been restored.
Discussion
SLE is a chronic systemic autoimmune disease in which the pathology affects multiple organ systems including the skin, rheumatic immune system, kidneys, heart, eyes, and nervous system. SLE can affect many parts of the eye, including the eyelid, sclera, cornea, uvea, retina, choroid, and optic nerve. The most common complication of SLE associated with ophthalmopathy is keratoconjunctivitis sicca. 2 SLE with bilateral angle-closure glaucoma as an ocular manifestation is rare. Since the first report of this condition by Wisotsky et al. 3 in 1998, there have only been four other reports.3–6 Sun et al. 5 and Han et al. 6 each reported a case of bilateral angle-closure glaucoma; one case had central nervous system vasculitis and uncontrolled nephropathy, and the other had polyserositis.
Dias-Santos et al. 7 et al. analyzed ocular lesions of 161 patients with SLE. These authors showed that the incidence of glaucoma was 3%, and its pathogenesis was related to long-term use of corticosteroids and the older age of the selected patients. Our patient was a young woman with no history of corticosteroid use. Therefore, the pathogenesis of glaucoma in our patient is different from that reported by Dias-Santos et al. 7 The possible mechanism of angle-closure glaucoma in our patient with SLE is that choroidal edema and edema of the ciliary body caused fluid to enter the suprachoroidal cavity from choroidal capillaries. This then resulted in anterior rotation of the ciliary body, anterior displacement of the septum of the iris and closure of the chamber angle, obstruction of aqueous humor outflow, and elevation of intraocular pressure. Ciliary edema leads to relaxation of the suspensory ligament, thickening of the lens, and a change in myopia. The transient myopia found in our patient supports this mechanism. Our patient with angle-closure glaucoma as the ocular manifestation of SLE was accompanied by eyelid edema, pleural effusion, and pericardial effusion, and the pathogenesis of these lesions may be the same.
In our case, SLE was complicated by choroidal leakage, fluid entered the subretinal neuroepithelium, and exudative retinal detachment occurred. There are many possible mechanisms for these findings, which can be summarized as follows. First, deposition of immune complexes in the choroid and choroidal capillary layer and production of autoantibodies against retinal pigment epithelium (RPE) can lead to low choroidal perfusion and destruction of the extraretinal barrier. 8 Furthermore, hypertension caused by SLE nephropathy reduces choroidal blood flow, which leads to choroidal ischemia and destruction of the extraretinal barrier. 9 Finally, thrombosis can cause microvascular disease and then choroid lesions. 8 The combination of these three factors leads to low perfusion of the choroidal capillary layer, resulting in damage to the RPE and fluid entering the subretinal neuroepithelium.
Our patient’s kidneys and choroid were involved at the same time in SLE because of their similarity in pathogenesis and structure. The kidney and choroid are highly sensitive to deposition of immune complexes. 10 Stefater et al. 11 performed suprachoroidal drainage for patients with SLE complicated by choroidal leakage and found that the drainage was exudate. They speculated that local choroidal inflammation caused by deposition of immune complex on the choroid was the mechanism of SLE with choroidal leakage. In terms of structure, Bruch’s-RPE-choriocapillary complex is similar to the renal glomerulus, the choroidal capillary layer is similar to the glomerular capillary blood vessels, the Bruch membrane is similar to the glomerular basement membrane, and the RPE is similar to the glomerular epithelial cell layer. 12 As a result, 64% of patients with SLE and choroidal lesions are associated with renal lesions. 8
Systemic immunosuppression and high-dose corticosteroids are the first choice for treating bilateral secondary acute angle closure caused by SLE. Systemic treatment is provided at the same time, with local use of intraocular pressure-lowering drugs and ciliary muscle paralysis agents. Ciliary paralysis is prohibited in traditional angle-closure glaucoma, but in the case of edema of the ciliary body, it can reduce intraocular pressure by inducing the ciliary body to rotate backward. If systemic treatment is ineffective for choroidal lesions, a laser can be used to treat leaking lesions. If the effect of drug control of intraocular pressure is not good, peripheral iridectomy and suprachoroidal drainage can be considered.
Footnotes
Acknowledgements
We thank all of the ophthalmologists and physicians from The First Hospital Affiliated with Shandong First Medical University who participated in the care of the patient.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics statement and informed consent
The study was approved by the Ethics Committee of Shandong Provincial Qianfoshan Hospital. Written informed consent was obtained from the patient for publication of this Case Report and the accompanying images. A copy of the written consent is available for review by the Editor of the journal.
