Abstract
Pulmonary artery pseudoaneurysm (PAP) is an uncommon entity associated with high mortality if left untreated. PAPs are most often acquired secondary to trauma, inflammation, infection, or malignancy. The most common presentation is hemoptysis, but many patients are asymptomatic. Because of the increased risk of enlargement and rupture, timely diagnosis and intervention is necessary. Imaging plays an essential role not only in diagnosis of this entity but also in intervention. We describe a case using gray-scale and color Doppler imaging providing sonographically aided diagnosis and follow-up of a clinically silent PAP in a pediatric patient.
Introduction
Pulmonary artery pseudoaneurysms (PAPs) are relatively uncommon but are associated with high mortality. Reports in the literature cite mortality rates following rupture of approximately 45% to 65%, with over 50% mortality in cases involving significant hemoptysis.1,2 In contrast to true aneurysms, which are outpouchings of vessels demonstrating a three-layer wall, a pseudoaneurysm represents a hematoma resulting from disruption of at least one vascular layer, weakening the vessel and therefore increasing susceptibility to rupture.
3
Pulmonary artery pseudoaneurysms are not frequently described in the pediatric population, but most common causes include trauma (including iatrogenic) and infection.4–8 Because of their rarity, the exact incidence is unknown. Diagnosis is most often made on computed tomographic angiography (CTA), which can depict the vessels involved. In this case report, we describe a case of a PAP in the setting of methicillin resistant
Case Report
A 6-month-old female presented to the emergency department with increased work of breathing, cough, and fever up to approximately 103°F. A chest radiograph at the time of admission showed opacification suspicious for pneumonia and possible pleural effusion. The patient was admitted to the pediatric intensive care unit with increasing respiratory distress, requiring endotracheal intubation. Because of persistent pleural fluid accumulation, the patient underwent a video assisted thoracoscopic surgery (VATS), with placement of a percutaneous drainage catheter. Cultures of the pleural fluid were positive for methicillin-resistant
Because of continued fevers and associated leukocytosis, elevated C-reactive protein (CRP), and difficulty weaning off mechanical ventilation, contrast enhanced computed tomography (CT) of the chest was obtained (Figure 1). A large right hydropneumothorax was seen, with evidence of consolidated and necrotic lung parenchyma, presumably secondary to MRSA pneumonia. Within the right lower lobe, a hyperdense lesion was seen, concerning for a PAP. Less likely, given the presence of multiple cavitations, mycetoma was considered. Transthoracic chest sonography was done to further evaluate this finding (Figure 2) using a Philips iU22 (Philips Ultrasound, Bothell, Washington, USA) and a curvilinear C5-1 probe. Within the visualized portion of the right lung, gray-scale imaging showed a circumscribed hypoechoic lesion corresponding to the CT lesion measuring approximately 1.6 cm in diameter. Color Doppler imaging showed a characteristic “yin yang” pattern of turbulent flow. Finally, spectral Doppler waveforms demonstrated a “to and fro” pattern (Figure 3). All of these findings supported the diagnosis of a pseudoaneurysm.
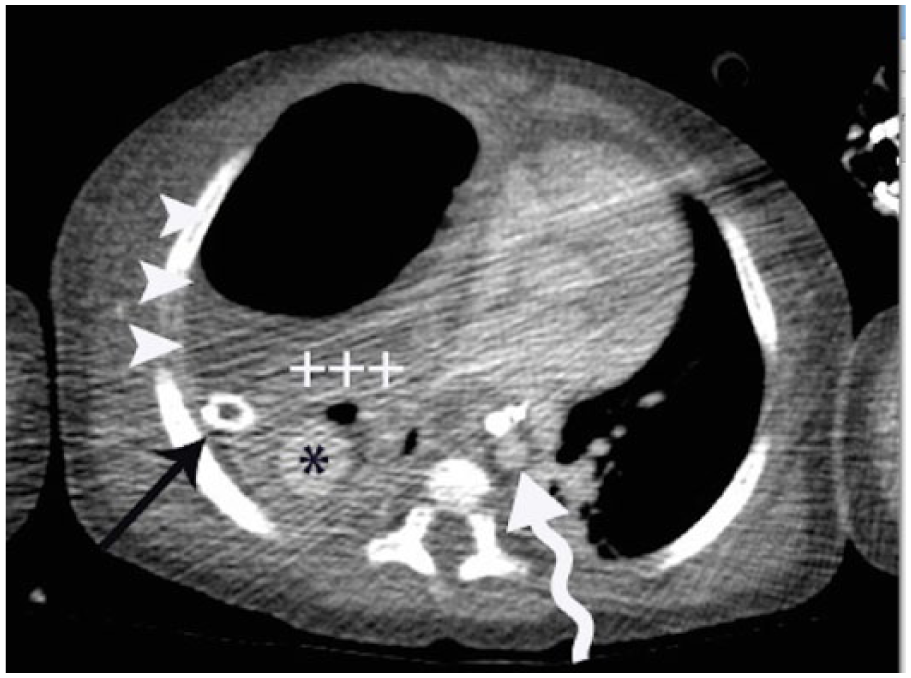
Axial contrast enhanced computed tomographic image of the chest showing a large hydropneumothorax (arrowheads). There is consolidated and necrotic parenchyma (plus signs) in the right lower lobe, within which there is a circumscribed hyperdensity (asterisk) with attenuation similar to that of the aorta (curved arrow). Straight arrow denotes presence of a chest tube.
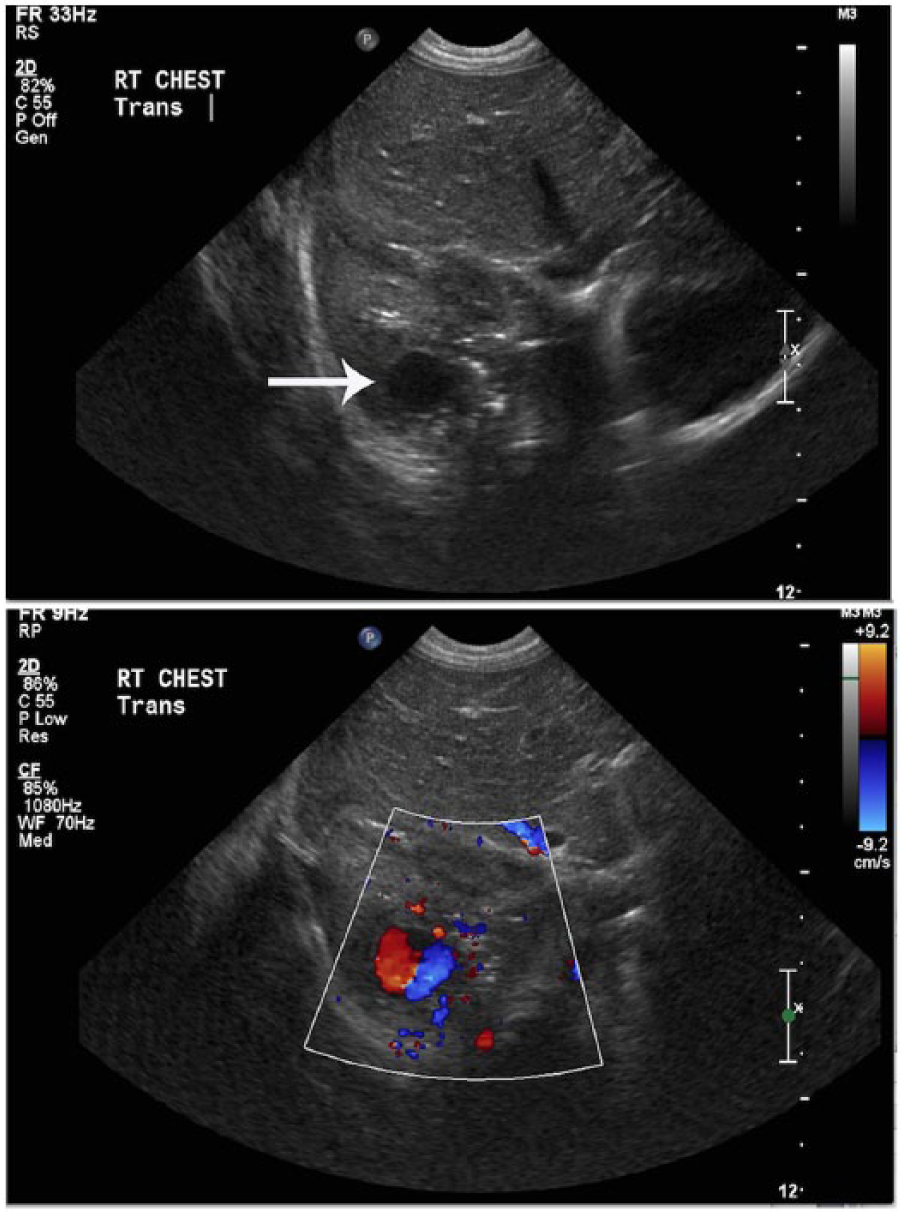
On the superior panel, 2D gray scale sonography demonstrates a hypoechoic lesion (arrow) corresponding to the lesion seen on computed tomography. On the inferior panel, color Doppler imaging shows the classic “yin yang” flow pattern characteristic of a pseudoaneurysm.
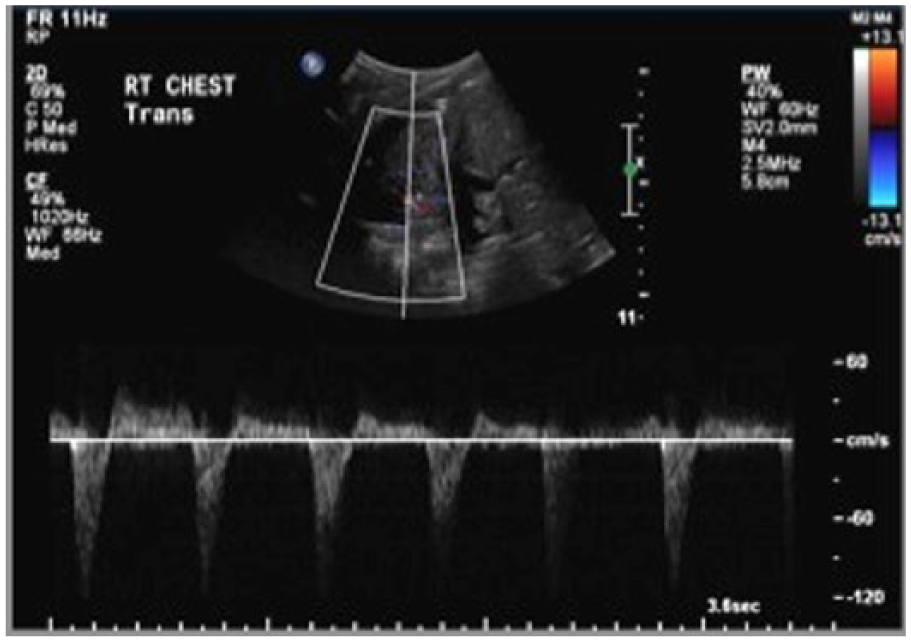
Spectral Doppler evaluation, with the sample volume placement fuided by color Doppler imaging, shows a bidirectional “to and fro” flow pattern characteristic of a pseudoaneurysm.
The patient was referred to interventional radiology for vascular embolization. After the lesion was depicted on pulmonary arteriography (Figure 4), the feeding vessel was subselected, and a vessel occlusion device was deployed. Post-embolization arteriography showed complete occlusion of the feeding vessel. A follow-up chest sonogram was done that confirmed resolution of the pseudoaneurysm (Figure 5).
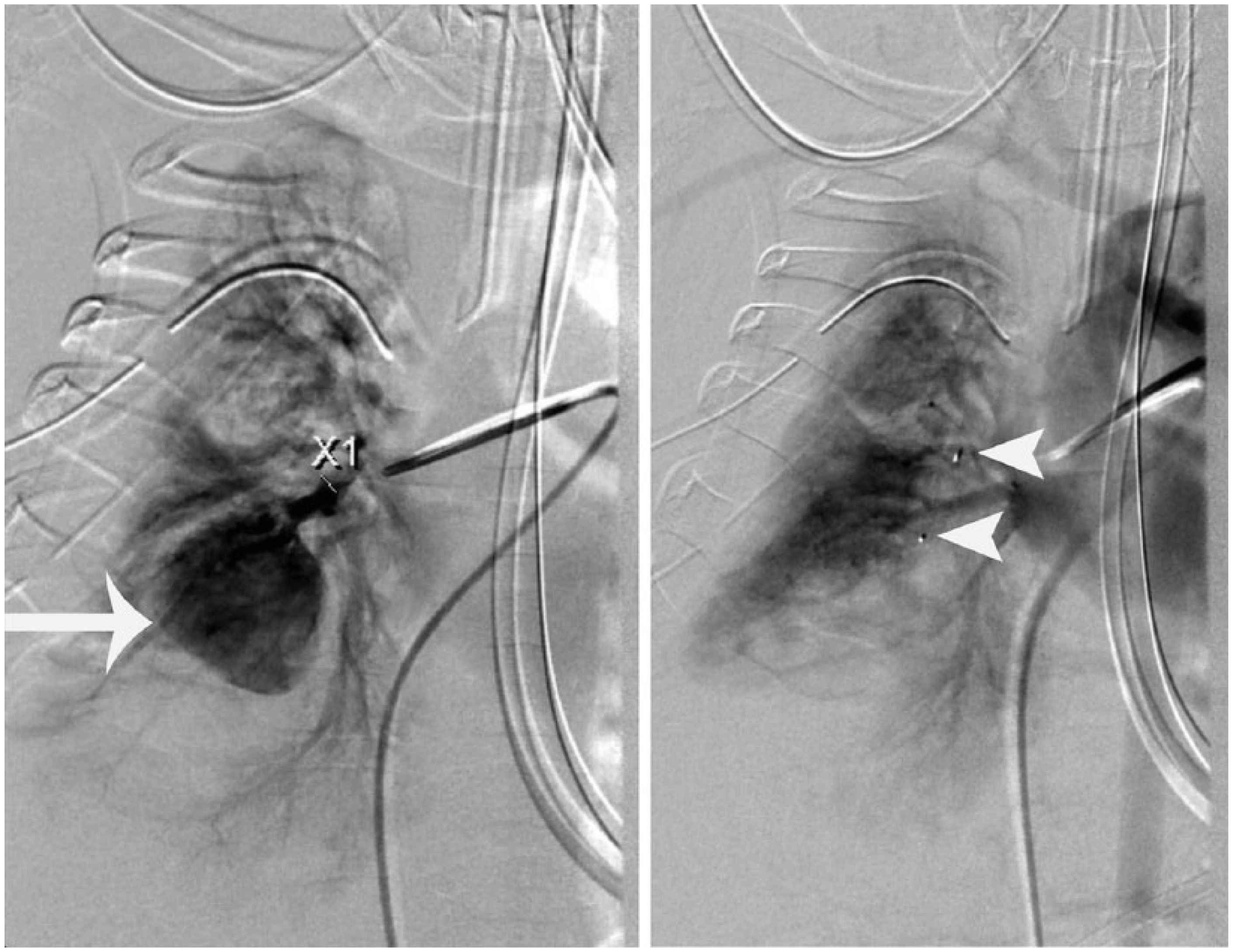
Digital subtraction angiographic image on the left shows contrast opacification of the pseudoaneurysm (arrow). Post-embolization angiographic image on the right shows that the pseudoaneurysm is no longer present. (Arrowheads point to embolization hardware.)
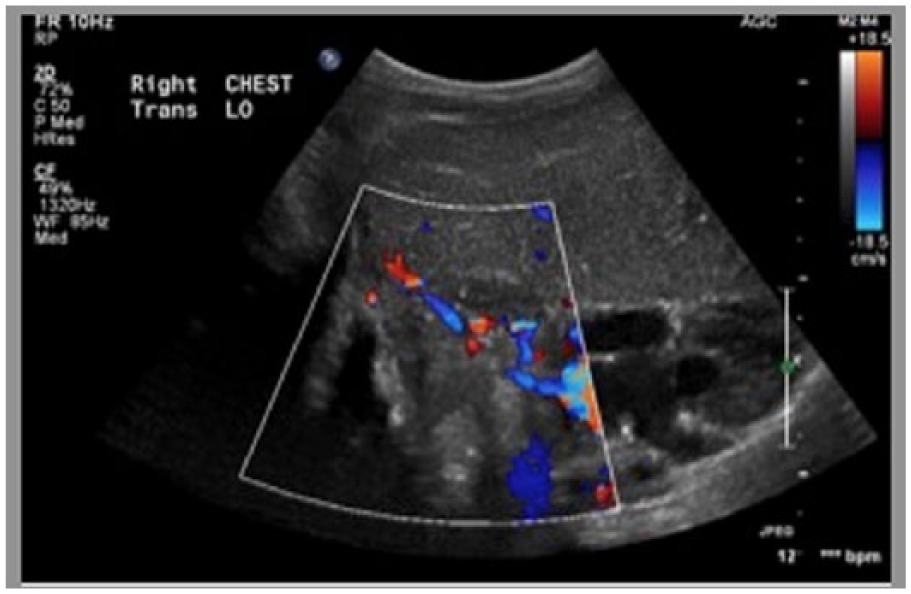
Color Doppler sonography following embolization of the area of the previously demonstrated pseudoaneurysm shows no similar flow abnormality.
Discussion
Pulmonary artery pseudoaneurysms are uncommon but are important to recognize because of high mortality related to rupture.1,4,7 In contrast to true aneurysms that involve dilatation of all three layers of a vessel wall, a pseudoaneurysm is essentially a hematoma resulting from disruption of at least one vascular layer, weakening the vessel wall and therefore at higher risk of rupture. This vascular disruption is usually acquired secondary to trauma (iatrogenic or otherwise), chronic inflammation, infection, or malignancy.3,7–10 With regards to infectious etiologies, historically tuberculous pulmonary artery pseudoaneurysms (Rasmussen aneurysms) have been described.
3
Non-tuberculous infections are reportedly a rarer cause of PAP but have been described more frequently in recent literature. Described pathogens include
Computed tomography angiogram with multiplanar reconstruction (MPR) has been advocated as the noninvasive modality of choice for diagnostic and therapy planning purposes.3,11 Computed tomography provides useful information regarding the size, location, and origin vessel of the pseudoaneurysm to allow for superselective therapy. Less described in literature, however, is the use of gray-scale, color Doppler, and spectral Doppler sonography to demonstrate expected pseudoaneurysm findings in PAPs, including the “yin-yang” sign and the typical “to-and-fro” spectral patterns. The presence of air generally limits the use of sonography in pulmonary evaluation; however, as shown in our case, targeted evaluation using these techniques can be helpful in problem solving. Use of color and spectral Doppler evaluation to aid in diagnosis of PAPs is especially important in pediatric patients, where minimizing radiation exposure is a strong consideration in choice of imaging. 7 Furthermore, ultrasound-guided localization for percutaneous embolization has been described in rare cases. 12
Currently, the treatment of choice for PAPs is by percutaneous endovascular embolization, either of the feeding vessel(s) if they can be identified or of the pseudoaneurysm sac itself.13–15 Endovascular embolization has proven to be a safe, minimally invasive, efficient, and cost-effective alternative to surgery. The procedure can be performed by selectively placing coils or plugs into or adjacent to the pseudoaneurysm. Alternatively, percutaneous embolization methods used include sclerosing agents (sobutyl-2-cyanoacrylate), absorbable gelatin sponges, or detachable balloons in the artery feeding the false aneurysm. 14
Conclusion
This case demonstrates a multimodality approach to the diagnosis and treatment of a pulmonary artery aneurysm, augmented by the use of sonography. While initial diagnostic approaches favor the use of computed tomography, we have described a case in which sonography, particularly color and spectral Doppler evaluation, proved to be an effective adjunct tool both to diagnose the initial finding and to confirm resolution after successful endovascular intervention.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
