Abstract
Gallbladder perforation is a rare and severe complication of acute cholecystitis. Sonography is typically the first modality of choice for assessing gallbladder pathology due to its high sensitivity, portability, real-time imaging capability, and nonionizing technique. Gray-scale and color Doppler sonographic imaging may be equivocal for diagnosing perforation. In the presented case, gallbladder wall perforation was definitively diagnosed utilizing contrast-enhanced ultrasound, allowing for prompt treatment and a successful patient outcome.
Asymptomatic cholelithiasis affects 10% to 20% of the adult population. 1 Approximately 1% to 2% of adults with cholelithiasis will develop acute cholecystitis in their lifetime. 2 Gallbladder (GB) perforation is a rare complication occurring in 2% to 11% of acute cholecystitis cases.3,4 Diagnosis is unfortunately often delayed due to nonspecific clinical signs and symptoms, resulting in high morbidity and mortality rates.5–7 Risk factors for GB perforation include male gender, age over 60 years, gallstones and GB disease, corticosteroid therapy, or systemic disease.7,8 Patients may present with fulminant sepsis and shock. 9 Clinical presentation can vary from right upper quadrant pain to signs of peritonitis including fever, elevated white blood cell count, abdominal guarding, and rebound tenderness.7,10 Familiarity with the clinical and imaging features of GB perforation is necessary to provide an early diagnosis and prompt treatment to reduce morbidity and mortality. The following case report illustrates the limitations of gray-scale and color Doppler sonography in the diagnosis of GB perforation, and presents a novel method for making the diagnosis using contrast-enhanced ultrasound (CEUS) when gray-scale and color Doppler imaging is uncertain.
Case Report
A 64-year-old Caucasian male presented to the emergency department for nausea, vomiting, and abdominal pain. His past medical history was significant for decompensated hepatic cirrhosis with portal hypertension and ascites. The patient was diagnosed with adynamic ileus of the bowel based on a computed tomography (CT) examination and was clinically encephalopathic. Laboratory data showed an elevated lactate level of 4.6 mmol/L (normal range, 0.5–2.2 mmol/L), bilirubin level of 3.0 mg/dL (normal, <1.2 mg/dL), and sepsis secondary to
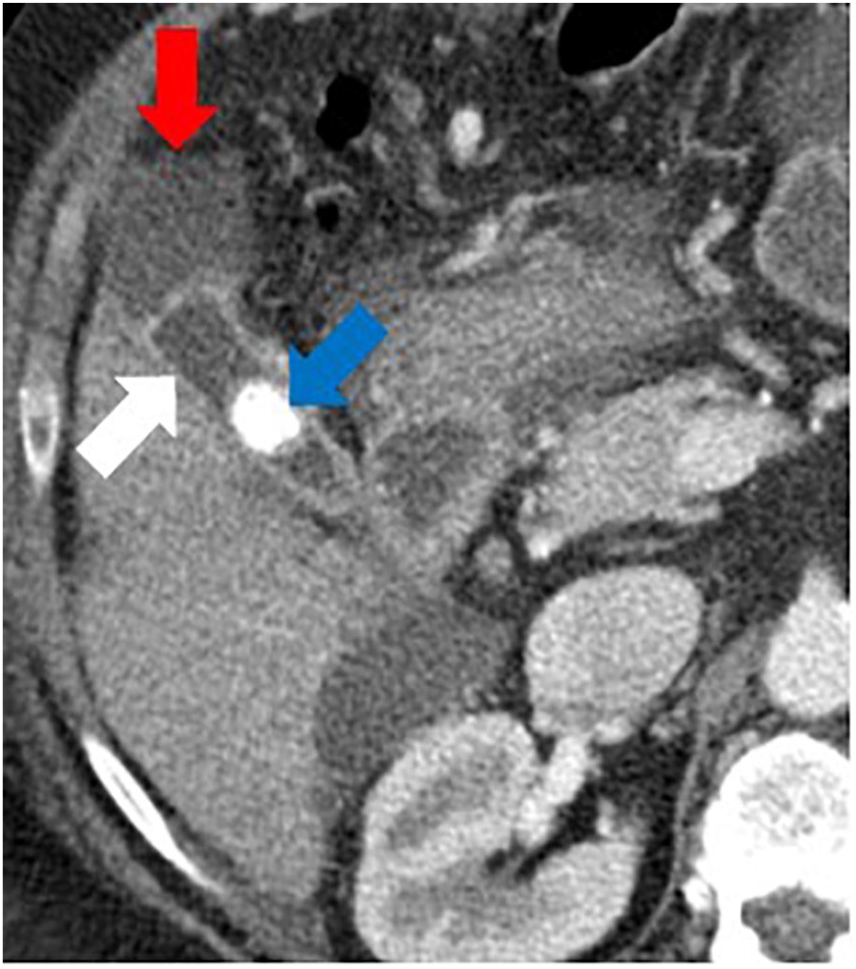
CT examination demonstrated a thickened GB wall (white arrow), a large gallstone (blue arrow), and a loculated fluid collection (red arrow) on this axial image.
Gray-scale sonography (LOGIQ E9; GE Health Care) using a 6-MHz curvilinear transducer demonstrated GB luminal decompression, a large gallstone, and a thickened, irregular GB wall measuring 4.6 mm (Figure 2). Internal debris was adherent to the GB fundal surface and there was extraluminal complex loculated fluid adjacent to the GB wall (Figure 3). Repeat imaging using a 9-MHz linear transducer demonstrated an apparent 4-mm focal defect within the anterior GB wall concerning for perforation (Figure 4).
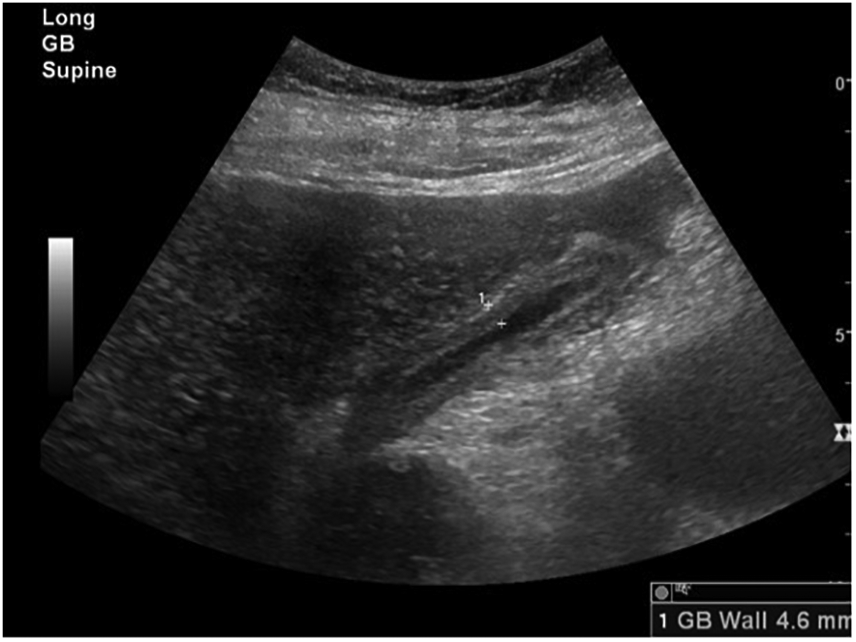
Longitudinal gray-scale sonographic image of the GB demonstrates an irregular thickened wall of 4.6 mm.
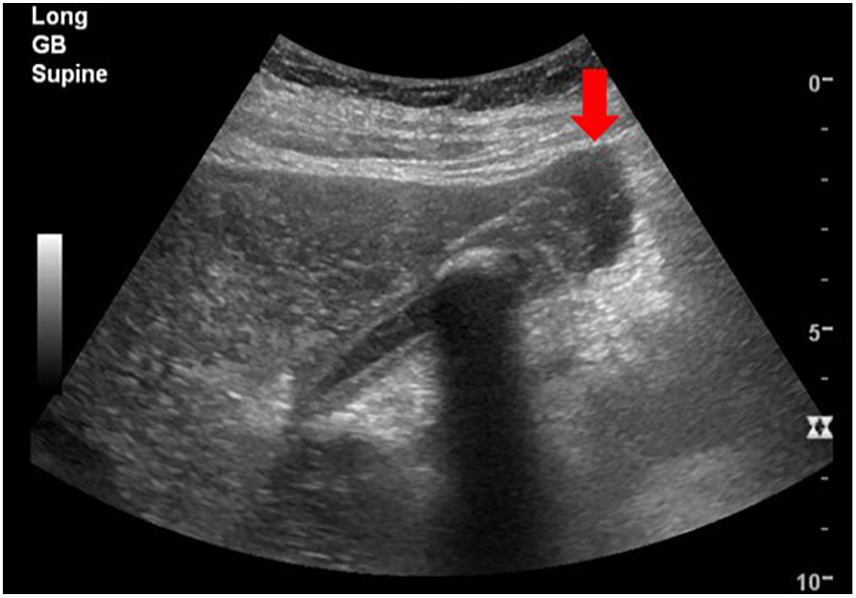
Longitudinal gray-scale sonographic image of the GB demonstrates a large gallstone and adjacent loculated fluid (red arrow).
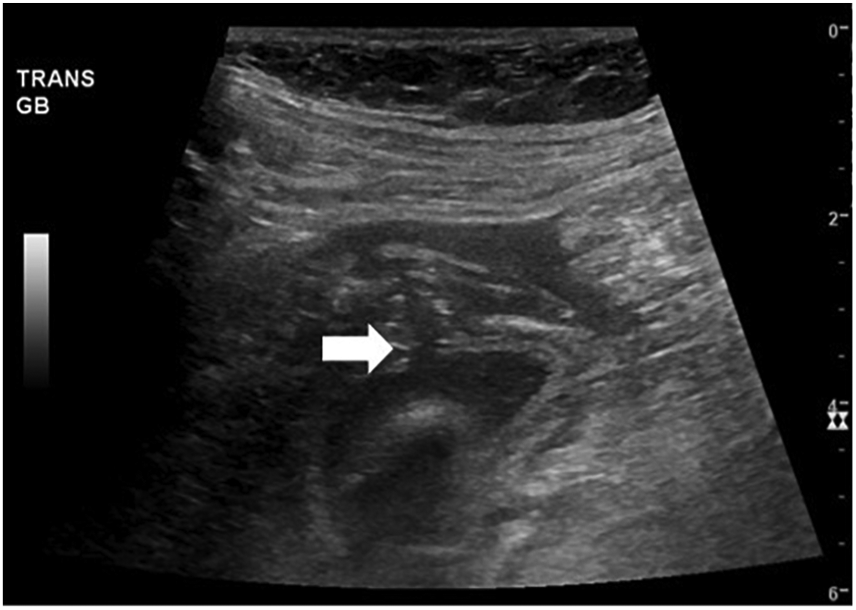
Transverse gray-scale sonographic image identifies a 4-mm defect within the anterior GB wall (white arrow).
For better delineation and to confirm the suspected gray-scale finding of perforation, CEUS was performed to assess for a focal, nonenhancing GB wall defect. Two milliliters of LUMASON was injected as off-label use. Prior to the administration of contrast, the sonographer located the optimal position for GB wall visualization. The patient was prescreened for any contraindications for contrast, namely, a prior allergic reaction. A 20-gauge intravenous (IV) catheter was accessed in the left antecubital fossa in the ICU, which was previously placed by the patient’s nurse. A 20- to 22-gauge needle is preferred as a smaller gauge potentially destroys the microbubbles. This location is optimal as the sonographer scans on the right side of the patient, thus making the right arm inaccessible. Two milliliters of LUMASON (sulfur hexafluoride lipid–type A microspheres; Bracco Diagnostics Inc) was injected slowly over 2 seconds intravenously followed by a 10-mL saline flush, which clears the IV tubing outside of the patient and ensures delivery of the contrast agent into the IV system. A fast injection of contrast should be avoided to prevent microbubble rupture. At this institution, sonographers were allowed to inject the IV contrast agent and saline, as part of their scope of practice.
Shortly following the injection, the sonographer and the radiologist monitored the enhancement pattern of the GB dynamically. CEUS images demonstrated enhancement of the GB wall with the exception of the 4-mm focal defect, confirming the gray-scale suspicion of GB wall perforation (Figures 5 and 6). Once confirmed, further interrogation with a 9-MHz linear transducer demonstrated intraluminal GB fluid passing through the defect, into the peritoneal cavity.
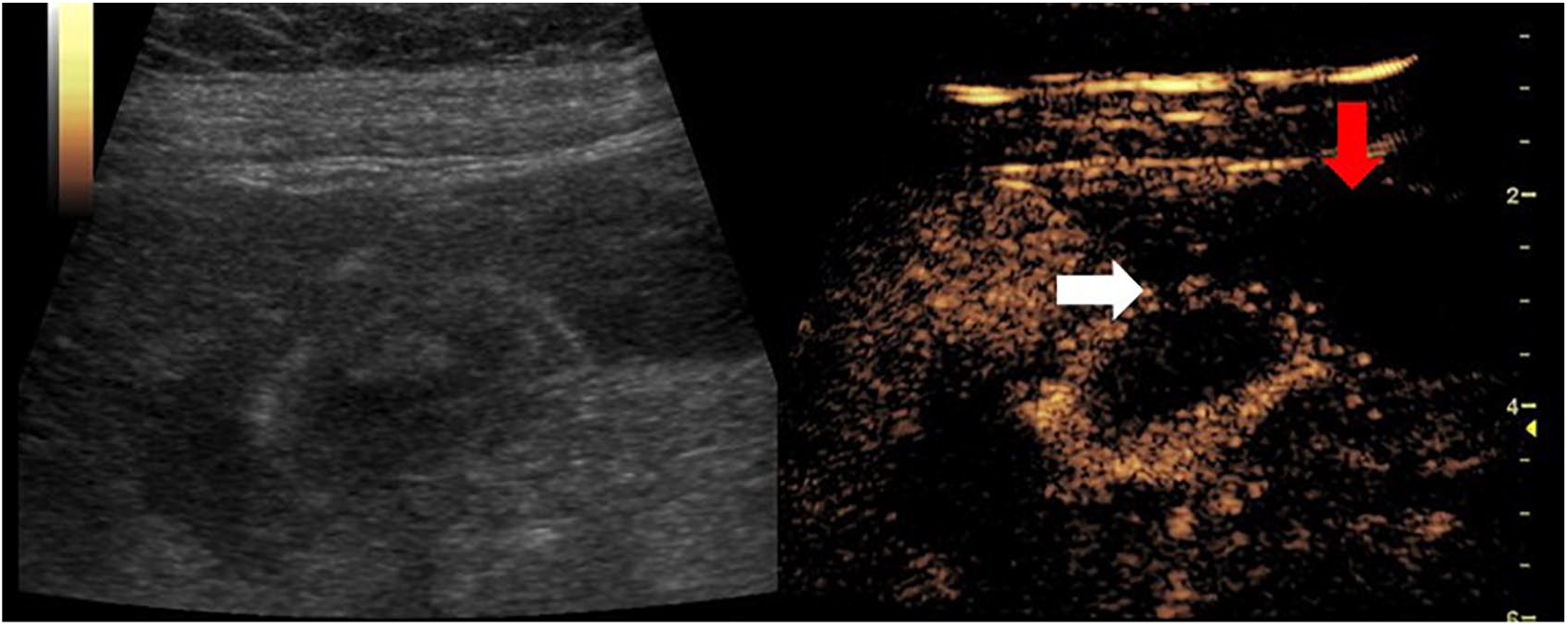
CEUS dual image demonstrated GB wall enhancement with the exception of the 4-mm defect (white arrow) and adjacent fluid collection (red arrow) at 18 seconds following the injection.
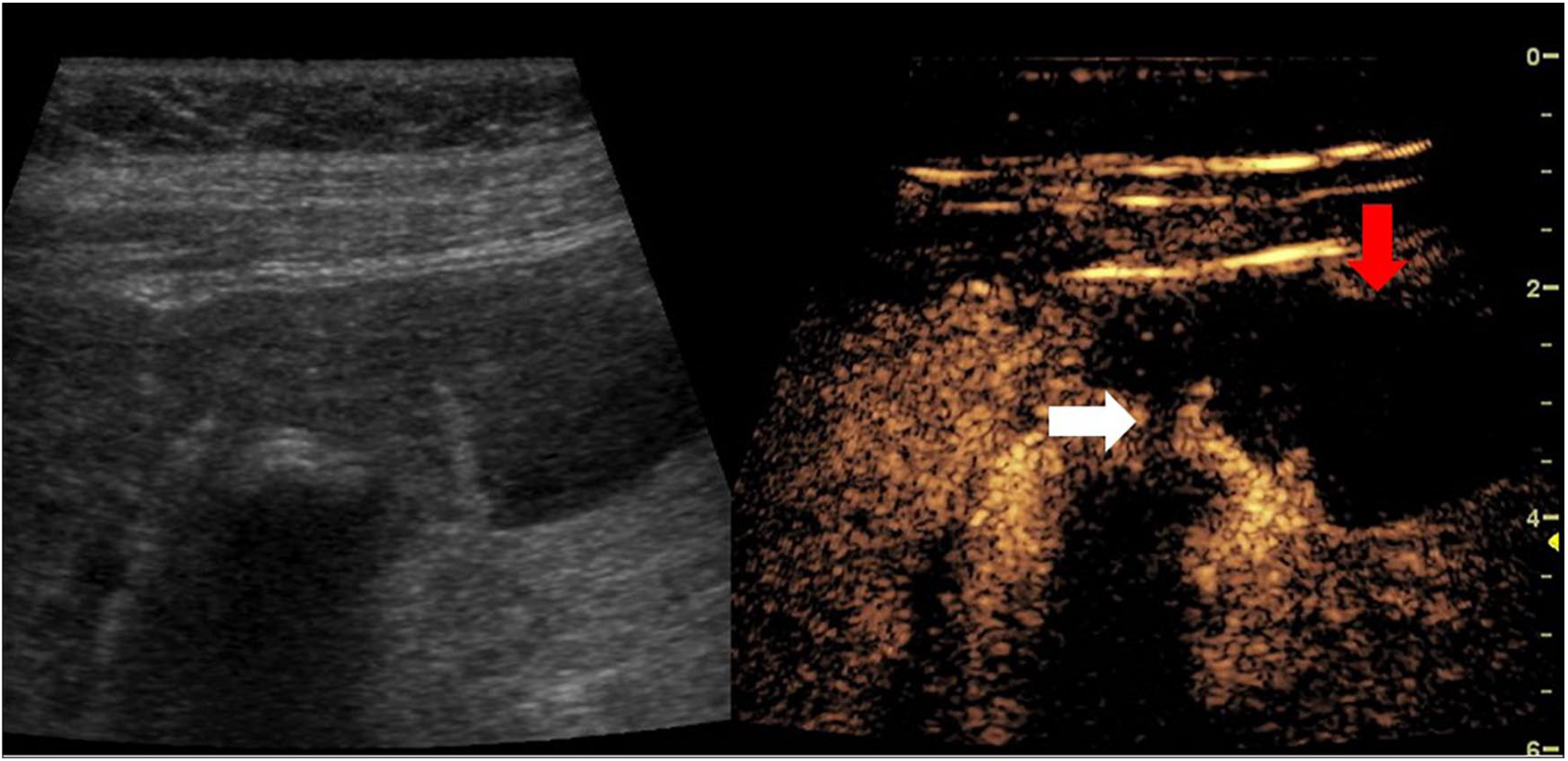
CEUS dual image demonstrated GB wall enhancement with the exception of the 4-mm defect (white arrow) and adjacent fluid collection (red arrow) at 31 seconds following the injection.
The patient subsequently underwent emergent percutaneous cholecystostomy tube placement by interventional radiology (IR) (Figure 7). Injected water-soluble iodinated contrast after tube placement confirmed a type II fundal GB wall perforation and demonstrated a patent cystic duct. Purulent, bloody bile was sent to the laboratory for culture and later confirmed the presence of
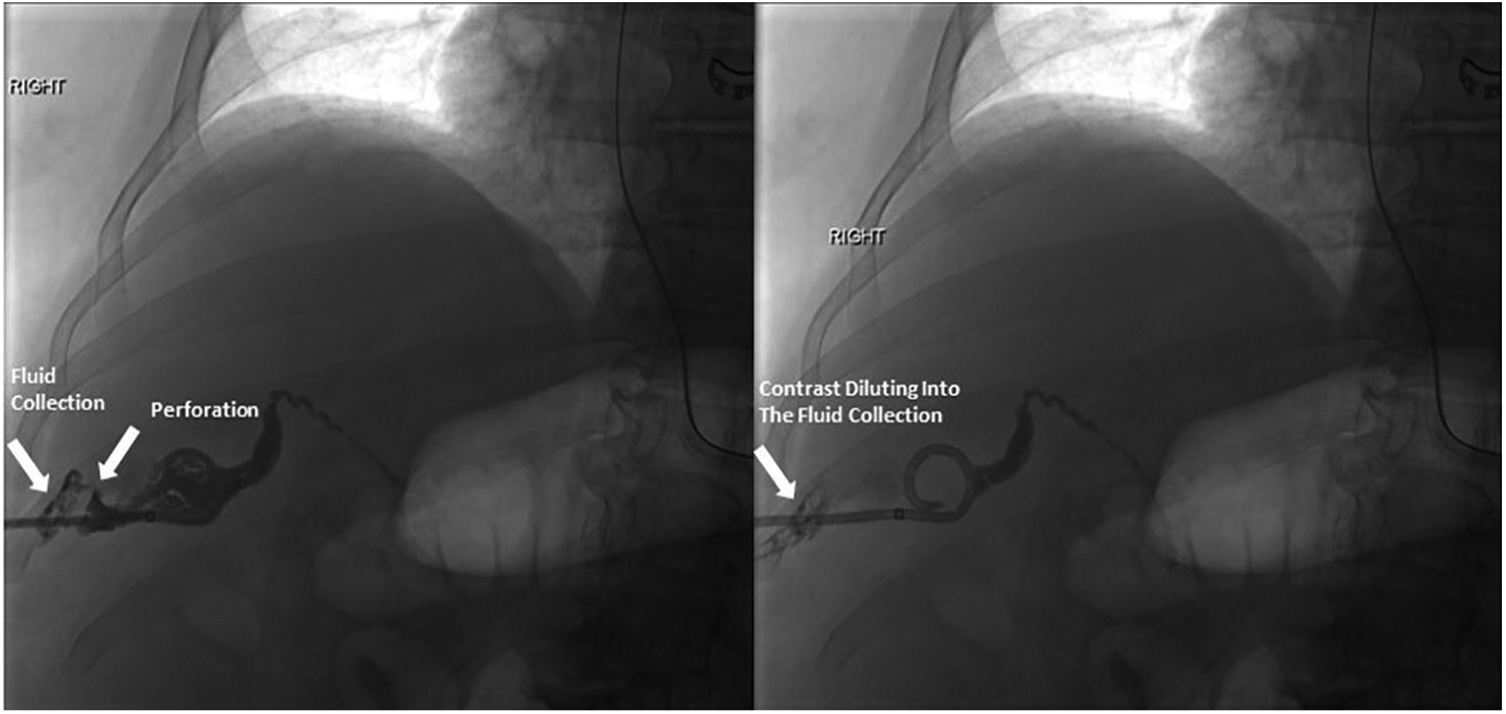
Percutaneous cholecystostomy tube placement performed in IR. The injected contrast confirmed GB perforation in the fundal wall.
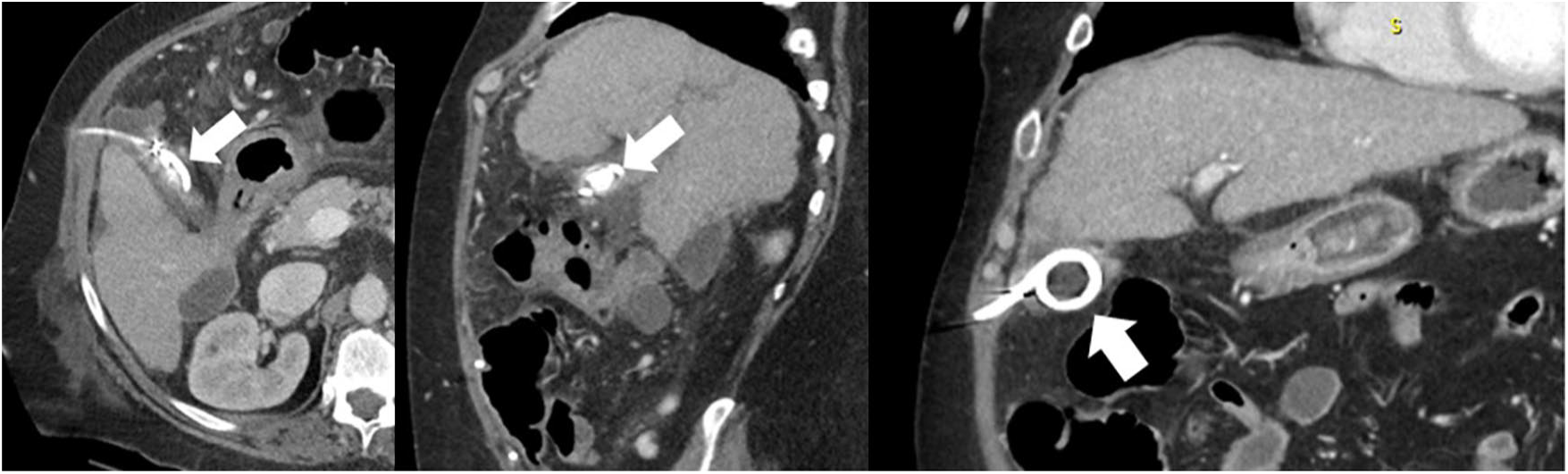
Follow-up CT examination demonstrated the appropriate position of the drain in these anteroposterior, sagittal, and coronal views (white arrows). The adjacent fluid collection decreased in size.
The patient improved clinically and was discharged from the hospital 3 weeks later with the cholecystostomy tube. An elective cholecystectomy was planned upon adequate outpatient recovery.
Discussion
GB perforation is a rare and severe complication usually secondary to acute cholecystitis. Acute cholecystitis is inflammation of the GB wall and results from obstruction of the cystic duct, commonly by gallstones (Figure 9). Patients typically present with severe right upper quadrant pain that may radiate to the upper back. The GB lumen distends and wall thickening >3 mm is often considered abnormal. It is important for the sonographer or clinician to evaluate Murphy’s sign. When positive, the patient is tender with transducer pressure directly over the GB. An unequivocally positive sonographic Murphy’s sign is highly suspicious that the GB is the etiology of the patient’s upper abdominal pain. Chronic cholecystitis can be present in patients who have had multiple acute inflammatory episodes over time. Sonographically, the GB wall is thickened greater than 3 mm and the GB lumen may be contracted. The loss of normal GB function usually follows this appearance.
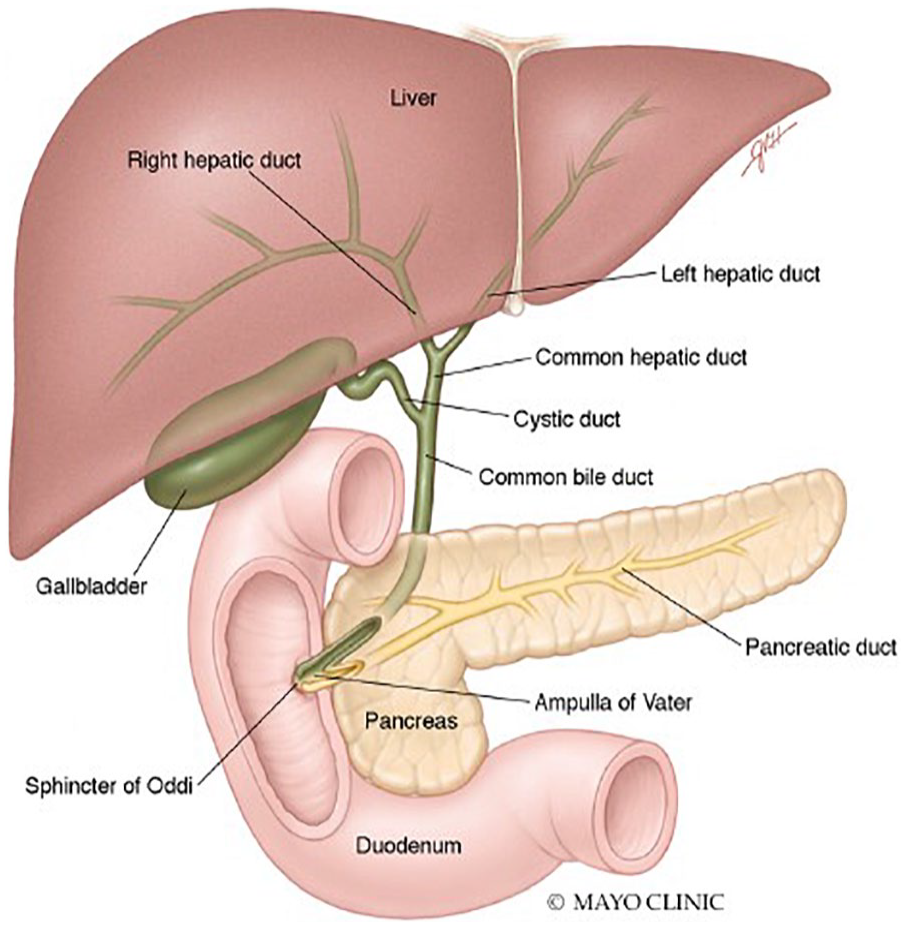
Diagram illustrating the normal GB and biliary tract anatomy. Used with permission of Mayo Foundation for Medical Education and Research. All rights reserved.
When severe enough, inflammation of the GB wall may lead to GB wall necrosis. Necrosis, or cell death, leads to break down of the GB wall and may ultimately result in perforation. Necrosis occurs secondary to ischemia, or inadequate blood flow. For the GB, obstruction of the cystic duct results in luminal distention and elevated pressure within the GB lumen. When blood perfusion is overcome by the elevated pressure in the GB wall, ischemic necrosis follows. 11 The fundal portion of the GB is the most common site of perforation because it has the weakest blood supply.6,7 Patients develop GB perforation or necrosis in 2% to 15% of acute cholecystitis cases. 6
Niemeier described three types of GB perforation in 1934 (Figure 10 and Table 1).5–8,12 Type I represents free perforation into the abdomen and generalized peritonitis following acute cholecystitis.5–8,12 Type I perforations usually occur in patients that do not have a history of chronic cholecystitis and may have heart disease, malignancy, diabetes, liver cirrhosis, or other immunosuppressive diseases. Subacute perforation, type II, is defined as a localized peritonitis with a pericholecystic abscess.5–8,12 Type II is the most common, followed by type I.6–8 Roslyn et al. reported that types I and II tend to occur in patients younger than 50 years of age, whereas type III patients were more common in the elderly population. 13 Chronic perforation, type III, represents fistula formation between the GB and adjacent bowel, or cholecystoenteric fistula.5–8,12 Type III perforations most commonly occur in patients with chronic GB disease or stones. 7
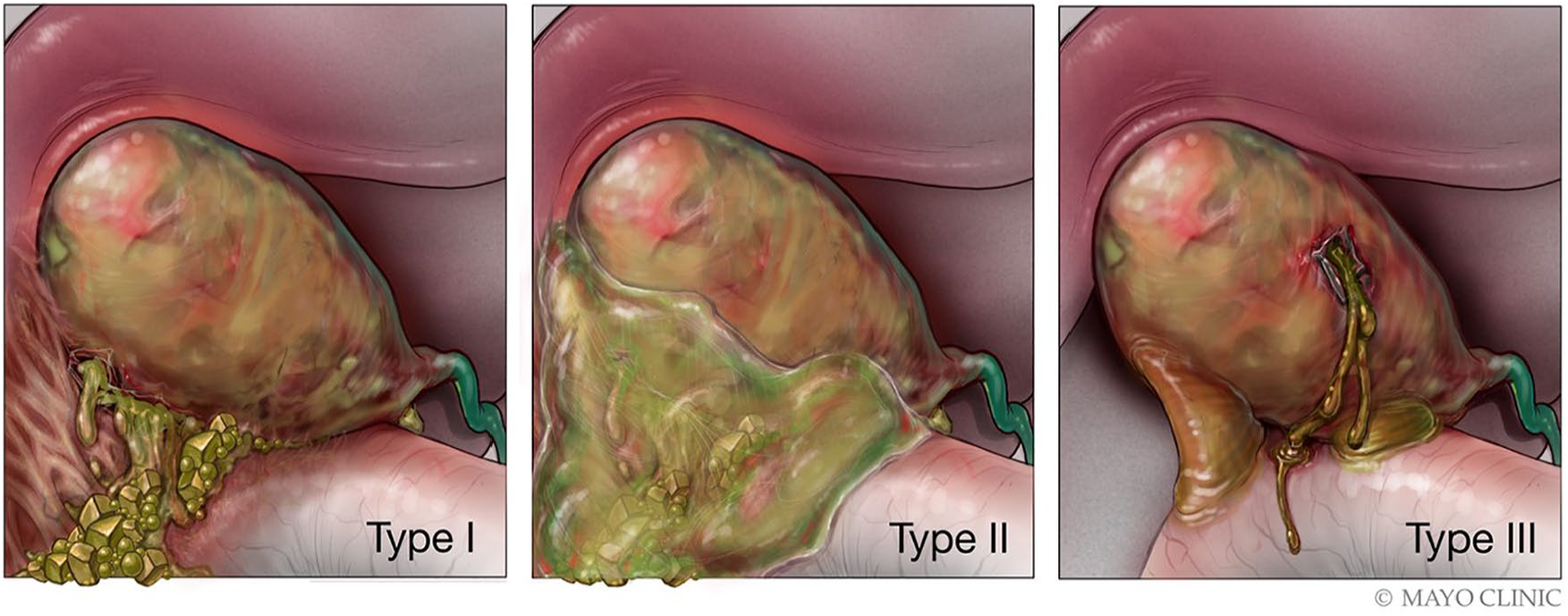
Diagram illustrating the three types of GB perforation. Used with permission of Mayo Foundation for Medical Education and Research. All rights reserved.
Three Types of GB Perforation Described by Niemeier.

Sonography is typically the first modality of choice assessing for GB pathology due to its high sensitivity and real-time imaging. The normal sonographic appearance of the GB is a completely anechoic fluid-filled lumen with a thin smooth wall <3 mm. Folds may be present and are considered normal. The patient should fast at least 4 to 6 hours prior to examination to prevent a contracted GB, which can obscure pathology. The sonographer should utilize multiple scanning windows including supine and left lateral decubitus patient positioning. A 2- to 6-MHz curvilinear transducer is typically used for imaging.
However, sonographic imaging is often challenging for accurately diagnosing GB wall perforation due to the site and size of the defect.6,8 Sood et al. reported that a GB wall defect could be demonstrated in 70% of patients using a high-resolution ultrasound imaging. 14 Kim et al. reported that a GB wall defect could not be identified in 54% of patients undergoing a CT examination. 15 When a distinct defect is not able to be identified by ultrasound, gray-scale sonographic imaging may demonstrate secondary findings such as layering of the GB wall and a pericholecystic fluid collection.
This case report illustrates that CEUS can be helpful to assess for GB perforation. CEUS is a relatively new, cost-effective, noninvasive technique that improves resolution limitations. Its application is now recognized as a useful tool in addition to sonography and other cross-sectional imaging in the assessment and diagnosis of GB disease. 11 Ultrasound contrast agents are gas-filled microbubbles injected intravenously and lasting 3 to 4 minutes for high-quality diagnostic imaging. As the contrast degrades due to bursting bubbles, the gas is expelled through the lungs. Currently, LUMASON is the only ultrasound contrast agent approved by the U.S. Food and Drug Administration (FDA) for abdominal application in adults and pediatrics, specifically focal liver lesion evaluation in adults. LUMASON comes as a nonrefrigerated kit. The ultrasound machine requires dedicated contrast software and/or hardware. A low power output and a low mechanical index (MI) between 0.05 and 0.4 should be used to minimize microbubble disturbance. 16 CEUS is a safe alternative for those with impaired renal function or an iodinated contrast allergy.17,18 The reported incidence of life-threatening anaphylactic reactions is extremely low at 0.001%. 19 CEUS may be limited due to the depth of the image target, obesity, motion, gas, shadowing calcification, or overlying bandages. 11
The hallmark sign using CEUS to assess for perforation is a focal nonenhancing full-thickness defect within the otherwise enhancing GB wall. Once perforation has occurred, adjacent fluid collections may be present. These collections may have mild enhancement appearing as a honeycomb pattern or may be completely anechoic demonstrating no enhancement.6,11
Because the diagnosis of GB perforation often proves difficult, treatment is often delayed, resulting in higher morbidity and mortality rates. The mortality rate of GB perforation reported by Glenn and Moore is 42%. 20 Other studies reported declining mortality rates of 12% to 16% due to recent developments in anesthesiology and the ICU.13,21 The high morbidity and mortality underscores the importance of a prompt imaging diagnosis.
Treatment options for GB perforation include cholecystectomy, percutaneous drainage, or abdominal washout. A cholecystectomy may be performed once the infection has cleared the body following a percutaneous drain placement in type II GB perforations. 7 In type III GB perforations, a cholecystectomy may be difficult and require additional surgical interventions to repair the fistula. 7
Conclusion
Sonography remains a cost-effective, accessible, nonnephrotoxic, and nonionizing tool for the diagnosis of GB disease and is the initial imaging modality of choice. GB perforation is a severe illness that requires a prompt imaging diagnosis, but unfortunately, traditional gray-scale and color Doppler imaging may not detect the perforation. High-frequency transducers should be utilized to scrutinize the wall for a focal defect. In equivocal cases of GB wall integrity and/or when secondary signs of perforation such as an adjacent fluid collection exist, further evaluation with CEUS may be helpful. The key CEUS finding of GB perforation is a focal area of GB wall nonenhancement.
Footnotes
Acknowledgements
The authors thank Britta Thomforde, RDMS, RVT, for her assistance and expertise in this clinical case.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
