Abstract
Purpose:
This study was planned to assess and compare the effectiveness of Dente91 DB toothpaste and conventional toothpaste (Colgate Total) on salivary glucose and pH in Indian patients with type 2 diabetes mellitus.
Methods:
This was a randomized, open-label, single-center, active-control, parallel-group clinical trial. Adult patients with established type 2 diabetes mellitus (HbA1c >8%) on treatment for ≥3 months were randomized to twice daily brushing with either Dente91 DB toothpaste or conventional toothpaste (Colgate Total) for 8 weeks. The primary endpoints were to assess the change in salivary glucose level and salivary pH from baseline to the end of 8 weeks.
Results:
Of 54 patients screened, 50 were enrolled in the study (25 in each group). At the end of 8 weeks, the mean change in salivary glucose level was significantly better in the Dente91 DB toothpaste group compared to the conventional toothpaste group (−3.20 ± 1.90 mg/dL vs −0.60 ± 1.76 mg/dL; P < .0001). The mean change in salivary pH did not differ significantly between Dente91 DB toothpaste and conventional toothpaste (0.32 ± 0.34 vs 0.17 ± 0.23; P = .09). The improvement in saliva flow rate was significantly better with Dente91 DB toothpaste (0.07 ± 0.03 mL/min vs 0.03 ± 0.03 mL/min; P < .0001), while the mean change in gingival index, plaque index, and dental hypersensitivity was not statistically different between the two groups at the end of 8 weeks.
Conclusions:
In patients with type 2 diabetes mellitus, Dente91 DB toothpaste was better than conventional toothpaste at reducing salivary glucose levels after 8 weeks. Dente91 DB toothpaste demonstrated significant improvement in salivary pH level; however, it was not better compared to conventional toothpaste.
Introduction
Diabetes mellitus, a chronic and complex metabolic disorder, virtually affects all organs and tissues of the body including the oral cavity, manifesting several complications like xerostomia, periodontitis, and tooth decay. Alteration in an oral environment in diabetes mellitus plays a significant role in the pathogenesis of such complications. Blood glucose level is positively correlated with salivary glucose level in diabetes mellitus. 1 Apart from high salivary glucose, patients with diabetes mellitus also have low salivary flow rate and pH; and these parameters adversely influence the periodontal status in diabetes. 2
Maintaining oral hygiene plays a vital role in preventing complications of oral cavity. Brushing twice daily for two minutes using a toothbrush with soft bristles is the recommended oral hygiene practice. 3 Conventional fluoride-based toothpastes are extensively studied for maintaining oral hygiene in healthy population 4 ; however, their effectiveness is not adequately studied in at-risk populations like diabetes mellitus with distinct oral cavity profile like high salivary glucose, low salivary pH, and poor salivary flow rate. Although numerous toothpastes containing ingredients with potential benefits for diabetes mellitus are available in India5-7; the literature proving their benefits, especially in comparison to conventional toothpaste is lacking.
Dente91 DB toothpaste is a specially designed formulation with endogenous ingredients (such as lactoferrin and nano-hydroxyapatite) and devoid of sodium lauryl sulfate, fluoride, and paraben. 8 In-vitro studies of Dente91 DB toothpaste have demonstrated numerous benefits like reduction in salivary glucose, prevention of xerostomia, antimicrobial activity against oral pathogenic microorganisms, survival of good oral microorganisms, accelerated remineralization and healing, decreased hypersensitivity and lastly, protection against dental caries/tooth decay; however, the effectiveness in human is not known. 9 To confirm these findings in a clinical setting, the present pilot study was planned to assess the effectiveness of Dente91 DB Toothpaste and compare it with conventional toothpaste (Colgate Total) among Indian patients with type 2 diabetes mellitus.
Materials and Methods
Study Design
This randomized, open-label, active-controlled, parallel-group, and comparative study was conducted at a single center (Shree Kanhoba Dental Clinic, Pune, India) in India after obtaining approval from the Royal Pune independent ethics committee (Registration No: ECR/45/Indt/MH/2013/RR-19). The study was conducted as per the study protocol in accordance with the Good Clinical Practice guidelines of the International Council for Harmonisation, ethical guidelines by ICMR, and other applicable Indian regulations. It was registered prospectively on CTRI (Reg No: CTRI/2023/ 07/055156). The written informed consent was obtained from all the patients before participation in the study.
Study Population
Eligible patients of either gender aged 18 to 65 years with type 2 diabetes mellitus (HbA1c >8%) on treatment for at least the last 3 months and having a minimum of 20 functional teeth in the oral cavity were enrolled in the study. Patients with systemic diseases (other than diabetes) which can hamper study participation as per investigator’s discretion; habits such as betel chewing, tobacco chewing, smoking, and alcohol consumption; history of systemic antibiotic use during the last 4 months; use of orthodontic appliance; or taking supplemental ascorbic acid were excluded.
Treatment Groups
After confirmation of the eligibility, patients were randomized in a 1:1 ratio to either the Dente91 DB Toothpaste group or the conventional toothpaste (Colgate Total) group as per a computer-generated randomization list. Patients were provided with assigned toothpaste during randomization. Patients were advised to brush their teeth with their respective toothpaste using a soft toothbrush for approximately 2 minutes at least twice daily for 8 weeks. They were also advised to brush at the correct angle for plaque removal, but not too hard to be careful with the soft tissues in the mouth. There were a total of three visits for each patient as follows: screening visit, randomization/baseline visit (Day 1), and end of study visit (at the end of Week 8; Day 56 ± 2).
Primary and Secondary Endpoints
The primary endpoints were a change in salivary glucose levels and a change in salivary pH from baseline to the end of 8 weeks. The secondary endpoints were changes in observation for Dry Mouth (by checking saliva flow rate [Sialometry]), gingival index (by Loe and Silness 10 ), plaque index (Silness-Löe Index 10 ), dental hypersensitivity (Schiff sensitivity score 11 ) from baseline to end of 8 weeks. All baseline assessments were performed before the first use of toothpaste during the randomization visit. The safety endpoint included the incidence of adverse events, treatment-emergent adverse events, and serious adverse events reported during the study.
Salivary Sample Collection and Analysis
Saliva collection was performed in the early morning. One hour before the collection of saliva, the patients were advised to refrain from eating, drinking, or engaging in other oral hygiene procedures. Patients were asked to hold the saliva and collect/spit it into a sterile container containing sodium fluoride until 1.5 ml of saliva was collected. After collection, samples were stored and transferred to the laboratory as soon as possible for analysis. Each unstimulated 1 mL saliva sample was centrifuged at 3000 rpm for 20 min in the semi-automatic analyzer. The clear supernatant was processed immediately for salivary glucose estimation using the GOD-POD kit. 12 Initially, 1000 µl of glucose reagent solution was taken in a test tube, and then 10 µl of the test salivary sample was added and stirred properly. After 15 minutes, the test sample was aspirated, and the readings were noted (ERBA CHEM-5 Plus). Salivary pH was estimated by a digital pH meter, Hanna Instrument (Hanna pH Tester Checker). 13
Statistical Analysis
Considering the pilot study, no formal hypothesis was planned for the study. Approximately 50 patients were planned to be enrolled to obtain 40 evaluable cases considering a 20% dropout rate. All study data was planned to be presented using descriptive statistical methods. Any analysis, wherever applicable, was performed using appropriate two-sided t-tests with alpha at 0.05. The statistical analysis was performed using SAS software version 9.4 or higher (SAS Institute Inc., Cary, NC, USA).
Results
A total of 54 patients were initially screened; however, 4 patients did not meet the eligibility criteria (considered screen failures). Accordingly, a total of 50 patients were enrolled in the study (25 patients in each arm) who were included in the analysis (Figure 1).
Study Flow Chart.
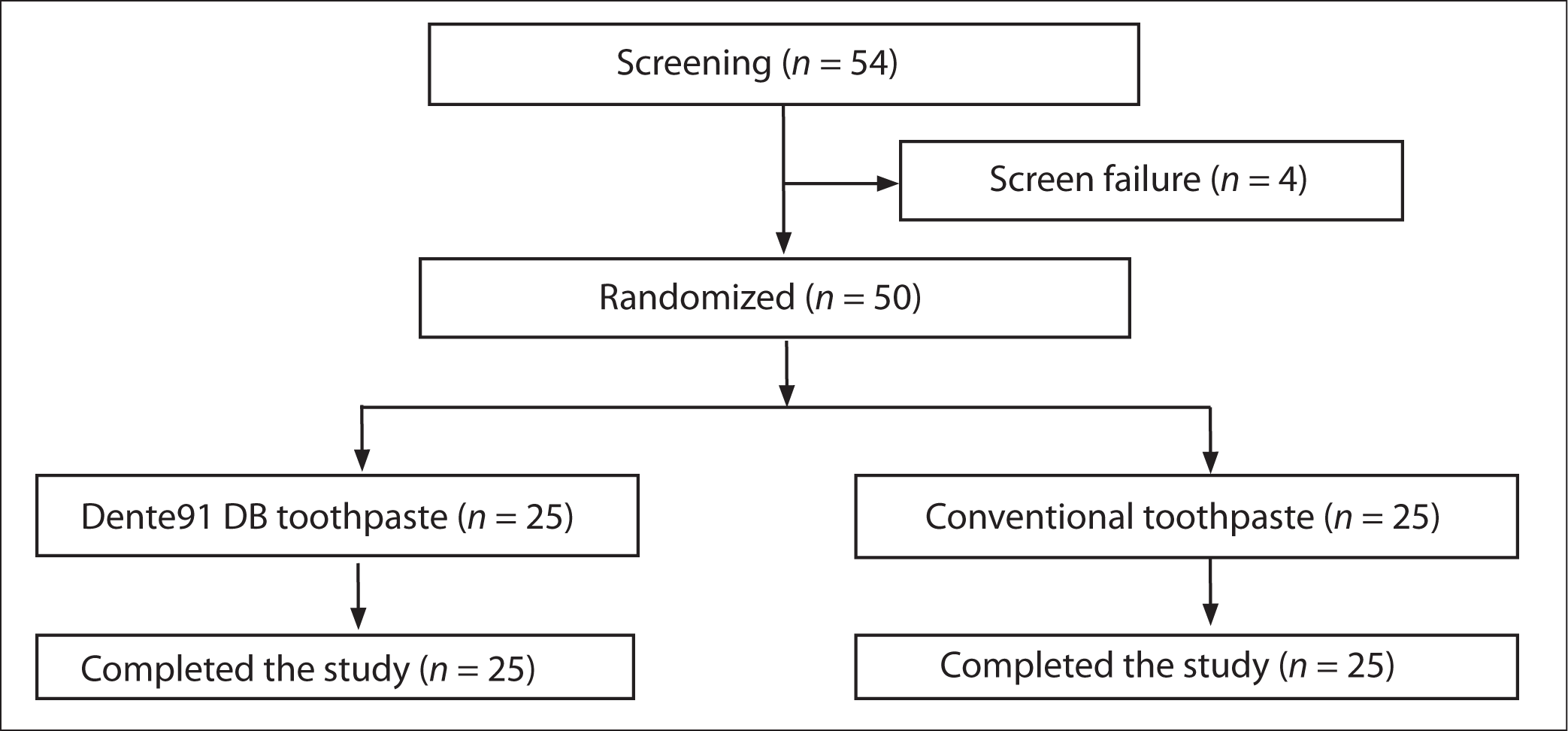
Baseline Characteristics
Overall, the baseline characteristics at screening were comparable in both groups except for male-to-female ratio (Table 1).
Baseline Characteristics.

Primary Endpoints
The baseline salivary glucose levels were comparable in both groups. At the end of 8 weeks, the reduction of salivary glucose levels in the Dente91 DB toothpaste group was significantly higher; while the same did not achieve significance in the conventional toothpaste group. The difference in the reduction in salivary glucose levels between the two groups was significant in favor of Dente91 DB toothpaste (Table 2).
Results of Salivary Glucose and pH Levels in Both Groups.
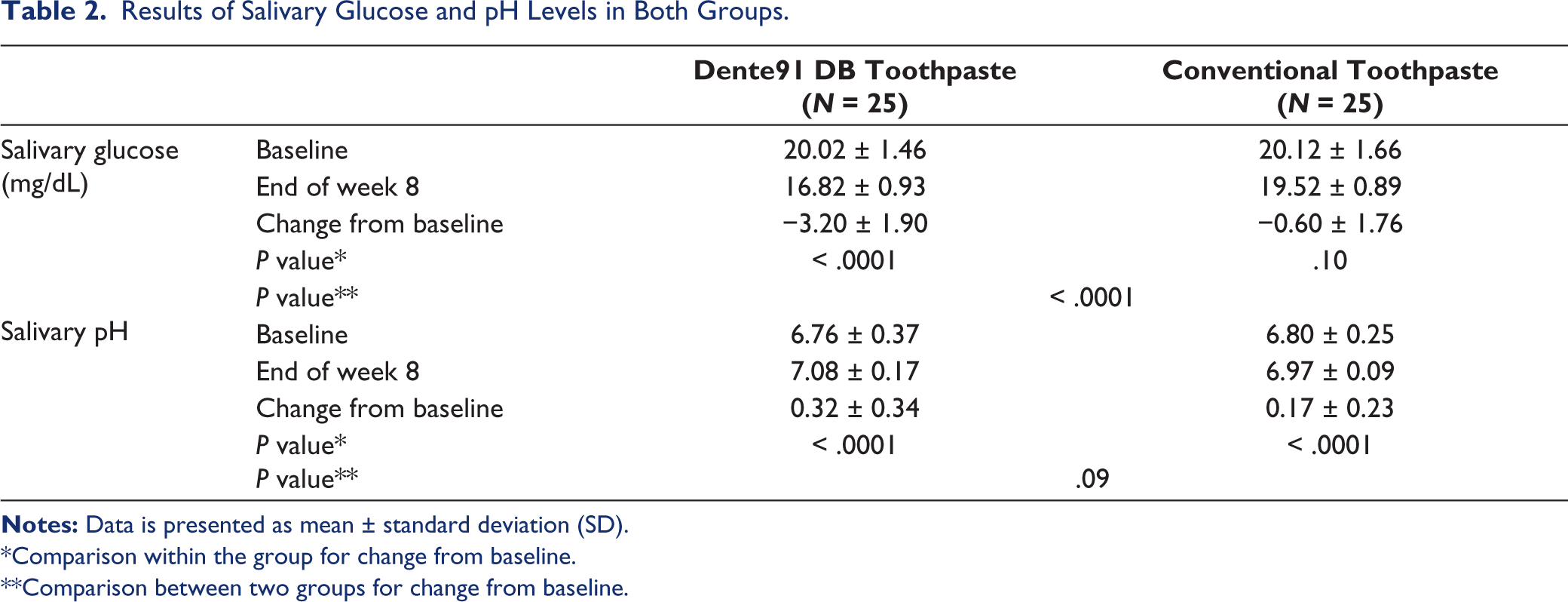
*Comparison within the group for change from baseline.
**Comparison between two groups for change from baseline.
The baseline salivary pH was comparable in both groups. After 8 weeks, there was a significant increase in salivary pH in both groups; however, the difference between the two groups did not achieve statistical significance (P = .09).
Secondary Endpoints
The baseline observations for all four secondary efficacy endpoints (saliva flow rate, gingival index, plaque index, and dental hypersensitivity) were comparable (Table 3). Saliva flow rate was significantly increased in both groups at the end of 8 weeks; however, the improvement in the Dente91 DB toothpaste group was significantly higher than conventional toothpaste. Gingival index score and Plaque index score were significantly reduced at the end of 8 weeks in both groups; the reduction was similar between the two groups for both parameters. Dental hypersensitivity was reduced significantly in both groups; and the reduction between the two groups, although statistically not different (P = .08), was numerically favorable in the Dente91 DB toothpaste group.
Results of Saliva Flow Rate, Gingival Index, Plaque Index, and Dental Hypersensitivity in Both Groups.
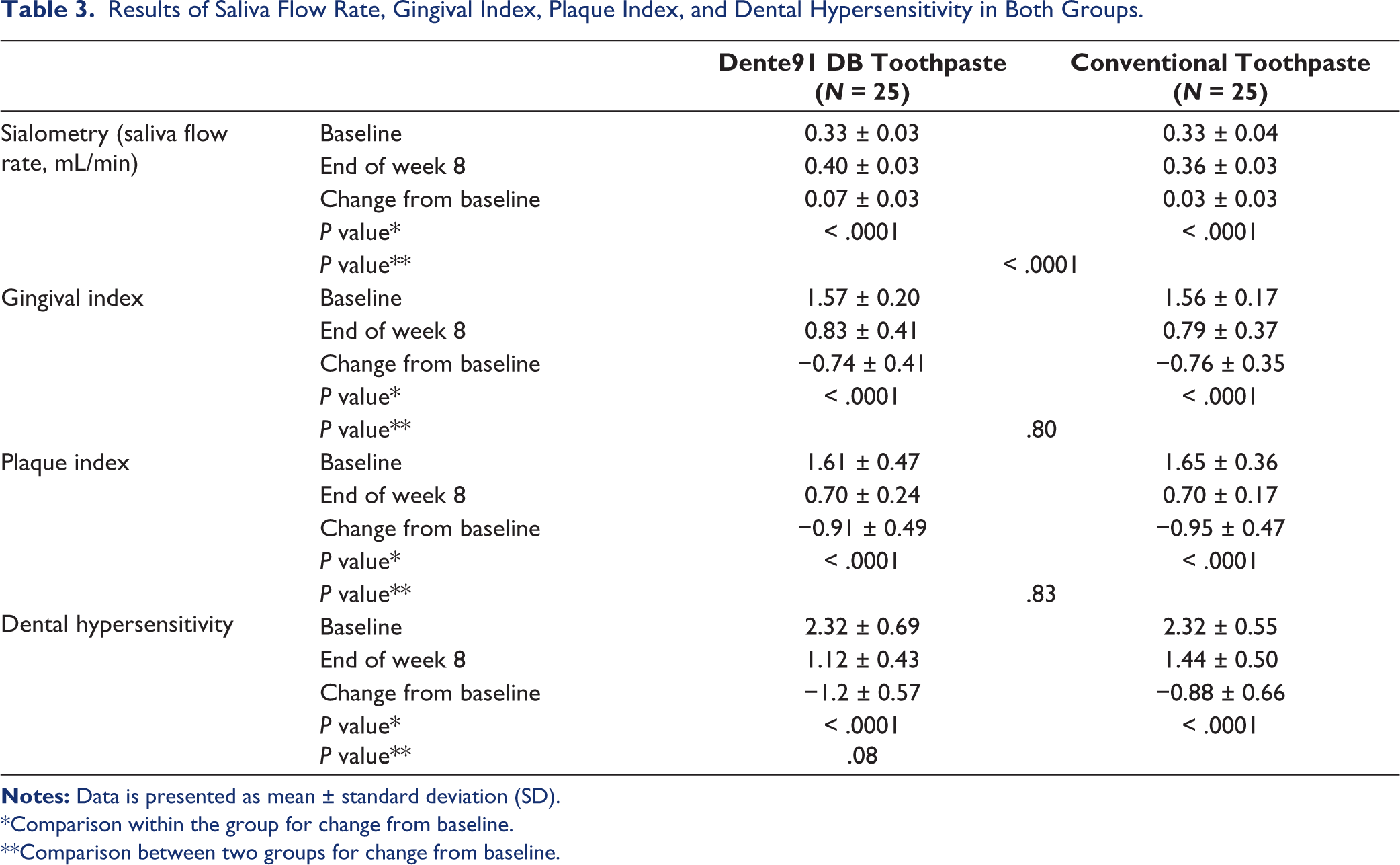
*Comparison within the group for change from baseline.
**Comparison between two groups for change from baseline.
Safety
Overall, 17 adverse events were reported during the entire study. The most commonly reported adverse events were pyrexia, headache, pain, and asthenia (Table 4). Out of these AEs, 16 adverse events (8 adverse events in each group) were of mild severity, and one adverse event (pyrexia in conventional toothpaste) was classified as moderate in severity. For all adverse events, the causality assessment was unlikely; and the outcome was reported as recovered/resolved. There were no severe or serious adverse events reported, or adverse events requiring discontinuation during the study.
Adverse Events Reported During the Study.

Discussion
This pilot study was planned to assess and compare the effectiveness of Dente91 DB toothpaste against conventional toothpaste (Colgate Total) in patients with diabetes mellitus. The primary endpoints of the study were the change in salivary glucose and pH. The observed baseline salivary glucose levels (~20 mg/dL) are in line with published studies with similar populations14-16; however, other studies have reported lower1,17 or higher 18 salivary glucose levels. A better reduction in salivary glucose (~16%) was observed with Dente91 DB toothpaste compared to conventional toothpaste (~3%). While we can consider any such reduction in salivary glucose beneficial in diabetes; its clinical significance in terms of impact on the incidence of oral complications in long-term studies needs to be further studied.
The baseline salivary pH in the current study was similar to published literature. 15 The improvement in salivary pH was observed in both groups, reaching to neutral pH (7), ideal for the oral cavity observed in the non-diabetic population. 15 The improvement of pH was numerically better in the Dente91 DB group compared to the other group (0.32 vs 0.17, respectively).
The improvement in unstimulated salivary flow rate was observed in both groups. The difference between the two groups was small, but statistically significant. Patients on Dente91 DB toothpaste achieved a 0.4 ml/min salivary flow rate, which is closer to the normal range observed in healthy populations (0.4 to 0.5 mL/min).19,20
The baseline dental hypersensitivity score was similar in both groups and it was high (>2) as expected for the diabetic population. The reduction in Schiff sensitivity score was similar and clinically meaningful in both groups. The difference between the two groups, although not statistically significant, was numerically better in the Dente91 DB group compared to the conventional group. A small study conducted on patients with dental hypersensitivity established the superiority of Dente91 toothpaste over other commercially available toothpaste (Vantej). 21
Both, the gingival index score and plaque index score were improved significantly in both groups and the improvement was comparable. Despite better improvement in other oral cavity parameters, Dente91 DB toothpaste did not achieve better gingival and plaque index scores compared to conventional toothpaste. The possible explanations are 1) improvement observed in these parameters (like salivary glucose, pH, and flow rate) is not sufficient to improve scores or 2) the duration of the study is quite small to translate these improvements into better scores. Long-term studies with clinical outcomes need to be conducted for better understanding.
The literature evaluating the effectiveness of various toothpastes on salivary glucose and pH in the diabetic population is limited. One study evaluated sugar-free toothpaste (Vicco SF) compared to conventional toothpaste (Colgate) in diabetic and non-diabetic populations for four weeks. 22 The reduction in salivary glucose with Vicco SF was 3 mg/dL as compared to 1.1 mg/dL in the conventional group, while the improvement in pH was 0.16 and 0.07, respectively in both groups. In comparison to our study, the baseline salivary glucose levels were different in that study; however, the absolute numerical reduction is comparable. The improvement in salivary pH in the diabetic population was two times better in our study with Dente91 DB toothpaste (0.32) compared to Vicco SF toothpaste (0.16). The other study evaluating three herbal toothpastes (Dant Kanti, Himalaya Complete Care, and Vicco Vajradanti) in the non-diabetic population demonstrated a small reduction in salivary glucose (up to 0.38 mg/dL) and pH (up to 0.04), which is quite lower than the observed results in our study, including the diabetic population. 23 Such differences are partly explained by differences in the study population; however, the effectiveness of different toothpastes cannot be ruled out.
There were limitations of this study. The study was planned as a pilot study with a limited sample size at a single center; therefore, the generalization of results needs to be considered carefully. Considering the objective nature of the primary endpoints and differences in the appearance of both toothpastes, the study was planned as open-label; however, the bias due to treatment awareness cannot be completely ignored, especially on subjective secondary efficacy endpoints. Further, the study assessments were limited to indicators of oral hygiene for up to 8 weeks duration; the long-term benefits especially on incidence of oral complications of type 2 diabetes mellitus (such as gingivitis, periodontitis, etc.) need to be explored in long-term studies on larger populations.
Conclusion
The pilot study results demonstrated that Dente91 DB toothpaste showed a better reduction in salivary glucose level compared to conventional toothpaste in patients with type 2 diabetes mellitus. The improvement in the salivary pH level was significant with Dente91 DB toothpaste after 8 weeks; however, it was not better compared to conventional toothpaste.
Footnotes
Acknowledgement (Financial Support and Sponsorship)
This study was funded by Frimline Private Limited, Ahmedabad, India.
CTRI Registration Number
CTRI/2023/07/055156 [Registered on: 13/07/2023].
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Statement of Ethical Approval and Informed Consent
Ethics Committee Registration Number ECR/45/Indt/MH/2013/RR-19. All the study participants signed the informed consent to participate in the study.
IEC Number
RPIEC090723.
