Abstract
Vascular trauma can be a life-threatening event. Various complications can arise when injury to an artery occurs. Thus, early diagnostic imaging may be necessary for emergency treatment. A case study is presented of a delayed arterial injury, after a gunshot wound, involving the right upper extremity. This case demonstrates the importance of continued clinical surveillance and management. Duplex sonography has been shown to have 94% to 97% sensitivity and 82.5% to 98% specificity, regarding vascular injury. This case study describes the use of duplex sonography for the identification and clinical follow-up of multiple arterial injuries that presented 1 month after the initial traumatic incident.
Vascular trauma coincides with a higher mortality rate and increased use of medical resources, compared with patients without vascular injury. A majority of these traumatic incidents result from penetrating injuries to an extremity and have several potential complications. 1 However, not all arterial injuries occur at the time of trauma.1,2 While computed tomography angiography (CTA) is the preferred diagnostic imaging choice for the initial evaluation of vascular trauma, and arteriography is considered the gold standard, duplex ultrasonography (DUS) has proven to be a useful diagnostic tool for the identification and surveillance of vascular injuries, which has proven to have 94% to 97% sensitivity and 82.5% to 98% sensitivity.2 –5 This case study presents the identification of delayed arterial injury with subsequent clinical follow-up evaluation using DUS.
Case Report
A 45-year-old man presented to a vascular surgery clinic 1 month after a gunshot wound (GSW), to the right upper extremity and chest. Initial CTA at the time of traumatic incident demonstrated multiple metallic fragments within the upper arm. Both CTA and DUS were performed at an outside medical facility. The subsequent diagnostic reports described 50% to 75% focal narrowing of the lumen in the distal brachial artery, thought to be a possible dissection. DUS had also shown nonocclusive arterial thrombus in the mid and distal segments of the ulnar artery and nonocclusive venous thrombus, which was incidentally noted in a radial vein. No treatment was performed at that time, and the patient was referred to the vascular surgery clinic for clinical follow-up.
At the subsequent clinical visit, the patient complained of pain and numbness in the right forearm and hand. He said his fingers were cold and the first and second digits were constantly numb. One week prior, an enlarged pulsatile lump presented near the right antecubital fossa. An upper extremity arterial duplex was performed, and multiple echogenic foreign bodies were seen, within the soft tissue of the upper arm (see Figure 1). At the distal brachial artery, a pseudoaneurysm (PSA) measuring 6.5 × 5.1 × 4.5 mm was visualized with a 4.2-mm open wall defect (see Figure 2). Just distal to this area, a 5.9-mm slight protrusion of the arterial wall was visualized, with an absence of the intimal layer (see Figure 3). Inferior to this, near the antecubital fossa and at the location of the patient’s pulsatile lump, a 9.0 × 11.2 × 6.8-mm PSA or possible saccular aneurysm was visualized. The open wall defect measured 4.1 mm (see Figure 4). Opposite of this was a second 2.9-mm mild protuberance of the vessel wall with absent intima. The distal ulnar artery was found to be occluded with thrombus. Figure 5 demonstrates the locations of the arterial injuries along the brachial artery and their relation to each another.
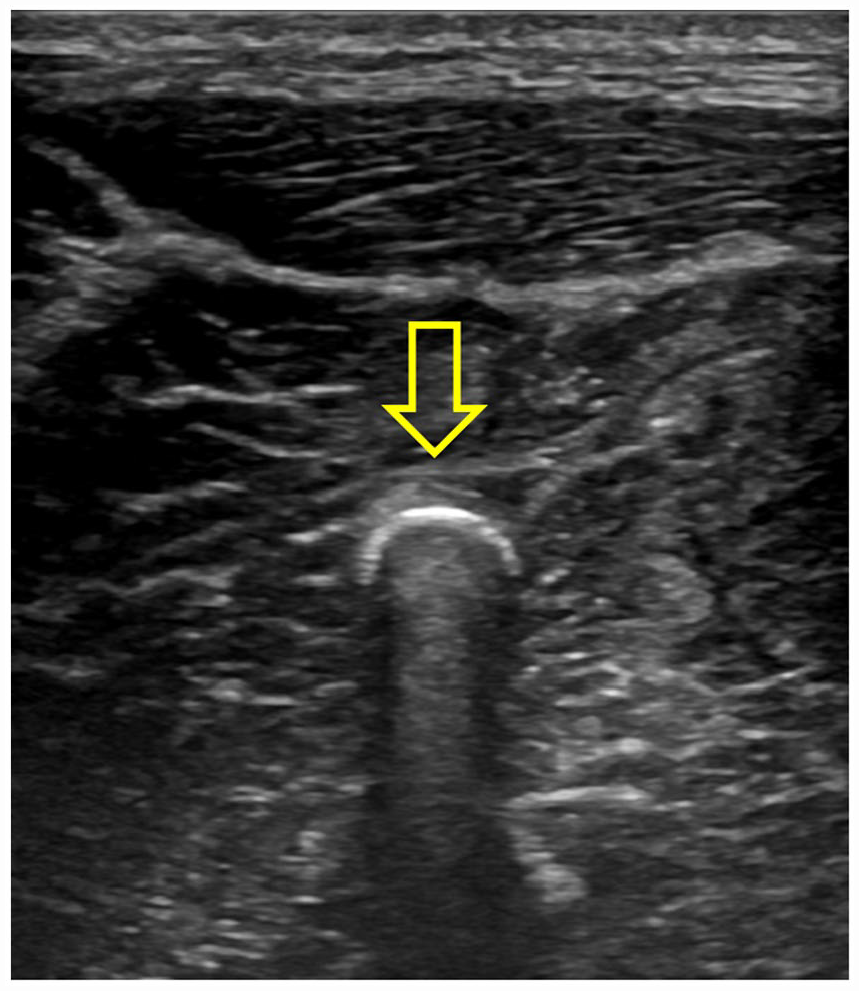
A sonogram of one of multiple echogenic shadowing pellets seen within the soft tissue of the patient’s right upper arm.
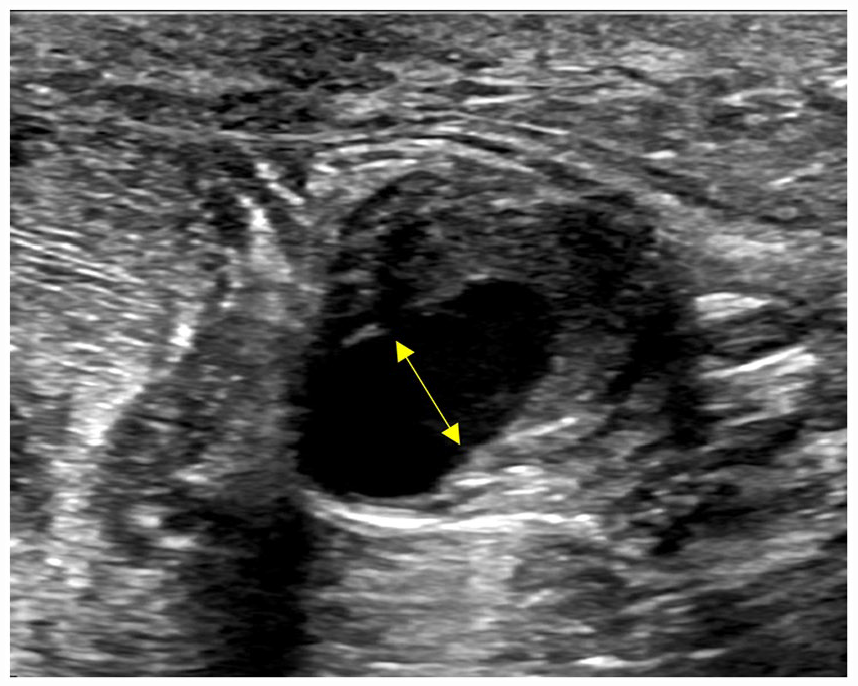
A transverse sonographic image of most superior pseudoaneurysm (1), 6.5 × 5.1 × 4.5 mm area of active flow with surrounding thrombus. The arrow demonstrates the 4.2-mm open wall defect.
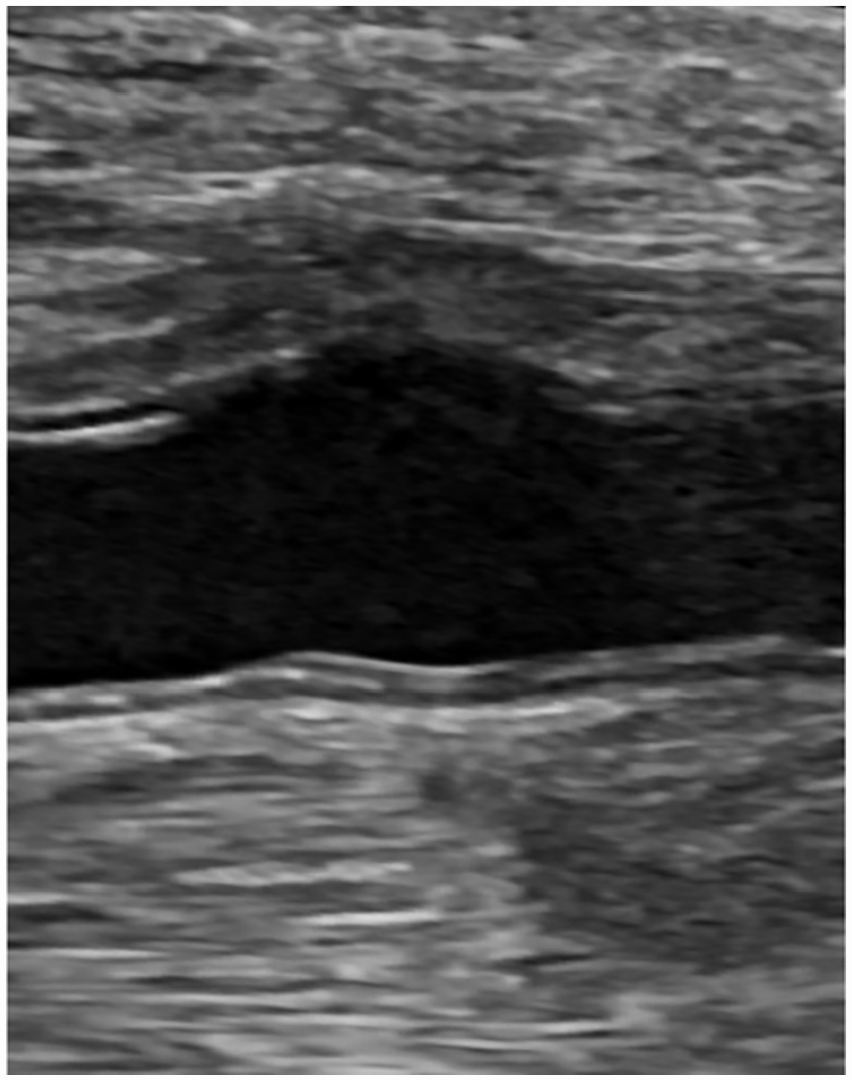
A longitudinal sonographic image of 5.9-mm mild protuberance of the distal brachial (2) demonstrating an absent intimal layer.
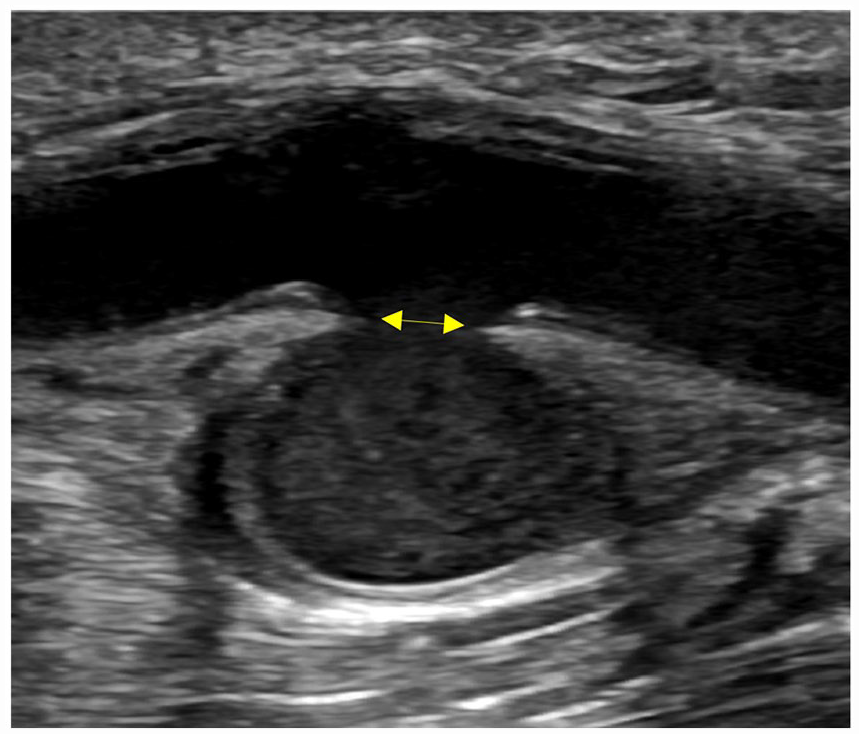
A longitudinal sonographic image of the arterial injury (3) at the pulsatile lump near the antecubital fossa, demonstrating a 9.0 × 11.2 × 6.8-mm pseudoaneurysm with a 4.1-mm open wall defect (see arrow provided).
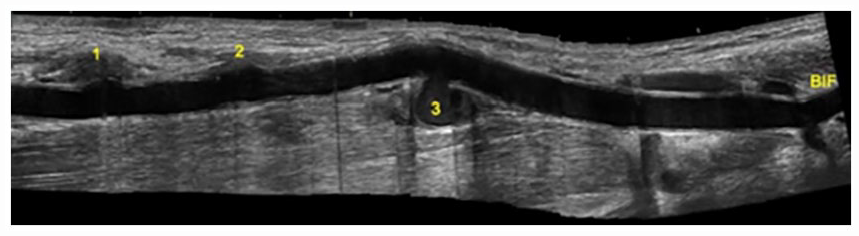
A longitudinal sonographic image of the right brachial artery during the initial examination at the host facility, which demonstrated the locations of the arterial injuries in relation to each other and the brachial bifurcation.
The patient was taken to surgery for repair of the injured brachial artery. Upon initial surgery, a PSA was seen adhered to an adjacent brachial vein. A segment of the brachial artery was removed, and an interposition graft was placed using the patient’s reversed greater saphenous vein. At the 7-week postoperative clinical follow-up, the patient complained of unchanged right forearm and hand numbness. On the current sonogram, the interposition vein graft was patent with elevated velocities at the proximal anastomosis and in the proximal segment, where a venous valve was located. The graft had replaced the most superior segment of the injured artery; however, the other injuries were still present. A new intimal flap was seen in the area labeled “2” (see Figure 6). The most inferior PSA vs saccular aneurysm (3) measured 10.4 × 9.8 × 7.5 mm. The change in size may have been attributed to a difference in measuring technique or change over time.
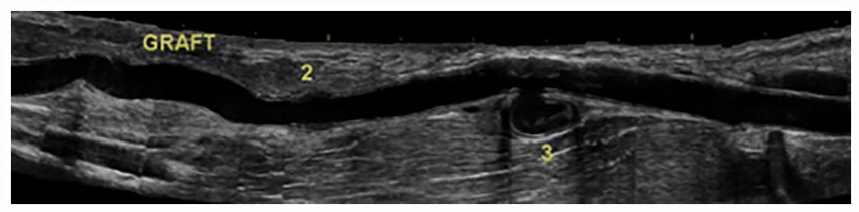
A longitudinal sonographic image of the right brachial artery after the interposition graft placement; a new intimal flap was seen at the area labeled “2,” and the remaining pseudoaneurysm (3) was resected during the second surgery.
The patient was again taken to surgery due to concerns for a mass effect of the remaining PSA causing his neurologic symptoms of right forearm and hand numbness. The brachial artery was exposed, and a PSA (3) was confirmed and resected. A 5-mm open wall defect was noted in the brachial artery, and a primary repair of the area was performed. The subsequent sonogram, after the second surgery, demonstrated successful repair of the PSA (3) and near-complete adherence of the previously visualized intimal flap (2).
Discussion
Dilatation of the arterial wall which includes all three layers is classified as a true aneurysm and is often a gradual process.6,7 In some cases of trauma, the arterial wall is bruised and weakened allowing for aneurysm formation. 8 GSW causes a temporary cavitation effect of the tissues around the high-velocity projectile which includes the surrounding vasculature.1,9 This can damage the arterial wall while leaving it intact, resulting in contusion, dissection, thrombosis, or vasospasm.1,9–11 Bulging of the arterial wall can occur with or after contusion and can mimic a PSA. 8 In this case, “saccular aneurysm” was given as a differential diagnosis for the inferior PSA (3), as platelet layering appeared similar to the vessel wall. Other potential complications of arterial trauma include complete or partial transection, as well as the formation of an arteriovenous fistula (AVF) or PSA.1,10,11
PSAs occur when blood flows outside of the artery through an open defect in the vessel wall (neck). This flow is not encapsulated by the arterial wall but contained in the surrounding soft tissue.6,7 While this can present with initial trauma, delayed development of a PSA also occur. 2 A large PSA can compress nearby vessels and nerves, causing onset or worsening of the patient symptoms. 1
Signs and Symptoms
Signs and symptoms of arterial injury are categorized as “hard” or “soft” diagnostic signs. Hard diagnostic signs include active arterial hemorrhage, shock, absent pulses, enlarging hematoma, and an active thrill. Soft diagnostic signs of arterial injury include a history of arterial bleeding at the site of trauma, diminished pulses, and trauma near an artery. Neurologic deficits in the area of injury are another soft diagnostic sign and should be carefully evaluated, as this is considered an early sign of vascular injury.1,10–12
Treatment
Treatment for arterial injuries varies based on the abnormality. Grossly damaged segments of an artery are resected, and the uninjured ends are then anastomosed. If the removed injured portion is too long to allow for a tension-free end-to-end anastomosis, an interposition graft can be placed. Autogenous grafts, such as the patient’s saphenous vein, are most commonly used. 1
Treatment for PSA is modified based on the diameter of the PSA and the length of the neck. It is possible for a PSA less than 2–3 cm to thrombose spontaneously; however, a larger PSA requires further action.1,13 Sonography-guided compression or thrombin injection are potential treatment options, though injection is not recommended for PSA less than 1 cm or with a neck less than 2 mm in length, due to the risk of arterial embolization. 13 Endovascular techniques include transcatheter embolization and stent-graft placement. 1 Patch angioplasty or primary repair may be necessary for large wall defects. 1 Left untreated, arterial injuries can lead to limited function or loss of the affected extremity. 10 Interposition vein graft placement demonstrates the best outcome for limb salvage after arterial injury in an extremity. 1 One study found short-term and long-term patency of these grafts as 93% and 97.6%, respectively. 14
Diagnostic Imaging
Arteriography is the gold standard for the evaluation of vascular injuries. However, there are other imaging options such as CTA, magnetic resonance angiography (MRA), and DUS. 1 In 2014, a study found that DUS had a 94% sensitivity and 82.5% specificity for the diagnosis of vascular injury. 3 Other studies have described 95% to 97% sensitivity and 95% to 98% specificity. 5 DUS not only allows proper evaluation of the injured limb but is an inexpensive and less invasive option than other modalities. 1
The sonographic protocol used to follow-up a right upper extremity arterial injury included grayscale, color, and Doppler evaluation of the innominate, subclavian, axillary, brachial, radial, and ulnar arteries. A limited evaluation of the veins in the area was also performed to document normal venous flow and rule out an AVF. Due to the proximity of the GSW, the axillary, brachial, and proximal radial and ulnar arteries were thoroughly evaluated in transverse. This technique allows full visualization of the arterial wall and aids in identifying abnormalities that can originate from a sidewall and may not be seen in the longitudinal plane (see Figure 7A and 7B). Transverse evaluation also aids in finding the best approach to elongate an abnormality that is not well seen in the typically used longitudinal sonographic window (see Figure 8).
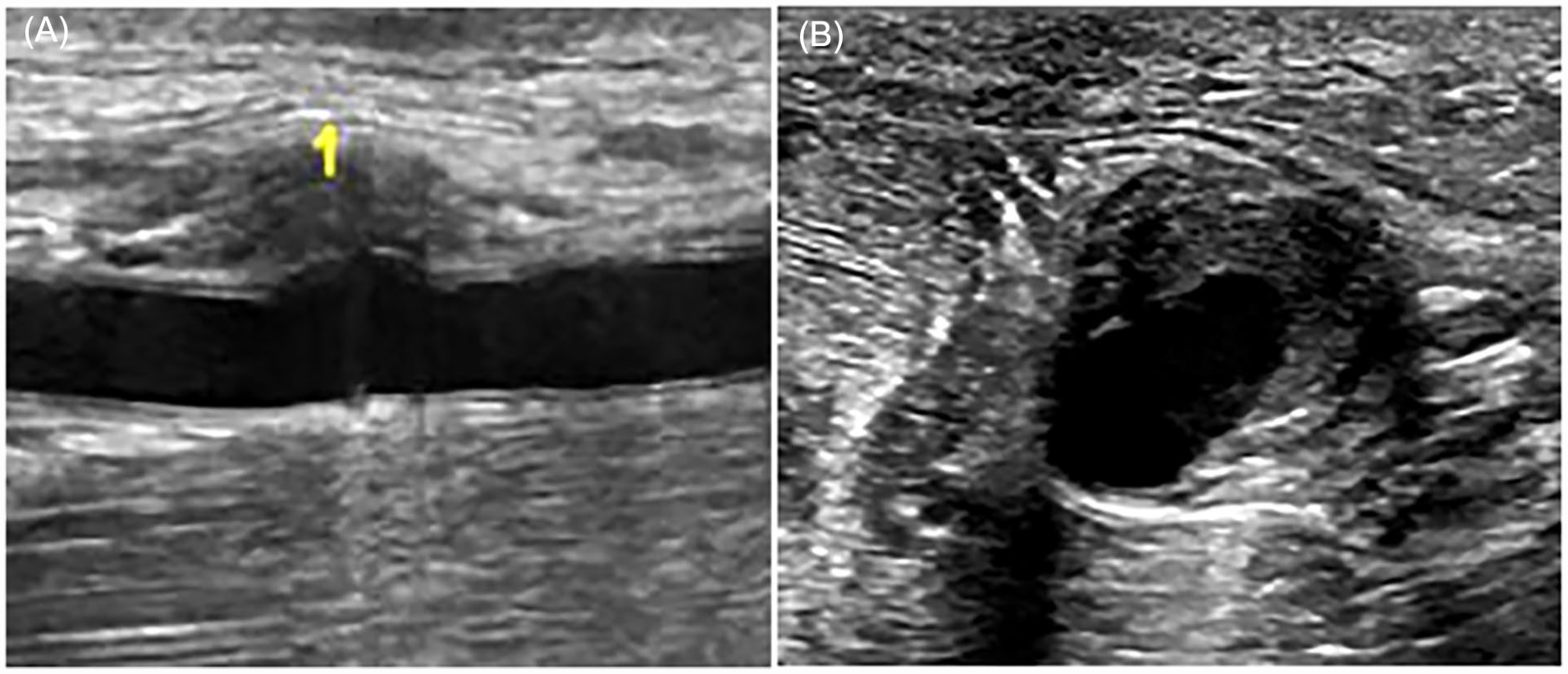
(A) A selected sonographic image from examination completed at the vascular surgery clinic showing the subtle appearance of the most superior PSA (1) in the longitudinal plane. (B) A comparison sonographic image of the most superior PSA (1) demonstrating its more obvious appearance in the transverse plane. PSA, pseudoaneurysm.
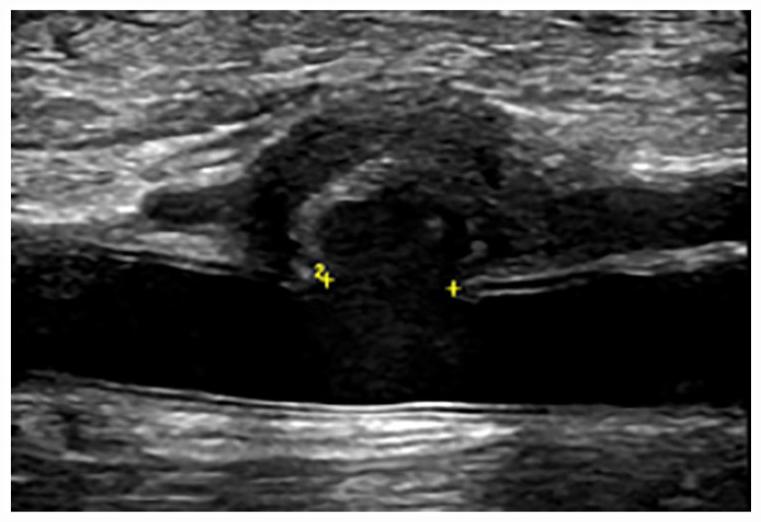
A pseudoaneurysm (1) elongated from a more posterior approach after visualization in transverse plane. Electronic calipers demonstrate the wall defect (neck) and measured 4.2 mm.
All PSAs were documented for size and a “ying-yang” sign was demonstrated with color flow when possible (see Figure 9). The size of the open wall defects was measured; however, the PSA in this case did not have a distinct neck, as the bleeding from the artery was contained directly adjacent to the vessel. This was noted in the “Sonographer’s Findings” on the preliminary report to allow for proper treatment decisions. The large open wall defects in this case minimized the classic to-and-fro flow that is commonly seen in the neck of a PSA (see Figure 10). 4 Serial surveillance imaging included these same DUS techniques to assess for variation of residual abnormalities and patency of repairs. Arterial wall irregularities such as dissections and abnormal wall contour should be thoroughly documented to evaluate for change over time (see Figure 11A–11C).
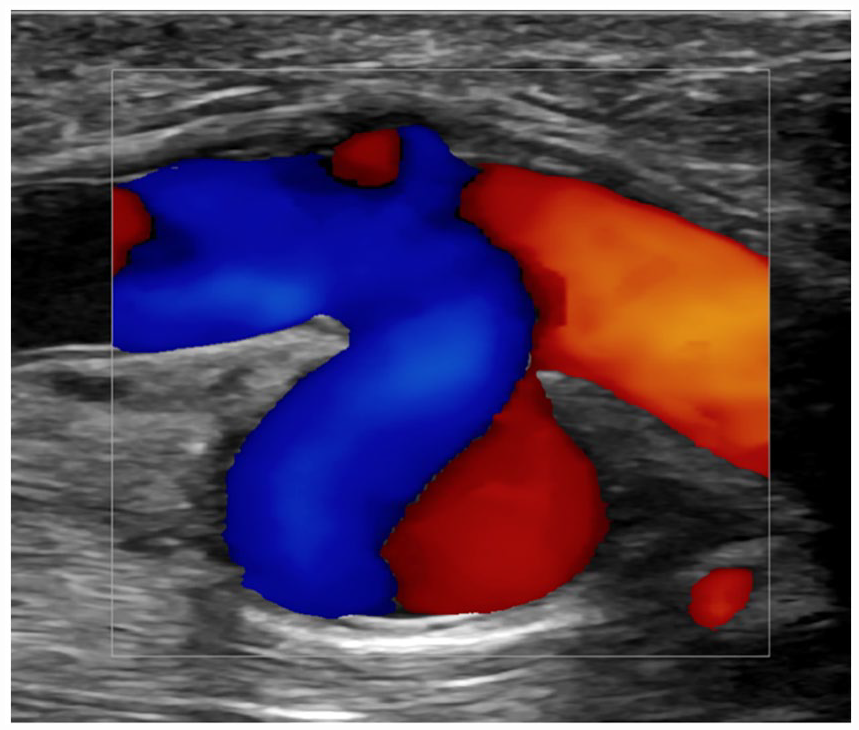
A color Doppler image that demonstrated the Ying-Yang diagnostic sign within a pseudoaneurysm (3) and a small protrusion of directly adjacent arterial wall.
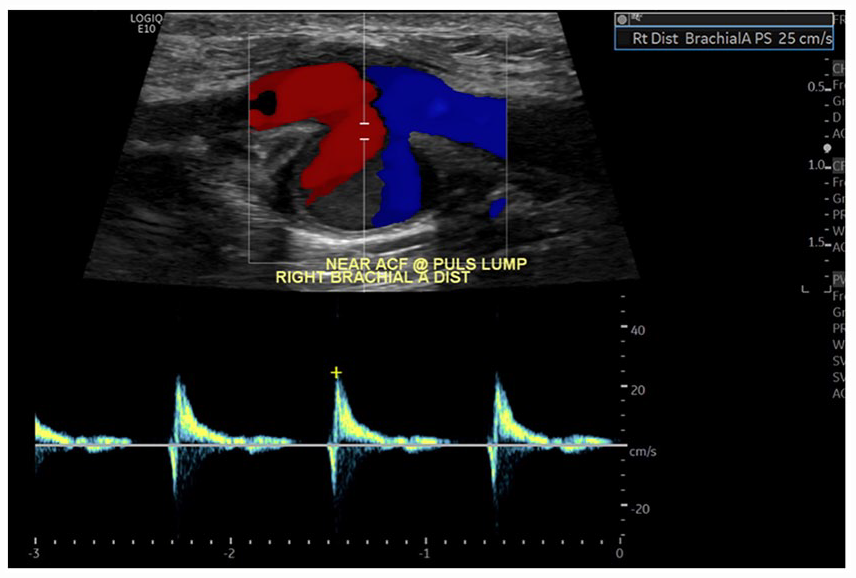
The spectral waveform analysis of the most inferior pseudoaneurysm (3) which demonstrated minimal to and fro flow, due to a large vessel wall defect.
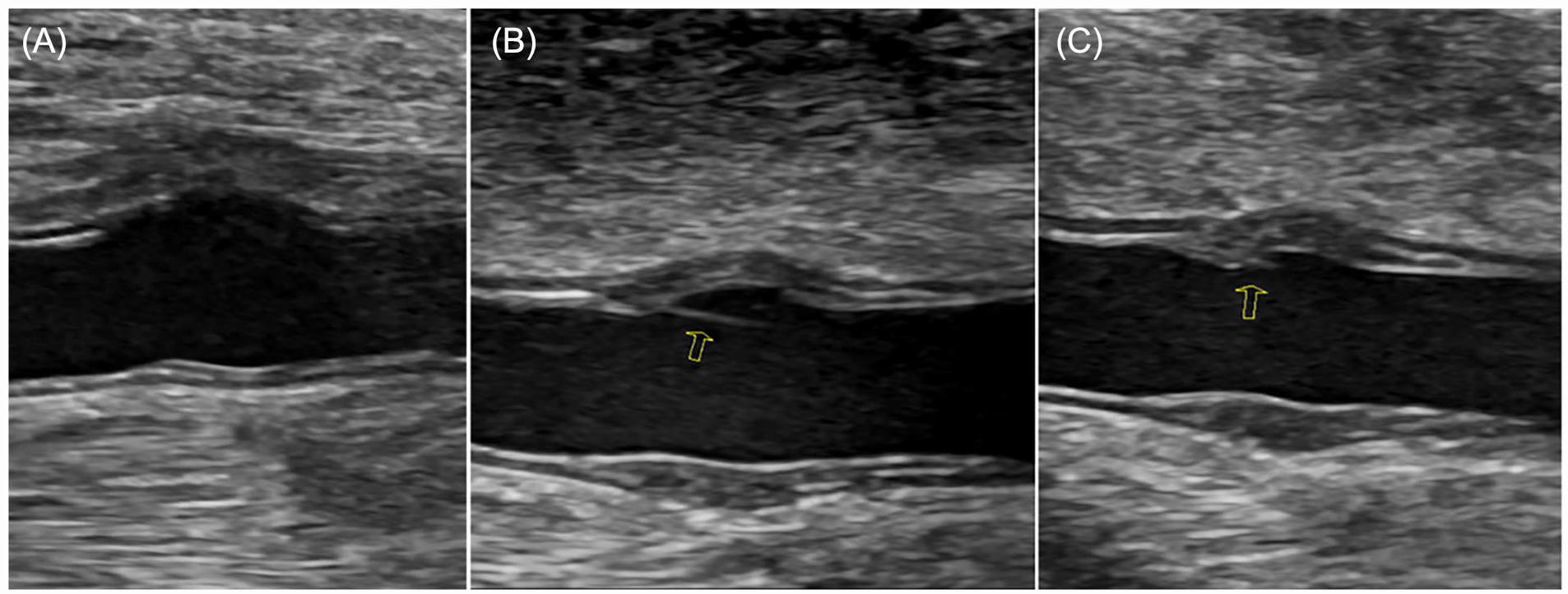
(A) An arterial injury (2) captured on the first DUS performed at the vascular surgery clinic, which demonstrated an abnormal wall contour and lack of the tunica intima. (B) Arterial injury (2) at the second DUS, 7 weeks later, showing an intimal flap in the same area. (C) An arterial injury (2) documented at the third DUS examination and documented a partial adhesion of the previous intimal flap. DUS, duplex ultrasonography.
Conclusion
Failure to diagnose and treat arterial injury risks further complications. However, obvious irregularities may not present until after the initial trauma takes place. The occurrence of delayed vascular injuries demonstrates the importance of clinical follow-up imaging. Thorough examination with duplex sonography can aid in the diagnosis of arterial injury development after trauma. As shown in this case report, duplex ultrasound is a noninvasive technology that can be utilized to serially monitor subsequent arterial injuries and can be more cost effective than other diagnostic imaging choices.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not sought for the present study becuase only anonymous information was used in this report.
Informed Consent
Informed consent was not sought for the present study because all case data were de-identified and/or aggregated and followed ethics committee or IRB guidelines (also referred to as the Honest Broker System).
