Abstract
This case study describes an obstetrical patient who presented for a routine second-trimester morphology sonogram in which the fetus was found to have arthrogryposis multiplex congenita. Other abnormalities visualized included a single umbilical artery, hypoplastic nasal bone, polyhydramnios, and poorly visualized lateral ventricles. Amniocentesis demonstrated a normal microarray with a negative result for cytomegalovirus and toxoplasmosis. This case study highlights the utility of prenatal sonography to identify functional abnormalities of the fetal musculoskeletal system.
Sonography has become an essential diagnostic tool for imaging in obstetrics. Sonography can identify abnormalities in both structure (i.e., short long bones, cardiac abnormalities) and function (e.g., lack of normal movement of extremities and abnormal cardiovasculature). Assessment of normal function of the musculoskeletal system is an essential part of the prenatal sonographic evaluation. Arthrogryposis multiplex congenita (AMC) is a syndrome that is defined by the abnormal position of two or more joints. The cause of AMC is believed to be decreased fetal movement or fetal akinesia. 1
Case Report
A 29-year-old woman presented for a routine second-trimester morphology sonogram at 20 weeks of pregnancy. The first-trimester sonogram was unremarkable. The sonogram revealed an intrauterine pregnancy with multiple fetal anomalies. The arms and hands were abnormally flexed and did not move from this position during the course of the examination (Figures 1–3). Both legs were persistently flexed at the hip and knee. The right leg was notable for the presence of a club foot (Figure 4), and there were additional abnormalities in the position of the toes (Figure 5). The left leg had only a single bone distal to the knee (Figure 6). There did not appear to be a fully formed left foot (Figure 7). The fetal brain was difficult to visualize because of fetal position. The bilateral ventricles were not well visualized at the time of the initial morphology scan but were felt to appear normal at the follow-up examination. The amniotic fluid appeared increased for the gestational age with the maximum vertical pocket (MVP) measuring 9.91 cm. A single umbilical artery, hypoplastic nasal bone, and micrognathia were also visualized (Figure 8). There were no obvious cardiac or renal anomalies. A genetic amniocentesis was performed under sonographic guidance, and results demonstrated normal microarray.
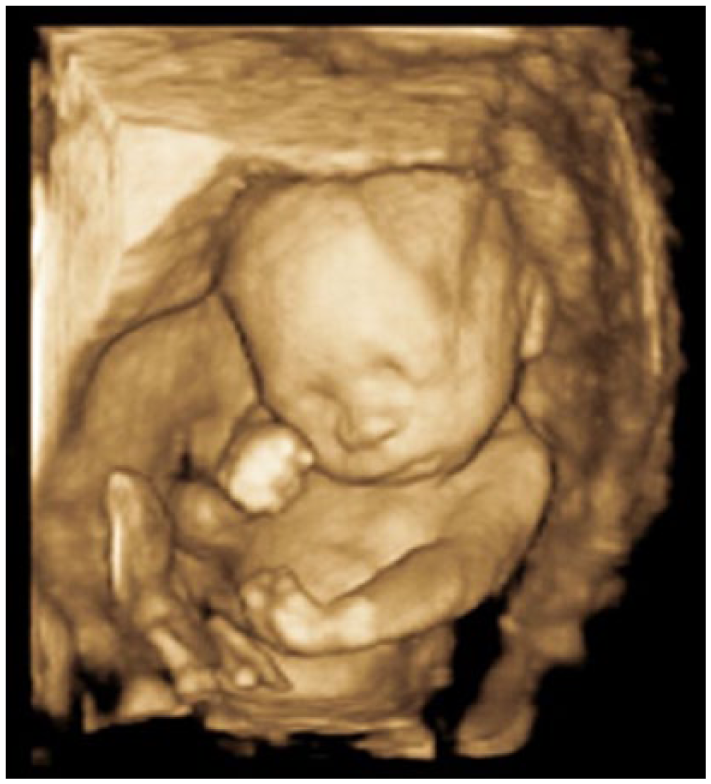
A 3D sonogram demonstrating abnormally flexed arms and hands.
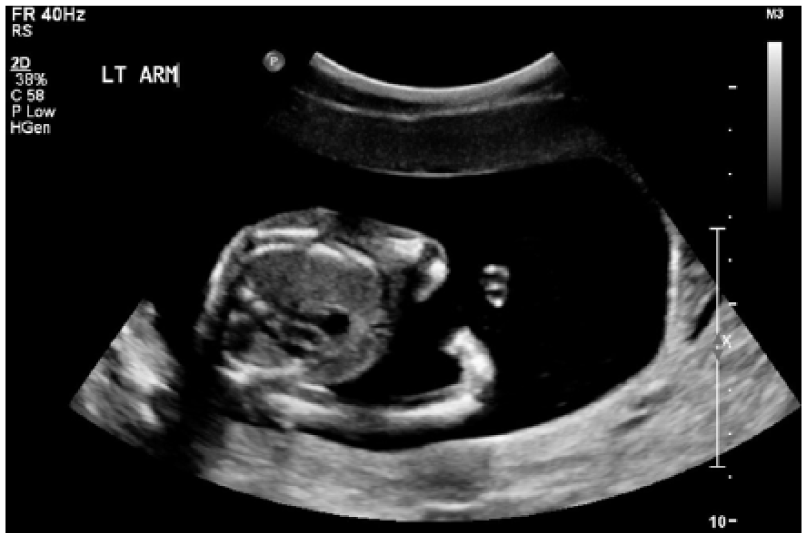
A 2D sonogram demonstrating an abnormally postured left arm.
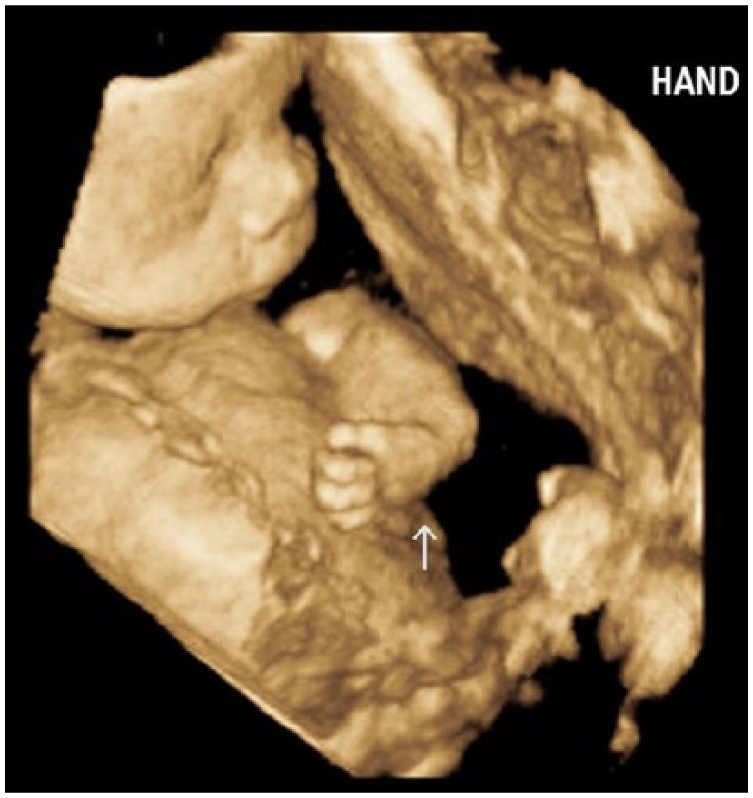
A 3D sonogram demonstrating the abnormally flexed left hand.
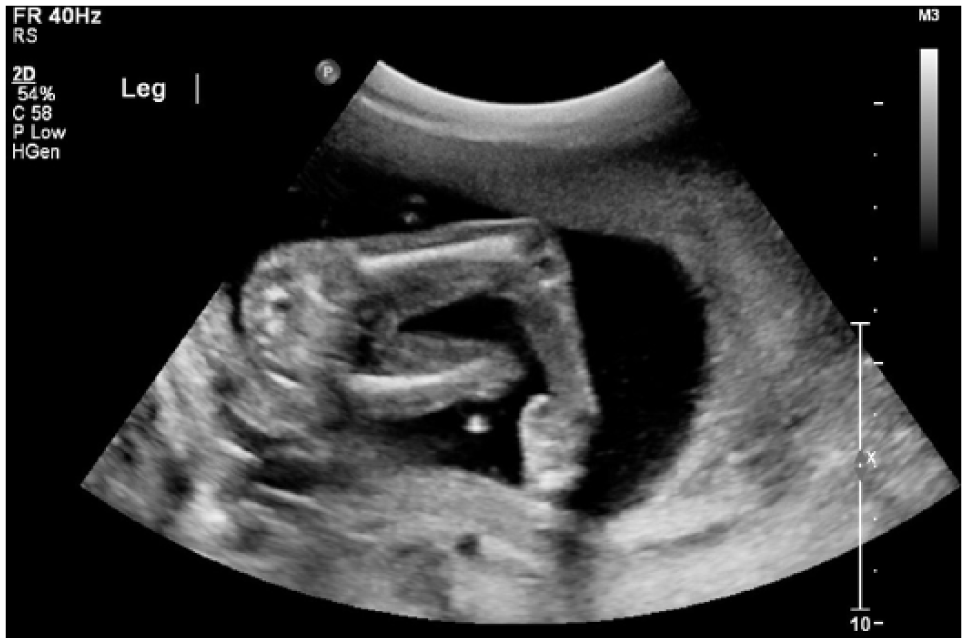
A 2D sonogram demonstrating a right clubbed foot.
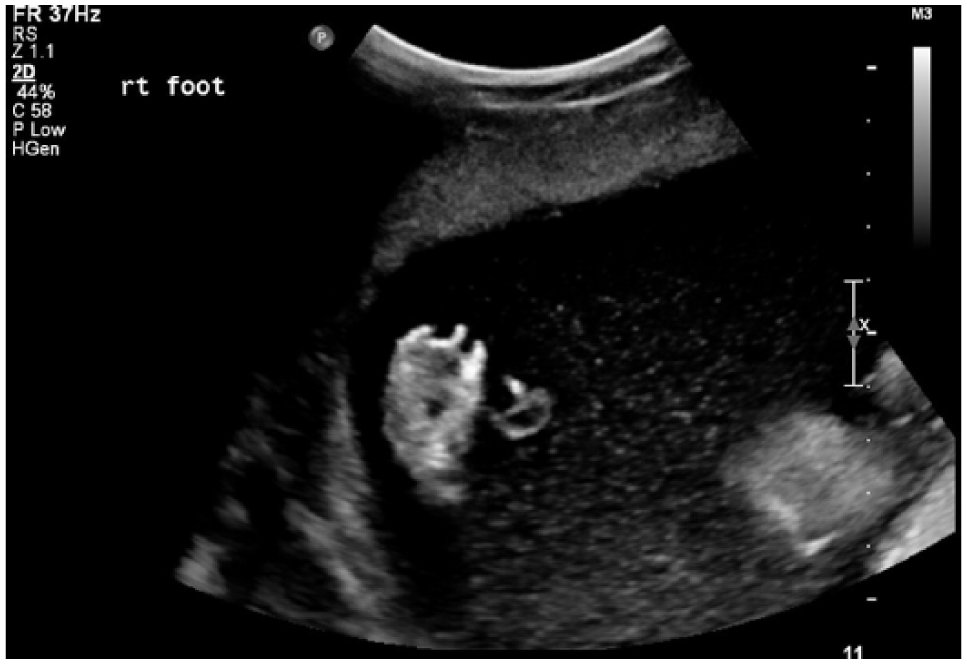
A 2D sonogram demonstrating abnormally positioned toes on the right foot.
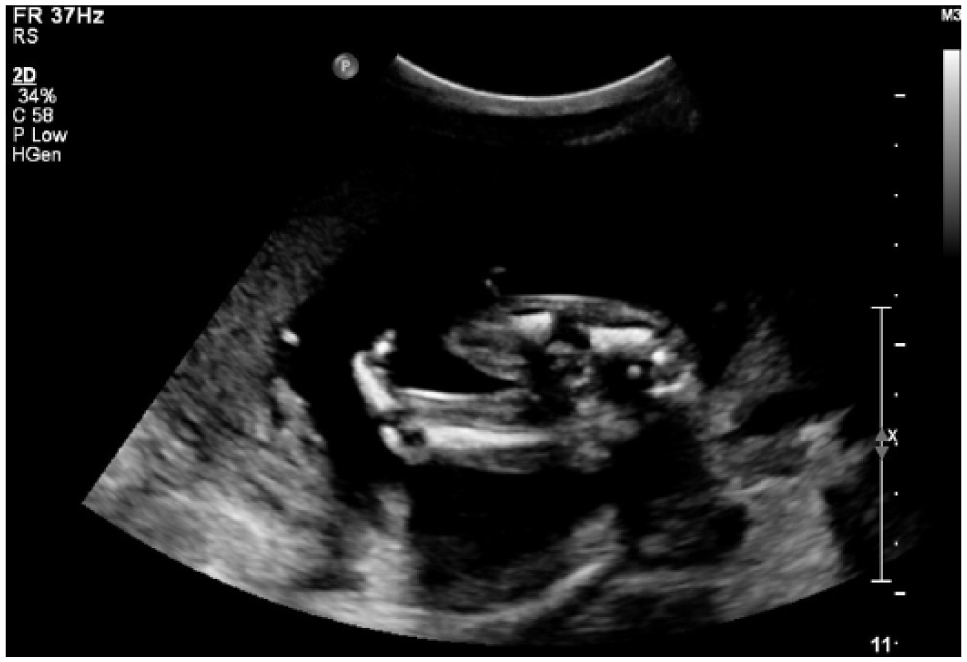
A 2D sonogram demonstrating the single bone distal to the left knee.
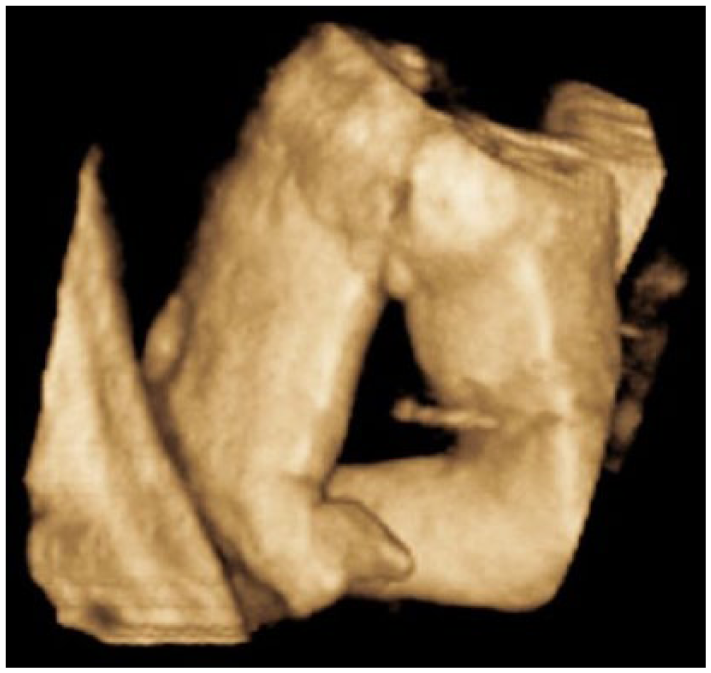
A 3D sonogram demonstrating hypoplasia of the left tibia/fibula.
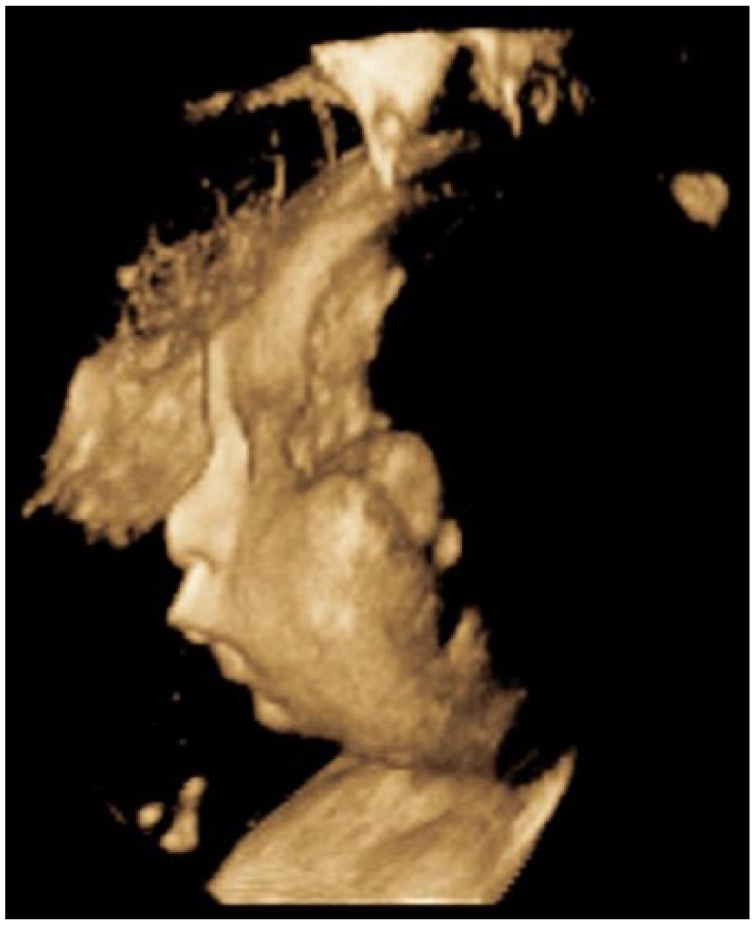
A 3D sonogram of the fetal profile demonstrating micrognathia.
The pregnancy was followed with monthly sonograms to assess fetal growth and weekly biophysical profiles (BPP) starting at 32 weeks’ gestation. Weekly umbilical artery Doppler studies were performed because of the finding of intrauterine growth restriction at 34 weeks’ gestation.
At 37 weeks’ gestation, a routine BPP was scored at 2/8 because of inadequate gross body movement, tone, and fetal breathing being observed during the examination. Polyhydramnios was present with a maximum vertical pocket of 8.83 cm and amniotic fluid index of 32 cm. The patient was sent to the labor and delivery area, where the fetal heart tracing was noted to be nonreassuring and cesarean delivery was performed. The surgery was uncomplicated with an estimated blood loss of 800 mL (deemed to be normal). Upon delivery, the infant was brought to the resuscitation room. The infant was placed under a radiant warmer and stimulated but was noted to be limp and breathing spontaneously. Multiple congenital anomalies were appreciated during this initial examination, including contractures in all four limbs and a short left lower limb. Pulse oxygen and a temperature probe were placed. At this time, the infant developed moderate subcostal and suprasternal retractions and coarse breathing sounds. The decision was made to transfer the patient to the neonatal intensive care unit due the infant’s respiratory distress. Otolaryngology was consulted for evaluation and establishment of a definitive airway. The patient did not respond to positive pressure ventilation or continuous positive airway pressure. Intubation was attempted, but secondary to poor jaw opening, a view of the epiglottis was unable to be obtained, and the airway was temporized emergently with a laryngeal mask airway (LMA).
Orthopedics, cardiology, neurology, and genetics consults were recommended. An echocardiogram was performed and revealed a small midmuscular ventricular septal defect (VSD). Also noted was a posterior pulmonary venous confluence with unobstructed drainage into the left atrium. Normal left ventricle systolic function was noted. The VSD and abnormal pulmonary venous anatomy did not seem to be hemodynamically significant at that time. A prenatal sonographic diagnosis of VSDs and pulmonary venous abnormalities can be difficult. Because of the lack of a pressure gradient between the ventricles in utero, small VSDs may not be visible on gray-scale echocardiography. 2 Given the patient’s history and presentation, it is likely that her respiratory distress was due to upper airway obstruction/micrognathia given good pulmonary compliance and functionally normal anatomy.
Orthopedics confirmed findings of severe contractures (Figure 9). The right arm appeared to be well developed with good range of motion of the shoulder and elbow. The left upper extremity was smaller than the right. Shoulder range of motion appeared to be adequate; however, the left elbow had limited range of motion from full extension to about 40° of flexion. There were flexion contractures of the wrist and fingers (Figures 10 and 11). Five fingers were present bilaterally. The right leg appeared clubbed with cavus abductus and equinus deformity (Figure 12). The left leg was dysplastic. There appeared to be a knee and a proximal segment of the tibia with a foot that was void of any bone or cartilage (Figures 13 and 14).
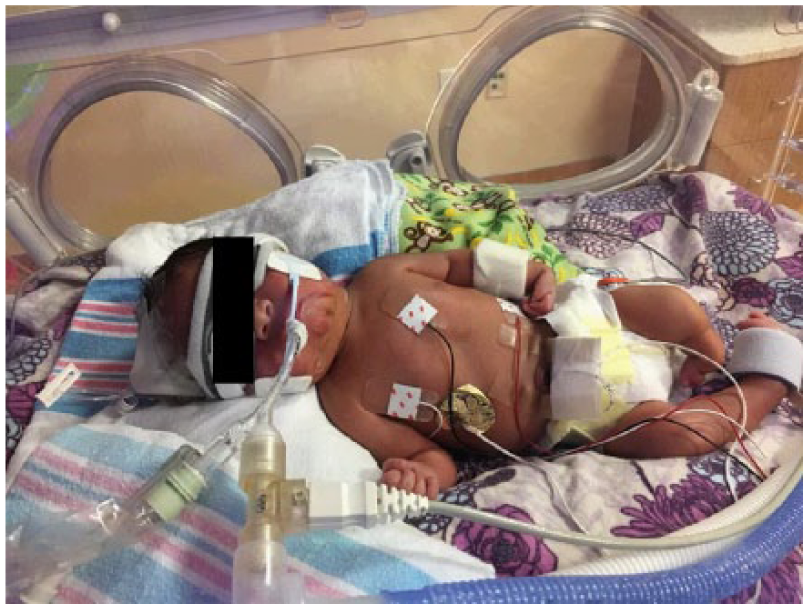
Postnatal image demonstrating contractures in all four limbs and a short left limb.
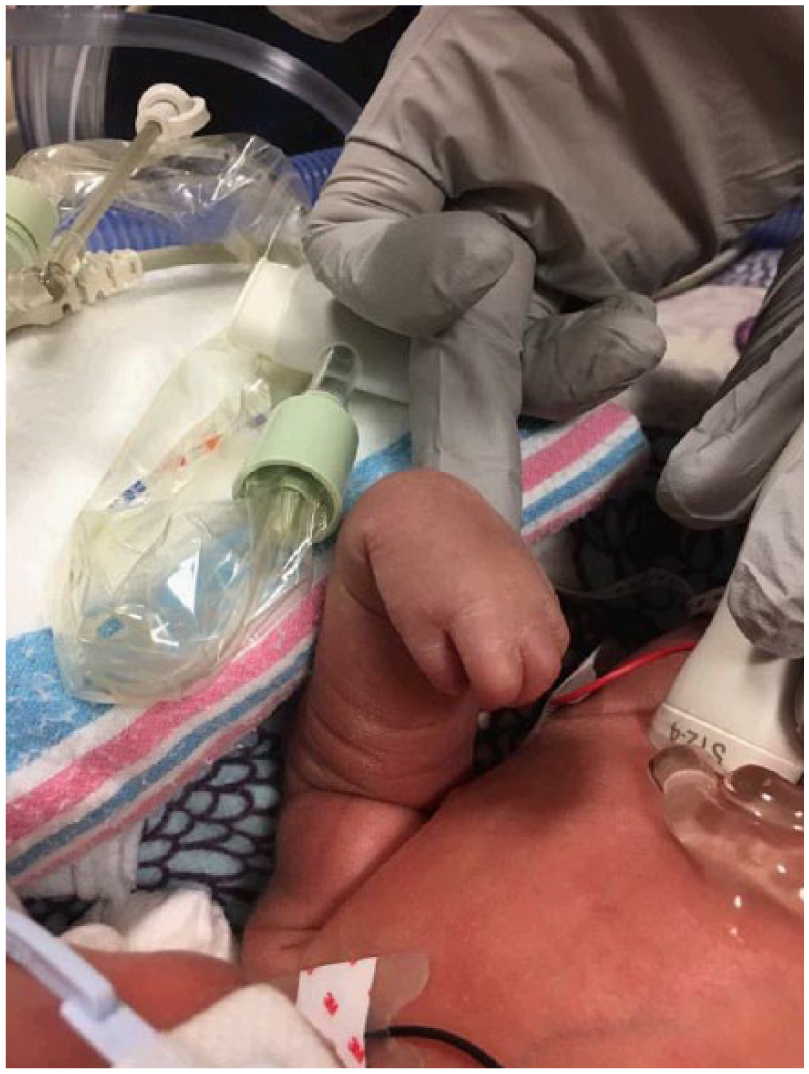
Postnatal image of the left arm demonstrating flexion contractures of the elbow, wrist, and fingers.
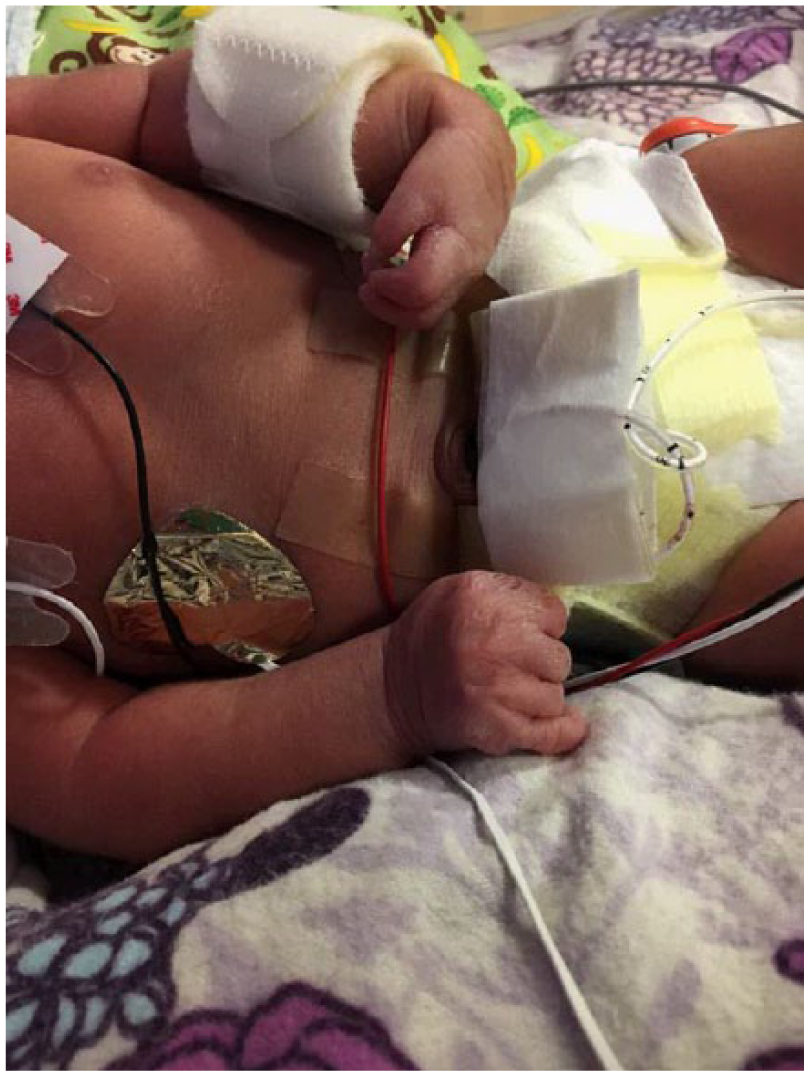
Postnatal image of the left wrist demonstrating flexion contractures of the wrist.
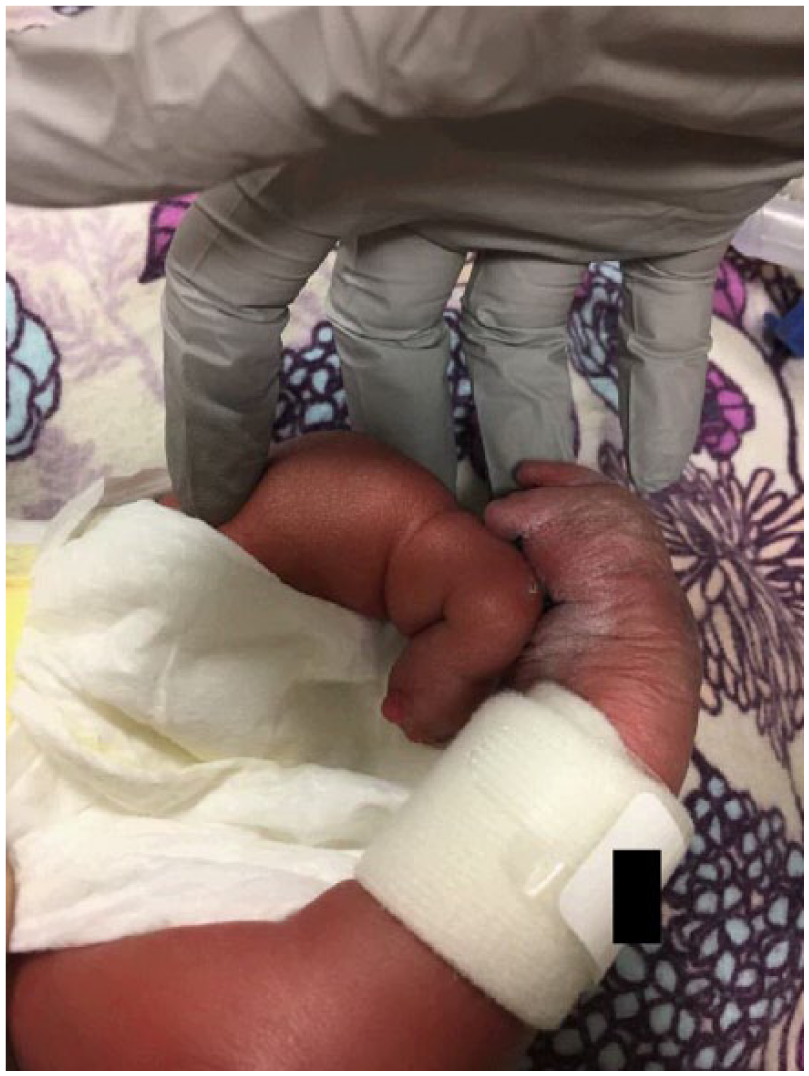
Postnatal image of the left and right legs. A short left limb and clubbed right foot is demonstrated in this postnatal image.
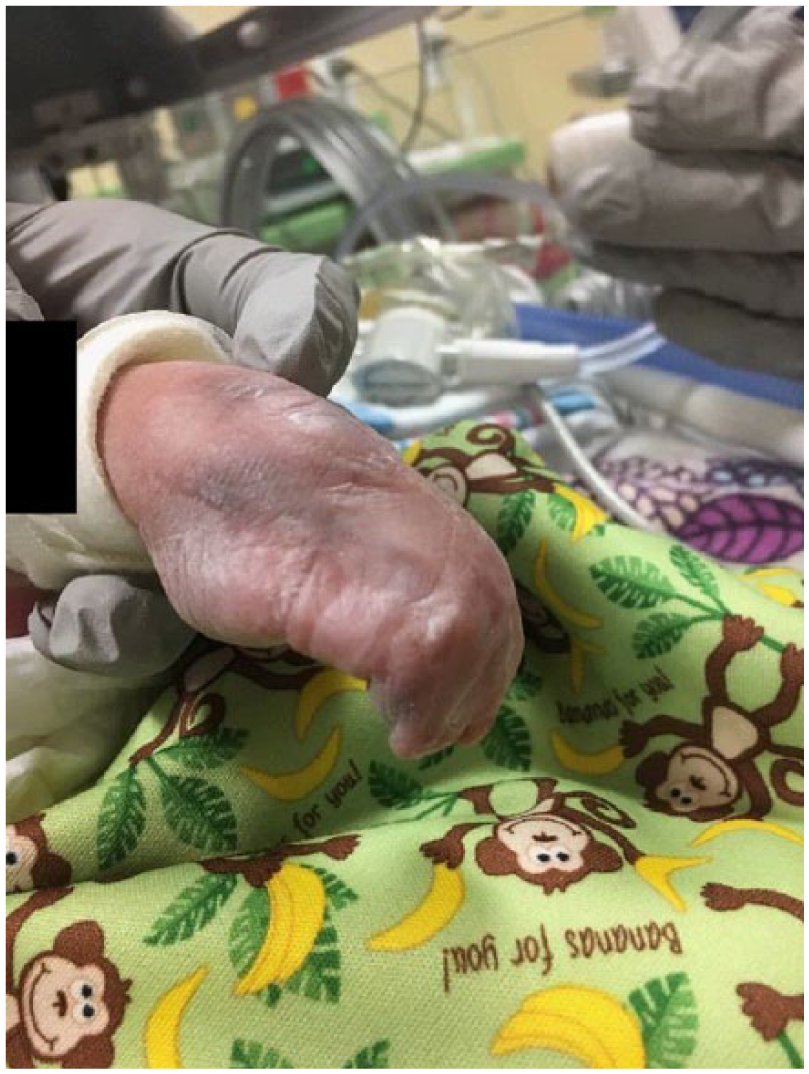
Postnatal image of the dysplastic left leg that felt void of any bone or cartilage.
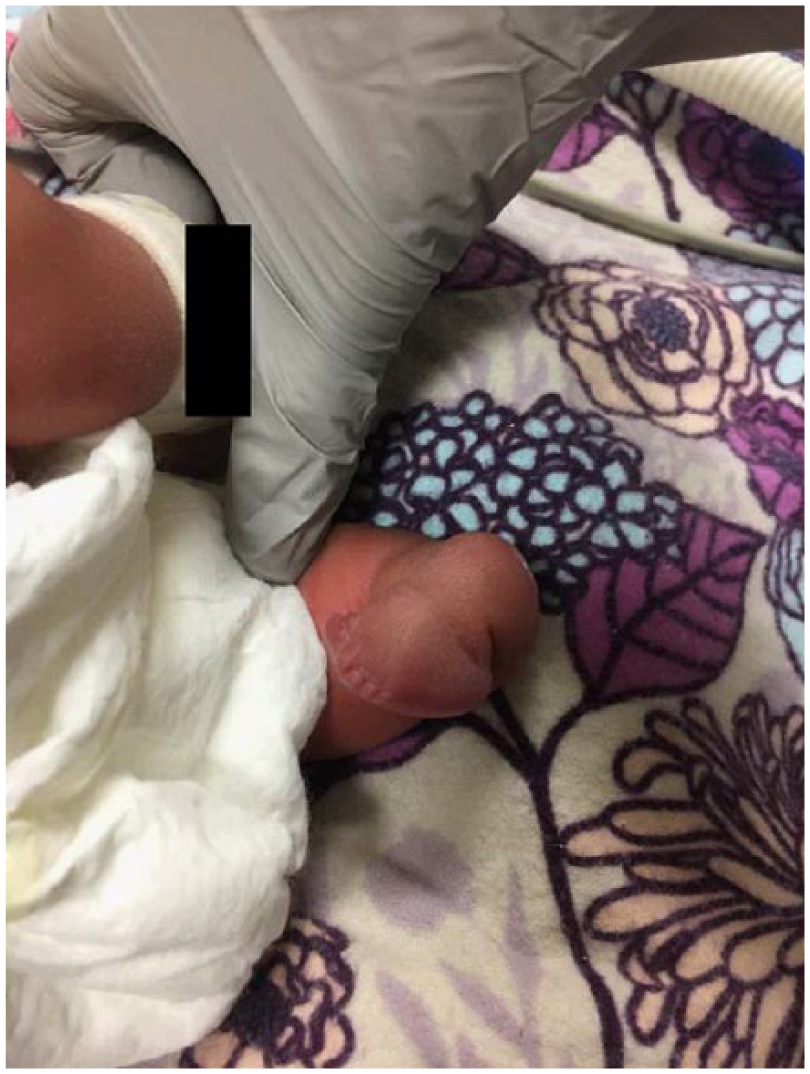
Postnatal image of the dysplastic left leg.
Radiographs of the arms and legs further confirmed these findings. The right humerus and forearm bones were completely formed and appeared normal in size. Radiographs of the right lower arm/hand revealed ulnar deviation of the second through fifth proximal phalanges at the metacarpophalangeal joints. There were flexion contractures of the second and fifth interphalangeal joints with diffusely hypoplastic distal phalanges. Radiographs of the left arm demonstrated a hypoplastic arm and forearm bones (Figure 15). The arm and forearm were asymmetrically shorter and thinner compared with the contralateral side. There were flexion contractures at the left wrist and distal interphlangeal joints with hypoplastic/malformed distal phalanges (Figure 16). There appeared to be a hypoplastic first metacarpal. Additional radiographs of the lower extremities revealed severe pes cavus of right lower extremity with dorsiflexion of the calcaneus and abnormally increased calcaneal pitch angle (Figure 17). A radiograph of the right foot was also obtained. The right foot was plantarflexed and inverted with abnormally positioned toes (Figure 18). The left lower leg radiograph demonstrated a femur and the proximal segment of tibia and fibula as well as a dislocated left hip (Figure 19).
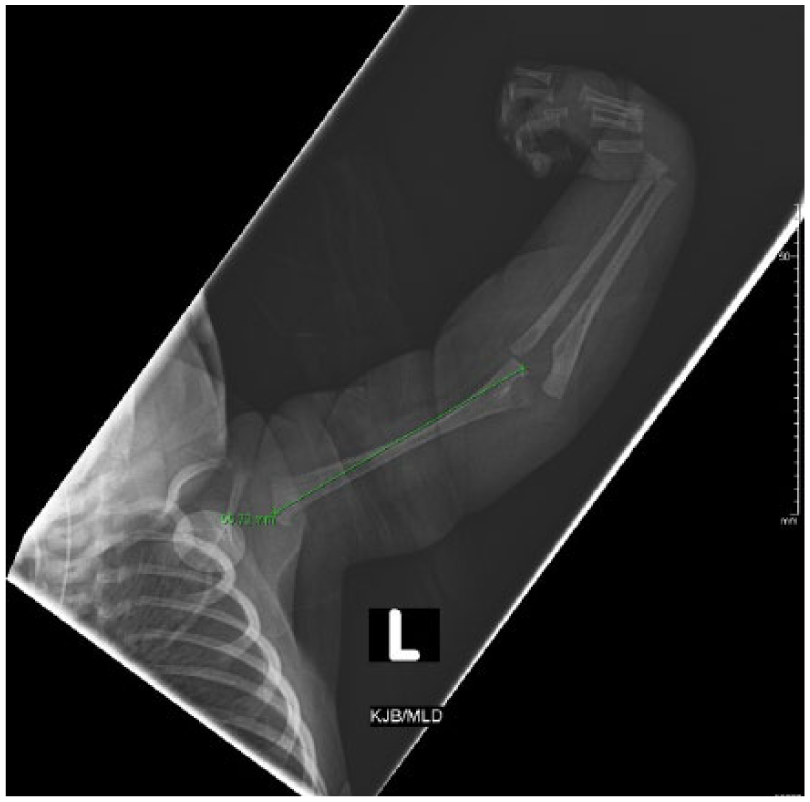
Radiograph of the dysplastic left arm and forearm with flexion contractures at the wrist.
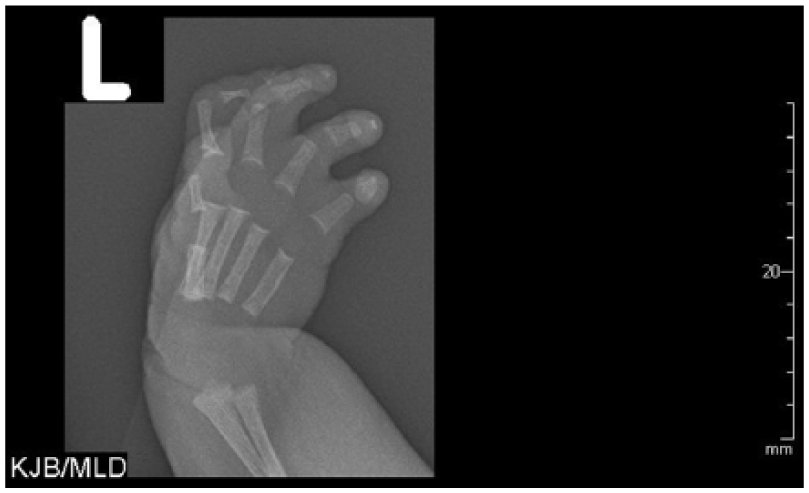
Radiograph of the left hand demonstrating flexion contractures of the wrist and distal interphlangeal joints with hypoplastic/malformed distal phalanges.
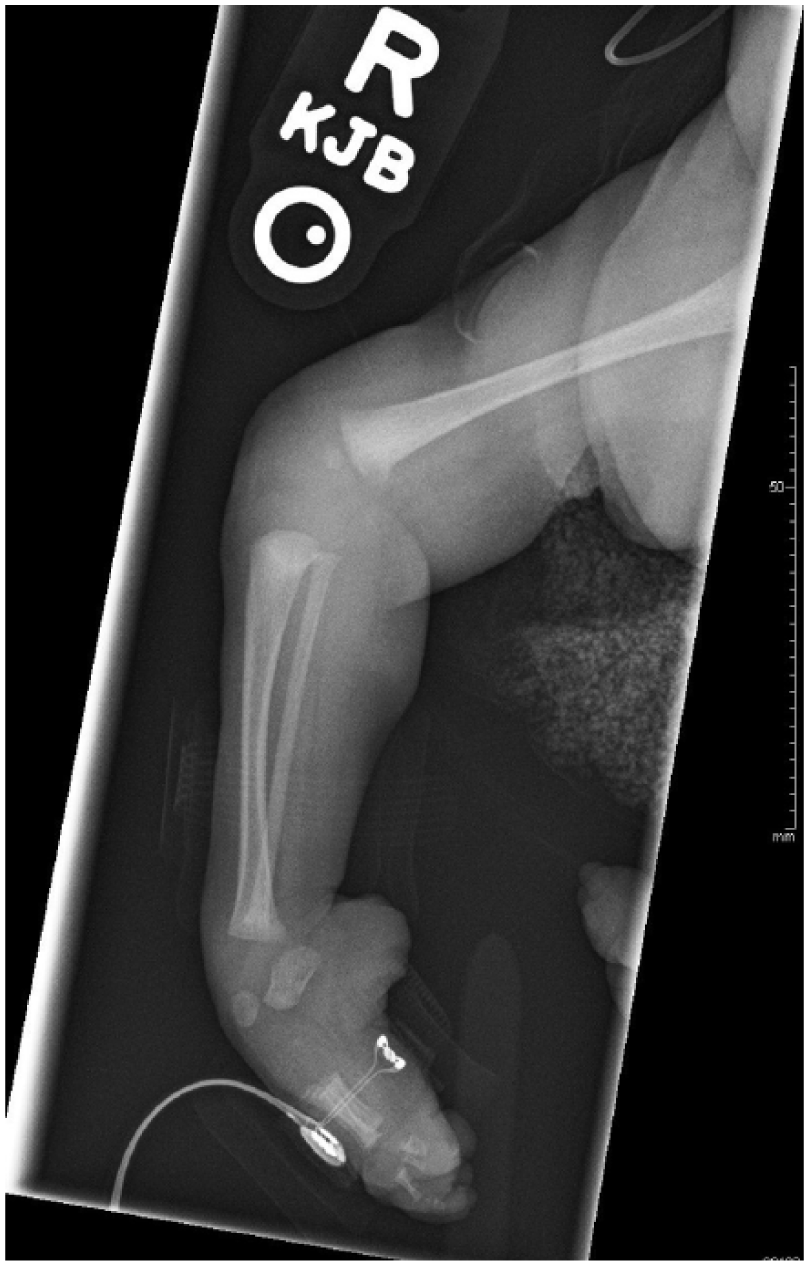
Radiograph of the right lower leg demonstrating severe pes cavarus with dorsiflexion of the calcaneus and abnormally increased calcaneal pitch angle.
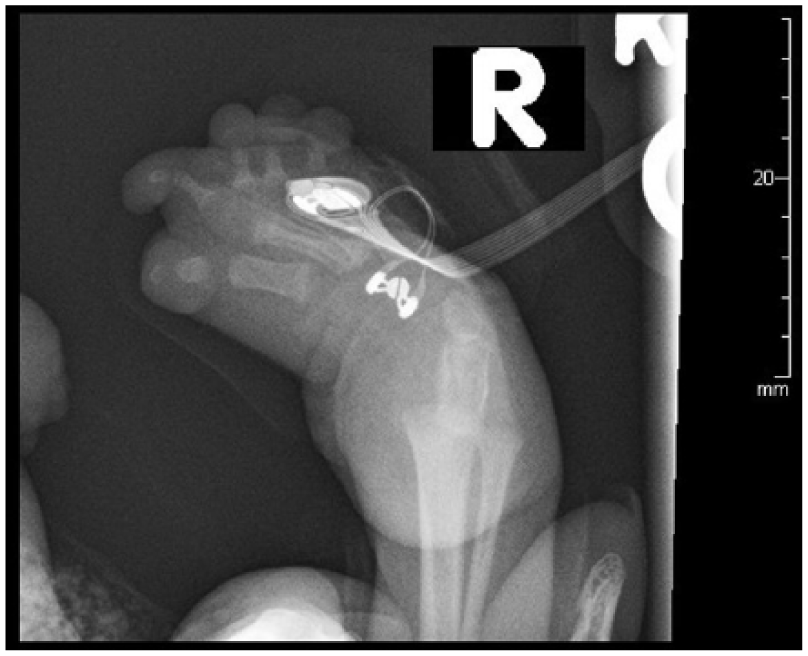
Radiograph of the plantarflexed and inverted right foot with abnormally positioned toes.
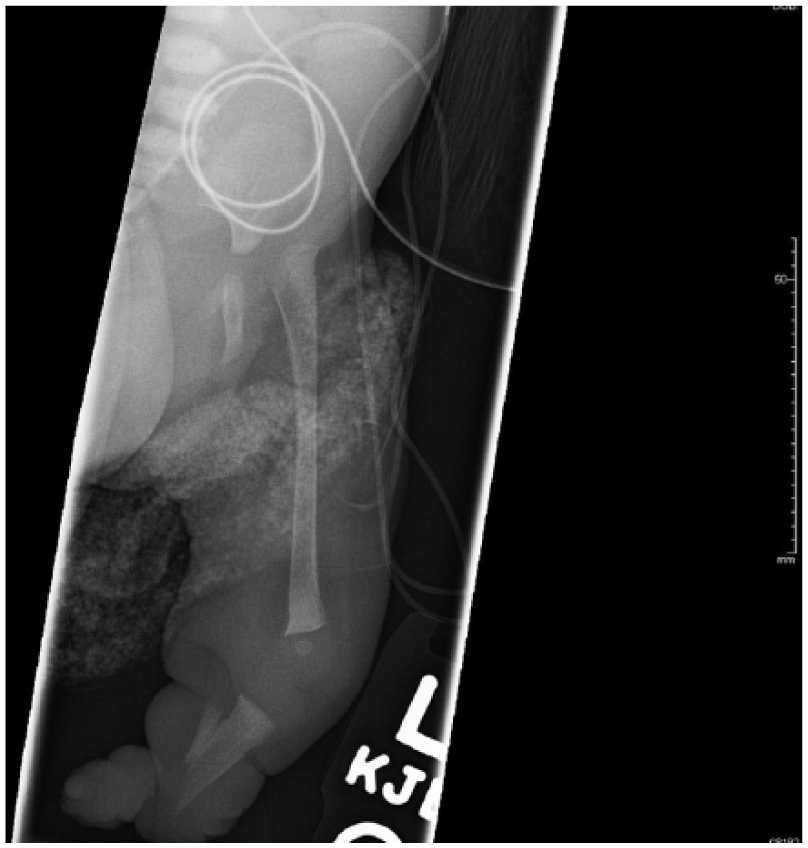
Radiograph of the left lower leg. A femur and proximal segment of the tibia and fibula are visualized as well as a dislocated left hip.
Neurology recommended pediatric magnetic resonance imaging (MRI) of the brain because of poor visualization of the lateral ventricles in utero and multiple fetal anomalies. The MRI demonstrated extensive bilateral supratentorial cortical abnormalities with predominant pattern of pachygyria. There was also focal polymicrogyria involving the left insula suggestive of closed-lip schizencephaly involving the inferior right frontal gyrus, right insula, and superior temporal lobe. Schizencephaly is defined by abnormal clefts in the cerebral hemispheres and can affect one or both sides of the brain.
Genetics further examined the patient. The patient was found to have dysmorphic facies, micrognatia, syndactyly of the toes, and camptodactyly of the fingers. Except for occasional recreational marijuana use and prenatal vitamins, the patient denied any prescription or over-the-counter medications, alcohol, infections, or other potential teratogens. Genetics recommended a whole exome sequencing. Whole exome sequencing evaluates all exomes in the genome. It is able to detect genetic disorders that would not be detected by a microarray. 3 Because of the potential for misinterpretation of the results and the increased cost, whole exome sequencing is not recommended as first-line genetic testing in obstetrics. 3 It can be useful in cases such as this with multiple fetal anomalies and a normal microarray. The whole exome sequencing came back with normal results. Therefore, it is difficult to combine these findings under a single unifying diagnosis. However, they are most consistent with a diagnosis of AMC.
Discussion
AMC is rare, with a prevalence of 1 in 3000 live births with equal gender ratios.1,4 The term arthrogryposis is derived from the Greek word meaning “curved or hooked joints.” 4 Contractures range in severity from a clubbed foot to the lethal Pena-Shokeir phenotype, which is characterized by facial anomalies, multiple joint contractures, polyhydramnios, intrauterine growth restriction, and pulmonary hypoplasia.5,6 Fetal movement can be recognized with sonography by 8 to 9 weeks’ gestation; however, contractures are difficult to identify in the first trimester. The most common time to diagnose contractures is during the second trimester. Arthrogryposis is often an incidental finding. A club foot is the most common isolated finding identified during a routine second-trimester sonogram, with a prevalence of 1 in 500 live births.1,4,5 If one joint contracture or anomaly is noted, a comprehensive evaluation of all limbs and joints should be performed. The differential diagnosis is broad, and the workup includes a recommendation for invasive genetic testing (microarray and karyotype analysis); complete anatomic survey with assessment for vertebral defects, anal atresia, cardiac defects, tracheoesophageal fistula, renal anomalies, and limb abnormalities; and a family history. Some cases are identified only later in pregnancy, when a sonogram is urged because of decreased fetal movement. 5 In this setting, it may be reasonable to defer genetic testing until after delivery.
Although the diagnosis of AMC is more common in the second and third trimester, there are reports of affected fetuses demonstrating anomalies in the first trimester. In a study of 27 cases of lethal congenital arthrogryposis, 26% of the fetuses presented with nuchal edema during second- or third-trimester sonography examinations. 7 Of these cases, two had previously demonstrated nuchal translucency thickness of greater than the 99th percentile for gestational age during the first-trimester sonogram. 7 Although this retrospective study was performed on a small sample size, it suggests the need for future, larger-scale studies exploring the relationship between abnormal nuchal translucency measurement in the first trimester and AMC. 7 Based on these limited findings, it may be important to consider lethal arthrogryposis in the differential diagnosis, given a thickened nuchal translucency. This highlights the importance of counseling patients that in the setting of a thickened nuchal translucency and normal microarray on chorionic villus sampling or amniocentesis, there remains an increased risk of adverse perinatal outcome. 7
The pathogenesis of AMC is unknown. The condition has been reproduced in animal models by “viruses, neuromuscular blocking agents, hyperthermia and limb immobilization.”1,4,5 In an early animal study from 1969, chickens and rats were immobilized early in fetal development. 8 This led to multiple anomalies, including intrauterine growth restriction, generalized contractures, shortened extremities, pulmonary hypoplasia, shortened and premature gut, and craniofacial changes. 8 These anomalies are features of the lethal Pena-Shokeir phenotype of arthrogryposis. 8 Broadly speaking, the differential diagnosis can be thought of in terms of extrinsic (oligohydramnios) and/or intrinsic (neurologic) insult resulting in a loss of muscle mass that produces connective tissue around the joint and causes fixation.4,5,8 Oligohydramnios may be due to early rupture of membranes, fetal renal agenesis, or bladder outlet obstruction. Fetal neurologic insult can be caused by a number of factors, including infection (cytomegalovirus, toxoplasmosis, or Zika virus), fetal cerebral vascular accident resulting in schizencephaly or porencepahly, aneuploidy, and or a number of syndromic conditions, the most common of which is spinal muscular atrophy type I Werdnig-Hoffmann disease. Recurrence risk depends on the underlying etiology. For example, spinal muscular atrophy (SMA) is inherited in an autosomal recessive pattern, and carrier parents would have a 25% risk of having another affected child. Genetics consultation and individualized assessment of recurrence risk is recommended.
The etiologies of AMC can be separated into subcategories, as a way of producing differential diagnosis. 8 This includes neurologic diseases, connective tissue defects, muscle abnormalities, limitations in utero, intrauterine fetal vascular compromise, and maternal disease.1,4,5,8 Neurologic anomalies comprise 70% to 80% of cases of AMC.1,4,5 The most common neurologic anomaly is anterior horn cell disease and SMA1,4 Maternal myasthenia gravis has been reported to cause decreased fetal movement due to the transfer of acetylcholinesterase receptor antibodies across the placenta. Other muscular disorders that may cause AMC include but are not limited to muscular dystrophy, myopathies, myositis, and mitochondrial disorders. 4 Regardless of the underlying mechanism, fetal motion is vital for appropriate growth of fetal joints. A lack of motion triggers a collagenic response around the joints, causing contractions or joint fixations. 4 Syndromes that may cause joint fixation and are most commonly associated with AMC include Larsen dysplasia, Beal contractural arachnodactyly, and pterygium syndrome. 4 Insufficient blood supply to the fetus during utero can also cause contractures. Inadequate blood supply can lead to fetal hypoxia, which leads to fetal cell death, most commonly anterior horn cell death. 4 This cell death causes damage in the neurons, muscles, and bones and as a result develops multiple contractures. 4 Lastly, maternal disease such as multiple sclerosis, myotonic dystrophy, diabetes mellitus, and infection have all been closely associated with akinesia or AMC. 4
Management and treatment of AMC must be initiated as early as possible to achieve optimal results. The primary long-term treatment goal is improved joint mobility and attaining the best quality of life, whether independent daily living or social involvement.1,9 Each treatment plan is exclusively tailored to each patient and most often includes a team of pediatricians, orthopedic surgeons, geneticist, occupational/physical therapists, and a case manager. 9 With surgical and therapy treatments available, significant improvements in mobility and joint function are possible. Casting or splinting is the most common treatment option. This involves repositioning the affected limb into the most normal position achievable and then immobilizing it for long periods of time. This process is repeated every 1 to 2 weeks or as recommend by occupational/physical therapist to increase range of motion. If repositioning and immobilization fail to move the involved extremity to a normal position, surgery may be recommended. Most commonly, contractures are an isolated finding; however, in more extreme cases, contractures are associated with lethal genetic disorders (Trisomy 18, Trisomy 13 Pena-Shokeir syndrome) and therefore have a poor prognosis. In this particular case, the infant received progressive thermoplastic splints. The left wrist was placed in a neutral position with the digits in extension. The right hand was also splinted to improve alignment and structural integrity. Routine passive range of motion numerous times per day with therapy was also performed. The right leg was casted, and the infant was placed in a Pavlik harness due to findings of bilateral hip dysplasia.
Conclusion
Prenatal diagnosis of AMC is made based on fetal sonographic findings. The very subtle abnormally postured limbs can be difficult to visualize, and therefore fetal movement is important to visualize during routine examinations. The differential diagnosis for AMC is broad and requires a substantial diagnostic workup. Establishment of an underlying etiology is essential to inform families regarding prognosis, arrange for appropriate treatment, and provide an estimation of recurrence risk for future pregnancies.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
