Abstract
The demand for highly sonographically visible breast biopsy markers is growing as an increasing number of physicians are using sonography for preoperative localization and single-step intraoperative ultrasound-guided excisional procedures. The objective of this study was to directly compare the sonographic visibility of five commercially available breast biopsy site markers. A single-site prospective comparative study was performed with 25 female participants, each assigned to receive one of five biopsy markers. Postdeployment, 6-week, and 12-week follow-up sonographic images were obtained and a survey was e-mailed to members of the Society of Breast Imaging asking them to grade marker visibility on a 1- to 5-point Likert-type scale. At the time of deployment, the SecurMark (Hologic, Bedford, Massachusetts) was rated with the highest sonographic visibility. At both 6 and 12 weeks after deployment, the HydroMARK (Biopsy Sciences, Clearwater, Florida) was found to be the most visible by sonography. The results can be used by physicians who require highly sonographically visible breast biopsy markers for ultrasound-guided interventional procedures. Further research is needed as biopsy marker manufacturers continue to refine their products.
It is estimated that in 2012, more than 226,000 women were diagnosed with and nearly 40,000 died of breast cancer in the United States. 1 With the widespread adoption of screening mammograms, there has been a surge in the discovery of occult nonpalpable breast lesions over the past several decades. Sonography was first used in breast imaging mostly to aid in the distinction between cystic and solid lesions. Today, sonography has a prominent and expanding role in the diagnosis of breast cancer and facilitation of interventional procedures. 2
Breast lesions that raise the concern for malignancy are routinely biopsied by stereotactic or ultrasound-guided techniques. It has become common practice to deploy a radiomarker at the time of biopsy so that the lesion can be easily identified on subsequent imaging examinations. Historically, breast biopsy markers were made of metal to optimize mammographic visualization. As ultrasound-guided wire localization procedures have gained popularity among radiologists, manufacturers are beginning to market biopsy markers with material properties specifically tailored to improve sonographic visibility.
There has already been experimentation with sonographically visible breast biopsy markers in operating rooms as a surrogate for wire localizations.3–5 In this intraoperative ultrasound (IOUS) technique, the surgeon is afforded real-time visibility of the target lesion and/or marker and can directly excise the area of concern. The markers play an important role when the target lesion itself or post–biopsy tissue changes are poorly seen sonographically. The main advantage to the IOUS technique is to eliminate the need for a separate wire localization or radioactive seed implantation procedure. This helps to reduce patient pain and anxiety prior to surgery. Additional potential benefits include cost savings, a lower incidence of operating room scheduling conflicts, and more effective and cosmetically pleasing excisional procedures.3–5
Regardless of whether the biopsy marker will be used for an ultrasound-guided localization procedure or lumpectomy using the IOUS technique, a highly sonographically visible marker is desired. The goal of this study was to compare the sonographic visibility of five commercially available breast biopsy site markers at the time of deployment, 6 weeks after marker placement, and 12 weeks after marker placement.
Method
Upon institutional review board approval, a single-site prospective comparative study was performed to evaluate the sonographic visibility of five different biopsy site markers in 25 patients (five groups, each group containing five patients with the same marker placed). The markers included the SenoMarkUltraCor MRI (SenoRx, Irvine, California), Gel Mark UltraCor (SenoRx), HydroMARK (Biopsy Sciences, Clearwater, Florida), SecurMark (Hologic, Bedford, Massachusetts), and UltraClip Enhanced Coil (C. R. Bard, Inc, Murray Hill, New Jersey).3,6–9 The final biopsy markers were selected based on which markers were currently in stock at our institution and, after contacting several manufacturers, those companies willing to donate markers for the purposes of the project. The biopsy markers were limited to five given the time necessary to obtain cohorts of five patients with the same biopsy marker who were willing to participate in the clinical study. The manufacturers of all five biopsy markers marketed their products as being sonographically visible. The material properties of the five biopsy markers are shown in Table 1.
Characteristics and Material Properties of the Five Tested Biopsy Markers.3,6–9
Abbreviations: PEG, poly(ethylene glycol); PGA, poly(glycolic acid); PLA, poly(lactic acid); PVA, poly(vinyl alcohol).
Each marker was placed after an ultrasound-guided core needle breast biopsy of a suspicious breast mass. A 14-gauge MaxCore biopsy device (C. R. Bard, Inc) and a 13-gauge introducer were used for the procedures. All biopsy markers were deployed in the transverse plane. Biopsy dates ranged from September 16, 2009, through February 10, 2011. The patient consented to return at 6 weeks (±4 days) and 12 weeks (±4 days) after marker placement for sonographic imaging of the marker, provided the pathology results were benign.
Sixty-three women were originally enrolled in the study. Sixteen had a positive cancer diagnosis and one had a diagnosis of intraductal papilloma that was excised before the completion of the study. One participant requested to have her breast biopsy marker removed. Twenty participants did not return for follow-up appointments. The distribution of biopsy markers for those who did not return for follow-up appointments was five for the SecurMark; four each for the SenoMark UltraCor MRI, Gel Mark UltraCor, and HydroMARK; and three for the UltraClip Enhanced Coil. The age and final pathology diagnosis of the 25 remaining women who participated in the full study are listed in Table 2.
Age and Pathology Diagnosis of the 25 Participants (Grouped by Which Biopsy Marker Was Deployed).
A Philips ATL HDI 5000 SonoCT ultrasound machine (Philips, Andover, Massachusetts) with the same transducer (L12-5 linear array probe) was used for image acquisition of all markers. The biopsy procedures and imaging studies were performed by a single breast imaging fellowship-trained radiologist. Per protocol at our institution, sonographic images of each biopsy site marker were obtained in both the sagittal and transverse planes. As all the biopsy markers were introduced in the transverse plane, this provided orthogonal views of the markers along both their long and short axes.
Once the clinical data had been obtained from all 25 patients, an online survey was e-mailed to the 2584 physician members of the Society of Breast Imaging (SBI). Reminder e-mails were sent to the nonresponding SBI members at two weeks. The survey began by asking members to estimate their percentage of time spent in breast imaging, whether they completed a breast or women’s imaging fellowship, the number of years they had been in practice, and if they were American Board of Radiology (ABR) or American Osteopathic Board of Radiology (AOBR) certified in diagnostic radiology. Society members who reported spending less than 25% of their workday in breast imaging were excluded from the study.
Next, five randomly assigned “cases” containing images from a single biopsy marker at each of the three specified time periods (for a total of 15 sets of images) were displayed on the screen (Figure 1). Each case was dedicated to one of the five biopsy markers, with all five biopsy markers represented in every survey but not all physicians necessarily seeing the same cases. The SBI members were asked to grade, on a 1- to 5-point Likert-type scale, the degree of visibility of each marker at the time of deployment, 6 weeks after marker placement, and 12 weeks after marker placement. The respondents were informed that a rating of 1 indicated that the biopsy marker was very difficult or impossible to see, and a rating of 5 meant that the marker was easily identified.
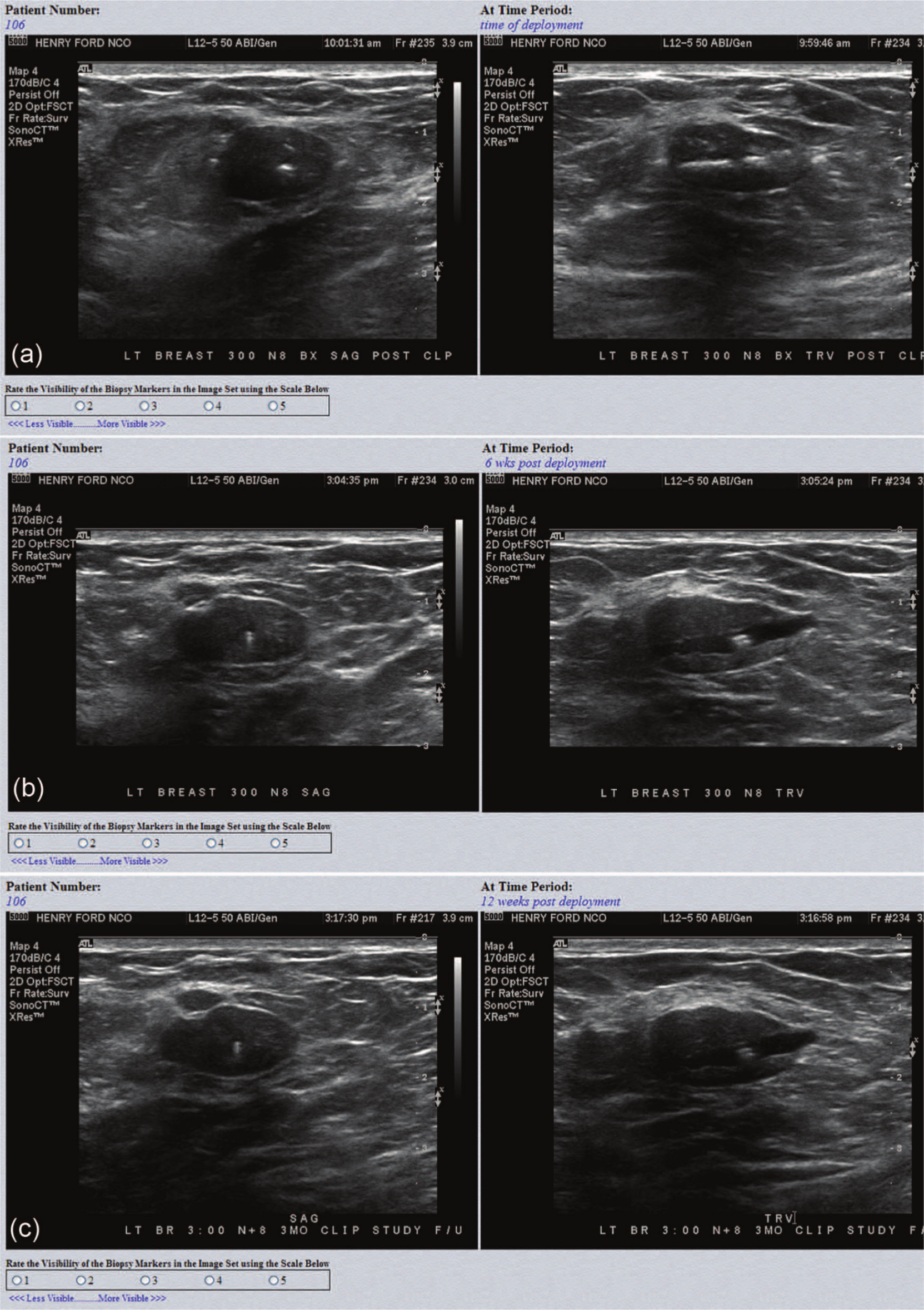
Typical screenshots of one of the “cases” sent to Society of Breast Imaging members. Respondents were asked to grade each set of images for each time point for degree of visibility on a 1- to 5-point Likert-type scale, with 1 being very poor or no visibility and 5 being very good visibility. Time points were (a) time of deployment, (b) 6 weeks after deployment, and (c) 12 weeks after deployment.
Statistical analysis was performed using SAS 9.2 software (SAS Institute, Cary, North Carolina). The proportion of respondents grading the ultrasound visibility of the biopsy markers as either a 4 or 5 at each time point was compared using McNemar tests with exact P values. When a test was significant overall, each pairwise comparison was analyzed to locate the difference and the P values were adjusted using the Hochberg method to control the type I error rate. Statistical significance was set at P < .05.
Results
Of the 2584 surveys sent, 158 SBI members replied, for a response rate of 6.1%. All respondents completed the entire survey. Eight respondents were excluded from the data pool as they indicated that they spent less than 25% of their workday in breast imaging. Of the remaining 150 responding members, the mean (SD) estimated percentage of time spent in breast imaging was 81.4% (22.3%). Of the respondents, 75.3% answered that they have been in practice for more than 5 years, and 30.7% said that they have been practicing radiologists for greater than 20 years. Of the SBI members, 55.3% had completed a breast imaging fellowship and 93.3% were either ABR or AOBR certified in diagnostic radiology.
A score of 4 or 5 on the survey was considered “highly visible.” Figure 2 shows a graph of the five tested breast biopsy markers and the corresponding percentage of SBI members rating the marker as highly visible at each of the three time points. Of the five tested breast biopsy markers, the most visible marker at the time of deployment was the SecurMark, for which 74.7% of the respondents rated the sonographic visibility as a 4 or 5. The HydroMARK received 4s or 5s by 52.7% of respondents. This difference was statistically significant (P < .05), indicating that there was a true difference in sonographic visibility (as judged by the SBI members) of the markers at the time of deployment. The least visible marker at the time of deployment was the UltraClip Enhanced Coil, with only 23.4% rating it as highly visible (Figure 3).
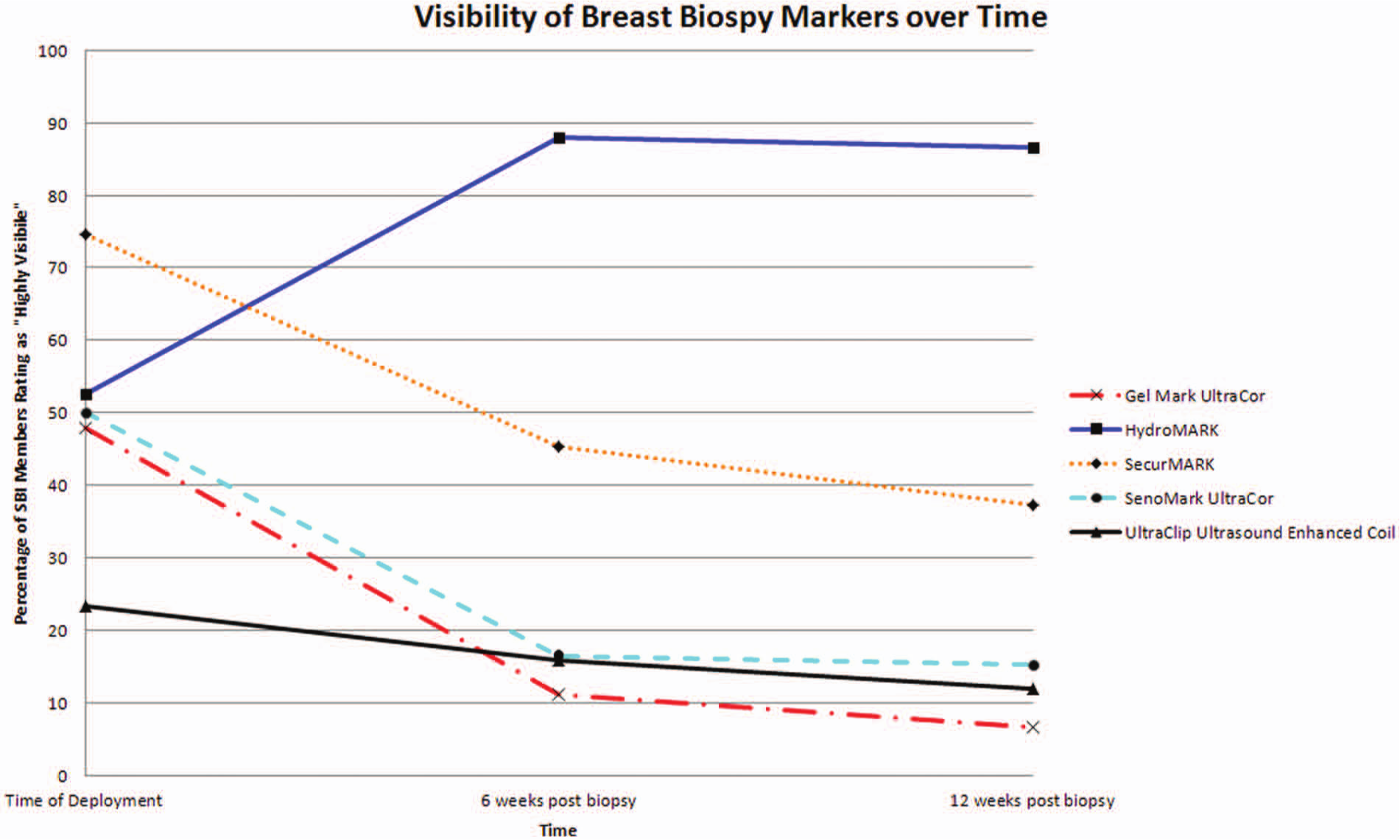
Percentage of responding Society of Breast Imaging members rating the sonographic visibility of the breast biopsy markers as a 4 or 5 (considered “highly visible”).
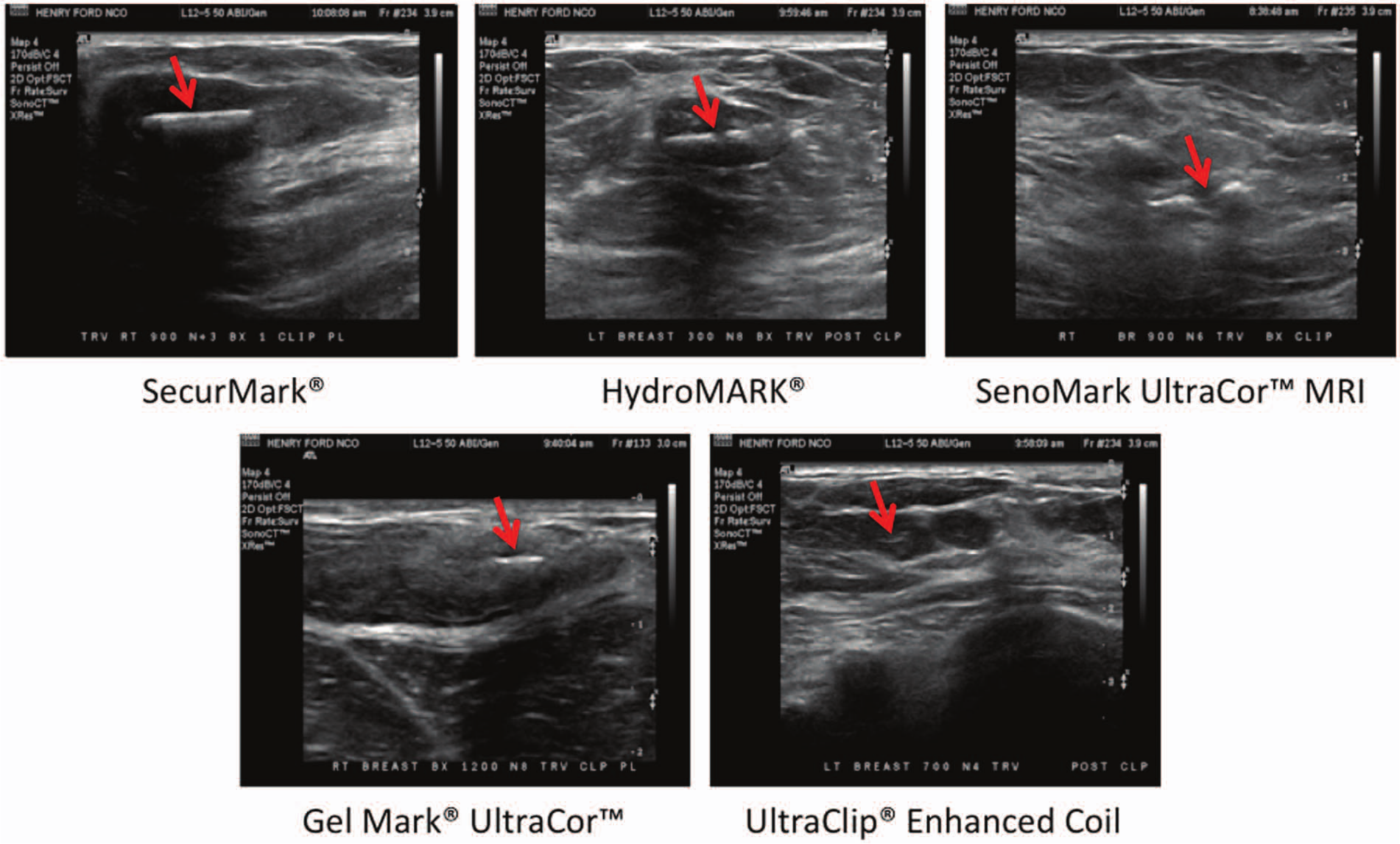
Representative transverse sonographic images of the five biopsy markers at the time of deployment (in order from most visible to least visible, as ranked by the responding Society of Breast Imaging members). Arrows mark the location of the biopsy markers.
At six weeks postprocedure, 88.0% of the SBI members indicated that the HydroMARK was highly visible (Figure 4a). The biopsy marker receiving the second most 4s and 5s (by 45.4% of SBI members) was the SecurMark. The difference between the proportion of those who considered the HydroMARK and SecurMark highly visible was statistically significant (P < .05). The Gel Mark UltraCor received the fewest 4s and 5s at six weeks, with 11.3% rating it highly visible.
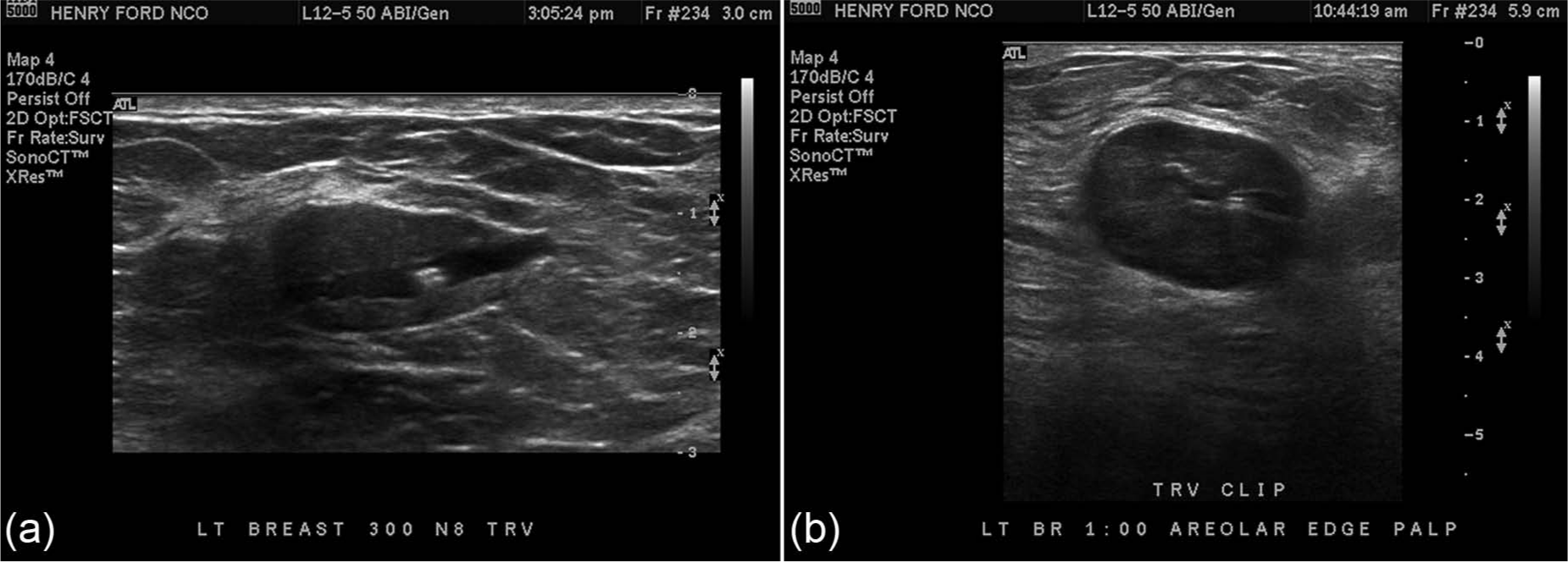
Representative transverse sonographic images of the HydroMARK from two different patients at 6 weeks after deployment (a) and at 12 weeks after deployment (b). At both time points, the HydroMARK was rated the most highly visible breast biopsy marker.
At 12 weeks postprocedure, the overall results were similar to the results at 6 weeks. The HydroMARK again had the largest proportion of SBI members considering it highly visible, with 86.7% rating it a 4 or 5 (Figure 4b). Of the responding SBI members, 37.3% rated the sonographic visibility of the SecurMark with 4s and 5s, a statistically significant difference when compared with the HydroMARK (P < .05). The Gel Mark UltraCor again received the fewest 4s and 5s, with 6.7% of respondents rating it highly visible and 56.0% responding that it was “very difficult or impossible” to see at 12 weeks.
Discussion
This study directly compared the sonographic visibility of five commercially available breast biopsy markers at three separate time points. Although surgical intervention for pathologically concerning lesions often occurs within a couple weeks after initial biopsy, many lesions are followed for longer periods, especially when neoadjuvant chemotherapy or cryoablation is used. Thus, in addition to the initial set of images taken at the time of deployment, 6- and 12-week follow-up images were obtained to assess for changes in sonographic visibility over time.
Based on image reviews by member radiologists of the SBI, the most sonographically visible marker at the time of deployment was the SecurMark. At both 6 and 12 weeks after biopsy, the HydroMARK was ranked the most highly visible biopsy marker. The visibility of the HydroMARK increased between time of deployment and 6 weeks and remained highly visible at 12 weeks. Conversely, the sonographic visibility of the other four biopsy markers steadily declined from time of deployment to 6- and 12-week follow-up examinations. The unique material properties of the poly(ethylene glycol) hydrogel component of the HydroMARK to attract water molecules (and in turn to create an anechoic structure) likely played a role in its increasing sonographic visibility over time.
Although this is the first known published study to directly compare the sonographic visibility of multiple breast biopsy markers, similar studies have been conducted by other groups using a single biopsy marker. Eby and colleagues 10 examined the sonographic visibility of the CorMark (Devicor, Cincinnati, Ohio) biopsy marker, a collagen-titanium alloy marker clip matrix, by surgeons and radiologists in the clinic and in the operating room within four weeks of biopsy marker deployment. A Likert scale similar to the one used in our study was employed with the only difference being that a rating of 3, 4, or 5 was considered “adequate” visibility. They found a nonsignificant trend toward decreasing visibility of the biopsy markers from the time of deployment to the day of the operation. Although the follow-up time intervals differed slightly between studies, these findings are congruent with the decreasing sonographic visibility of the biopsy markers in our study, with the notable exception of the HydroMARK.
The use of intraoperative ultrasound for localization of breast masses at the time of surgery (as opposed to a separate preoperative localization procedure) has been studied by multiple authors with generally favorable results.3–5,11–13 However, in most of these studies, the surgeon targeted the residual breast lesion and/or post–biopsy tissue changes (such as hematoma or cavity) in at least a portion of the cases. These are inherently unreliable targets and, although useful if present, cannot be depended on for accurate and consistent intraoperative lesion localization. Highly visible breast biopsy markers provide an opportunity to improve the success rate of intraoperative ultrasound-guided excisional procedures.
Klein et al 3 performed a single-institution retrospective review of the use of intraoperative ultrasound evaluation of the HydroMARK biopsy marker as a potential alternative to preoperative localization procedures. On the basis of postoperative notes, they found that at the time of surgery, 93.6% of HydroMARK markers examined using the IOUS technique were “adequately” identified by the surgeon. Despite the high intraoperative visibility of the biopsy markers, most patients had undergone preoperative localization procedures. However, six lesions were successfully excised without a preoperative localization procedure, using IOUS alone. Although these early results are promising, the authors noted that the surgeons encountered problems with migration or extrusion of the marker through the biopsy tract in more than half of the cases. Marker migration and extrusion have been described in the literature, and design improvements in marker tissue adherence have received considerable attention by manufacturers.14–16 Adequate and reliable intraoperative ultrasound visualization of the biopsy marker, decreased incidence of marker migration, and prevention of intraoperative extrusion are fundamental requirements before separate localization procedures can be eliminated and stand-alone IOUS localization and excisional procedures become common practice.
Although this study showed that the SecurMark was the most sonographically visible biopsy marker at the time of deployment and the HydroMARK was most visible on subsequent follow-up examinations, it is unclear when, if follow-up imaging had been performed at a shorter time interval, the HydroMARK would have overtaken the SecurMark in terms of visibility. In addition, it is unknown if the improved resolution of a newer generation ultrasound machine would have influenced the outcome. Although the results showed statistically significant differences in the proportion of SBI members ranking the biopsy markers as highly sonographically visible, the response rate to the survey was quite low at 6.1%. It is conceivable that with a higher response rate, the results may have changed (nonresponse bias). An additional limitation of the study pertains to the variability in imaging characteristics of a given patient. For example, differences in body habitus, breast density, conspicuity of the breast mass, and depth of the mass/marker could affect the sonographic visibility of the biopsy marker. An attempt to control for these factors was made by placing the same type of biopsy marker in five different patients, although clearly much larger cohorts of patients would be needed to negate this influence. Finally, as it was not logistically possible to test all available biopsy markers marketed as being sonographically visible, it is feasible that there are markers that have equal or better sonographic visibility than those examined in this study. This is especially true as newer generation biopsy markers with improved material properties are released.
Conclusion
Five commercially available breast biopsy markers were directly compared to assess their sonographic visibility at the time of deployment, a 6-week follow-up, and a 12-week follow-up through a survey sent to members of the SBI. The results of the survey show that there was variability in the sonographic visibility of the markers and that this variability changed with time. Further research into additional biopsy markers is needed, particularly as new technologies are developed to improve visibility and tissue adherence.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Portions of this research project were funded by SenoRx (Irvine, California), which was later purchased by C. R. Bard, Inc. (Murray Hill, New Jersey), including the supply of biopsy markers and the financing of follow-up imaging examinations. Biopsy Sciences (Clearwater, Florida), manufacturer of the HydroMARK, also donated biopsy markers for this study.
