Abstract
Skene’s glands are glands situated in the caudal two-thirds of the urethra. Infection of the gland by bacteria leads to inflammatory obstruction of the paraurethral ducts, causing the formation of a cyst or abscess. Patients typically present with dysuria, dyspareunia, vaginal discharge, and pain. On physical examination, a palpable mass is present and purulent material can be expressed from the ductal orifice. Clinical assessment of women with paraurethral gland symptoms is difficult, necessitating further evaluation with imaging. Recent advances in sonographic and magnetic resonance imaging have dramatically improved evaluation of the female urethra and the paraurethral glands, clarifying findings at physical examination and providing accurate road maps for surgeons. Even though magnetic resonance imaging is the gold standard, transperineal sonography is an emerging imaging technique that is being used to evaluate female urethral and periurethral disease in a more cost-effective manner.
Introduction
Despite Skene’s glands being first described in the mid-17th century and named after the Scottish gynecologist Alexander Skene in 1880, little is known about their function and the potential complications associated with them. 1 Skene’s glands are glands situated in the caudal two-thirds of the urethra, mainly in the dorsal and lateral mucosal stroma. 2 In some cases, these glands extend to the smooth musculature of the septum urethrovaginale. They are tubuloaveolar formations on long ductal structures and resemble the male prostate gland prior to puberty and androgenic stimulation. They contain prostate-specific antigen and prostatic acid phosphatase on immunostaining.2–5 Cysts of the gland are uncommon, and when infection occurs, it can cause dysuria, dyspareunia, vaginal discharge, and pain.6–8 On physical examination, a palpable mass is present next to the distal urethra, and purulent material can be expressed from the ductal orifice. 4 It has been suggested that Skene’s gland infection is more common than previously appreciated. We report a case using transperineal sonographic imaging to confirm the diagnosis and to help formulate an appropriate treatment plan.
Case Report
A woman in her early 20s presented to the emergency department complaining of pelvic pressure, vaginal pain, and vaginal discharge. Her symptoms had been continuous for three days. She had no personal history of an abnormal pap smear. Previously, the patient had been seen at an outpatient doctor’s office, and the diagnosis was suspected to be urethritis, gonorrhea, and/or chlamydia. At that time, she was given the oral antibiotic levofloxacin (Levaquin) and an intramuscular injection of the antibacterial drugs azithromycin and ceftriaxone (Rocephin). On her current visit, she stated her symptoms were unresolved. Vaginal examination showed a 2-cm swollen area visualized on the left side lateral to the urethra. Yellow-colored discharge was seen, and the swollen area was nontender upon palpation. A urinalysis, gonorrhea and chlamydia cultures, and a complete blood count (CBC) were done. The gonorrhea and chlamydia culture results were negative. The CBC revealed an elevated white blood cell count, especially neutrophils. An abscess was suspected, and intravenous antibiotics were initiated. The patient was admitted into the hospital, and a transvaginal sonogram was ordered.
Transvaginal sonography of the pelvic anatomy was performed with a GE LOGIQ E9 system (GE Healthcare-Ultrasound, Waukesha, WI) using a 5- to 9-MHz bandwidth intracavitary probe. The transvaginal examination showed that the pelvic organs measured within normal limits. There were no pelvic masses or free fluid, and the ovaries demonstrated normal blood flow. Transperineal sonography was then done using a 9-MHz linear array transducer. The transperineal examination showed a 1.6-cm complex, septated fluid collection located along the midline of the perineum anterior to the Foley catheter (Figure 1). On color Doppler imaging, the complex fluid collection demonstrated a small trace of vascularity around the periphery of the structure (Figure 2). Based on the patient’s symptoms, along with the results of the CBC, physical examination, and sonogram, Skene’s gland abscess was diagnosed and treated.
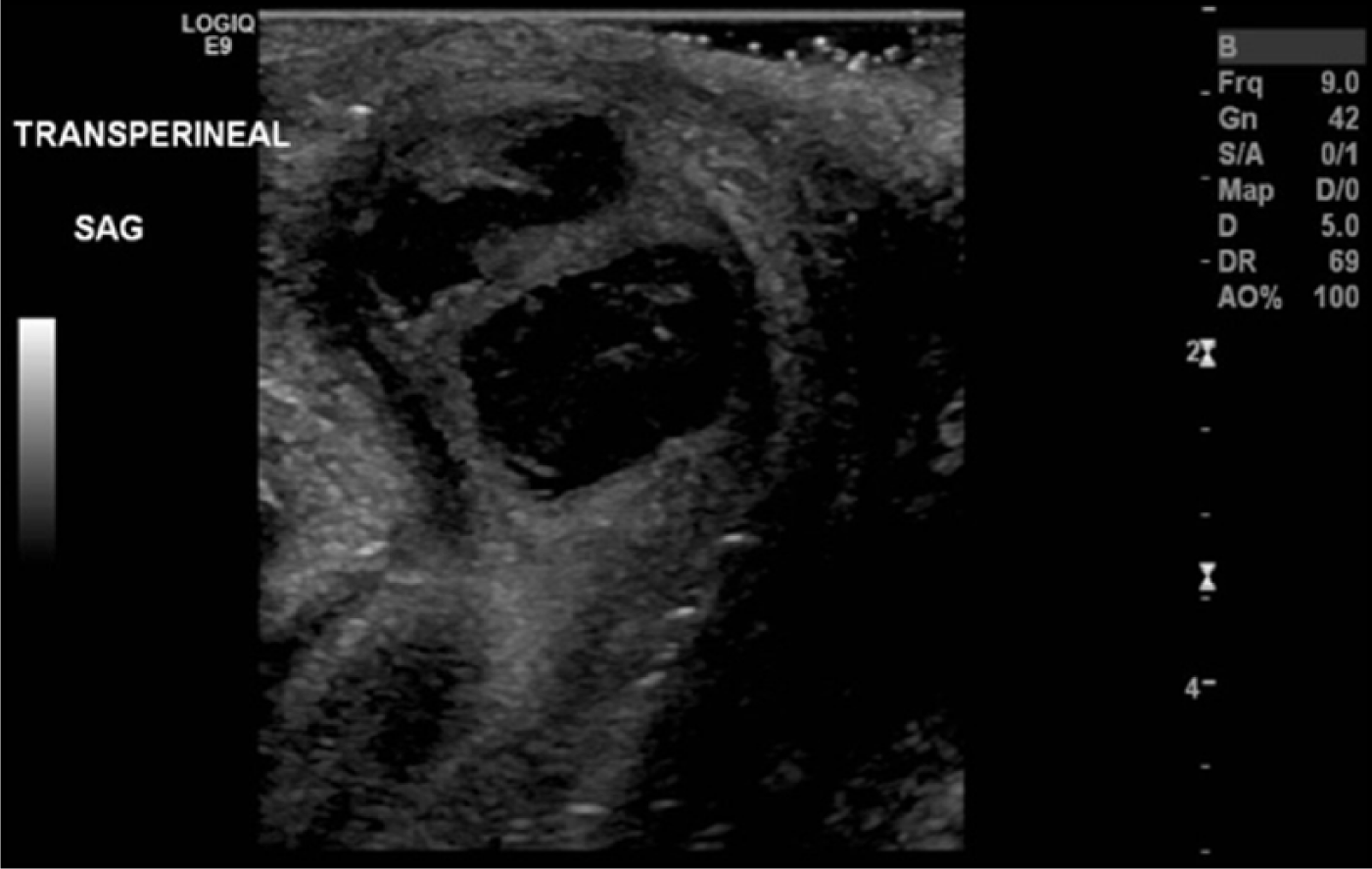
Transperineal sagittal gray-scale image showing a complex, septated fluid collection located along the midline of the perineum.
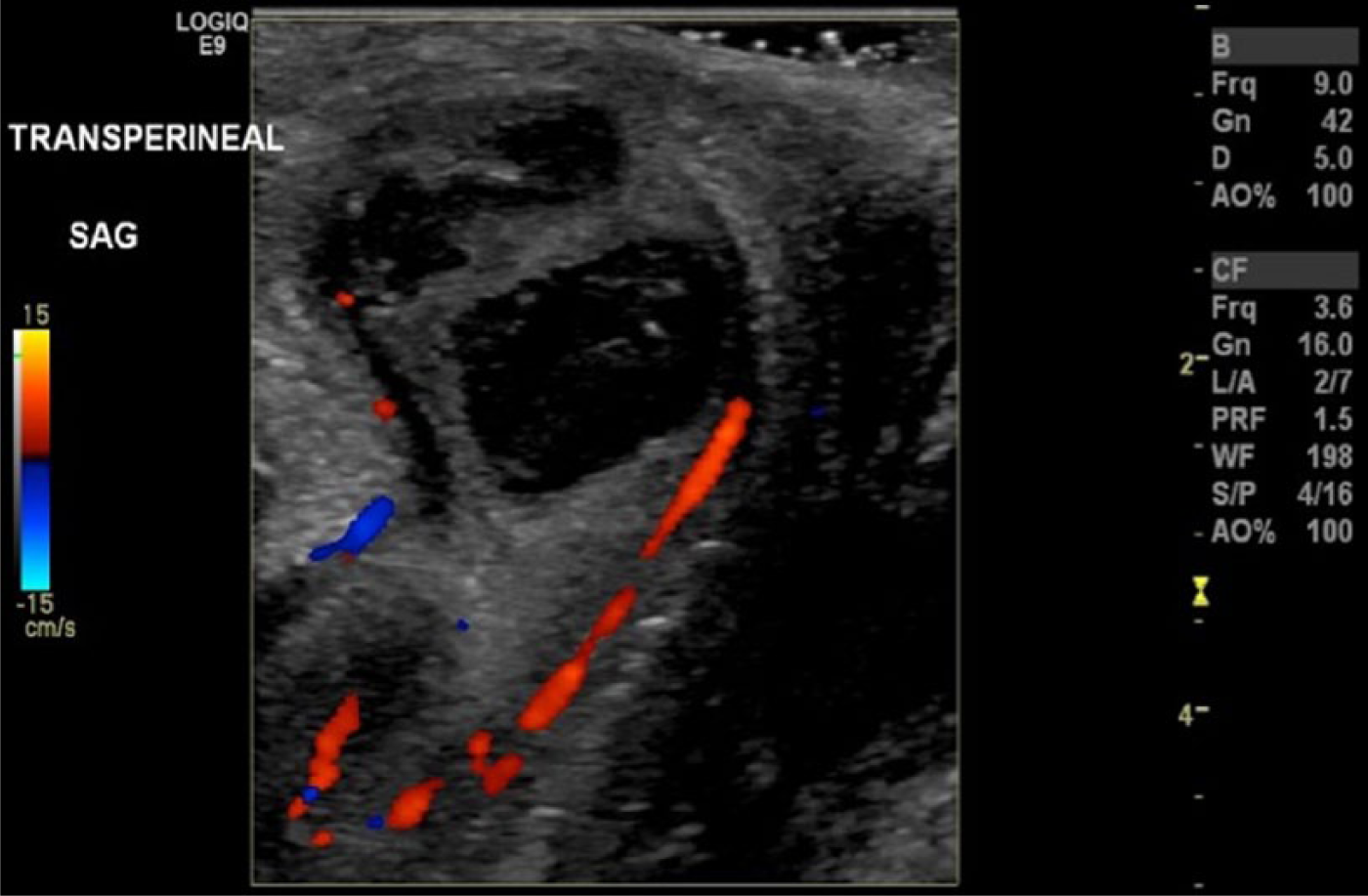
Transperineal sagittal color Doppler image showing the complex, septated fluid collection with peripheral vascularity.
Discussion
Skene’s glands were first described by Regnier de Graaf (1641–1693) but are named after the Scottish gynecologist Alexander Skene, who published his findings in the Western medical literature in 1880.1,3 The Skene’s glands, or paraurethral glands, are found in the caudal two-thirds of the urethra, mainly in the dorsal and lateral mucosal stroma.2,4 They are the female homologues of the prostate gland in the male and arise from the urogenital sinus. Wernert et al. 2 presented a histological study and found that these glands are not always present and were found in only two-thirds of the 33 women they studied.
Abscesses of the Skene’s gland most commonly present in the third to fourth decades, and it is uncommon to find Skene’s gland abscesses in pediatric patients.9,10 Most cases occur without predisposing risks, but predisposing factors include diabetes, pregnancy, trauma, and previous impetigo. 8 Presenting symptoms of Skene’s gland abscess include urethral pain, dysuria, dyspareunia, presence of an asymptomatic mass, recurrent urinary tract infection, urethral drainage, and voiding symptoms.6,7,9,10 Infection of the gland by bacteria leads to inflammatory obstruction of the paraurethral ducts, which give rise to the formation of a cyst or abscess. Bacteria is the pathogen commonly associated with a Skene’s gland abscess.8,9 Bacteria associated with a Skene’s gland abscess include Escherichia coli, Neisseria gonorrhoeae, vaginal flora, and other coliform bacteria. 10 Antibiotic therapy is typically initiated to cover these common pathogens, as was done in the case described. If a response is seen to antibiotic therapy but symptoms recur, then a repeat course is given. If there is no response to antibiotics, then surgical therapy is offered. It is reasonable to consider surgical intervention after a failure or recurrence of symptoms following one or two courses of antibiotics if the patient is symptomatic and appropriately counseled. Surgical excision, marsupialization, and needle aspiration have all been described for the surgical management of a Skene’s gland abscess.6,10,11
The diagnosis of Skene’s gland abscess can generally be considered based on the history and physical examination. However, clinical assessment of women with paraurethral gland symptoms may be difficult, necessitating further evaluation with imaging. When the diagnosis is in doubt, further workup with sonography, magnetic resonance imaging, voiding cystourethrography, or cystourethroscopy is warranted. 10
Recent advances in sonography and magnetic resonance imaging have dramatically improved evaluation of the female urethra and the paraurethral glands, clarifying findings at physical examination and providing accurate roadmaps for surgeons.12–15 High-resolution multiplanar magnetic resonance imaging (MRI) with phased-array pelvic and endovaginal coils allows greater soft-tissue differentiation of the paraurethral region (Figure 3). Endovaginal-specific MRI coils provide high-resolution images of the female urethra and periurethral anatomy, since the receiving antenna is within a centimeter of the urethra. 13 MRI has been consistently reported to have higher sensitivity and higher positive and negative predictive values than either voiding cystourethrography or retrograde double-balloon positive-pressure urethrography in the detection of female urethral and paraurethral disorders.15,16 Because of its high resolution, increased signal-to-noise ratio, and mulitiplanar capability, MRI has become the imaging modality of choice for diagnosis and preoperative planning in female patients with urethral and periurethral disease. Important additional advantages of magnetic resonance imaging include its capability to help detect noncommunicating urethral diverticula and to demonstrate the radial and circumferential extent of paraurethral disease without the need for catheterization or urethral contrast material injection.15,16 The limitations of MRI, despite its excellent characterization of female urethral disease, remain its lack of generalized accessibility and high cost.
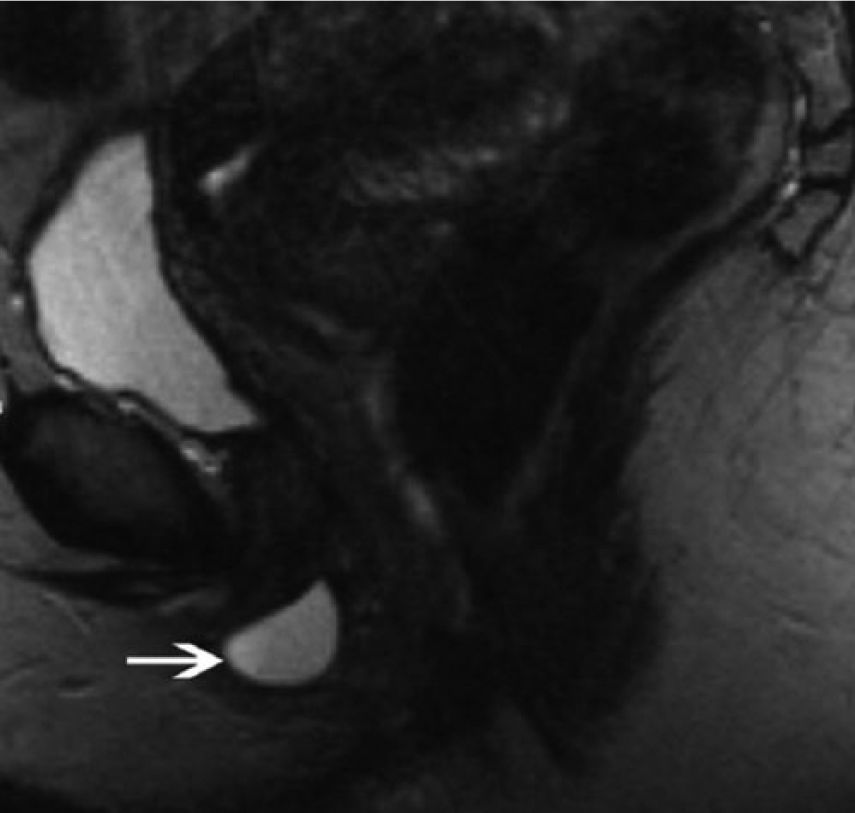
T2-weighted magnetic resonance image showing a cystic lesion (arrow) lateral to the expected location of the external urethral meatus inferior to the pubic symphysis.
Transperineal sonography is an emerging imaging technique that is being used to evaluate female urethral and periurethral disease in a more cost-effective manner.12–14 In addition to lower costs, the advantages of sonography include the ability to perform real-time imaging during straining, imaging in different orientations, differentiation between solid and cystic lesions, and evaluation of the paraurethral anatomy. The limitation of sonography is its operator dependence. 13 To evaluate the paraurethral glands with transperineal sonography, gel is placed on the footprint of a high-frequency linear array transducer. A protective transducer covering is applied around the transducer and secured with a rubber band. Sterile ultrasound gel is then applied to the outer surface of the protective covering. The examination should be carried out with the patient lying in the supine position with the thighs abducted sufficiently to allow placement and lateral angulation of the transducer. The transducer should be placed directly over the perineum between the labia majora, usually directly over the labia minora, but occasionally between them. 13 The urethra, vagina, and anal canal will be seen in the near field of the screen. The uterus will be demonstrated in the far field of the screen. Sonographically, abscesses are usually round or oval complex masses with associated hypervascularity, thickened walls, and internal echoes (Figures 1 and 2). The presence of echogenic material with acoustic shadowing indicates the presence of gas.
Surgical excision, marsupialization (in which the cystic space is opened, incised, and drained but the incised wall is left open to heal by granulation tissue forming from the base of the pouch), and needle aspiration have all been described for the surgical management of the Skene’s glands. An argument for surgical excision, rather than marsupialization or needle aspiration, is the fact that malignancy has been reported to occur in paraurethral cysts.3,7,10,17 In most cases, the patient is sent home after the surgical excision without a urethral catheter. For larger reconstructions of the urethral meatus, a catheter may be kept in place for several days. Nitti et al. 10 reported a study of 34 women who underwent Skene’s gland excision. After the initial excision, 88.2% of the patients had resolution of symptoms. However, symptoms recurred in 9 of 30 women (30%) who were initially cured, with a mean time to recurrence of 24 months. Of these nine women, eight were treated successfully with either further conservative management or repeat surgical treatment, for a total cure rate of 85% after two interventions.
Conclusion
Skene’s glands are glands that resemble the male prostate gland and are situated in the caudal two-thirds of the urethra, mainly in the dorsal and lateral mucosal stroma. Infection of the gland by bacteria leads to inflammatory obstruction of the paraurethral ducts, causing the formation of an abscess. The diagnosis of Skene’s gland abscess can generally be made by history and physical examination, but when the diagnosis is in doubt, further workup with sonography and magnetic resonance imaging may be warranted. Even though magnetic resonance imaging is the gold standard in the detection of female urethral and paraurethral disorders, transperineal sonography is an emerging imaging technique that is being used to evaluate female urethral and periurethral disease in a more cost-effective manner.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
