Abstract
Three-dimensional automated breast sonography is currently an investigational modality for breast cancer screening not yet accepted clinically in the United States. In this pilot study, a prospective study of 24 asymptomatic female patients with more than 50% breast density by digital screening mammography was performed using automated breast sonography and compared with 3D contrast breast magnetic resonance imaging (MRI) correlation (the gold standard). This study was performed as part of a multimodality approach to histologically proven breast lesions classified as categories 4 and 5 according to the American College of Radiology Breast Imaging, Reporting, and Data System (BI-RADS). Comparable diagnostic reliability (kappa values between 0.95 and 0.98) was observed between automated breast sonography and MRI in differentiating benign from malignant breast lesions based on similar morphologic criteria. This preliminary study offers automated breast sonography as a promising cost-effective adjunct diagnostic modality to MRI in the evaluation women with increased breast density by digital mammography.
Breast cancer is a significant health risk in otherwise healthy, asymptomatic women. The epidemiology in the United States ranks breast cancer second only to lung in cancer incidence, with 207,090 new cases in 2010 among women. 1 Although beneficial, the use of screening mammography has historically been very limited in the setting of increased breast density. In the United States, automated breast sonography is a Food and Drug Administration (FDA)–approved diagnostic modality for mammographically dense breasts but is generally not yet accepted clinically. Recently, an FDA Radiologic Devices Panel recommended approval of automated breast sonography for use in screening of women with dense breast parenchyma. 2 Unlike handheld sonography, automated breast sonography produces a 3D volume acquisition using a 10- to 14-MHz phased array linear transducer. Frontal, medial-frontal, and lateral frontal scans in 3-mm coronal volume acquisitions produce more than 350 high-quality images for each breast that are digitally reformatted on a dedicated workstation to produce real-time axial and sagittal reformatted images. Although automated breast sonography does not statistically differ significantly from HHUS in terms of detection rate 3 (Table 1), significant practical advantages may apply to large screening populations. These include short examination times of only 10 minutes or less per breast, simplicity of use, less technical training, less user variability, application for potential screening of large numbers of patients with dense breast parenchyma, and volumetric data with multiplanar capability. We are reporting the results of a pilot study correlating automated breast sonography and magnetic resonance imaging (MRI) in the detection of histologically proven breast lesions in a dense breast screening population of asymptomatic patients with mammographically occult lesions.
Detection Rates of Automated Breast Sonography Compared to Handheld Sonography 3

Materials and Methods
The automated breast sonography system consists of a scan station that automatically surveys/scans the breasts and a view station that is used to review the images and to manipulate the volumetric data. The image workstation enables the viewer to analyze the breasts using a cine function for specific regions of interest on the gray-scale images, also known as maximum intensity projection (MIP). The 3D volumetric data can be analyzed in different formats and also can be converted into thin tomographic slices. By using a virtual probe function, various scanning planes can be viewed to demonstrate the relationship of the lesion to the surrounding anatomic structures. The scan station is an automated sonography image acquisition system that acquires volumetric data for specialized imaging processing. The base system contains an engine, a wide-aperture high-frequency linear transducer, a 15-inch LCD touch screen, a computer, and Windows-based software to provide automated scanning parameters, automated image acquisition, and 3D digital image data storage.
The automated breast sonography system has a linear transducer of wide aperture. However, to provide good contact between probe and tissue, the length of the transducer is not able to cover the whole breast. Three views are generally required for each breast, including frontal, medial-frontal, and lateral-frontal. In each view, six scan sweeps are obtained, providing thick tomographic slices with MIP of reversed format from skin toward the chest wall. Because of the shadows resulting from Cooper’s ligaments, an irregular linear/reticular pattern of high signal intensity (high level of brightness) is a common finding on both thick and thin tomographic views. The breast stroma can be of variable signal intensity based on the fibroglandular density. The ribs of the chest wall are also of high signal intensity.
A prospective pilot study of 24 female screening breast patients was evaluated using digital mammography (Selenia; Hologic, Inc, Bedford, Massachusetts) and automated breast sonography (SomoVu; U-Systems, Inc, San Jose, California) equipped with a 10-MHz transducer. The selection criterion for automated breast sonography was patients with greater than 50% breast density on mammography. Patients also underwent subsequent contrast MRI examinations based on positive automated breast sonography scans and mammographically occult lesions. MRI examinations were performed within 1 week of the automated breast sonography examinations. Automated breast sonography examinations were performed by experienced American Registry of Diagnostic Medical Sonography (ARDMS) certified sonographers with 1 year or more experience using the automated breast sonography system. Breast MRI examinations were performed in a high-field imaging system using breast-specific imaging software (3D VIBRANT; GE Medical Systems, Waukesha, Wisconsin) in combination with an 8-channel phased-array breast coil within a 1.5T HDe imaging system (GE Medical Systems). Diagnostic images were independently evaluated by 2 American Board of Radiology (ABR)–certified radiologists, also credentialed as ARDMS-certified breast sonographers. The radiologist is responsible for image capture and archiving; selecting specific regions of interest on the gray-scale images transferred from the base system to the workstation, using the cine function on the automated breast sonography workstation; and measuring lesion dimensions. With automated breast sonography, the radiologist performs many of the traditional roles of image capture performed by the breast sonographer using handheld sonography.
Lesions detected as Breast Imaging, Reporting, and Data System (BI-RADS) 4 and 5 were made on the basis of automated breast sonography and MRI, as these lesions were occult to mammography due to breast density. All detected lesions classified as final BI-RADS 4 and 5 underwent sonography-guided stereotactic 12-gauge core biopsies using handheld sonography performed by the breast surgeon in an outpatient setting. (Automated breast sonography is presently not equipped with biopsy capability.) Final pathology results were obtained within 4 weeks of initial automated breast sonography diagnosis. The diagnostic imaging features of histologically proven benign and malignant lesions were compared between automated breast sonography and MRI.
Results
BI-RADS 4 and 5 lesions (N = 24) underwent tissue diagnosis with histologic evaluation, which revealed 15 malignant tumors and 9 benign pathologies (Table 2). There was excellent interobserver agreement, with all breast cancers found by automated breast sonography and MRI correctly classified as BI-RADS 4 or 5 (kappa values between 0.95 and 0.98).
Comparing Automated Breast Sonography to Magnetic Resonance Imaging (MRI) in Classification of Benign Versus Malignant

Morphologic characterization (Table 3) with automated breast sonography showed the following diagnostic features of malignancy: irregular shape/margins (Figure 1); nonparallel orientation (Figure 2); hypoechoic halo sign, representing the tumor interface (Figure 3); noncircumscribed margin; and shadowing. These correlated with MRI features of breast cancer, including irregular shape, noncircumscribed margin, and halo of contrast enhancement. Low T2 intratumoral signal intensity on MRI corresponded to intratumoral hypoechogenicity seen by automated breast sonography (Figure 4), representing fibrotic and/or desmoplastic changes on the histologic specimen. The most common causes of false-positive results with benign pathologies were focal fibrosis, fibroadenomas, intramammary lipoma, and fibrocystic changes, with and without inflammatory change.
Morphologic Features of Breast Cancer Comparing Automated Breast Sonography to Breast Magnetic Resonance Imaging (MRI)

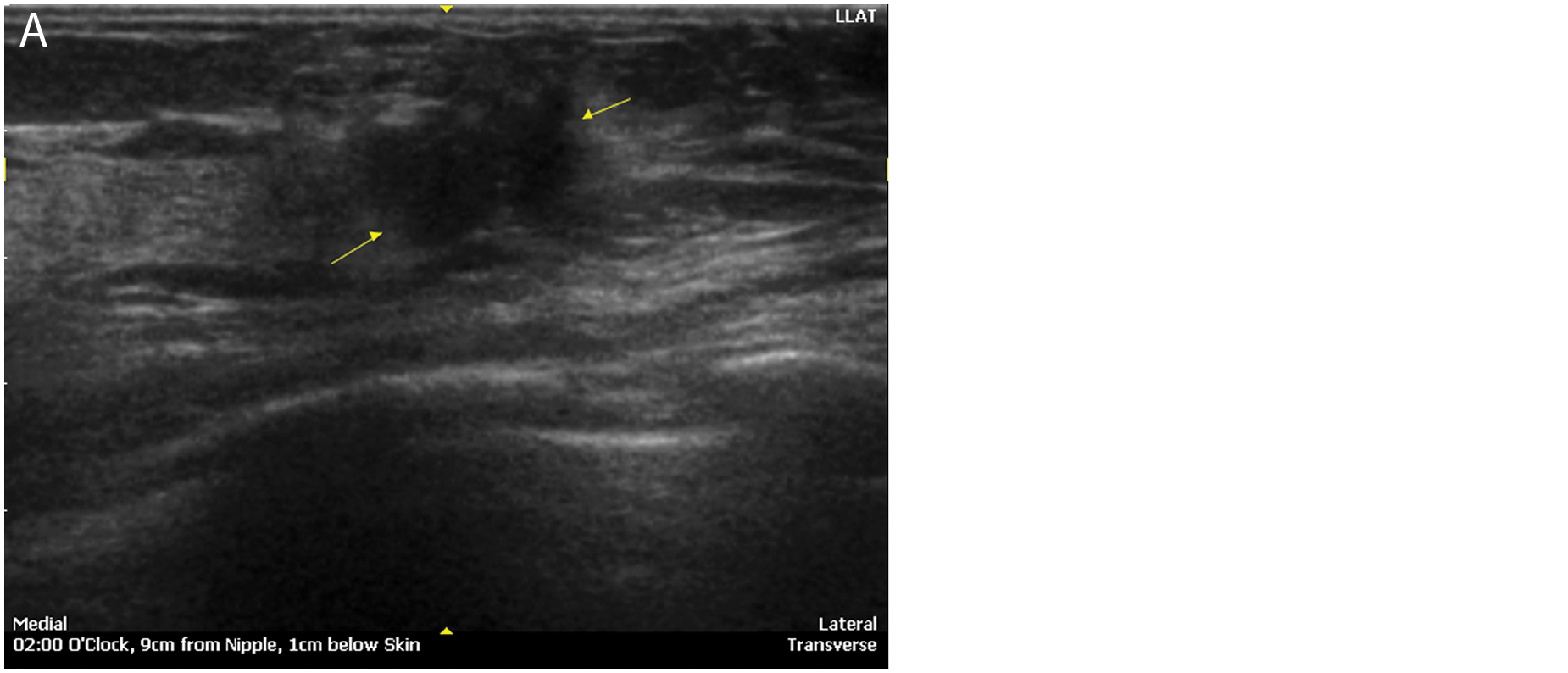
Morphologic characterization of breast cancer—irregular shape/margins. (A) Irregularly shaped heterogeneous mass on automated breast sonography. (B) Corresponding spiculated, avidly enhancing mass on contrast magnetic resonance imaging scan.
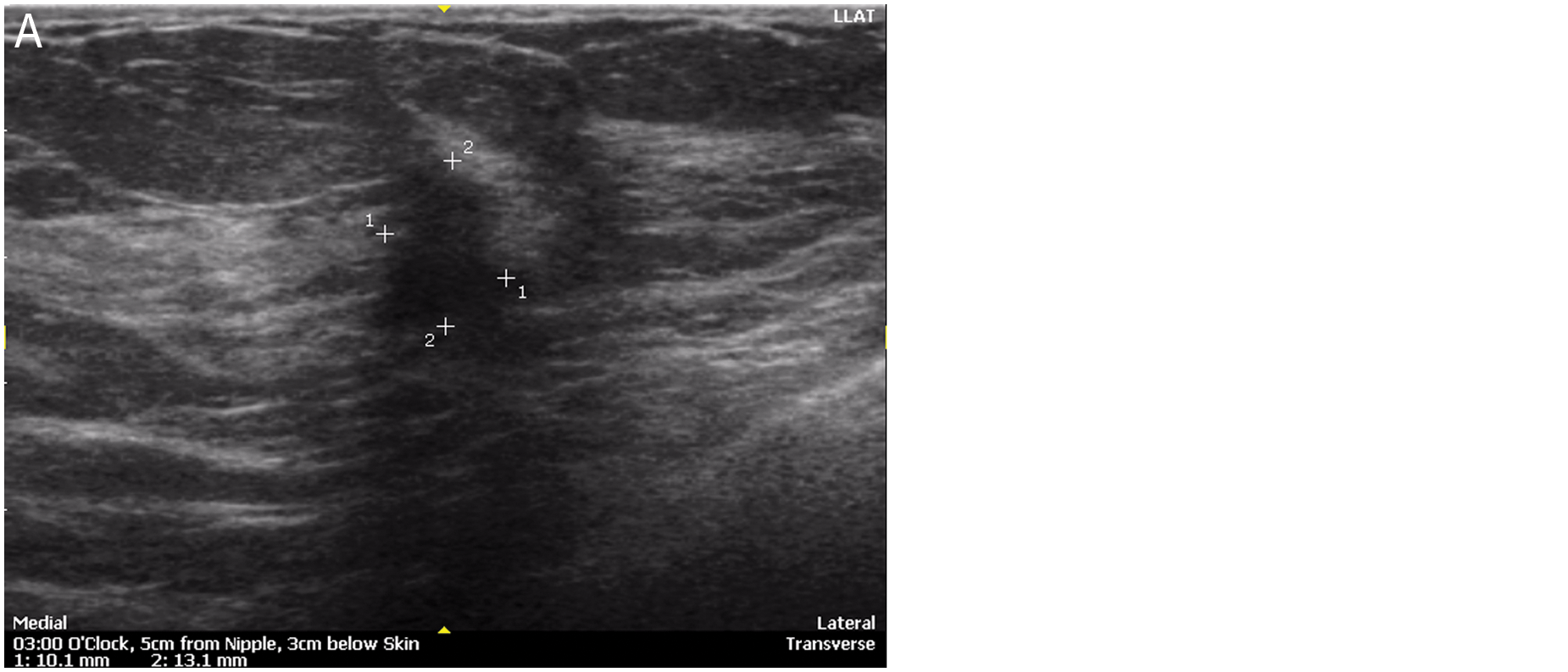
Morphologic characterization of breast cancer—nonparallel orientation. (A) Automated breast sonography shows a heterogeneous mass taller than wide. (B) Corresponding contrast magnetic resonance imaging scan.
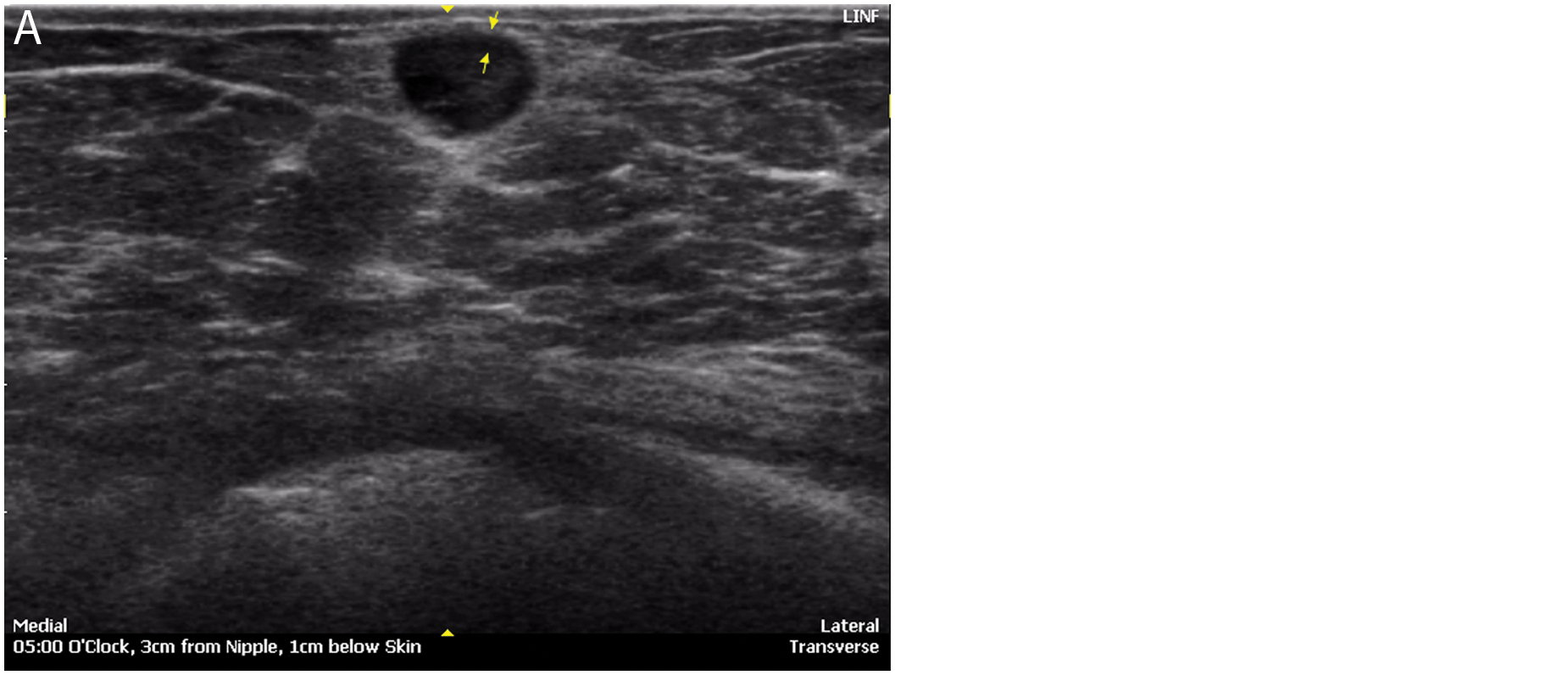
Morphologic characterization of breast cancer—the halo sign. (A) Automated breast sonography shows a characteristic hypoechoic halo at the tumor interface. (B) Magnetic resonance imaging shows characteristic rim enhancement correlating with hypoechoic halo by automated breast sonography.
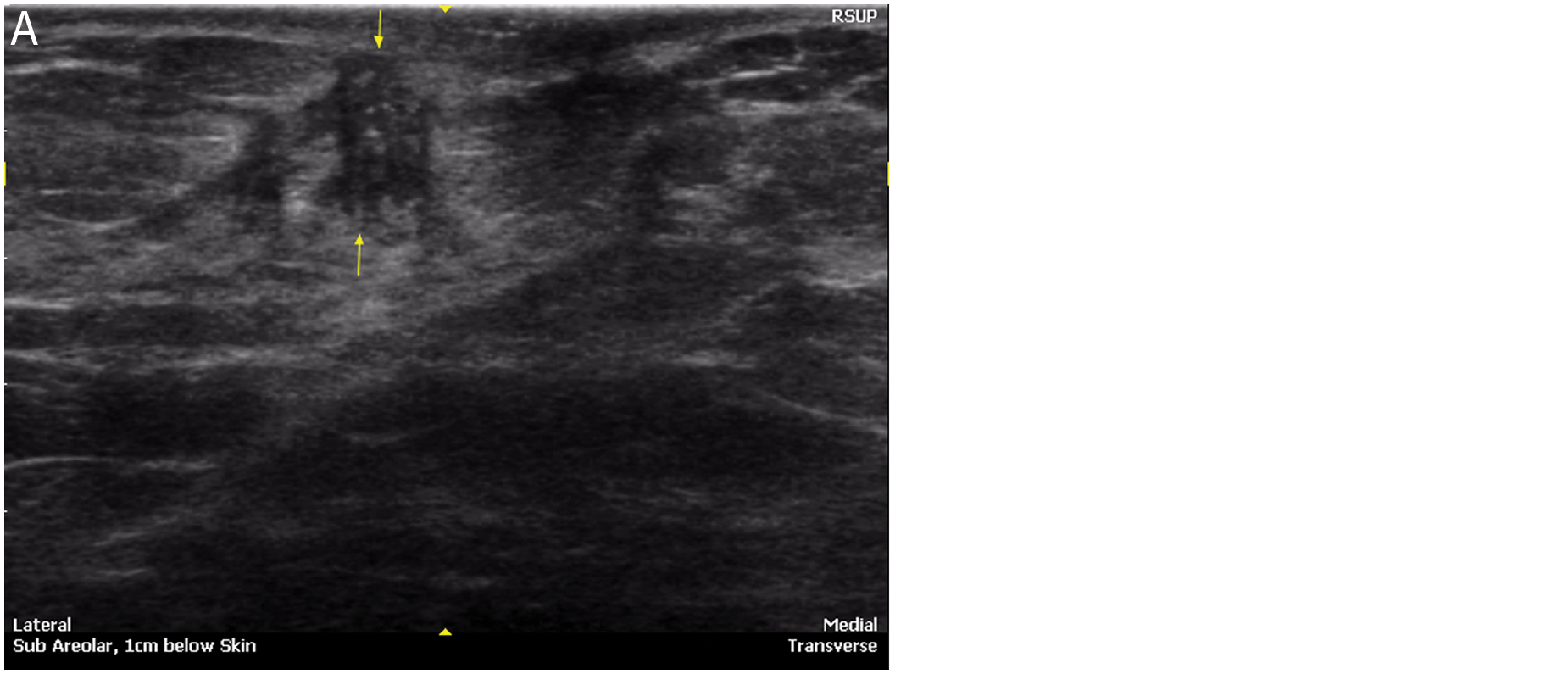
Morphologic characterization of breast cancer—tumoral fibrosis/desmoplasia. (A) Automated breast sonography shows intratumoral hypoechogenicity, representing tumoral fibrosis and desmoplasia. (B) Corresponding magnetic resonance imaging shows low intratumor signal on T2-weighted scans.
Discussion
Despite progress in identifying risk factors and genetic markers for breast cancer, approximately 70% to 80% of cases occur in women without known major predictors. 3 Population-based screening for early detection of breast cancer therefore provides the best primary strategy for reducing breast cancer mortality. Routine screening mammograms are recommended by the American Cancer Society for women 40 years of age and older and are the current standard of care for breast cancer screening. 4 One of the significant limitations of screening mammography is breast density, which can obscure small masses. Women younger than age 50 tend to have more density in their breasts than women who are older, but density may be present in women of all ages and is estimated to exist in 40% to 60% of all women who have mammograms. Women who have more fibrous connective and glandular tissue than fatty tissue in their breasts have more breast density. There are also multiple literature references showing that women who have dense breast tissue are 4 to 6 times more likely to develop breast cancer in their lifetime than women who do not have dense breast tissue. Not only do women with increased breast density have a higher risk for breast cancer, but their cancers are much harder to detect in early stages with mammography. 5
Automated breast sonography is an FDA-approved diagnostic breast imaging technology that is less affected by breast density. Multiple white paper studies, including the Somo-Insight study, a prospective multicenter study, show significant improvement in breast cancer detection rates when automated breast sonography is performed in addition to a digital mammogram, as part of annual routine breast cancer screening, in women with increased breast density.6–9 Recently, the FDA has recommended approval of automated breast sonography for use in screening of women with dense breast parenchyma. 2 However, despite these encouraging results, automated breast sonography is not widely accepted clinically in the United States. The American Institute of Ultrasound in Medicine (AIUM) currently is the only organization in the United States currently accrediting and certifying automated breast sonography in the United States.
Automated breast sonography offers no advantage to handheld sonography in terms of detection rate but may have significant practical advantages when applied to large screening populations. These include short examination times of only 10 minutes or less per breast, simplicity of use, less technical training, and less user variability. In our practice, even inexperienced sonographers have been able to establish a comfortable learning curve from as few as 50 automated breast sonography examinations.
The automated breast sonography procedure also has some disadvantages and limitations compared with handheld sonography, including the inability to perform compression or elastography maneuvers, lack of color or spectral Doppler capability for tissue or lesion vascularity, and a limited ability to evaluate the axilla and tail of Spence. In addition, automated breast sonography currently is not programmed for use in sonography-guided procedures. At the present time, AIUM accreditation is requiring certification in breast sonography by either the American Registry of Radiologic Technologists (ARRT) or the ARDMS to perform automated breast sonography examinations. In future applications of automated breast sonography, the sonographer would likely play more of a supervisory role in the performance of these examinations or alternatively might be called upon to perform more focused diagnostic breast sonography studies, with a limited role in screening.
MRI has recently been recommended by the American Cancer Society (ACS) to screen women at very high risk of breast cancer. 10 Although highly sensitive, MRI is costly and does have some drawbacks, such as the risks from the required contrast media. MRI for breast cancer screening has also been characterized by lower specificity, as compared with mammography, with a higher rate of false positives, leading to further follow-up MRI and/or imaging-guided biopsy costs. For example, a study by Leach et al 11 reported MRI specificity of 81% compared with 93% specificity in mammography. Griebsch et al 12 reported MRI as having almost 4 times more recalls than mammography for women with high familial breast cancer risk, and 70% of the recalls did not involve cancer. Because of lower specificity and higher cost compared with mammography, MRI may not be optimal for breast cancer screening.
Automated breast sonography is an attractive supplement to mammography because it is well tolerated by patients and is relatively inexpensive. Previous studies have shown that automated breast sonography has excellent interobserver variability compared with handheld sonography for both benign and malignant lesions. 13 However, although contrast MRI breast studies are now regarded as the gold standard for breast cancer detection, there are few studies comparing automated breast sonography with MRI on biopsy-proven cases. The primary end point of this study was a comparison of automated breast sonography and MRI in detecting and diagnosing nonpalpable suspicious breast lesions in asymptomatic women with dense breast parenchyma. In our study, excellent interobserver agreement and similar morphologic features were seen in both benign and malignant lesions when comparing automated breast sonography with MRI. Automated breast sonography also offered the same multiplanar imaging capabilities as MRI, was well tolerated by participants, and was easily integrated into the breast imaging practice. Our institutional protocol now follows a three-step diagnostic algorithm and protocol for the workup of patients with mammographically dense breast parenchyma (Table 4). All patients with dense mammograms of greater than 50% breast density proceed to automated breast sonography and are subsequently referred for elective surgical evaluation for workup of BI-RADS category 4 and 5 lesions. Ideally, patient workup might also include the option of MRI as an intermediary step to intervention to stage positive breast cancer cases, as appropriate.
Proposed Workup of Dense Mammograms Using Automated Breast Sonography

Many women who are at increased risk of breast cancer do not meet the ACS criteria for annual MRI but could benefit from more effective screening than mammography alone. 14 The ease of use and cost at approximately $185 make automated breast sonography an attractive alternative to MRI for women with dense breasts, family or personal history of breast cancer, or other risk factors. Automated breast sonography is well accepted by participants because of less breast compression than mammography, as well as no exposure to ionizing radiation or contrast media administration.
Two limitations to this pilot study are noted. The first relates to the automated breast sonography technique itself. Automated breast sonography images were collected in the coronal imaging plane. However, 3D reconstruction in the axial, sagittal, or any off-axis planes was possible for any discovered possible abnormality. The technique itself is limited, as are other sonography techniques, in women with large breasts. Patient cooperation is also necessary as motion beyond quiet breathing will degrade the examination. Continuing future modification of the automated breast sonography system, including computer-assisted detection (CAD) capabilities, should standardize this modality in a way similar to current state-of-the art technologies, such as digital mammography and breast MRI. The second limitation of this study is the relatively small number (N = 24) of histologically proven cases using automated breast sonography with corresponding MRI studies. The reason for this in the present study is primarily reimbursement driven, as most insurance carriers regard biopsy as the direct end point of workup, rather than MRI characterization as an intermediary step. Another obstacle is that insurance carriers presently regard automated breast sonography as an experimental procedure, and therefore it is not reimbursable as a distinct and separate charge from handheld sonography.
Conclusions
These preliminary results showed that automated breast sonography and MRI are complementary diagnostic modalities in the mammographically dense breast screening population. Very similar morphologic assessment was seen comparing automated breast sonography to MRI evaluation of breast lesions, with excellent interobserver agreement between trained diagnostic radiologists experienced in multimodality breast imaging. There was comparable diagnostic reliability between automated breast sonography and MRI in differentiating benign from malignant breast lesions based on similar morphologic criteria. In summary, this pilot study offers a platform for further research in using the automated breast sonography procedure as part of a larger study of asymptomatic patients with mammographically dense breasts. In the future, automated breast sonography may be more integrated into a general diagnostic algorithm in a large screening population compared with breast MRI, which is significantly limited by cost and equipment requirements.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
