Abstract
We report a case of a 69-year-old gentleman who developed an acutely painful eye with loss of visual acuity whilst on the critical care unit. He was admitted three days previously with an infective exacerbation of chronic obstructive pulmonary disease requiring invasive mechanical ventilation. In addition, he received intravenous antibiotics, steroids, nebulised bronchodilators and intravenous aminophylline, together with noradrenaline for blood pressure support. On development of visual symptoms, an emergency ophthalmology review diagnosed acute angle closure glaucoma. Treatment with pilocarpine eye drops, intravenous acetazolamide and bilateral YAG laser iridotomies provided immediate symptom relief and he went on to make an excellent recovery. Acute angle closure glaucoma is a potentially devastating ophthalmic emergency. Critical care patients are at particular risk for the development of this condition due to the use of predisposing medications, such as sympathomimetics and beta agonists. Sedated patients also run a risk of delayed diagnosis so a high index of suspicion is required.
Introduction
Acute angle closure glaucoma (AACG) is an ophthalmic emergency that progresses to blindness if untreated. Raised intraocular pressure (IOP) resulting in damage to the optic nerve occurs secondary to obstruction of the drainage of aqueous humour from narrowing or closure of the anterior chamber angle. Critically ill patients are at particular risk of developing this sight-threatening condition due to premorbid comorbidities, the development of critical illness and its pharmacological management along with challenging diagnostic circumstances associated with low conscious states.
Case presentation
A 69-year-old gentleman presented via the emergency department (ED) in type 2 respiratory failure with a four-day history of cough and increasing shortness of breath. He presented in extremis with tachycardia, hypotension, hypoxia and hypercapnia and had a mixed respiratory and metabolic acidosis. His medical history includes chronic obstructive pulmonary disease (COPD) and peripheral vascular disease.
He was intubated in the ED and transferred to the intensive care unit (ICU) where he was treated for a community-acquired pneumonia, exacerbation of COPD and septic shock. His treatment included intravenous pipercillin/tazobactam, aminophylline and steroids, nebulised salbutamol and ipratropium bromide and cardiovascular support with noradrenaline. His ventilation improved, and within 24 h, he was extubated and aminophylline and noradrenaline were weaned. He remained on nebulised bronchodilators for a further day.
On the evening of day three of admission, he developed right-sided eye pain, redness and decreased visual acuity. This worsened throughout the course of the night and by the morning began to also affect his left eye. He was seen urgently in the onsite ophthalmology clinic the same morning. On examination, his visual acuity was assessed as counting fingers in the right eye and 6/18 in the left. His right eye had a fixed and dilated pupil, with a shallow anterior chamber and a hazy cornea. The IOPs were 45 mmHg and 20 mmHg in the right and left eyes, respectively. A diagnosis of AACG of the right eye was made, and the left eye was also considered to be at risk of AACG with a markedly narrow angle. Immediate treatment was commenced with pilocarpine eye drops, intravenous acetazolamide and bilateral YAG laser peripheral iridotomies. This decreased the IOPs to 22 mmHg in the right eye and 16 mmHg in the left eye with immediate symptomatic relief.
Clinically, he continued to improve both from his ophthalmic and respiratory conditions and was discharged to the ward following a seven-day ICU stay and subsequently home.
Discussion
AACG is an ophthalmic emergency and, if left untreated, can lead to blindness. It is defined as the appositional or synechial closure of the anterior chamber angle. 1 The narrowing or closure of the anterior chamber angle blocks the drainage of aqueous humour, resulting in elevated IOP and damage to the optic nerve.
Pathophysiology
Aqueous humour is produced by the ciliary body in the posterior chamber of the eye and diffuses from the posterior chamber through the pupil and in to the anterior chamber. From the anterior chamber, the fluid is drained into the vascular system via the trabecular meshwork and Schlemm canal contained within the angle (the junction of the iris and cornea at the periphery of the anterior chamber).
AACG is caused by factors that either push or pull the iris up into the angle thus blocking the drainage of aqueous humour, raising the IOP and damaging the optic nerve (see Figure 1).
Image replicated with the kind permission of the Glaucoma Research Foundation (www.glaucoma.org).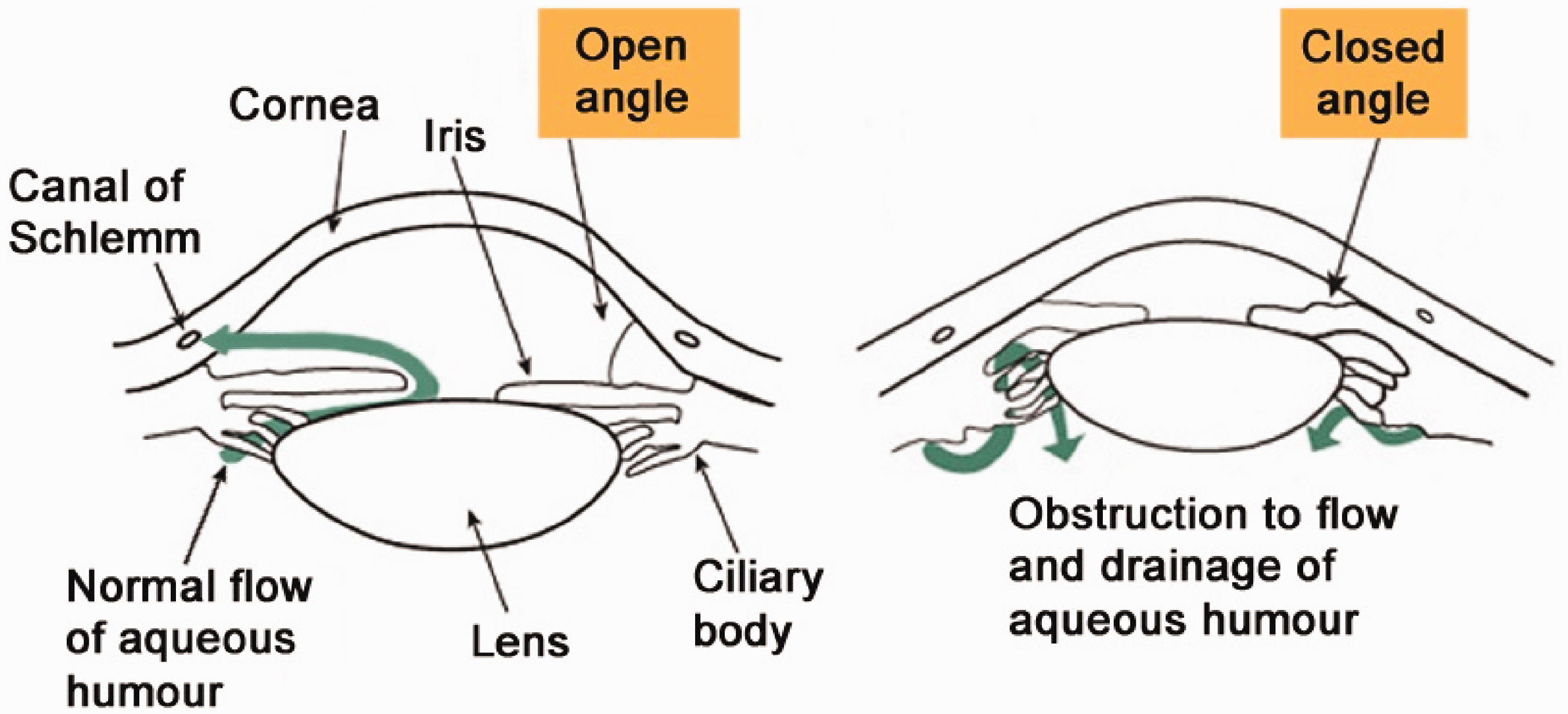
AACG arises chiefly through a mechanism named pupillary block secondary to mydriasis. As the iris dilates and is pulled centripetally and posteriorly causing iris-lens contact, aqueous humour is prevented from passing between the lens and the iris into the anterior chamber. However, aqueous humour continues to be produced by the ciliary body pushing the peripheral iris anteriorly and thus closing the angle. The resultant effect is to obstruct aqueous outflow and cause a rapid onset of severe intraocular hypertension.
A further cause of acute angle closure occurs via the non-pupillary block mechanism, whereby the ciliary body is displaced forward shallowing the peripheral anterior chamber.
Signs and symptoms
AACG usually presents as a rapid progressive visual impairment in one or both eyes with severe ocular and periocular pain and patients may complain of nausea and vomiting. The pupil is often fixed and semi-dilated with corneal oedema and ocular redness. On examination, the IOP will be raised (above 21 mmHg) and the optic disc appears cupped.1,2
AACG in the critically unwell patient and patients in the post-operative period can be particularly challenging to diagnose. Often patients are sedated or have an altered mental status rendering the patient either unaware or unable to communicate pain. In these instances, a clinician must rely on the physiological signs often associated with a painful stimulus, such as a rise in heart rate or blood pressure along with characteristic ocular findings on examination. Even in awake patients, the early non-specific symptoms such as nausea, vomiting and headache are often attributed to other, more common pathologies.
Causes
Risk factors: Patient factors
Predisposing factors include being female, of East Asian descent, having a short axial length (hypermetropia) and a shallow anterior chamber, history of glaucoma, increasing age, thick lens and having a small corneal diameter and a positive family history.1–3
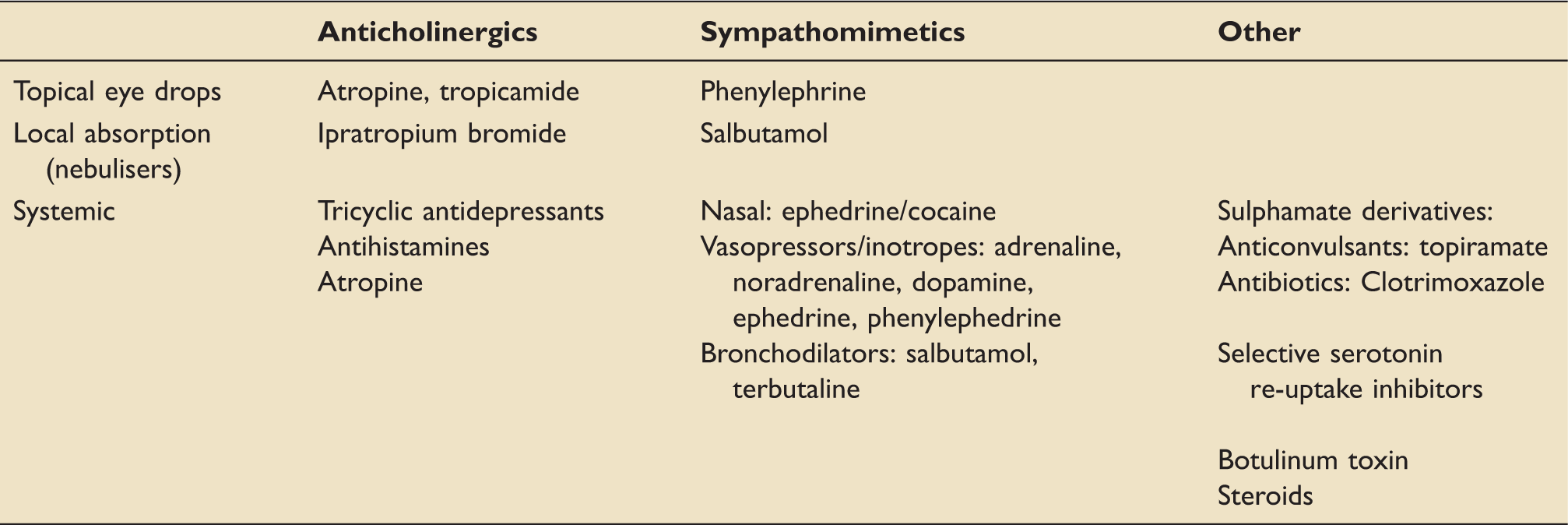
Common agents implicated in the development of AACG.
Conclusion
AACG is a rapidly developing ophthalmic emergency with potentially disastrous effects. This rare but treatable condition can cause major morbidity in our patients and requires a high index of suspicion and vigilance for successful diagnosis and prompt management.
Many critically ill patients fall in to high-risk categories and predisposing risk factors should be considered during history taking. These include patient factors such as advanced age and short sightedness, and medical history including pre-existing eye disease and the use of topical medications. The precipitating cause for critical care admission and the pharmacological agents used for their treatment can precipitate AACG, in particular sepsis and obstructive airways disease. In addition, our patients are particularly vulnerable as they are often unable to communicate their symptoms due to sedation, delirium or weakness.
Clinicians should consider adapting routine pain and eye assessments on critical care for susceptible patients to take in to account eye pain and a global eye examination in addition to routine pupillary checks. Referral and treatment should be sought early in suspected cases of AACG and consideration should be given to the potential difficulty in accessing out of hours emergency ophthalmology services in a critical care environment.
Footnotes
Acknowledgements
We would like to thank Ms Bina Patel for her ophthalmology expertise and advice.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
