Abstract
Radiation is a standard of care in the treatment of locally advanced cervical cancer. Radiation consists of external beam radiation and brachytherapy to optimize local control and cure. High-dose-rate (HDR) brachytherapy is most commonly used in the United States. Advantages of HDR are outpatient treatment, elimination of exposure to medical staff, patient convenience, and the ability to modify the treatment because multiple fractions are used. Accurate placement of the tandem in the uterus is critical to maximize tumor control and minimize complications. Transabdominal sonography offers real-time imaging of the tandem placement, which allows the radiation oncologist to adjust the tandem position, thus ensuring correct dosimetry and treatment. This article discusses the etiology, diagnosis, and treatment of cervical cancer. This article further explains the importance of the brachytherapy technique, the role of transabdominal sonography, and how both can lead to a successful outcome for the patient.
Incidence, Causes, and Pathophysiology
In the United States, the annual incidence of cervical cancer is 12,200, with 4210 expected deaths for 2010. 1 The incidence in the United States is declining due to screening, early detection and treatment of preinvasive disease, and, potentially, the implementation of cervical cancer vaccines.
Almost all cervical cancer is caused by sexually transmitted diseases, particularly a few high-risk human papillomavirus (HPV) strains (subtypes 16, 18, 31, and 33).2–4 In fact, about 70% of cervical cancer is caused by HPV strains 16 and 18. A vaccine containing the seven most common HPV types (16, 18, 45, 31, 33, 52, 58) may potentially prevent nearly 90% of cervical cancer. 5 Once inside the cervix, HPV damages the cervical epithelial cells, resulting in dysplasia, a precancerous condition. 6 This condition can be detected with the Papanicolaou test (Pap test) and successfully treated. If cervical dysplasia is not detected and treated, neoplastic transformation may occur.
Risk Factors
Risk factors for cervical cancer include early onset of sexual activity, multiple partners, smoking, and, most important, HPV exposure. Vaccines currently exist and can potentially prevent the neoplastic transformation of HPV.7,8 Women from lower socioeconomic status are also more likely to have HPV and/or cervical cancer. This may be due to lack of screening. 9
Symptoms and Detections
Patients with cervical cancer often present with vaginal bleeding, dyspareunia, pelvic pain, and, when more advanced, weight loss, pain radiating down the leg, and back pain. Detection of cervical cancer begins with the use of the Pap test to screen for abnormal cervical cells. In addition, pelvic examinations with visual inspection are critical in checking for lesions, sexually transmitted disease (STDs), and other abnormalities of the cervix. Vaginal and cervical smears are also beneficial in testing for the presence HPV.
Once a lesion has been found, a punch biopsy, loop electrosurgical excision, endocervical curettage, or conization is usually completed to determine the lesion’s histopathological qualities. 10 Colposcopy is used to further determine if a subtle lesion is present by applying acetic acid to the cervix, which guides where the biopsy should be taken. If the lesion is cancerous, staging of the cancer is the next step.
Staging consists of a thorough history and physical examination, cervical biopsies, pelvic computed tomography (CT), and often a positron emission tomography (PET) scan to determine the extent of disease. Cystoscopy and sigmoidoscopy are performed as well if there is a suspicion for bladder and/or rectal invasion. Although staging is primarily clinical (determined by pelvic examination), radiographic findings of pelvic and/or para-aortic adenopathy influence treatment and prognosis. Staging and treatment options are detailed in Table 1.
Stage and Treatment Recommendations 11
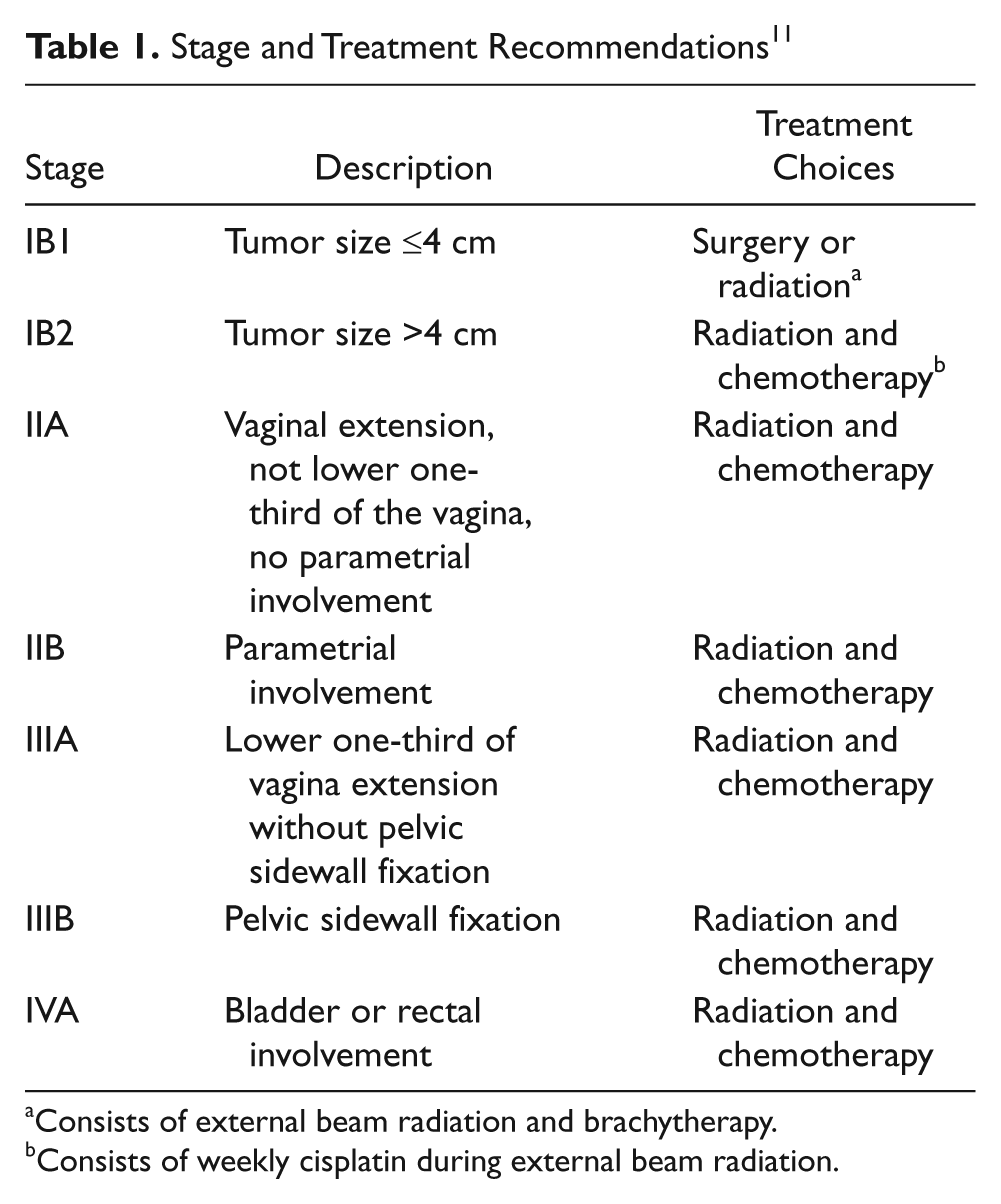
Consists of external beam radiation and brachytherapy.
Consists of weekly cisplatin during external beam radiation.
Treatments
Cervical cancer can be treated with surgery, radiation, or a combination of both. 11 Chemotherapy is routinely given with radiation for advanced stages of cervical cancer. About 15% to 20% of surgical patients require adjuvant radiation because of high-risk pathologic features such as positive lymph nodes, positive margins, and/or deeply invasive disease. 11
Surgery and radiation yield an equivalently high cure rate for stage I disease (tumor size <4 cm).8,9 Surgery consists of a radical hysterectomy and pelvic lymph node dissection. The ovaries and fallopian tubes are preserved in younger patients but typically removed in postmenopausal patients. A radical trachelectomy (removal of the cervix only) is sometimes offered to younger patients to preserve fertility. Surgery is often the treatment of choice for younger patients because ovarian function is preserved and sexual function is better.11,12 Potential side effects from surgery may include urinary incontinence, ureteral strictures, small bowel obstructions, and vaginal shortening. 13
Radiation therapy targets the DNA of the cancer, resulting in a mitotic death. Radiation therapy consists of external beam radiation in combination with brachytherapy (internal radiation).11,14 Potential side effects of radiation include skin irritation, fatigue, nausea and vomiting, cystitis, vaginitis, diarrhea, ureteral strictures, small bowel obstructions, fistulas, and sexual dysfunction. 15
Concurrent chemoradiotherapy is the standard of care for more advanced cervical cancer (stage IB2–IVA disease; Table 1). Cisplatinum is the most commonly used chemotherapy agent. It is given intravenously weekly and sensitizes the cancer cells to radiation. Combining chemotherapy with radiation improves the cure rate by nearly 10%. 16 Side effects of chemotherapy include myelosuppression, nausea, vomiting, diarrhea, and weight loss.
External beam radiation is delivered by a linear accelerator, where high-energy photons are used to treat the pelvic lymph nodes as at least 20% to 30% of patients have microscopic nodal disease and to shrink the cervical cancer in preparation for brachytherapy. 14 Twenty-five to 28 external beam treatments are routinely given to accomplish these goals (45–50.4 gray [Gy]).
Treatment Using Low-Dose-Rate Brachytherapy
Brachytherapy is radiation delivered within the tumor and is a mandatory part of curative therapy. 14 Brachytherapy is delivered by one of two main methods: low-dose-rate (LDR) brachytherapy or high-dose-rate (HDR) brachytherapy.17,18 Technically, LDR is radiation delivered at a dose rate <2 Gy/h, whereas HDR is delivered at >12 Gy/h, similar to the dose rate of linear accelerators. Historically, cervical cancer was treated with LDR in which gynecologic applicators and cesium sources were placed in the uterine cavity for 48 to 72 hours. This procedure was often repeated again one week later.
The advantage of LDR brachytherapy is that by extending the treatment over time, radiation-affected normal tissues will have more time to repair the DNA damage.14, 19 Disadvantages of this approach include patient inconvenience and discomfort as applicators are in for extended periods of time (two to three days). Therefore, the patient is on strict bed rest and cannot move as applicator movement may result in inadequate radiation delivery. Other disadvantages include the risk of thromboembolism, radiation exposure to hospital personnel, and applicator movement.
Treatment Using HDR Brachytherapy
HDR brachytherapy is delivered on an outpatient basis and requires three to five insertions during the latter part of external beam radiation. Advantages of HDR include patient convenience, outpatient therapy, no applicator movement, and elimination of radiation exposure to hospital personnel. A potential disadvantage of HDR is lack of normal tissue repair during radiation because the treatments are given over a short period. This potential radiobiologic disadvantage is overcome through fractionation (three to five treatments), aggressive use of packing to displace the bladder and rectum, and rigid immobilization because treatment times are short compared to LDR (see Petereit et al., 14 Petereit and Fowler, 18 and Petereit19,20 for more details). Of critical importance, the total dose is decreased to adjust for the dose rate effect. When the dose rate is increased, the percentage of cells killed also increases, both for the cancer and normal cells. The dose rate effect was not adequately recognized in the early 1980s, with one study reporting much higher complication rates because the total dose was not lowered. 21 For example, 6 Gy × 5 (total dose 30 Gy) to the cervical region given via HDR is equivalent to 40 Gy given through LDR. 18 Long-term data have shown no difference in either cure or complication rates between both methods of brachytherapy. HDR is now the most common method of brachytherapy delivery for reasons discussed above.
There is a positive correlation between number of cervical cancer patients treated and outcome. The Patterns of Care Study reported that patients treated at low-volume facilities were significantly more likely to be improperly dosed and have a treatment span greater than 70 days. In addition, survival rates were lower for facilities that treated fewer than six cervical cancer patients a year versus those facilities that treated more than six a year. 22 In other words, the outcome of the brachytherapy treatment is also dependent on the experience and skill level of the radiation oncologist.
Uterine perforation rates of 2% to 14% have been reported.23,24 Watkins et al. 25 reported no uterine perforations when sonography was used to guide the tandem placement. Accurate placement of gyn applicators (tandem and ovoids; Figure 1A) at the time of brachytherapy is critical to achieving an adequate dose distribution and ultimate cure. 26 This process will be further described.
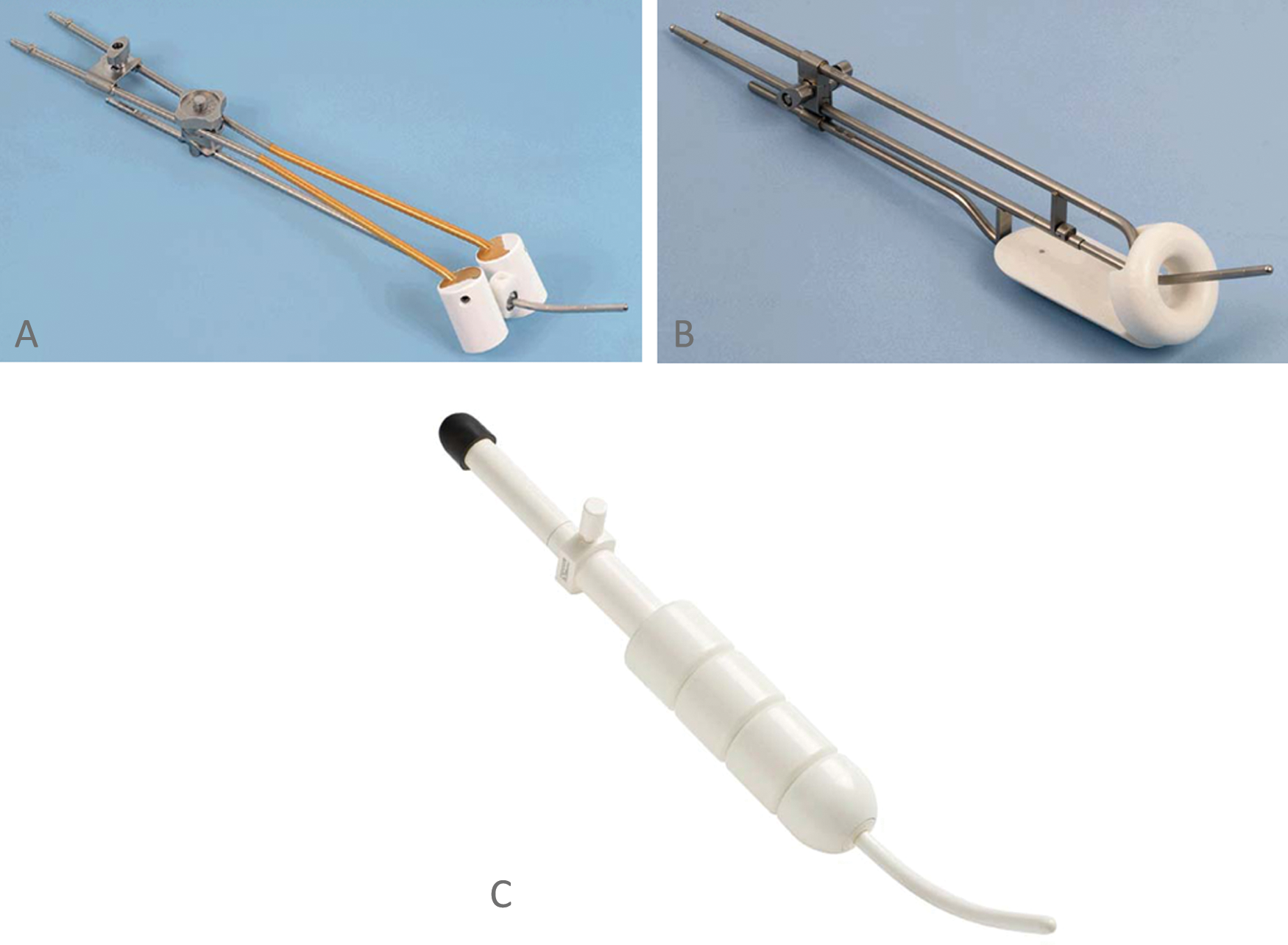
(A) Tandem and ovoids, (B) tandem and ring, and (C) tandem and cylinders. Figures are courtesy of Nucletron and are used with permission.
Brachytherapy Procedure
Proper tandem placement is critical to deliver a tumoricidal dose of radiation while sparing the adjacent normal tissues: small bowel, bladder, and rectum. The possibility of a uterine perforation is a major concern and can often happen because the cervix and lower uterine segment are friable after external beam radiation and tumor necrosis. If the radiation is delivered through a perforate uterus, a small bowel obstruction or fistula may develop, which is debilitating and potentially life threatening. 27 More commonly, the tandem may not be midline and as a result is too close to either the bladder or rectum: in either case, the tumor will be underdosed and the normal tissue will be overdosed—a double jeopardy phenomenon. When these significant complications develop, surgical intervention may be required.
The HDR procedure is often performed under a general anesthetic but can also be done using intravenous (IV) conscious sedation. Once the patient is properly sedated, the radiation oncologist performs the procedure under sterile conditions. A gynecologic oncologist frequently assists the radiation oncologist as well. Gynecologic applicators used include tandem and ovoids (Figure 1A), tandem and ring (Figure 1B), and tandem and cylinders (Figure 1C). Applicator selection depends on the patient’s anatomy, extent of disease, and physician preference. The uterine cavity is initially sounded (cannulated and measured) and often requires dilatation to facilitate sounding. Next a treatment tandem is placed in the uterus with a cervical stopper secured near the cervix to prevent a perforation (Figure 2). Transabdominal sonography is performed to facilitate uterine sounding and to confirm midline positioning of the tandem.
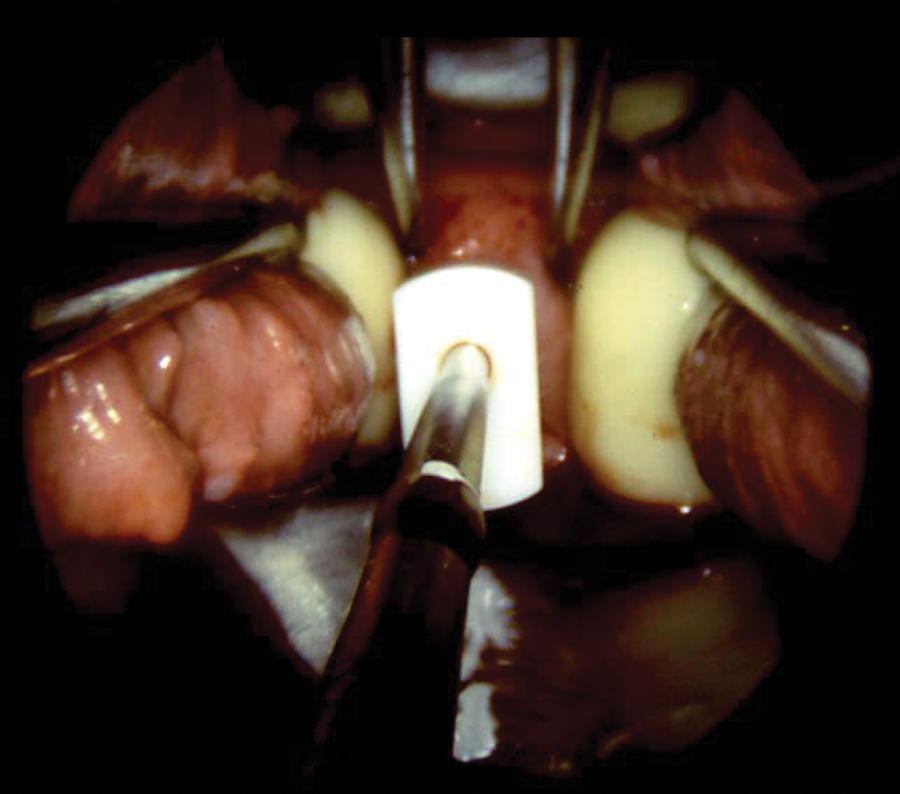
Intravaginal view of a tandem and ovoid insertion.
Transabdominal sonography offers real-time imaging of the tandem placement, which allows the radiation oncologist to immediately assess the tandem position. If the uterus is perforated, the tandem is repositioned under sonographic guidance until it is midline. 27 Sonography is also useful for determining where the dose should be prescribed as measurements are routinely taken of the cervix, uterine thickness, and distance from the tandem tip to the uterine serosal surface/bowel interface (Figure 3).
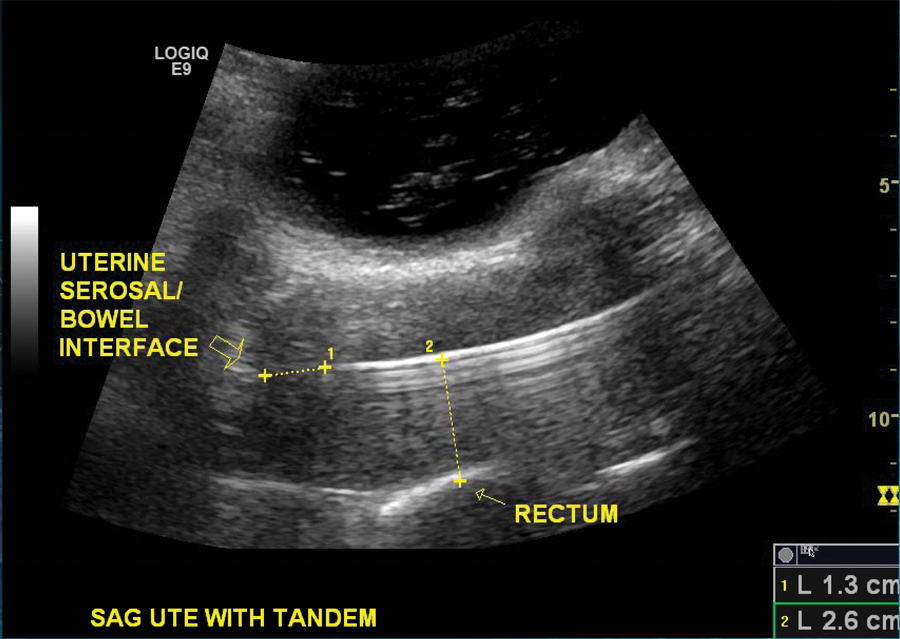
Sonographic longitudinal view of a well-positioned tandem in the uterus.
Techniques for Sonographic Evaluation of Tandem Placement
The anesthesiologist should position the patient’s arm at either 90 degrees or “tuck” the arm in so the sonographer has better access to the patient. The sonographer may need to use a step stool as the operating room table is raised to the desired height of the radiation oncologist.
For optimal transabdominal imaging of the insertion, the bladder is filled with enough normal saline to completely visualize the uterine fundus. For patients with an anteverted uterus, filling the bladder will also facilitate uterine sounding as the uterus will straighten out. If the bladder is fully distended and does not cover the fundus, the sonographer can move the transducer more inferiorly and angle superiorly to view the fundus. It is imperative that the sonographer be able to show the entire uterus to confirm correct tandem placement and accurate measurements.
The sonographer images the tandem as it enters the cervix and uterus to ensure that it is midline. The uterus appears as a hypoechoic pear-shaped organ with a hyperechoic line in the middle, which represents the endometrial stripe (Figure 3). The tandem will appear as a bright hyperechoic line in the endometrium with a ring-down artifact. B-mode imaging should be optimized by using the highest frequency that the patient’s body habitus will permit. The use of harmonics will facilitate accurate identification of the metal tandem. Some sonography machines have special settings to aid in needle biopsies. These settings are also applicable for imaging tandem placement. For example, using the max intensity crossbeam on the GE Logic E9 (GE Healthcare, Piscataway, New Jersey) improves image quality. Decreasing the dynamic range to about 65 also helps enhance the hyperechoic property of the tandem, making it easier to identify.
The first measurement taken is the distance from the tandem tip to the uterine serosal surface/bowel interface, which is the most superior part of the fundus (Figure 3). This measurement should be a minimum of 1 cm. Other measurements may be requested such as the distance between the tandem and the rectum at various points along the uterine/rectal interface. The radiation oncologist may ask for tumor dimensions of the cervix as well.
Imaging and Radiation Planning
Once the gyn applicators are well positioned, packing is placed above and below the ovoids to minimize the dose to the bladder and rectum, respectively (Figure 4). Imaging of the insertions is obtained by an X-ray (anterior-posterior and lateral view) or by a CT scan of the pelvis. In either case, computerized planning is performed next to determine how long the iridium isotope will travel inside the tandem and each ovoid to deliver the radiation dose. The dose is prescribed to point A: a historical point that represents where the uterine artery and ureter cross (paracervical triangle). It is 2 cm above the external cervical os and 2 cm lateral (Figure 5a,b). The radiation distribution is pear shaped, mimics the geometry of the cervical cancer, and delivers a high dose over a few minutes. Upon completion of the radiation, the applicators are removed and the patient goes homes once medically stable.
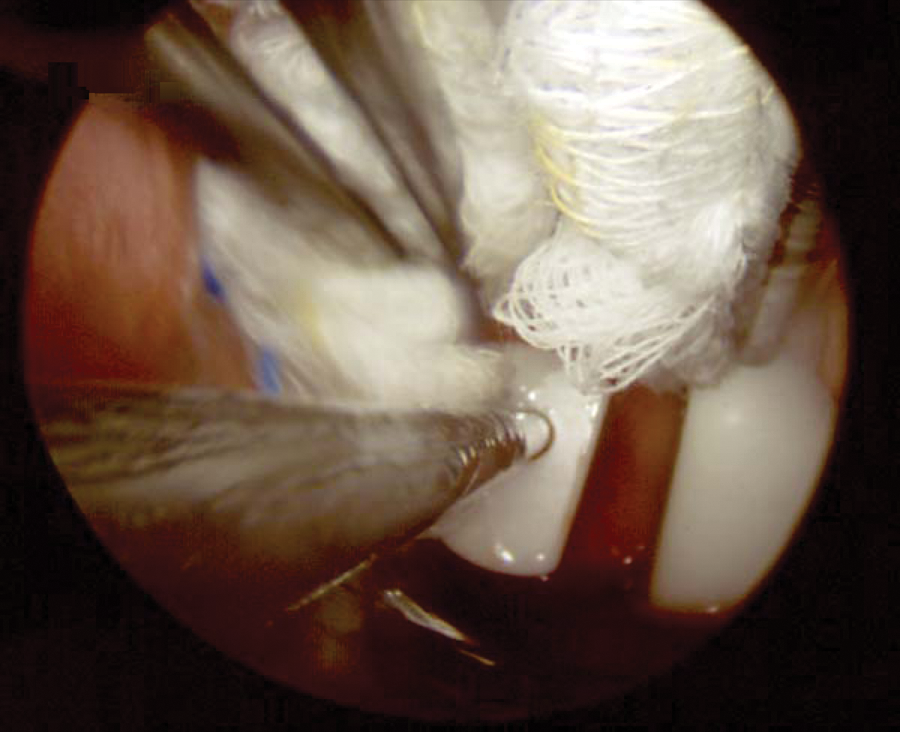
Intravaginal view of a tandem and ovoid insertion with packing to displace the bladder.
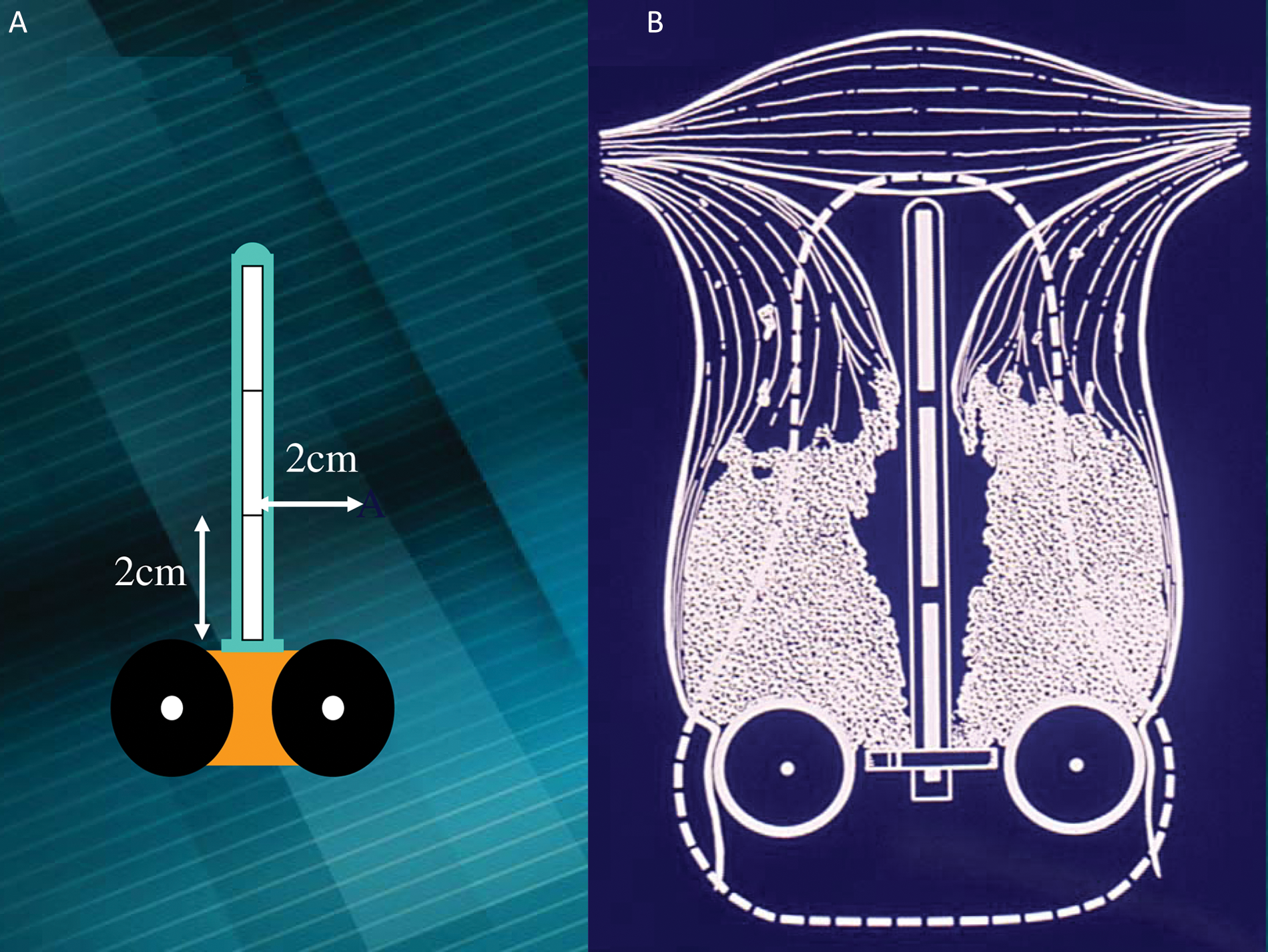
(A, B) Definition of point A and the radiation dose distribution.
Interstitial Brachytherapy
The brachytherapy techniques described so far are intracavitary. For patients with extensive parametrial and/or vaginal involvement that does not adequately regress after external beam radiation, interstitial brachytherapy is used to more adequately cover the tumor volume (Figure 6a,b). Interstitial brachytherapy is a technique where several catheters are inserted into the vagina to improve the radiation dose distribution. These catheters are sonographically guided, very similar to how Bubb and Hooper 28 described the use of sonography for interstitial breast brachytherapy. During gynecologic interstitial brachytherapy, the sonographer guides the radiation oncologist by imaging the catheters transabdominally so they are placed parallel to one another and to determine the most cephalad extent to prevent perforation of the adjacent bowel.
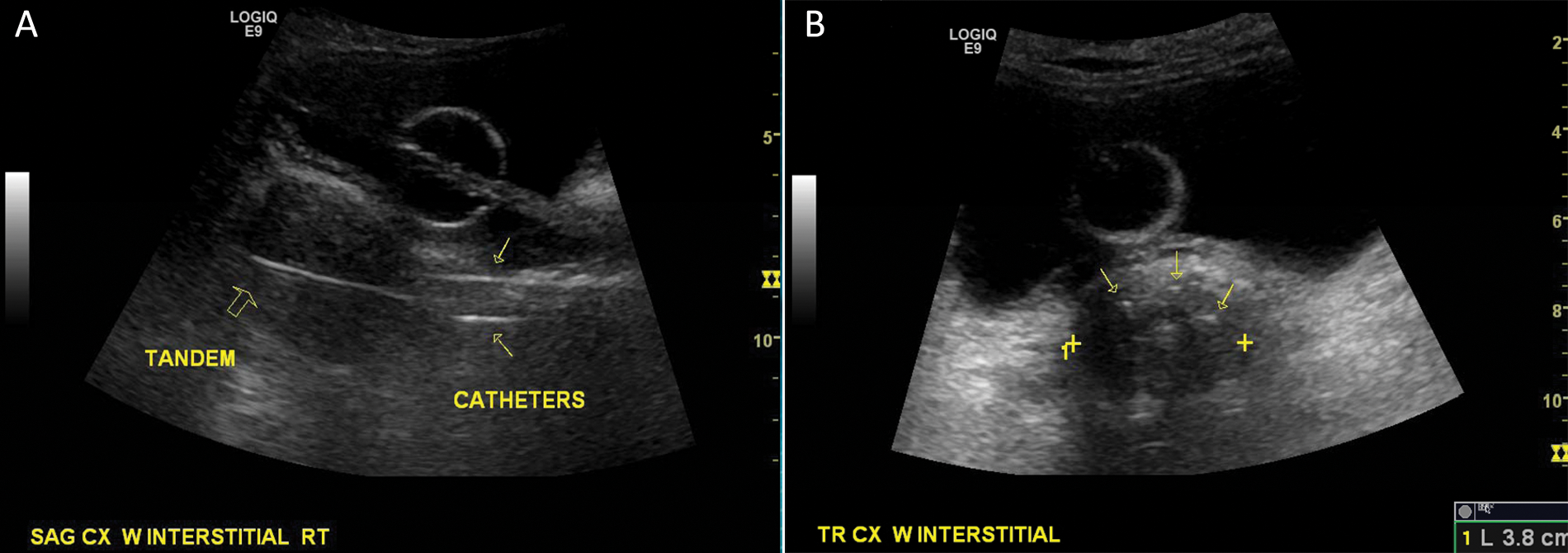
Sonographic view of a vaginal interstitial catheter placement: (A) longitudinal view and (B) axial view.
Discussion
The incidence and mortality rates of cervical cancer continue to decline in the United States due to education and screening. For women who are not surgical candidates, chemoradiotherapy yields relatively high cure rates, even for stage III disease—unlike other disease sites such as lung cancer, where cure rates are less than 20% for advanced stages of disease. This is in part due to the patterns of spread with cervical cancer, and the ability to significantly dose escalate with brachytherapy. Chemoradiation using fractionated HDR brachytherapy is now the standard of care for treating cervical cancer patients whose tumors are >4 cm (Table 1). HDR brachytherapy allows for outpatient treatment, elimination of radiation exposure to hospital personnel, adjustment in the radiation plan and dose as the tumor regresses, and patient convenience.
Similar to other surgical procedures, the outcome is better if cervical cancer patients are treated at higher volume institutions by an experienced radiation oncologist. 20 In addition, transabdominal sonography improves the efficacy rate of HDR brachytherapy by facilitating accurate tandem placement and radiation delivery while minimizing uterine perforation rates. 25 Sonography provides immediate feedback to the radiation oncologist. If uterine perforation or improper placement of the tandem is detected, it can be corrected immediately, leading to a successful outcome for the patient.
Conclusion
The incidence and mortality rates of cervical cancer in the United States are declining due to education, screening, and vaccinations against HPV. Surgery and/or radiation are highly effective treatments for those women who are diagnosed with cervical cancer. Surgery and radiation are equally effective treatments for women with earlier stage cervical cancer, whereas chemoradiotherapy using brachytherapy is an established standard of care for those with more advanced stages of disease. Brachytherapy is an essential part of the radiation treatment and is associated with better outcomes when used at higher volume facilities. Transabdominal sonography plays a critical role in accurate brachytherapy placement, radiation delivery, and patient outcome by facilitating proper tandem placement and decreasing uterine perforation rates.
Footnotes
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The authors disclosed receipt of the following financial support for the research and/or authorship of this article: Work funded by in part by National Institutes of Health grant (RFA 1U56CA99010-01).
