Abstract
Objective:
A systematic search was conducted to categorize targeted ultrasound contrast agents used in the detection of myocardial ischemia.
Methods:
The search identified 14 primary research articles published from 2000 to August 2019 that fulfilled the selection criteria. All studies were conducted in animal models ranging from mice to rhesus monkeys, with the most common targets being P-selectin and E-selectin.
Results:
These studies show that targeted ultrasound contrast agents produced greater signal enhancement in regions with prolonged ischemia and maintained enhancement hours after reperfusion.
Conclusion:
This review identified gaps in the literature, such as a need for comparative studies among different molecular markers and between current standard of care with the use of targeted contrast agents in cardiac ultrasound.
Acute coronary syndrome (ACS) contributes to more than 1 million hospital admissions in the United States annually and is a major cause of morbidity and mortality worldwide. 1 The most common mechanism for ACS involves the rupture of an atherosclerotic plaque, resulting in partial or complete occlusion of a coronary artery. Plaque disruption exposes subendothelial collagen, which activates platelets and the coagulation cascade, leading to thrombus formation. Reduction in blood flow due to coronary occlusion or embolization of thrombus into coronary vasculature results in ischemic chest discomfort. 2
The initial workup of ACS includes electrocardiography (ECG) and the measurement of cardiac enzymes. However, these tests possess certain limitations, as they may produce abnormal results in conditions other than ACS, leading to delayed initiation of appropriate therapy. 3 Therefore, new techniques are developed to more accurately diagnose ACS. One method involves determining the presence and extent of ischemic myocardium by detecting molecular changes. Quantitative molecular imaging has been described for vascular cell adhesion protein-1 and P-selectin, a member of a cellular adhesion molecule family, by examining the distribution of endothelial activation to measure the extent of cardiac ischemia. 4 Combining molecular and imaging techniques may make it possible to identify patients with active disease and to monitor therapy response.
Sonography offers certain benefits: it is noninvasive, widely available, portable, and inexpensive, and it allows real-time imaging without ionizing radiation. Microbubble ultrasound contrast agents (UCAs) are safe and well-tolerated in humans and utilized for their ability to enhance echogenicity and signal production. 5 UCAs contain a high molecular weight inert gas such as perfluorocarbons or sulfur hexafluoride, and have a shell composed of lipids, protein, or biocompatible polymers. Due to their 1-10 microns size, microbubbles remain within the vascular space and do not diffuse into the extravascular interstitial space 5 which allows them to be well suited for detecting and monitoring disease that could be characterized by a differential expression of molecules on the vasculature.
Novel UCAs are currently being developed to improve cardiac sonography by coating the surfaces of microbubbles with antibodies or peptides that target markers of ischemia. In preclinical studies, these agents produced greater signal enhancement in regions with prolonged ischemia compared with controls, and they maintained enhancement hours even after reperfusion. Contrast-enhanced ultrasound therefore is advantageous compared with other forms of molecular imaging, because images can be obtained rapidly, in real time, without the need for postprocessing. 5
The goal of this scoping review was to examine the extent, range, and nature of research on the use of targeted UCAs, specifically in the detection of cardiac ischemia. Innovative targeted contrast agents can potentially allow the noninvasive detection and evaluation of the ischemic response on a molecular scale. This review helps determine the value of undertaking a full systematic review and identifies gaps in the existing literature. For the purposes of clarity, this review covers the use of targeted UCAs in cardiac imaging, also known as molecular echocardiography.
Materials and Methods
The methodology for this scoping review was based on the protocol outlined by Arksey and O’Malley. 6 This was a review of preexisting literature and did not require the use of patient data. Therefore, ethics approval or institutional review board approval was not obtained for this study.
The goal of this scoping review was to conduct a systematic search of the existing literature and summarize the current state of research regarding the use of targeted UCAs in the detection of myocardial ischemia. The literature search, scope, and reporting of findings were guided by the following questions:
Have any clinical studies used targeted UCAs in patients with ACS?
How are targeted UCAs being applied to animal models in detecting myocardial ischemia, and what are the general trends and findings of these studies?
What are the gaps in the literature involving targeted UCAs and their application in myocardial ischemia detection?
Search Strategy
The initial literature search was conducted in August 2019 by a team of librarians, using the search terms “contrast-enhanced ultrasound,” “contrast-agent,” “myocardial,” “cardiac,” “ischemia,” and “microbubbles.” A systematic review search strategy was developed by librarian scientists (P.H., T.S.). The search was performed in the following databases on August 17, 2019: PubMed, Ovid MEDLINE, Embase, Cochrane Library, Database of Abstracts of Reviews of Effects, Health Technology Assessment Database, NHS Economic Evaluation Database, and ClinicalTrials.gov. A preliminary search of the literature revealed that Lindner et al. (2000) 7 was the first article published that fulfilled the search terms. Prior articles discussed UCAs but not in the setting of cardiac imaging. Because of this, the systematic search was conducted on literature published in electronic databases from January 1, 2000, to August 19, 2019. This does not include unpublished ongoing studies as of August 19, 2019, in addition to gray literature such as conferences, newsletters, and book chapters.
All citations (895) were imported into EndNote X8.0.1 (Thomson Reuters, Toronto, ON, Canada). The citations were then deduplicated and uploaded into Covidence (Covidence, Melbourne, Australia), an online tool to help organize and facilitate the literature selection process.
Study Selection
Two team members independently reviewed each title and abstract for the initial screening process. A third member assisted with providing consensus for cases of disagreement.
The screening process was an iterative process. Only articles that discussed targeted UCAs in early myocardial ischemia detection were included. Articles discussing untargeted UCAs or molecular contrast agents of other imaging modalities (i.e., magnetic resonance imaging, computed tomography, positron emission tomography) were excluded. Additionally, studies that used UCAs therapeutically (i.e., theranostics), such as directed drug delivery or microbubble cavitation, were not included in this review. Finally, studies that were not written in English were excluded. Full articles were obtained, and their references were screened for additional relevant articles.
Charting the Data
The data from primary research articles (studies that generate new data) were compiled in a single spreadsheet and imported into Excel 2010 (Microsoft, Redmond, WA). Secondary research articles were not directly included in the data collection process. However, these reviews were consulted to locate additional studies that may not have been included through our screening process. The Excel spreadsheet was organized to allowed pattern recognition and summarization of the literature.
Results
The total number of articles obtained from the initial database search was 895. After eliminating duplications, we included 555 articles. After reviewing titles, we excluded 215 articles because they did not specifically address UCAs. These articles included magnetic resonance imaging and computed tomographic contrast agents. A total of 340 abstracts were then reviewed, and 186 articles were subsequently excluded because they did not discuss molecular UCAs or cardiac applications. This reduced the number of potentially relevant articles to 154. Upon review, 66 studies were selected that used targeted UCAs for the purpose of evaluating myocardial ischemic processes. On closer inspection, 88 articles were excluded because they did not discuss UCAs in cardiac imaging that fulfilled the inclusion criteria. For example, these articles discussed targeted UCAs in thrombus, ischemia, or angiogenesis imaging that did not relate to cardiac physiology. The retained studies comprise 14 primary research articles and 52 reviews. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses flowchart details the review process (Figure 1).
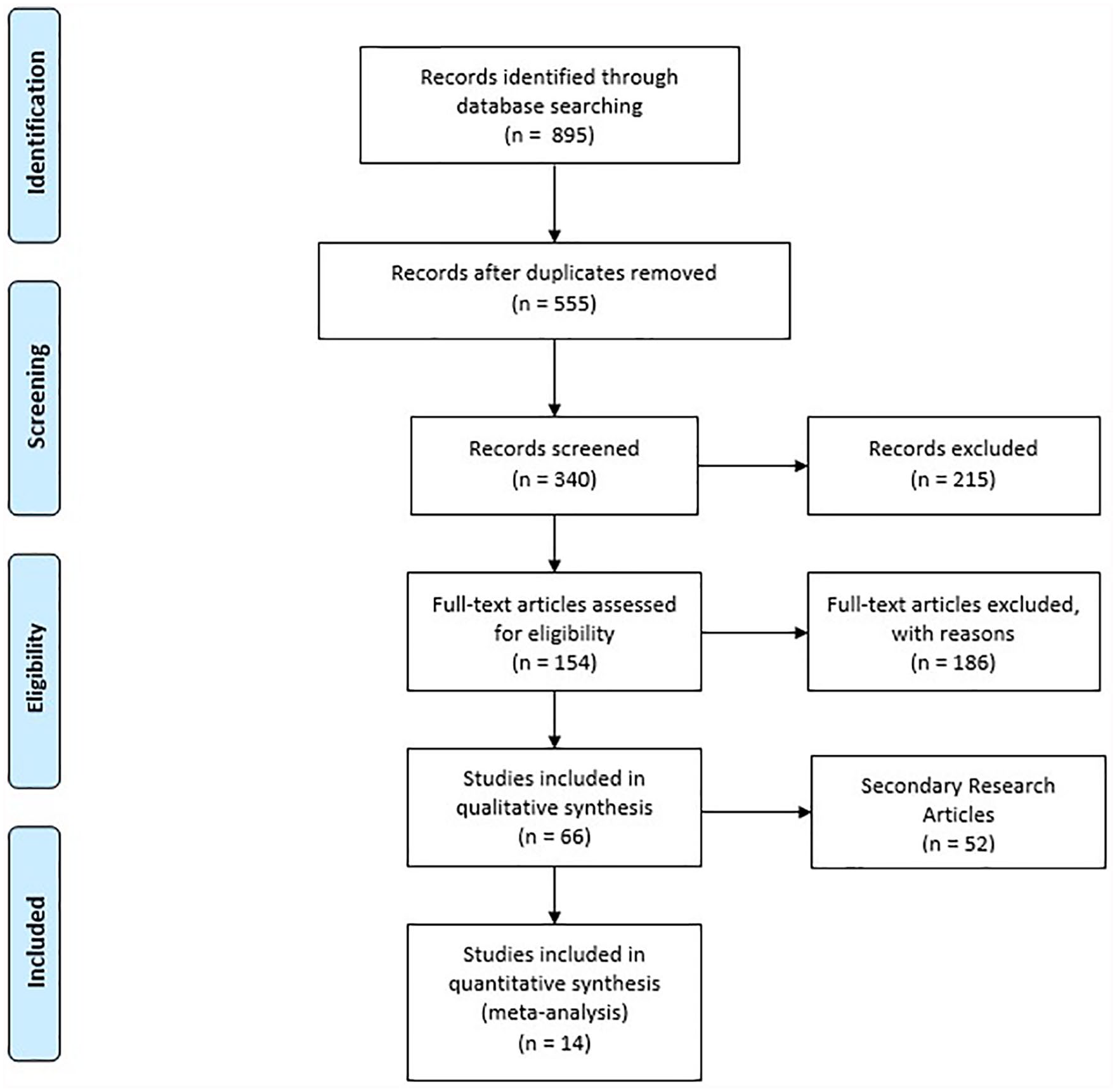
Preferred Reporting Items for Systematic Reviews and Meta-analyses flow diagram illustrating the source selection process.
The 14 primary research studies identified in this review specifically investigated the application of targeted UCAs in myocardial ischemia induced in animal models. A significant proportion of articles included in this study are reviews that discuss targeted UCAs in detecting ischemia, with the anticipation of applying this imaging technology to improve the diagnosis of myocardial ischemia.
Descriptive Summary of the Studies
The 14 primary research studies included in this review were published between May 17, 2000, and March 5, 2018, with 10 articles (71%) published within the past 10 years. Almost all the studies were conducted either in the United States (n = 8 [57%]) or China (n = 5 [35%]), with 1 study from Switzerland (n = 1 [7%]).
As of September 2019, no clinical studies have investigated the use of targeted UCAs in the detection of myocardial ischemia in humans. However, Davidson et al. 8 used human P-selectin glycoprotein ligand 1 fusion protein (YSPSL) molecular contrast echography in nonhuman primates. YSPSL binds to both early P-selectin and late E-selectin. Because tissue ischemia produces an endothelial P-selectin and E-selectin response, Davidson et al. used YSPSL as a targeting ligand with the goal of applying it to patients to detect early ischemia. The safety of nonconjugated YSPSL in humans has been established in phase 2 and 3 clinical trials in which high doses were used as a possible treatment for renal or liver allograft rejection. 9 The investigators demonstrated increased signal enhancement due to P-selectin activity from 1.5 to 6 hours. However, E-selectin demonstrated prolonged enhancement up to the 18-hour time point. They concluded that it is possible to detect myocardial ischemia in nonhuman primates by using targeted contrast ultrasound with a selectin-targeted microbubble contrast agent bearing a recombinant human P-selectin glycoprotein ligand 1.
Currently, most targeted contrast ultrasound designs involve inducing myocardial ischemia in animal models, most notably in mice. The most common method of inducing ischemia is to perform a thoracotomy and to constrict the left anterior descending coronary artery (n = 11) with suture for a designated time period. Some studies induced ischemia by constricting the left common artery (n = 2). The duration of ischemia varied depending on the animal model. For example, mouse models usually experienced ischemia for 10 to 15 minutes, while rat models (n = 3) varied from 15 to 20 to 30 minutes. Other animal models used include rabbits (n = 1) and dogs (n = 1). A summary of these findings is displayed in Table 1.8,9–21
Summary of the 14 Primary Research Articles Included in This Scoping Review. a
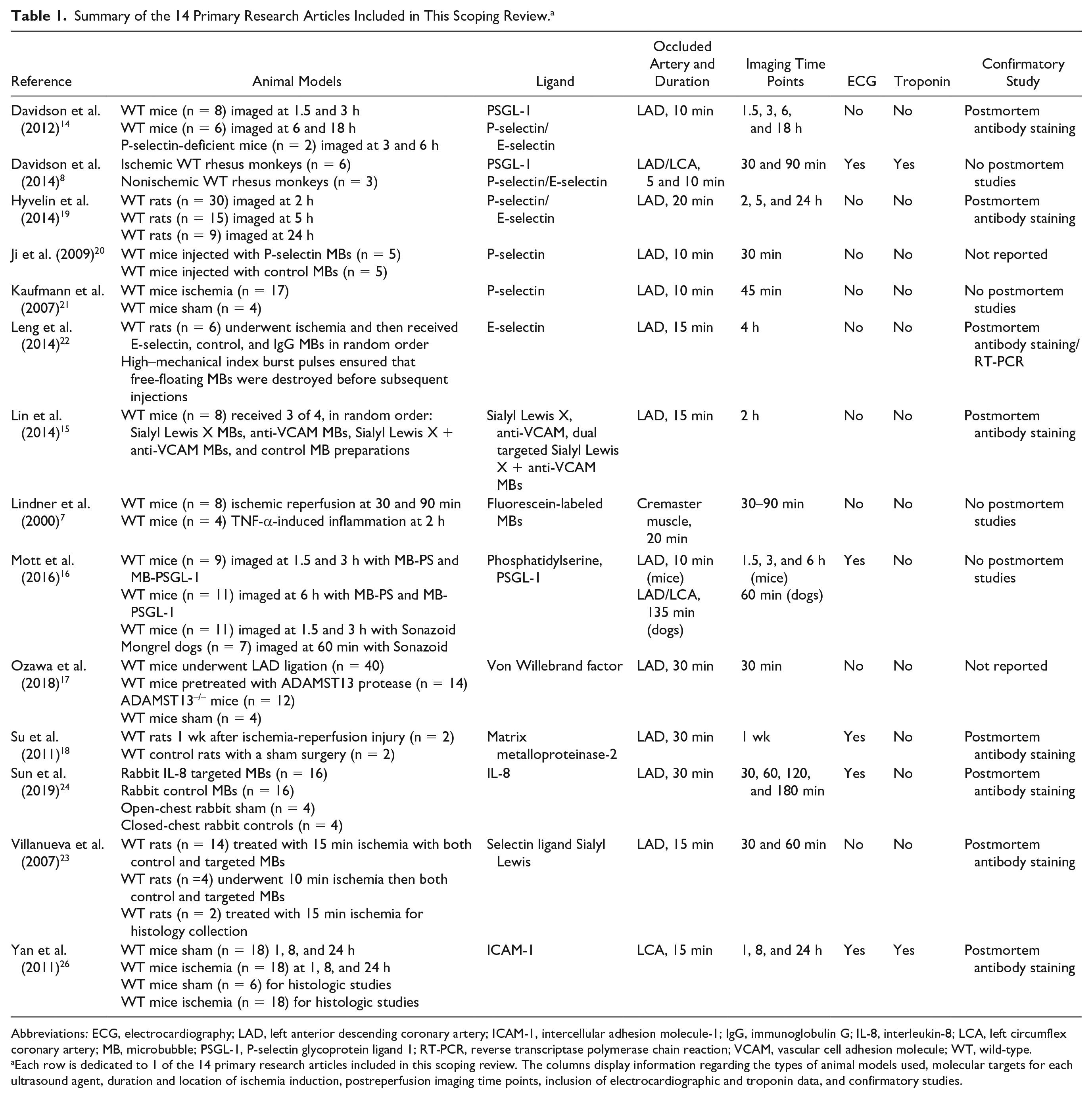
Abbreviations: ECG, electrocardiography; LAD, left anterior descending coronary artery; ICAM-1, intercellular adhesion molecule-1; IgG, immunoglobulin G; IL-8, interleukin-8; LCA, left circumflex coronary artery; MB, microbubble; PSGL-1, P-selectin glycoprotein ligand 1; RT-PCR, reverse transcriptase polymerase chain reaction; VCAM, vascular cell adhesion molecule; WT, wild-type.
Each row is dedicated to 1 of the 14 primary research articles included in this scoping review. The columns display information regarding the types of animal models used, molecular targets for each ultrasound agent, duration and location of ischemia induction, postreperfusion imaging time points, inclusion of electrocardiographic and troponin data, and confirmatory studies.
After a period of ischemia, the animal models targeted contrast ultrasound at set time points. For example, some studies imaged their animal models at 30 and 90 minutes after ischemia. Other studies imaged their animal models after reperfusion up to 24 hours after the ischemic event. Most studies concluded with immunohistochemistry tissue staining to confirm the presence of molecular ligands in control versus ischemic tissue. All studies showed that targeted UCAs produced stronger signals associated with myocardial ischemia.
Discussion
Ultrasound molecular imaging relies on the detection of targeted microbubbles to examine cellular events at the blood-endothelial interface. Because contrast agents are contained within the vasculature, they can provide unique information on atherosclerosis, ischemia reperfusion injury, angiogenesis, inflammation, and thrombus formation. 10 In addition to being well suited to evaluate the progression of cardiovascular disease, targeted UCAs are currently used in oncologic imaging.
Aberrant angiogenesis has been implicated in supporting the growth of cancer cells. Many tumors depend on increased neovascularization, once they grow beyond 1 to 2 mm, for oxygen and nutrient delivery. Several biomarkers have been studied for tumor detection at early stages by identifying these signals for angiogenesis. Novel contrast agents for these markers can enable visualization of disease at the molecular level and provide invaluable insight on early diagnosis of malignancy. 11
Through the iterative process of conducting a scoping review, we have identified other novel applications of ultrasound microbubbles outside the realm of diagnostics. Ultrasound microbubbles can be used therapeutically in drug delivery and treatment. The oscillatory property that makes microbubbles echogenic can be leveraged for therapeutic purposes. When microbubbles oscillate, the surrounding fluid can be set into motion, which can significantly enhance convection of drugs from the bloodstream into tissue. 12 Microbubbles are currently studied as a tool to deliver drugs past the blood-brain barrier and to enhance cellular uptake of drugs through sonoporation.
We did not include theranostic studies in the present review, because they did not fulfill the inclusion criteria. For example, the microbubbles being studied for therapeutic purposes are often involved with thrombolysis or drug delivery in the peripheral circulatory system and not within the heart. A future scoping review regarding the state of theranostic research will be beneficial.
The goal of this scoping review was to develop an understanding of the state of research regarding the use of targeted contrast ultrasound agents in the diagnosis of myocardial ischemia.
The final review included 14 primary research articles, with 10 studies conducted within the past 10 years. Of these, 1 study evaluated the safety and efficacy of a selectin-targeted UCA in a primate model after inducing brief myocardial ischemia. 8 The primate models tolerated selectin-targeted UCAs with no complications, bringing this new imaging modality one step closer to human applications.
Of the 14 studies that were included in this review, half used selectins (P- and E-selectin) as their primary molecular markers. Selectin is a popular marker because it is directly involved with leukocyte recruitment due to ischemic processes. 13 Additionally, there are selectin-targeting UCAs already in clinical trials for human application. 9 Other markers include interleukin-8, von Willebrand factor, and intercellular adhesion molecule-1, which are summarized in Table 2 with their mechanisms of action.
Summary of the Molecular Targets Included in This Study. a
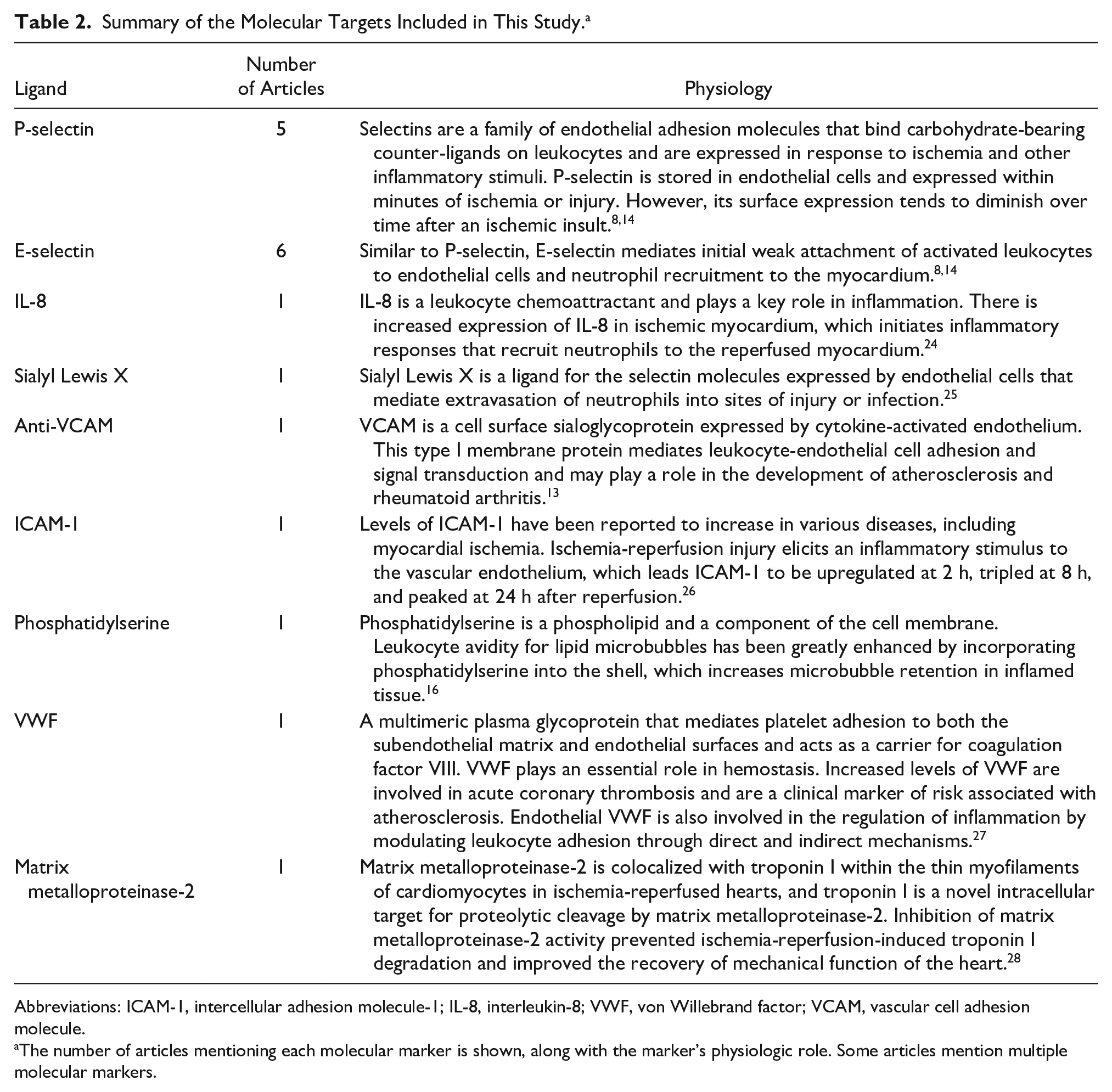
Abbreviations: ICAM-1, intercellular adhesion molecule-1; IL-8, interleukin-8; VWF, von Willebrand factor; VCAM, vascular cell adhesion molecule.
The number of articles mentioning each molecular marker is shown, along with the marker’s physiologic role. Some articles mention multiple molecular markers.
All studies included in this review showed that signal enhancement is greater in regions with prolonged ischemia compared with controls in animal studies. Additionally, ischemic regions showed ultrasound enhancement even hours after reperfusion. 14 Lin et al. 15 investigated a dual-targeting UCA in molecular contrast echocardiography and concluded that dual-targeted microbubbles yield greater signals than single-targeted agents, improving both the sensitivity and accuracy of this technique to measure risk regions in intact mice.
There is significant variability in the experimental designs that may limit cross-comparisons among different agents. For example, some studies8,16,17,18 used ECG to detect ST-segment changes when inducing ischemia in their animal models. However, other studies19,20,21,22 induced ischemia for predetermined time periods. This makes it challenging to compare studies, as the extent of ischemia induced in their animal models may not be the same. Additionally, not all studies obtained postmortem histologic data to compare with their imaging data. This may limit the validity of stating that the increased signal seen with targeted UCAs correlated with increased molecular expression.
One of the goals of targeted UCAs is to allow more accurate diagnosis and functional imaging of ACS. A question posed during this study was how well targeted contrast cardiac ultrasound performs compared with the current standard of care involving ECG and troponins. Five studies used ECG and 2 studies used troponin to detect ischemic changes. ECG was used predominantly as a confirmatory tool to indicate the presence of ischemia through ST-segment changes. Two studies collected troponin data on their ischemic models. Interestingly, in Davidson et al. 8 , troponin levels did not increase at study completion, even in the ischemic primate group. This suggests that molecular contrast imaging may be more sensitive to early ischemic process than current enzyme markers. Additionally, Davidson et al. 8 reported that microbubbles targeting human P-selectin were well tolerated and produced signal enhancement in the postischemic risk area at a time when both myocardial blood flow and wall motion had returned to normal. These are promising signs that targeted UCAs may be more sensitive than current standard of care in detecting early ACS. However, the amount of current data is limited.
We noticed that several gaps exist in the literature regarding the application of targeted UCAs in detection of myocardial ischemia:
E- and P-selectin are the most popular molecular targets used in cardiovascular targeted contrast ultrasound, contributing to half of the primary research articles included in this review. There is less attention placed on other molecular targets such as intercellular adhesion molecule-1 and interleukin-8 and an absence on L-selectin and vascular cell adhesion protein-1, other potential molecular targets. One could hypothesize that there is increased interest in the selectins compared with other targets because selectin-targeting microbubbles can be synthesized reliably and have a safety profile approved for clinical research.
There is a lack of comparative studies among different UCAs due to the variation of experimental design (e.g., varying ischemia induction times and postperfusion time points).
The goal of targeted contrast ultrasound is to be able to detect early ACS. Only 2 studies performed ECG or measured troponin levels, thus limiting the utility of this imaging modality compared with the current standard of care.
Limitations
This scoping review was limited by study selection from electronic databases and peer-reviewed journal articles. This may exclude interprofessional literature that may exist in publications such as book chapters, newsletters, and conference abstracts that are not published online.
The inclusion criteria resulted in more than 800 articles. A vast majority of these articles were reviews discussing the application of molecular contrast imaging in ischemia and angiogenesis and physiologic changes involved in a variety of pathologies ranging from thrombus formation in atherosclerotic disease to arteriogenesis in cancer. While conducting this review, we noted that several significant fields of targeted UCA research were related to the inclusion criteria and would benefit from a systematic review:
ischemia-targeting contrast agents in other imaging modalities (i.e., magnetic resonance imaging, computed tomography, or positron emission tomography);
targeted UCAs in the detection of arteriogenesis in thrombus; and
the therapeutic abilities of UCAs, such as thrombectomy with microbubble cavitation or directed microbubble facilitated drug delivery.
Conclusion
This review was focused on the role of targeted UCAs in the detection of myocardial ischemia. We found that the most studies used P- and E-selectin-targeting UCAs in mouse models, with myocardial ischemia induced by occluding the left anterior descending coronary artery. All studies concluded that targeted contrast ultrasound demonstrated ischemic memory. Myocardial regions that underwent ischemic events showed increased UCA enhancement compared with nonischemic regions for up to several hours after the inciting event. However, because this is a burgeoning field of sonographic research, there were notable limitations in the current state of research. There is a lack of standardized experimental design, which makes comparison studies among different targeted contrast UCAs difficult. Additionally, only 2 studies incorporated both ECG and troponin assessment, which makes it difficult to determine if molecular contrast echocardiography offers additional utility compared with the current standard of care for ACS.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
