Abstract
Infectious endophthalmitis after ocular surgery is a rare complication that can result in severe vision loss. Moraxella nonliquefaciens is an opportunistic pathogen that rarely causes ocular infections. We report a rare case of Moraxella nonliquefaciens endophthalmitis complicated by secondary angle-closure glaucoma occurring years after trabeculectomy and cataract surgery. Cycloplegia resulted in deepening of the anterior chamber and opening of the angles. Metagenomic next-generation sequencing of the aqueous humor identified Moraxella nonliquefaciens. Following multiple intravitreal vancomycin injections and vitrectomy with posterior capsulectomy, direct communication between the anterior chamber and the vitreous cavity was established, leading to resolution of the condition. The patient’s visual acuity was fully restored. We hypothesize that persistent infection with Moraxella nonliquefaciens may damage the lens zonules, resulting in zonular laxity and weakness; however, this represents only one possible mechanistic explanation and may play a secondary role in the development of secondary angle-closure glaucoma. Ophthalmologists should be aware that patients with Moraxella nonliquefaciens endophthalmitis may be at risk of zonular laxity and secondary angle-closure glaucoma, particularly years after trabeculectomy and cataract surgery. Prompt recognition and intervention may be vision-saving.
Introduction
Infectious endophthalmitis after trabeculectomy is a rare complication that can lead to severe visual loss. The affected eyes are often characterized by a leaking bleb and associated hypotony. 1 Streptococci, Haemophilus influenzae, and enterococci are commonly associated with late-onset endophthalmitis. 1
Moraxella species are part of the normal ocular and respiratory flora. Moraxella nonliquefaciens, a rare subtype of Moraxella, has low pathogenicity and rarely causes ocular infections. 2
Postoperative endophthalmitis caused by this bacterium is rare and has been associated with poor outcomes. 3 Herein, we described a case of M. nonliquefaciens endophthalmitis combined with secondary angle-closure glaucoma (SACG) occurring years after trabeculectomy and cataract surgery. After intravitreal vancomycin administration and pars plana vitrectomy (PPV) with posterior capsulectomy, the patient’s visual acuity was completely restored. To our knowledge, this is the first report of M. nonliquefaciens endophthalmitis complicated by SACG and the report with the best visual outcome among published cases of M. nonliquefaciens endophthalmitis.
Case report
A man in his 50 s presented to the Department of Ophthalmology at Tongji Hospital in February 2024 with a 2-day history of ocular redness, pain, and decreased visual acuity in the left eye. Four years earlier, he had been diagnosed with angle-closure glaucoma and had undergone trabeculectomy in the left eye with subconjunctival administration of 5-fluorouracil (5-Fu). Postoperatively, malignant glaucoma developed, and cataract surgery of the left eye was subsequently performed 3 years ago. At the most recent follow-up before presentation, the left eye showed a normal anterior chamber depth (ACD) with open angles. There was no history of systematic disease or ocular trauma.
On admission, visual acuity was 20/20 in the right eye and “hand motion” in the left eye. Intraocular pressure (IOP) was 19 mmHg in the right eye and 45 mmHg in the left eye. Slit-lamp examination of the left eye revealed conjunctival hyperemia, corneal edema, a 3-mm hypopyon, and a closed anterior chamber angle (Figure 1(a)). The filtering bleb was thin-walled and Seidel-negative, with no evidence of purulent discharge (Figure 1(b) and (c)). Fundus examination revealed vitreous clouding, with the fundus not visible.
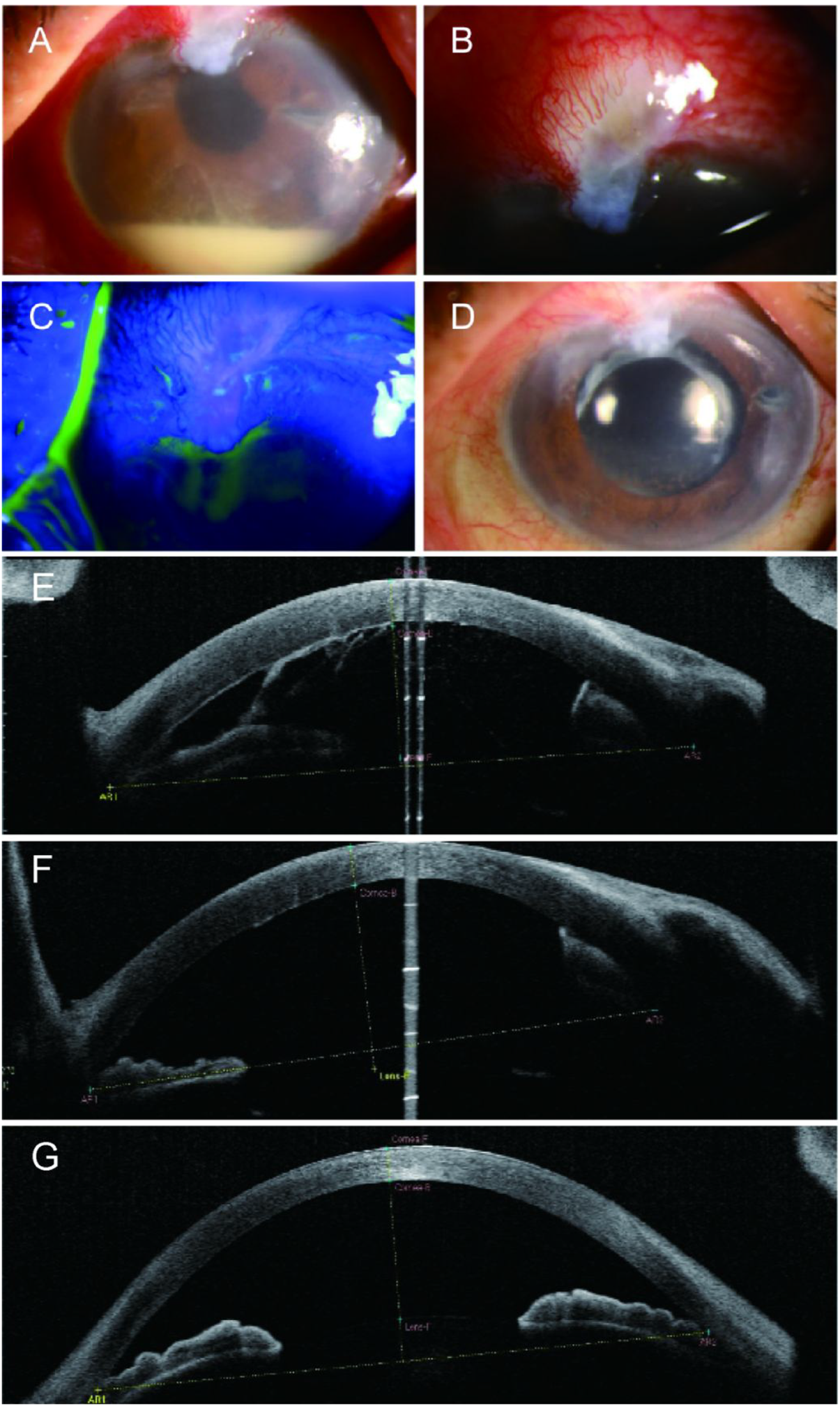
(a) The anterior segment image during the initial visit revealed hyperemic conjunctiva, cornea edema, and hypopyon. (b) The filtering bleb was observed to be thin-walled, with no purulent discharge. (c) The Seidel phenomenon was negative. (d) Three months later, the anterior segment image showed the resolution of endophthalmitis. (e) AS-OCT of the left eye indicated a shallow anterior chamber and closed angles during the initial visit. (f) After the ciliary muscle was completely paralyzed, the anterior chamber in the left eye clearly deepened and (g) AS-OCT of the right eye. AS-OCT: anterior segment optical coherence tomography.
The anterior chamber in the left eye (pseudophakic) was significantly shallower than that in the right eye (phakic). Anterior segment optical coherence tomography (AS-OCT) confirmed angle closure in the left eye. The ACD measured 2.26 mm in the left eye and 2.36 mm in the right eye (Figure 1(e) and (g)). After complete cycloplegia, the anterior chamber in the left eye deepened markedly, with the ACD increasing to 3.02 mm (Figure 1(f)).
Ultrasound B-scan demonstrated dense punctate hyperreflective echoes in the vitreous cavity, without evidence of choroidal abnormality or vitreous abscess. A-scan biometry and optical biometry using IOLMaster showed that axial length, corneal diameter, and corneal curvature were comparable between the two eyes and within normal ranges. In view of the thin-walled filtering bleb, ultrasound biomicroscopy (UBM) was not performed.
Based on these findings, the patient was diagnosed with endophthalmitis complicated by SACG. An anterior chamber paracentesis was performed on the first day for smear, culture, and metagenomic next-generation sequencing (mNGS) testing.
Considering the absence of an organized vitreous abscess, we initially opted for aggressive medical management. Intravenous mannitol (20%, 1 g/kg) was administered to reduce IOP. Tobramycin 0.3%/dexamethasone 0.1% eye drops, bimatoprost 0.03% eye drops, and cycloplegic agents for ciliary muscle paralysis (0.5% compound tropicamide eye drops and 1% atropine gel) were administered topically to the left eye. After 3 days, the hypopyon had completely resolved, and corneal edema had improved, suggesting that anterior chamber inflammation was adequately controlled. IOP returned to normal levels, and bimatoprost was discontinued.
To further identify the cause of the patient’s endophthalmitis and SACG, a comprehensive systemic evaluation was performed. Laboratory tests revealed a normal white blood cell count; an elevated C-reactive protein level (1.2 mg/L); decreased plasma total protein (58.5 g/L), albumin (38.9 g/L), and globulin (19.6 g/L) levels; and a reduced total T-cell count (556/µL; CD4+ T cells, 295/µL; CD8+ T cells, 197/µL). No endogenous source of infection was identified.
As routine pathogen detection failed to yield positive results, antimicrobial susceptibility testing could not be performed. A sample of aqueous humor was therefore sent to Beijing GiantMed Diagnostics Lab for mNGS and cytokine analysis. mNGS identified M. nonliquefaciens (Figure 2(a)), while cytokine analysis demonstrated a severe inflammatory response in the left eye. Unlike most Moraxella species, M. nonliquefaciens is susceptible to vancomycin in addition to cephalosporins. 4

(a) mNGS findings at diagnosis: mNGS detected the total DNA reads, of which 99.84% were from M. nonliquefaciens and 0.03% from Neisseria meningitidis and (b) mNGS findings after adequate intravitreal antibiotics: mNGS detected the total DNA reads, of which 3.8% were from M. nonliquefaciens. mNGS: metagenomic next-generation sequencing.
On day 4, because of the lack of significant visual improvement and persistent vitreous inflammation on B-scan ultrasonography, the patient received an initial intravitreal injection of vancomycin (1 mg/0.1 ml) and a 1-week course of intravenous ceftazidime and dexamethasone, followed by oral methylprednisolone. The patient responded well and showed gradual visual recovery over the following days. On days 9 and 12, second and third intravitreal injections of vancomycin (1 mg/0.1 ml) combined with ceftazidime (2 mg/0.1 ml) and ceftazidime alone, respectively, were administered. As a result, visual acuity improved to 20/200, and the ACD returned to normal (peripheral ACD (PACD) = 1 corneal thickness (CT)). However, macroscopic flake-like vitreous opacities were still observed on B-scan echography. On days 14 and 17, the patient received fourth and fifth intravitreal injections of vancomycin, after which the vitreous inflammation gradually subsided.
On day 24, a sixth intravitreal injection of vancomycin was administered, after which visual acuity improved to 20/60 and IOP in the left eye was 20 mmHg. Ultrasonography showed small residual patches of vitreous haze, and no choroidal effusion or detachment was observed throughout the treatment course. A repeat mNGS analysis of the aqueous humor was performed and continued to detect M. nonliquefaciens, with a relative abundance of 3.8% (Figure 2( b)). The patient was subsequently discharged.
Two months later, the patient re-presented with pain in the left eye and a shallow anterior chamber. At that time, IOP was 38 mmHg and visual acuity was 20/50. B-scan ultrasonography again revealed no choroidal effusion or detachment. The patient underwent pars plana vitrectomy with vancomycin added to the irrigating solution, combined with posterior capsulectomy. Intraoperatively, no retinal hemorrhage, vasculitis, or purulent material was observed in the fundus. On postoperative day 1, IOP and ACD had returned to normal levels.
At 6-month follow-up, visual acuity was 20/40 with stable IOP. The cornea was clear, and no anterior chamber inflammation or SACG recurrence was observed (Figure 1(d)). Figure 3 illustrates clinical progression. The study conforms to the Case Report (CARE) guidelines, 5 and informed consent was obtained.

The timeline of the case development. (Using Figdraw). VA: visual acuity; IOP: intraocular pressure; HM: hand motions; FC: finger counting; CAZ: ceftazidime; VAN: vancomycin; DEX: dexamethasone.
Discussion
M. nonliquefaciens is gram-negative coccobacillus that is typically non-pathogenic and generally requires predisposing factors to cause infection. 2 Previous studies have identified older age, immunocompromised states, diabetes mellitus, and prior ocular surgery as risk factors for ocular infections caused by Moraxella species. 2
Ocular infections due to M. nonliquefaciens most commonly involve the conjunctiva and cornea. 4 Endophthalmitis caused by this organism is rare, with only 10 cases reported in the literature. In one patient, infection occurred following corneal erosion associated with contact lens use. 6 Another report described delayed-onset endophthalmitis in an 84-year-old man after intravitreal injection for age-related macular degeneration. 7 The remaining eight cases involved patients who had undergone trabeculectomy, six of whom also had cataract surgery. The average time to presentation was approximately 5 years postoperatively. The mean age of affected patients was 74 years (range, 62–90 years), and three had underlying systemic diseases.3,8–13
However, previously reported intraocular cases are limited and lack large-scale data. A recent cohort study of 106 cases of Moraxella keratitis demonstrated that the disease often presents with severe inflammation, slow response to antimicrobial therapy, a high rate of surgical intervention (47.2%), and poor prognosis. 14 Older age, hypertension, and corneal perforation were identified as independent predictors of poor prognosis and need for surgery. 14 These features are consistent with the clinical course observed in our patient. Fortunately, the final visual outcome in this case was favorable, with recovery to 20/40. These findings highlight that both corneal and intraocular Moraxella infections require early etiologic diagnosis and prompt, aggressive antimicrobial and surgical management to optimize visual outcomes.
Unlike previous cases of endophthalmitis alone, the present case involved the rare combination of endophthalmitis with SACG. To our knowledge, this is the first such case reported in the literature. SACG is an uncommon condition that may result from inflammation, trauma, lens-related abnormalities, or certain medications. 15 Key mechanisms include choroidal expansion, anterior rotation of the ciliary body, and zonular laxity. 16 In these eyes, cholinergic agonists such as pilocarpine—the first-line therapy for primary angle-closure glaucoma—can exacerbate forward displacement of the lens–iris diaphragm through miosis, potentially worsening the condition. Cycloplegic agents such as atropine can theoretically counteract this mechanism by relaxing the ciliary body and deepening the anterior chamber. 17
In the present case, IOP in the left eye increased to 45 mmHg on initial presentation. In contrast, patients with endophthalmitis typically exhibit normal or low IOP due to ciliary body inflammation and reduced aqueous humor production. 18 This atypical finding prompted careful evaluation. The left eye anterior chamber was significantly shallower than at the cataract surgery 3 years earlier and compared with the contralateral eye. After complete cycloplegia, the anterior chamber deepened and the angles opened, supporting the diagnosis of SACG.
Several mechanisms may explain the angle closure in this case. Choroidal effusion or detachment was unlikely, as repeated B-scans during both initial and recurrent presentations showed no such abnormalities. The patient’s history of trabeculectomy and cataract surgery may have led to subtle, long-term zonular weakness, predisposing the eye to structural instability. Additionally, previous malignant glaucoma in the same eye suggests an inherent anatomical susceptibility to SACG.
Secondly, intraocular infection and inflammation can lead to choroidal expansion and ciliary body swelling, which may cause anterior rotation of the ciliary body and forward displacement of the lens and iris. This process can result in secondary angle closure and a marked increase in IOP. In the present case, following intravitreal antibiotic injections and control of the infection, the anterior chamber gradually deepened. After pars plana vitrectomy and posterior capsulotomy, the condition resolved. These observations support this proposed mechanism. Therefore, it is likely that the SACG in this case was triggered by a combination of infectious inflammatory response and anatomical predisposition.
Additionally, previous study have reported that bacterial biofilms can form on intraocular lens surfaces in eyes after long-term, uncomplicated cataract surgery. 19 Moraxella DNA was detected in intraocular fluid from such eyes. 19 Given the ability of Moraxella species to produce biofilms and induce protease-mediated tissue degradation, 20 we propose the theoretical hypothesis that chronic intraocular colonization by M. nonliquefaciens may lead to zonular damage.
However, this remains a mechanistic hypothesis without direct evidence and likely played only a secondary role in this case. Inflammation and anatomical predisposition appear to have been the primary contributors to the development of SACG.
mNGS is an advanced pathogen detection technique that enables rapid identification of all microorganisms within a clinical specimen. It is particularly useful for detecting rare or culture-negative pathogens that are difficult to identify by conventional methods. However, this method cannot distinguish between viable and nonviable microorganisms. In our case, the 3.8% relative abundance of M. nonliquefaciens on repeat mNGS likely represented residual microbial DNA. Nevertheless, mNGS remains a powerful adjunctive tool for etiological diagnosis in infectious endophthalmitis.
Footnotes
Acknowledgments
The authors used ChatGPT to assist with grammar correction and language polishing during manuscript preparation.
Authors’ contributions
Consent for publication
Informed consent for publication of this case report and accompanying images was obtained from the patient.
Data availability
Not applicable.
Declaration of conflicting interests
The authors declare no conflict of interest.
Ethical considerations
According to the policies of our institution, ethics review committee approval is not required for the publication of case reports.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
