Abstract
Ewing sarcoma is a malignant tumor that typically affects the appendicular skeleton; skull involvement is rare. Its clinical and radiological features are non-specific, making its diagnosis challenging. Confirmation is primarily achieved through immunohistochemistry. We report the case of a child who presented with facial swelling. MRI revealed an aggressive lesion involving the right sphenoid and temporal bones. Thoracic and abdominal CT scans showed no evidence of distant metastases. Biopsy of the lesion confirmed the diagnosis of Ewing sarcoma.
Keywords
Introduction
Ewing sarcoma (EWS) is a malignant round-cell bone tumor and the second most common bone malignancy in children, after osteosarcoma.1,2 It most frequently affects the long bones and pelvis. 3 In this report, we present a rare case of Ewing sarcoma localized in the spheno-temporal region. Such atypical presentations may lead to delays or misdiagnoses, which can significantly impact prognosis and treatment, as they can mimic other conditions and pose considerable diagnostic challenges. This is illustrated by a previously reported case of Ewing sarcoma of the ulna mimicking a typical lateral epicondylitis in a young patient with elbow pain without recognizing red flag symptoms such as persistent pain and swelling. 4
Case Report
A 6-year-old male presented with progressive swelling in the right temporal region, with no history of trauma or infection. Clinical examination was notable only for a non-pulsatile, fixed, and tender mass measuring approximately 6 cm in diameter, which was painful on palpation. The overlying skin appeared normal, with no signs of inflammation or infection. No neurological deficits or other significant findings were noted. Routine blood tests, including complete blood count and inflammatory markers, were within normal limits
The clinical differential diagnosis for this case includes several conditions that may present with swelling in the temporal region. These include osteomyelitis, which typically manifests with signs of infection, as well as Langerhans cell histiocytosis, fibrous dysplasia, primary bone tumors (such as osteosarcoma and Ewing sarcoma), and metastatic tumors.
Given the progressive nature of the swelling and the absence of trauma or infection, imaging played a crucial role in identifying the underlying cause. Head and neck MRI was performed to evaluate the nature, extent, and precise location of the lesion. The imaging revealed an expansive process involving the right sphenoid and temporal bones, with irregular contours in some areas. The lesion showed intermediate signal intensity on T1-weighted, T2-weighted (Figures 1 and 2), and FLAIR sequences (Figure 3). No diffusion restriction was observed on diffusion-weighted imaging (Figure 4). Significant contrast enhancement was noted following contrast administration (Figures 5 and 6). Loco-regional evaluation revealed invasion of the meninges and the outer wall of the parotid gland, along with compression of the temporal lobe.

Cerebral MRI showing a spheno-temporal lesion (arrow) with intermediate signal intensity on T2- weighted axial images. The lesion presents irregular contours in some areas, invades the meninges, exerts mass effect on the temporal lobe, and displaces the lateral wall of the right orbit.

Cerebral MRI showing a spheno-temporal lesion (arrow) with intermediate signal intensity on T2- weighted coronal images with irregular contours in some areas. The lesion invaded the meninges and compressed the temporal lobe.
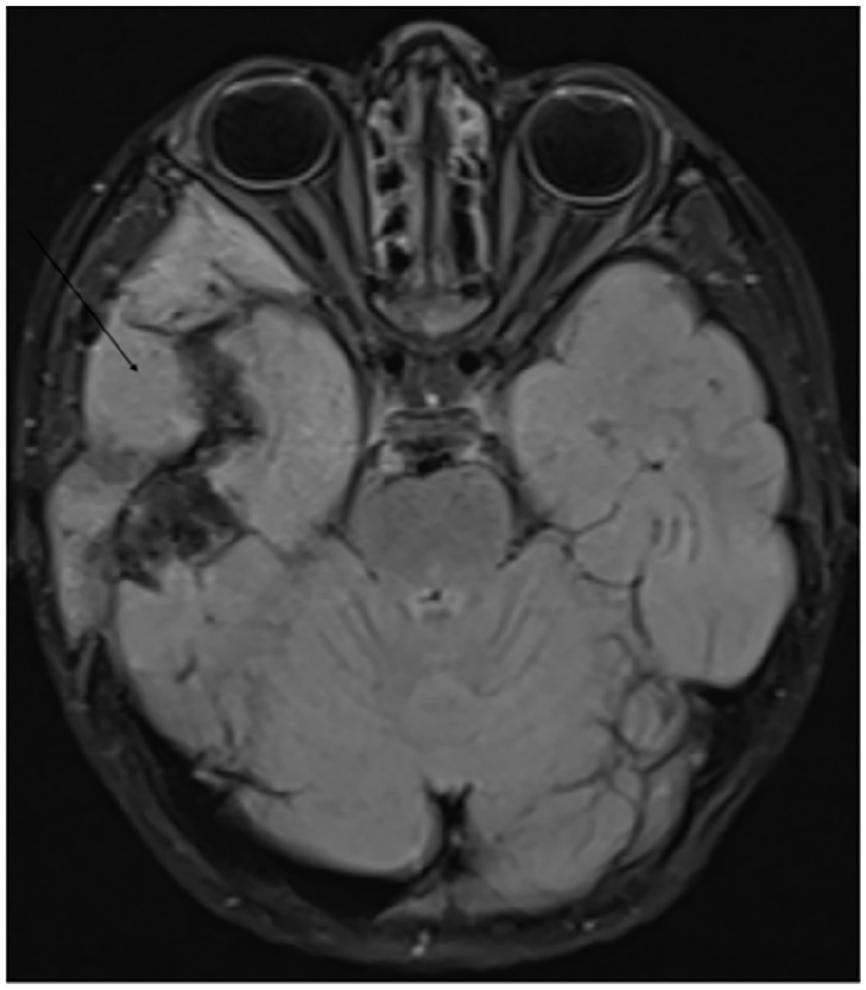
Cerebral MRI showing a spheno-temporal process (arrow) with intermediate signal intensity in Flair.

Cerebral MRI showing a spheno-temporal process (arrow) with no restriction in diffusion.
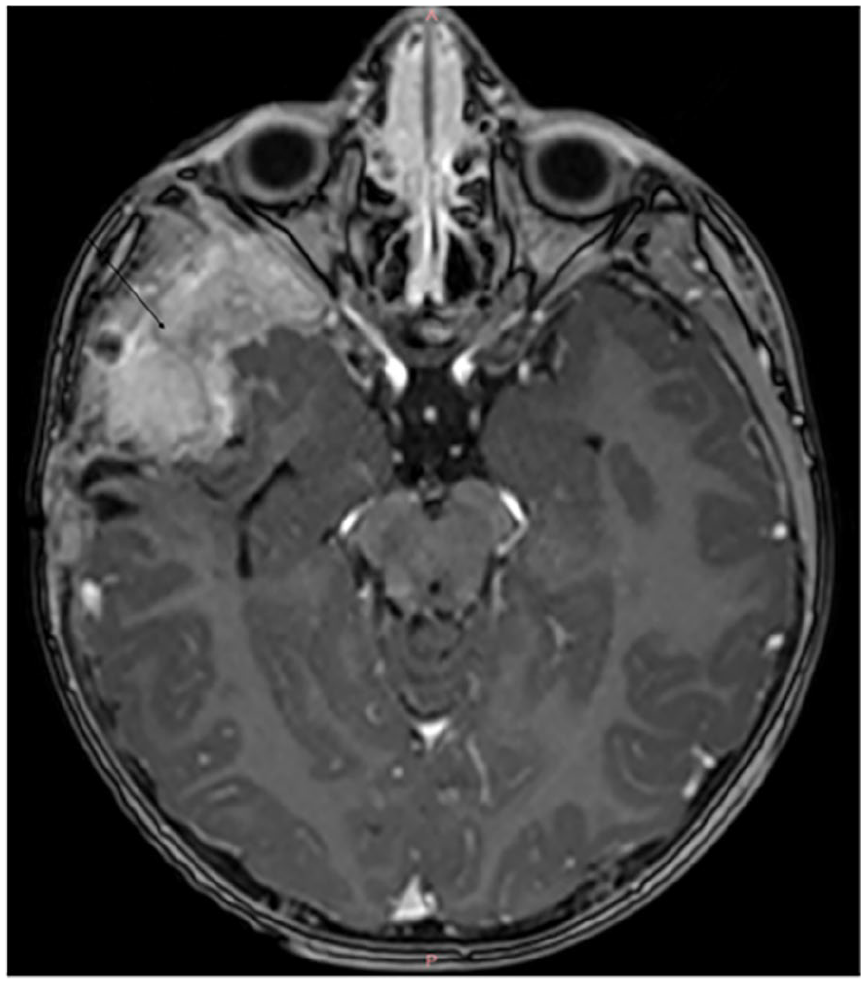
Cerebral MRI showing a spheno-temporal process (arrow) with significant enhancement after contrast injection (axial slice).

Cerebral MRI showing a spheno-temporal process (arrow) with significant enhancement after contrast injection (sagittal slice). At this stage, in the clinical context of our patient, several diagnoses can be considered, including malignant tumors such as osteosarcoma, Ewing sarcoma, lymphoma, as well as inflammatory lesions like Langerhans cell histiocytosis.
Given the location and characteristics of the lesion, a primary bone tumor or soft tissue mass was suspected.
Biopsy of the swelling revealed a neoplastic proliferation of small to medium-sized, non-cohesive round cells. The nuclei were round with finely dispersed chromatin, and the cytoplasm was clear and poorly defined. Immunohistochemistry showed positivity for CD99. These findings are consistent with a diagnosis of Ewing sarcoma.
To assess for distant metastasis, a CT scan of the chest and abdomen was performed, which showed no signs of distant metastasis.
Following confirmation of the diagnosis, the patient was started on a chemotherapy protocol, consisting of doxorubicin, vincristine, ifosfamide, and cyclophosphamide, with the primary goal of reducing tumor size and preventing metastasis. Given the tumor’s aggressive nature, radiation therapy was also considered to target residual disease and minimize the risk of recurrence. Surgical intervention was not initially planned. The patient is currently under close surveillance, including regular imaging studies such as MRI to monitor tumor response and detect distant metastases. Pain management strategies and supportive care were implemented to address treatment-related side effects and complications, aiming to ensure the best possible outcome
Discussion
Primary Ewing sarcoma is the second most common pediatric bone tumor.1,2 However, skull involvement is rare, accounting for only about 1% of all Ewing sarcoma cases. 3 Among these rare cranial cases, Temporal region is the most commonly affected, followed by the parietal and occipital bones. Involvement of the sphenoid bone is less frequent. 3 The disease predominantly affects children and young adults. 5
The clinical presentation of Ewing sarcoma is non-specific and typically involves a progressively enlarging, painful swelling, as seen in the case we report. 6 The absence of specific clinical features poses a diagnostic challenge, as it can be confused with other conditions such as osteomyelitis, primary bone tumors, Langerhans cell histiocytosis, fibrous dysplasia, and metastatic tumors. Although some patients may exhibit signs of increased intracranial pressure, such as nausea, vomiting, headache, and papilledema, along with neurological deficits when there is intracranial extension, our patient showed no neurological symptoms. 6
Biologically, anemia and leukocytosis can be found in Ewing sarcoma cases, however, these are non-specific findings and do not contribute significantly to the diagnosis. 7,8
Imaging plays a crucial role in the diagnosis and staging of Ewing sarcoma. Conventional radiography typically reveals aggressive osteolytic lesions with a wide zone of transition and a permeative pattern. 9 Complex periosteal reactions, such as lamellated (57%) and spiculated (28%) patterns, are commonly observed. CT scans are more sensitive than radiographs for detecting bone destruction. 10 The most frequent CT finding is an isodense lesion with heterogeneous enhancement. 10
Other imaging modalities, such as spectral CT, offer advantages over conventional CT. However, no studies have yet been published specifically on the use of spectral CT in Ewing sarcoma. Nevertheless, several studies have demonstrated the benefits of spectral CT in malignant diseases. For instance, in patients with bone marrow involvement, spectral CT can accurately detect both focal bone marrow lesions larger than 1 cm and the pattern of involvement, with accuracy comparable to MRI, including precise identification of osteolytic bone lesions. 11
Magnetic resonance imaging (MRI) is the most effective imaging modality for assessing local staging in Ewing sarcoma, as it provides precise visualization of tumor extent and can detect skip lesions. Typically, Ewing sarcoma tissue appears hypointense on T1-weighted images and hyperintense on T2-weighted images, with a prominent soft tissue component observed in approximately 96% of cases. 9 The tumor also shows enhancement following contrast administration. 12 Focal areas of cortical bone destruction are common, occurring in about 92% of cases. 9
Recent advances in medical imaging, particularly radiomic MRI, offer promising opportunities for improving the diagnosis and management of sarcomas. Although radiomic studies focusing on bone sarcomas—such as Ewing sarcoma—remain limited, recent research on soft tissue sarcomas 13 has demonstrated the potential of radiomic MRI to enhance patient stratification and prognostic evaluation. Notably, radiomic features extracted from T2-weighted (T2w) sequences have revealed significant differences between low- and high-proliferative sarcomas, suggesting greater intratumoral heterogeneity in tumors with higher proliferative activity.
Although the imaging characteristics of Ewing sarcoma can be suggestive, they are not pathognomonic. Several other conditions—including metastatic tumors such as neuroblastoma, lymphoma, and osteosarcoma, as well as benign lesions like Langerhans cell histiocytosis, meningioma, desmoplastic small round cell tumor, and plasmacytoma—can present with similar imaging features. This was shown in our case, where imaging findings were suggestive of a malignant lesion but did not allow for a definitive etiological diagnosis, particularly given the unusual location.
Therefore, biopsy and immunohistochemical analysis are essential for establishing a definitive diagnosis. Histologically, Ewing sarcoma is characterized by small, round cells with a high nuclear-to-cytoplasmic ratio and clear, poorly defined cytoplasm. Immunohistochemistry typically demonstrates strong membranous positivity for CD99, a highly sensitive marker for Ewing sarcoma. 14 Additional markers that may show positivity include neuron-specific enolase, synaptophysin, and S-100 protein. Nuclear staining with anti-FLI1 antibody is also considered specific for tumors of the Ewing family. Furthermore, the presence of the t(11;22)(q24;q12) chromosomal translocation—detected in up to 85% of cases—provides strong molecular confirmation of the diagnosis.15,16
When feasible, surgical resection is the preferred treatment for Ewing sarcoma. In cases where surgery is not feasible, radiotherapy combined with multi-agent chemotherapy—typically using the VAC (vincristine, doxorubicin, cyclophosphamide) regimen alternating with IE (ifosfamide, etoposide)—represents the standard therapeutic approach.17,18 The use of multi-agent chemotherapy is essential for reducing the risk of metastasis and recurrence. 19
Conclusion
Skull involvement in Ewing sarcoma is rare, particularly in the sphenoid bone. This case highlights the importance of including Ewing sarcoma in the differential diagnosis of unexplained swelling in atypical anatomical locations. MRI remains the imaging modality of choice, while definitive diagnosis relies on biopsy and immunohistochemical analysis.
Footnotes
Acknowledgements
I would like to express my gratitude to my professors and all the colleagues who participated in the completion of this work.
Ethics Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent to Participate
Written informed consent was obtained from the patient and the legal authorized representative (LAR) for their anonymized information to be published in this article.
Consent for Publication
Written informed consent was obtained from the patient and the legal authorized representative (LAR) of the patient for the publication of this case report.
Author Contributions
TO: Contributed to conception and design; Contributed to analysis; Drafted the manuscript; Gave final approval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy. BR: Contributed to conception and design; Contributed to analysis; Agrees to be accountable for all aspects of work ensuring integrity and accuracy. BL: Critically revised the manuscript; Gave final approval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy. NA and LC: critically revised the manuscript; Gave final approval; Agreed to be accountable for all aspects of work ensuring integrity and accuracy. SEL: Contributed to acquisition, analysis, or interpretation; Critically revised the manuscript; Gave final approval.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
