Abstract
Uterine artery pseudoaneurysm is an uncommon vascular lesion that can be life threatening. A ruptured uterine artery pseudoaneurysm can cause acute massive hemorrhaging, creating a true emergency for the postpartum patient. The etiology is more commonly seen with traumatic procedures such as cesarean delivery or curettage. This case report is of a patient with a history of HELLP (hemolysis, elevated liver enzymes, low platelet count) syndrome who presented with heavy bleeding after a cesarean delivery. Sonography was primarily used to diagnose the uterine artery pseudoaneurysm. Because of the massive hemorrhaging in this case, a hysterectomy was performed.
Uterine artery pseudoaneurysm (UAP) is an uncommon vascular pathology with an incidence of 0.3% to 0.6%.1,2 It forms from a defect in the artery through which blood leaks and creates a hematoma that remains in communication with the uterine artery. A UAP is a complication usually due to vascular trauma during vaginal delivery, cesarean delivery, curettage, or myomectomy, but it has also been associated with endometrial or cervical cancers and myomas.3,4 It is life threatening because of the potential of rupture and subsequent hemorrhaging. Postpartum hemorrhage accounts for 15% of maternal deaths, so identifying the underlying cause is important for proper patient management. 5 While angiography is the gold standard, sonography is a primary tool to assess the pelvis and can visualize a pseudoaneurysm when present. Because acute massive hemorrhaging has been documented when a pseudoaneurysm ruptures, it should be considered an obstetric emergency. 6
Case Report
A woman in her early 20s was diagnosed with HELLP syndrome, or hemolysis, elevated liver enzymes, and low platelet count. A cesarean delivery at full term was performed because of complications related to the HELLP syndrome. Her recovery was routine, and she was released within one week of delivery. She then presented one month later to the emergency department with heavy postpartum bleeding and severe anemia. The patient was admitted, and three units of blood was transfused that evening. A pelvic sonogram was ordered for the following morning.
The sonogram showed a slightly enlarged uterus with heterogeneous myometrium. An anechoic to hypoechoic lesion in the lower uterine segment measured 4.8 cm × 3.5 cm × 4.1 cm (Figure 1a and b). Turbulent blood flow within the lesion was noted with color Doppler (Figure 2). It contained heterogeneous debris likely representing thrombus formation. There was slightly heterogeneous, hypoechoic fluid within the endometrial canal, possibly reflecting liquefied hematoma. The ovaries were unremarkable and within normal range. No free fluid was present. The lesion was identified as a UAP.
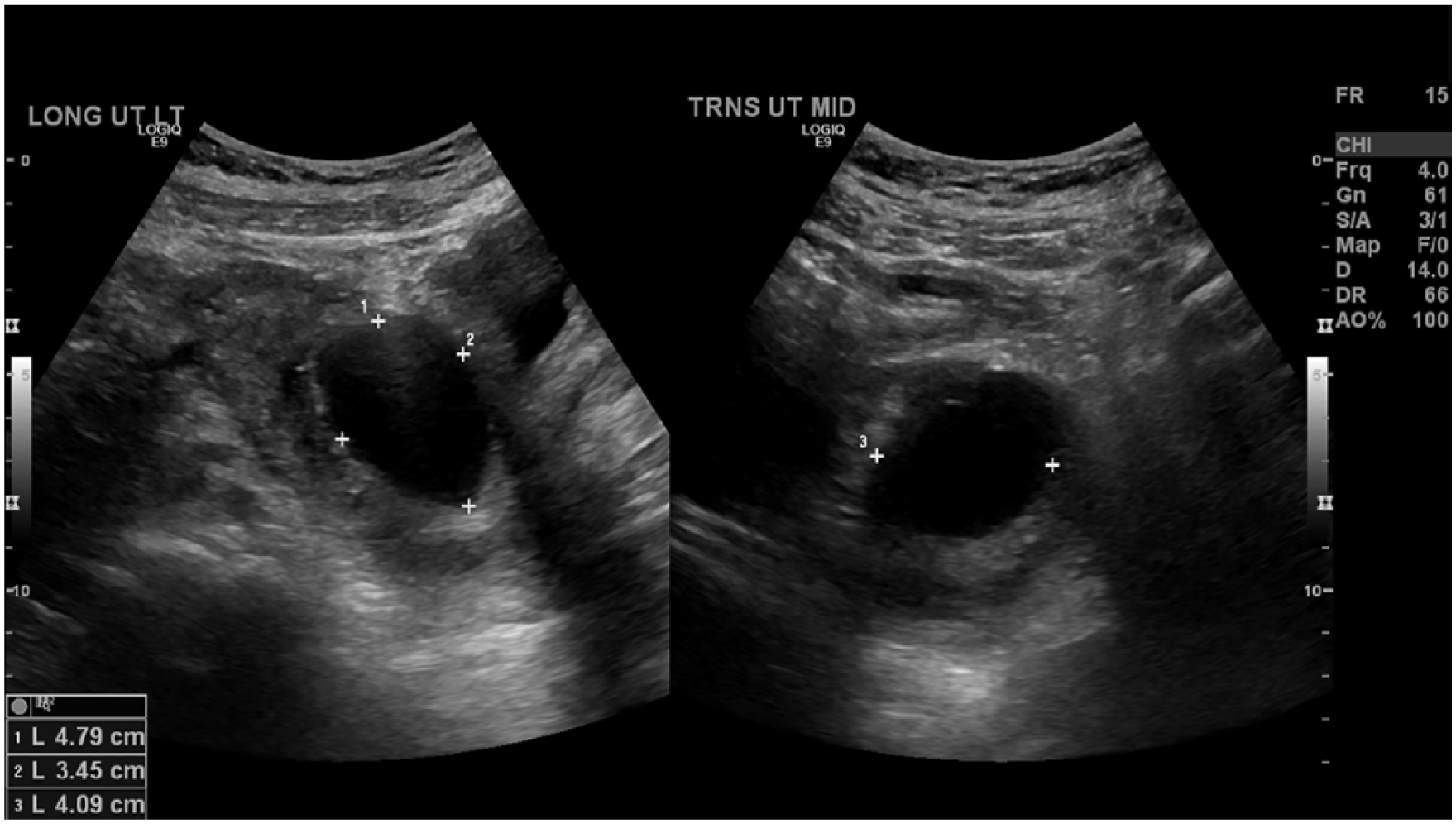
Longitudinal (A) and transverse (B) images demonstrating anechoic structure within the uterus.
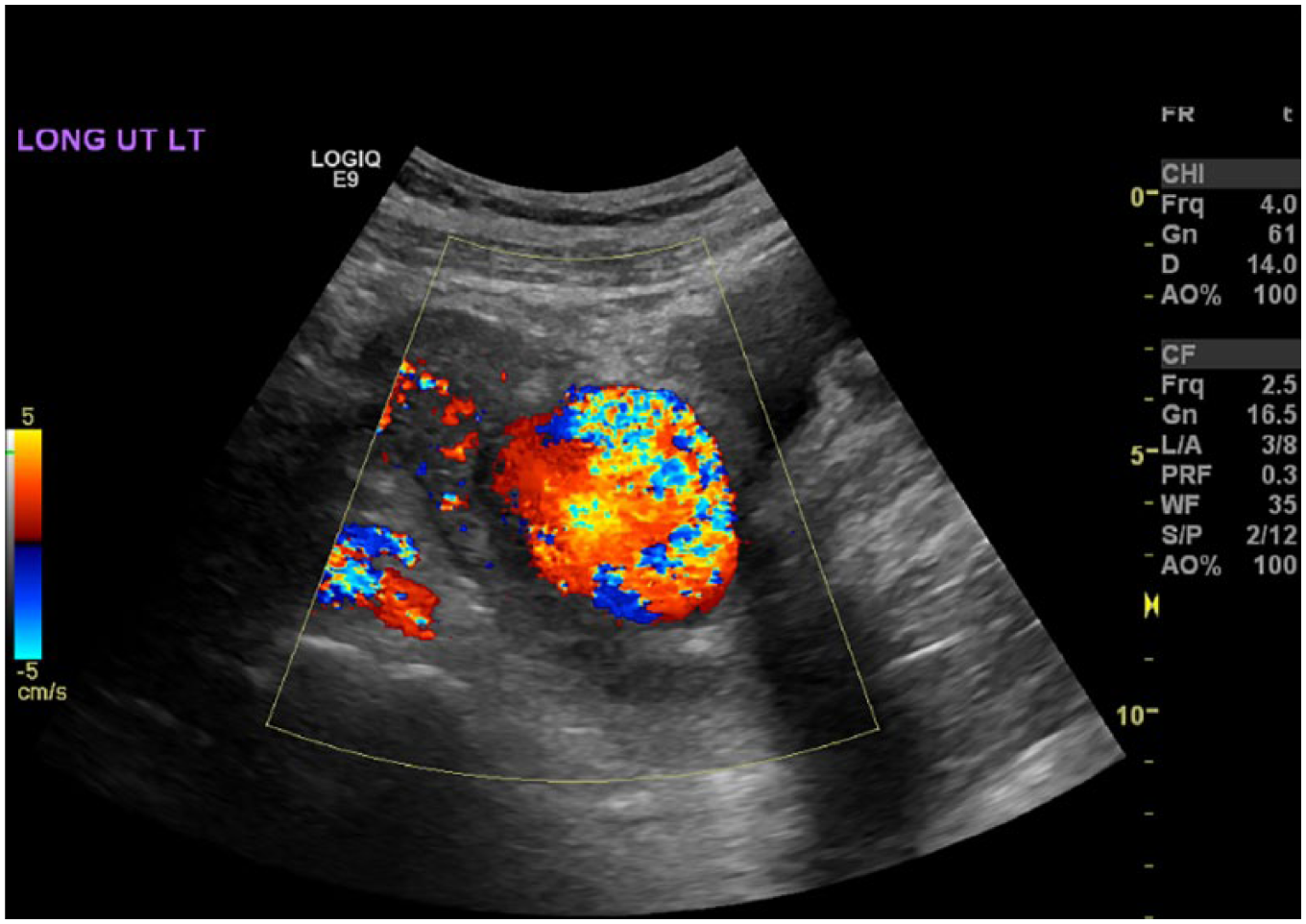
Swirling blood flow within the lesion documented with color Doppler.
The patient was counseled and treatment options given. The patient chose to be transferred to a tertiary hospital to have a transcatheter arterial embolization procedure. Prior to being transferred, the patient had an acute onset of massive vaginal hemorrhaging with a vagal response demonstrating dizziness, tunnel vision, and pallor. An immediate hysterectomy was performed. The patient was stabilized and recovered without being transferred. Four days later, she remained stable with laboratory values returning to normal and was released.
Discussion
Postpartum hemorrhage is a common cause of maternal mortality worldwide and is one of the leading causes of postpartum hysterectomy.6,7 Secondary postpartum hemorrhage is blood loss greater than 500 mL between 24 hours to 12 weeks after delivery. 7 Cesarean delivery is the most likely cause of postpartum hemorrhage, accounting for 47% of cases. 8 Other pathologies that may cause postpartum bleeding include retained products of conception, endometritis, placental bed subinvolution, arteriovenous malformation, pseudoaneurysm of the uterine artery, choriocarcinoma, and cesarean scar dehiscence.6,9 In general, the common causes of hemorrhage can be summarized by the 4Ts: tone, trauma, tissue, and thrombin. The most prominent of these factors is the loss of uterine tone. Atony is poorly contracted uterine musculature and may be responsible for 80% of postpartum hemorrhage cases.10,11 The incidence of postpartum hemorrhage due to atonia has increased in the past two decades, but the increase cannot necessarily be associated with a specific cause. 12 Trauma to the uterus could include inversion, episiotomy, lacerations, or rupture, especially during a cesarean delivery. Tissue refers to retained products of conception, and thrombin references different types of coagulopathies that consist of various blood clotting disorders such as HELLP syndrome, as was the condition in this case report.
HELLP, which stands for hemolysis, elevated liver enzymes, and low platelets, manifests as a severe form of preeclampsia with additional laboratory abnormalities. Hypertension and proteinuria are seen along with hemolytic anemia, thrombocytopenia, and abnormal liver function tests. 13 These components trigger the release of factors causing platelet activation, vasoconstriction, and loss of normal vascular relaxation during pregnancy. 13 The reaction can injure vessel endothelium, weakening the vessel wall. Trauma to the uterine artery during cesarean delivery may also cause weakening or tearing in the vessel wall, allowing blood to leak into adjacent tissue, forming a pseudoaneurysm. Usually, a UAP is detected when a postpartum patient reports vaginal bleeding, which relates to a delayed rupture. It is important to note that 22% of UAPs may be ruptured without active vaginal bleeding, so a complete patient history should be obtained beyond acute symptoms. 1 Detection rates have increased in recent years likely because of a corresponding increase in cesarean delivery rates but also in part to detection from improved imaging technology.1,2
A sonogram and a complete blood workup with a comparison of fever and tenderness are initially performed to diagnose a UAP. Sonographically, a pseudoaneurysm appears as an anechoic or a hypoechoic mass usually in the lower uterine wall. 14 There may be debris within it representing thrombosed blood. It demonstrates turbulent blood flow with color filling that swirls, often in a “yin-yang pattern,” as blood from the artery enters or leaves the pseudoaneurysm. 15 The Doppler flow is likely to alias and be bidirectional with a to-and-fro pattern 95% of the time.16,17 Using Doppler helps improve the diagnostic specificity and rules out differentials such as retained products of conception. 18 Sonography can show blood flow patterns and assess wall thickness of the pseudoaneurysm. Three-dimensional (3D) sonography may play a role to better define the lesion in relationship to cesarean scar and to obtain an accurate volume of the lesion. 19 Reconstruction of 3D Doppler data sets may provide different views of the aneurysm, offering more precise analysis of the vascular anatomy. 19 Differential pathologies such as hypermenorrhea, endometritis, and retained products of conception appear similar to a UAP sonographically and can lead to misdiagnosis. 20
Follow-up computed tomography (CT) is used to confirm the diagnosis identified on the sonogram. Pseudoaneurysms of the uterine artery as small as 2 cm have been identified on CT, confirmed by visualizing contrast media leakage from the parent artery. 21 An active bleed may be documented by performing a two-phase CT scan using contrast media, which shows dilatation in the arterial phase and enhancement of the hematoma surrounding the vessel during the venous phase—both of which indicate an active bleed.22,23 While CT is useful in identifying and diagnosing UAP, percutaneous angiography is considered the gold standard to diagnose a pseudoaneurysm and plays a role in the treatment as well. 24 When CT is equivocal, an angiogram can be used to better delineate a UAP from the differentials of arteriovenous malformation and cesarean scar dehiscence. A UAP appears as a saclike structure within the uterus with a connection to the uterine artery.23,25,26
Conservative treatment may be to monitor the pseudoaneurysm if it has not ruptured, because pseudoaneurysms have been reported to resolve spontaneously. 27 Rupture is prone to occur if the UAP diameter exceeds 2 cm. 28 Clinical treatments include transcatheter arterial embolization (TAE), uterine artery ligation, ultrasound-guided thrombin injection, and hysterectomy. TAE has become the preferred treatment method because it is minimally invasive, highly effective with few side effects, and preserves fertility.15,24 Uterine artery embolization blocks the blood supply to the pseudoaneurysm by using angiography/fluoroscopy to guide a catheter into the vessel that feeds the pseudoaneurysm. Particles are released from the catheter and travel to block the blood flow to that artery. TAE has been reported to be 97% effective. 29
Other treatment options include uterine artery ligation and percutaneous thrombin injection. Uterine artery ligation is performed by laparotomy and is sometimes used to control uterine bleeding while other interventions are being attempted. A suture is placed through the lower uterine segment near the cervix and circled through the broad ligament. It is tied to compress the ruptured vessels. Bilateral ligation has been reported to control hemorrhaging in 90% of cases30,31 but has had varied success rates from 42% to 100%, likely because of extensive pelvic collateral circulation. 32 Subsequent pregnancies have been reported successful with this treatment.31,33 The overall ultrasound-guided thrombin injection success rate is 97% effective and involves thrombin being injected directly into the pseudoaneurysm. 34 A needle is placed into the pseudoaneurysm using real-time two-dimensional imaging, and the thrombin is injected. The thrombin will seal the pseudoaneurysm defect site, blocking the pseudoaneurysm’s access to the artery. The pseudoaneurysm is broken down and absorbed over time. This option can preserve fertility. 35 If adequate hemostasis cannot be achieved, a hysterectomy may be performed. It may also be used as a primary treatment in cases in which preservation of fertility is not a factor.
Conclusion
UAP can be a life-threatening complication after a gynecologic or obstetric procedure, when massive hemorrhaging occurs days to weeks after the procedure. Sonography plays a primary role in postpartum imaging. Identifying the turbulent flow on Doppler along with the two-dimensional anechoic-hypoechoic features is essential to help differentiate UAP from other pathologies. CT and angiography can help differentiate when sonography is equivocal. If the pseudoaneurysm ruptures and massive hemorrhaging begins, treatment preferences become limited, and fertility preservation may not be an option.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
