Abstract
Multifaceted disparities exist in the natural history of aortic valvular disease (AVD) between men and women. Despite a similar prevalence of aortic stenosis (AS) in both sexes, women face marked disadvantages in time to diagnosis, referral to specialised care, intervention frequency, and survival. Importantly, the pathophysiology of and myocardial compensation for AS in women exhibit distinct features that warrant intentional sex-tailored investigations and haemodynamic stratifications to ensure diagnostic accuracy and optimal timing of intervention. Furthermore, women with AS represent a patient population with unique biological, physiological, and anatomical considerations that render lifetime disease management particularly challenging. Recognising and rigorously investigating these sex-specific differences across the disease course will be essential to advancing evidence-based and patient-tailored clinical practice. Ultimately, implementing a personalised, lifetime approach to AS in women will help bridge the gap in their care and improve long-term outcomes.
This is a visual representation of the abstract.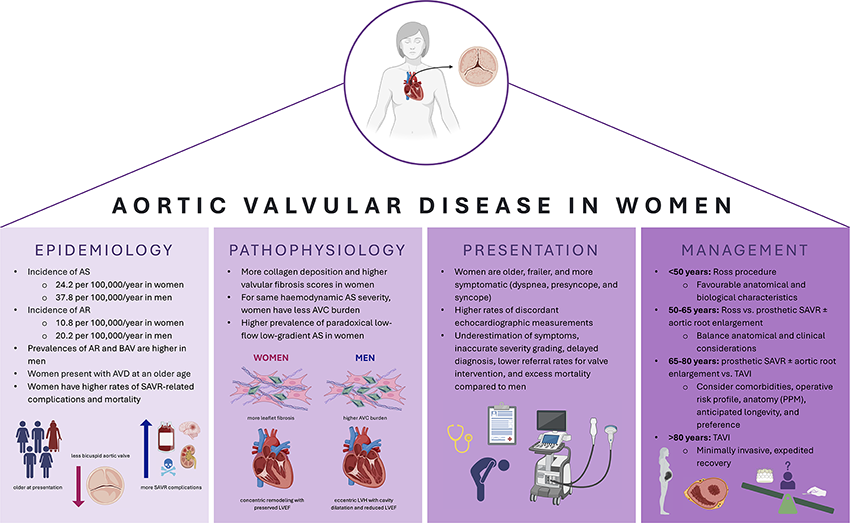
Keywords
Key Points
Women with aortic stenosis are underdiagnosed and undertreated compared with men, and they face consequently higher rates of AS-associated morbidity and mortality.
Sex differences in valvular pathology and resultant myocardial compensation can lead to systematic underestimation of disease severity in women when conventional haemodynamic criteria are applied.
Lifetime management of aortic stenosis in women is uniquely complex and influenced by factors including smaller aortic anatomy, childbearing potential, and burden of comorbidity at presentation.
Optimal and durable care for women with aortic stenosis requires personalised, sex-tailored diagnostic strategies and anticipatory lifetime management planning.
Introduction
Valvular heart disease (VHD) represents a significant cause of global morbidity and mortality. While rheumatic post-inflammatory etiology remains predominant in low- and middle-income countries, degenerative fibrocalcific remodeling processes constitute the primary cause among high-income populations.1–3 In developed countries, aortic stenosis is the most common primary VHD, 4 and it is characterised by inflammatory processes that lead to valvular fibrosis and calcification. Over time, leaflet stiffening results in progressive valve orifice narrowing with consequent haemodynamic aberrations and compensatory myocardial remodeling.
As with many cardiovascular disorders, aortic stenosis (AS) exhibits sex differences in anatomical presentation, clinical manifestation, diagnostic and management practices, as well as short- and long-term therapeutic outcomes. Contemporary surgical and interventional innovations have significantly expanded the arsenal of therapeutic options available for AS, yet each approach carries inherent limitations. Moreover, women with AS represent a patient population with a unique convergence of important biological, physiological, and anatomical considerations that require thoughtful deliberation for optimal lifetime management. With the aim of emphasising the critical importance of sex-specific personalised care, this article will summarise the unique features of AS in women and discuss their influence on the selection of optimal management strategies while highlighting opportunities for the improvement of clinical practice.
Epidemiology and Natural History
Pooling of multiple American population-based studies determined that the overall prevalences of clinically significant (i.e., severity grade ≥ moderate) AS and aortic regurgitation (AR) were 0.4% and 0.5% in the general population, respectively. 3 A Norwegian population-based study additionally found that the prevalence of AS (here defined as an echocardiographic transvalvular mean gradient ≥15 mm Hg) increases exponentially with age, with the following stratifications: 1.3% in people of ages 60 to 69 years, 3.9% in people of ages 70 to 79 years, and up to 9.8% in people 80 to 89 years. 5 Based on a study of the Swedish population between 2003 and 2010, the sex-stratified incidence of AS was 24.2 and 37.8 per 100 000/year in women and men, respectively, while that of AR was 10.8 and 20.2 per 100 000/year. 2 Interestingly, women were consistently older at disease diagnosis, with a median age of 80 versus 74 for AS and 71 versus 63 for AR. 2 Across all ages, however, the occurrence of AR was significantly higher in men.2,3 This can be explained by their estimated threefold higher prevalence of congenital bicuspid aortic valve (BAV), which is often associated with proximal thoracic aortic dilatation or aneurysm, and the fact that BAV-mediated aortic valvular disease (AVD) occurs more frequently via AR in men (48% vs 29%) and AS in women (78% vs 65%).6–9
Aortic valvular lesions do not necessarily occur in isolation. Indeed, patients can experience the concomitant failure of multiple native valves or, more commonly, the failure of one valve by multiple mechanisms. 10 For example, while rheumatic heart disease primarily targets the mitral valve, concomitant aortic valvular involvement occurs in up to 30% of cases.11–13 This can be in the form of AS, AR, or mixed disease due to some combination of post-inflammatory cusp thickening, fibrosis, retraction, commissural fusion, and calcification. 12 Alternatively, mixed aortic valvular disease can also occur due to degenerative remodeling, and patients with this complex entity remain inadequately investigated. Nonetheless, studies have shown that patients with mixed moderate AR and moderate AS have higher adverse event (i.e., cardiac death or hospitalisation) rates than those with isolated moderate AS and comparable rates to patients with isolated severe AS.14,15 Multiple and mixed VHDs represent challenging pathological entities with critical implications on decisions regarding management and intervention. However, given the significantly greater impact of AS than AR on women, the remainder of this article will focus on aortic stenosis as the primary valvular lesion.
Fibrocalcific AS is a slowly progressing lesion that is associated with a long asymptomatic phase. Once it becomes severe, the risk of all-cause mortality rises significantly.16–19 A study of 1,085,850 patients from 24 institutions found that, if left untreated, the estimated 4-year all-cause mortality rates of moderate, moderate-to-severe, and severe AS diagnoses were 33.5%, 45.7%, and 44.9%, respectively. 17 Another study, analysing the National Echocardiographic Database of Australia and encompassing 350,000 patients, found that those with moderate AS had a long-term mortality risk similar to that of patients with severe AS, and that the pivotal threshold for this link was indeed a mean gradient of ≥20 mm Hg. 16 Interestingly, in 2017, a novel AS classification system based on the extent of associated extravalvular cardiac damage was proposed and validated. 20 Using this system, a study of 1974 patients with severe AS found extent of cardiac damage to be one of the strongest predictors of 2-year mortality despite adjustment for known prognostic variables. 21 Moreover, in 57.9% of patients, the extent of cardiac damage remained unchanged at one year following either surgical aortic valve replacement or transcatheter aortic valve implantation, while in 26.5% of patients, it in fact worsened with an associated mortality risk increase. 21 The process of AS-associated extravalvular cardiac damage can begin well before the valvular lesion becomes haemodynamically severe. 22 Thus, these studies, along with others, challenged pre-existing dogma by demonstrating that even less-than-severe AS has an associated survival penalty.
This evidence underscores the importance of early detection, routine surveillance imaging, and prompt symptom reporting with the aim of mitigating long-term consequences through timely intervention. Unfortunately, despite a similar overall prevalence of AS between men and women, and despite the unequivocally advantageous effects of timely aortic valve replacement on survival, symptom alleviation, and left ventricular function in patients of low procedural risk,19,23–26 women were diagnosed significantly less frequently and were less commonly referred for specialised care and definitive treatment.3,27,28 This represents a significant gap in the care for women with AS, and elucidating its underlying causes will be central to strategising improvement.
Anatomy and Pathophysiology
Sex differences exist in native aortic root anatomy, with women having smaller annular diameters and shorter coronary ostial heights. 29 The molecular mechanisms underpinning aortic valvular degeneration also exhibit important sex differences. Early in the AS disease course, endothelial injury due to mechanical stress facilitates valvular lipid deposition, inflammatory infiltration, and the consequent release of pro-inflammatory mediators.30,31 Over time, chronic inflammation as well as the localised turnover of immune and inflammatory cells, leads to microcalcification at the sites of initial lipid deposition. 30 In the later stages of disease, extracellular matrix remodeling – including collagen deposition and upregulation of proteins associated with osteoblast function – generates a pro-calcific environment that promotes dystrophic mineralisation in and by valvular interstitial cells.30,31 Though AS is primarily characterised by aortic valvular calcification (AVC), myofibroblast activation and leaflet fibrosis also contribute significantly to leaflet stiffening in women.31–33 In fact, an analysis of surgically excised aortic valve leaflet tissue identified higher collagen content and greater valvular fibrosis scores in women than men after adjustment for age, body mass index, annular diameter, and AVC density. 32 Moreover, for a similar AVC burden, women have a higher associated haemodynamic AS severity, even after adjustment for body surface and annular areas (Figure 1).34,35 With these data in mind, the American College of Cardiology and American Heart Association guidelines on VHD management set sex-specific Agatston unit thresholds for the diagnosis of severe AS by computed tomography (CT) as 1300 for women and 2000 for men. 19 The European Society of Cardiology and European Association for Cardio-Thoracic Surgery guidelines set a slightly lower threshold for women at 1200 Agatston units. 10
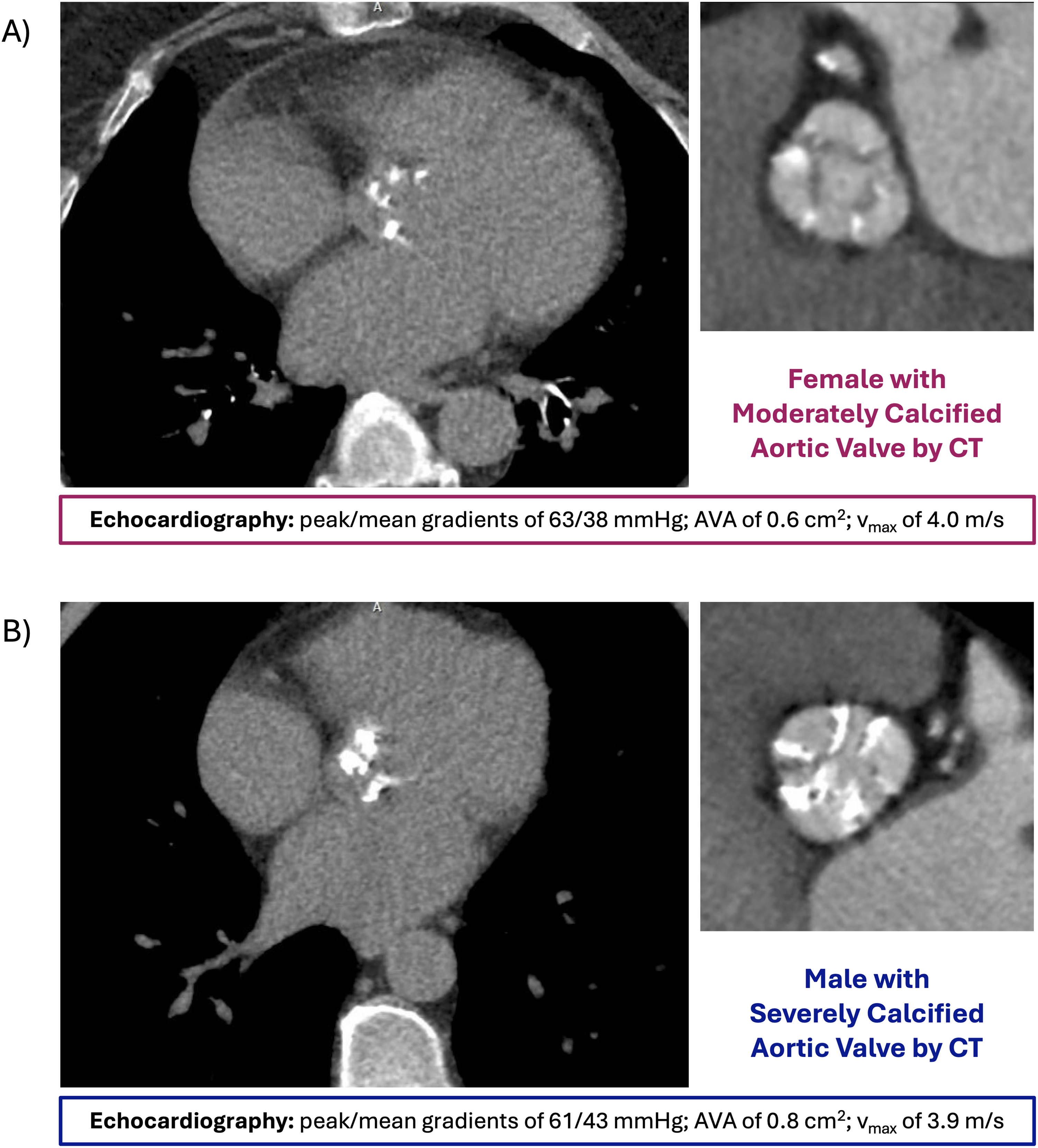
Representative CT images illustrating sex-specific patterns of aortic valvular calcification in severe aortic stenosis. Both patients exhibit comparable haemodynamics by echocardiography, but the female patient (A) demonstrates less extensive AVC burden than the male patient (B). This disparity underscores sex differences in the mechanism of AS pathogenesis. AS, aortic stenosis; AVA, aortic valve area; AVC, aortic valvular calcification; CT, computed tomography.
In addition to mechanistic differences in AS pathogenesis, sex-specific differences are also seen in patterns of left ventricular (LV) compensation as the disease progresses. Following calcific and fibrotic valve narrowing and consequent pressure overload, women tend to undergo concentric remodeling with a preserved left ventricular ejection fraction (LVEF), while men tend to undergo eccentric LV hypertrophy with cavity dilatation and a reduction in LVEF.36–38 Though the myocardial response to AS in men is more obviously maladaptive, concentric remodeling in women can progress to concentric LV hypertrophy and diastolic dysfunction, which have been linked to a significantly higher risk of all-cause mortality in women, specifically. 39 This may be due to the induced insufficiency of oxygen supply to the hypertrophied muscle wall, which can lead to impaired myocardial perfusion reserve and consequent fibrosis.40–42 Importantly, postmenopausal women also have a higher prevalence of increased arterial stiffness, aberrant pulsatile haemodynamics, and consequent isolated systolic hypertension.43,44 These cardiovascular changes have been shown to exacerbate concentric LV hypertrophy and diastolic dysfunction as well as impair ventricular-arterial coupling in women but not in men.45–47
Interestingly, the constellation of concentric remodeling, preserved systolic LV function, and myocardial fibrosis is commonly seen in an entity termed paradoxical low-flow, low-gradient (pLFLG) AS, which affects women more frequently and is associated with reduced survival compared to normal-flow, high-gradient severe AS.43,48 This subtype of AS is characterised by a reduced normalised stroke volume (i.e., stroke volume index [SVi] ≤35 mL/m2) due to extensive LV wall thickening and cavity obliteration, and thus, reduced maximal transvalvular jet velocity (<4 m/s) and mean gradient (<40 mm Hg) despite severe orifice narrowing (i.e., aortic valve area [AVA] ≤1 cm2).48–51
These sex-specific profiles of AS progression and consequent myocardial compensation have important implications on how the disease presents echocardiographically, and as a result, how it is subsequently diagnosed and managed.
Clinical Presentation and Diagnosis
Given the same degree of aortic valve narrowing and haemodynamic impairment, women were found to be older, frailer, and more symptomatic, with higher associated procedural risks.28,52,53 Their reported symptoms were more likely to include dyspnea, presyncope, and syncope, whereas those of men were more likely to include angina.1,52 At presentation with AS, comorbidities in women commonly included hypertension, diastolic dysfunction, concomitant atrioventricular valve disease, renal impairment, and anaemia, whereas in men, they commonly included atherosclerotic coronary artery disease.28,52
Transthoracic Doppler echocardiography is the standard diagnostic modality for VHD. Mean pressure gradient and maximum velocity are the key standard haemodynamic parameters used for grading AS, with peak velocity ≥4 m/s and mean pressure gradient ≥40 mm Hg constituting severe AS.10,19 An AVA measuring ≤1 cm2 is often considered in, but not required for, defining severity, except in nuanced cases of low-flow, low-gradient AS.10,19 Importantly, pLFLG AS has been shown to represent advanced disease and have a prognosis worse than that of concordant high-gradient severe AS.48,50,54 Indeed, symptomatic pLFLG AS (also known as stage D3 disease) is a class 1 recommendation for aortic valve replacement. 19 The higher prevalence of pLFLG AS among women translates into higher rates of discordant echocardiographic measurements in women, and this discordance has been associated with underestimation of symptoms, inaccurate severity grading, delayed diagnosis, lower referral rates for valve intervention, and excess mortality compared to men.28,40,53
This problem is unfortunately perpetuated by the vast and ubiquitous reliance on transvalvular pressure gradients and velocities for defining AS and tracking its progression when, indeed, declining SVi may be blunting changes in these parameters. 51 A study analysing 2429 patients with severe AS found that, despite having similar mean gradients, women had significantly smaller aortic valve areas than men. 53 Moreover, women in this cohort experienced significantly reduced 5-year survival rates compared to men in an age-matched analysis, had worse relative survival compared to the general population than men, and were significantly less likely to undergo aortic valve replacement despite more often being symptomatic. 53 As such, these cases demand meticulous investigation to detect masked yet bona fide disease progression, and they warrant assessment of additional haemodynamic and clinical parameters for the sake of appropriate decision-making. 10 In fact, another retrospective study on 1490 patients with severe AS and preserved LVEF undergoing surgical aortic valve replacement identified low flow as a marker of disease severity and demonstrated that setting sex-specific thresholds for defining low flow significantly improved risk stratification as well as diagnostic and therapeutic accuracy and efficiency. 54 The SVi thresholds proposed by this study were <32 mL/m2 for women and <40 mL/m2 for men. 54 Collectively, these findings ought to be further explored and potentially implemented into clinical practice guidelines accordingly.
Lastly, in the context of discordant echocardiographic measurements (i.e., AVA ≤1 cm2 with low transvalvular pressure gradients and low flow), alternative diagnostic imaging modalities ought to be employed. These can include CT for aortic valvular calcium scoring (with American and European sex-specific Agatston unit thresholds as detailed in Anatomy and Pathophysiology) and/or dobutamine stress echocardiography with quantification of the patient's responsive change in stroke volume as well as their “projected AVA” (an estimate of their AVA at a standardised normal flow rate).10,19,55,56 Dobutamine stress testing offers greater accuracy in patients with significantly reduced LVEF and is, therefore, better suited for cases of classical LFLG AS (LVEF <50%). 57 On the other hand, patients with pLFLG AS (LVEF ≥50%) stand to derive much greater benefit from CT imaging as this modality has a high sensitivity and specificity for detecting severe AS independently of flow.58,59
Therapeutic Arsenal and Outcomes
The only treatments for aortic stenosis are surgical aortic valve replacement (SAVR) and transcatheter aortic valve implantation (TAVI) (Figure 2). Current guidelines recommend these interventions when patients have echocardiographically severe AS as well as symptoms, reduced LVEF (i.e., <50%), or other concomitant indications for cardiac surgery.10,19
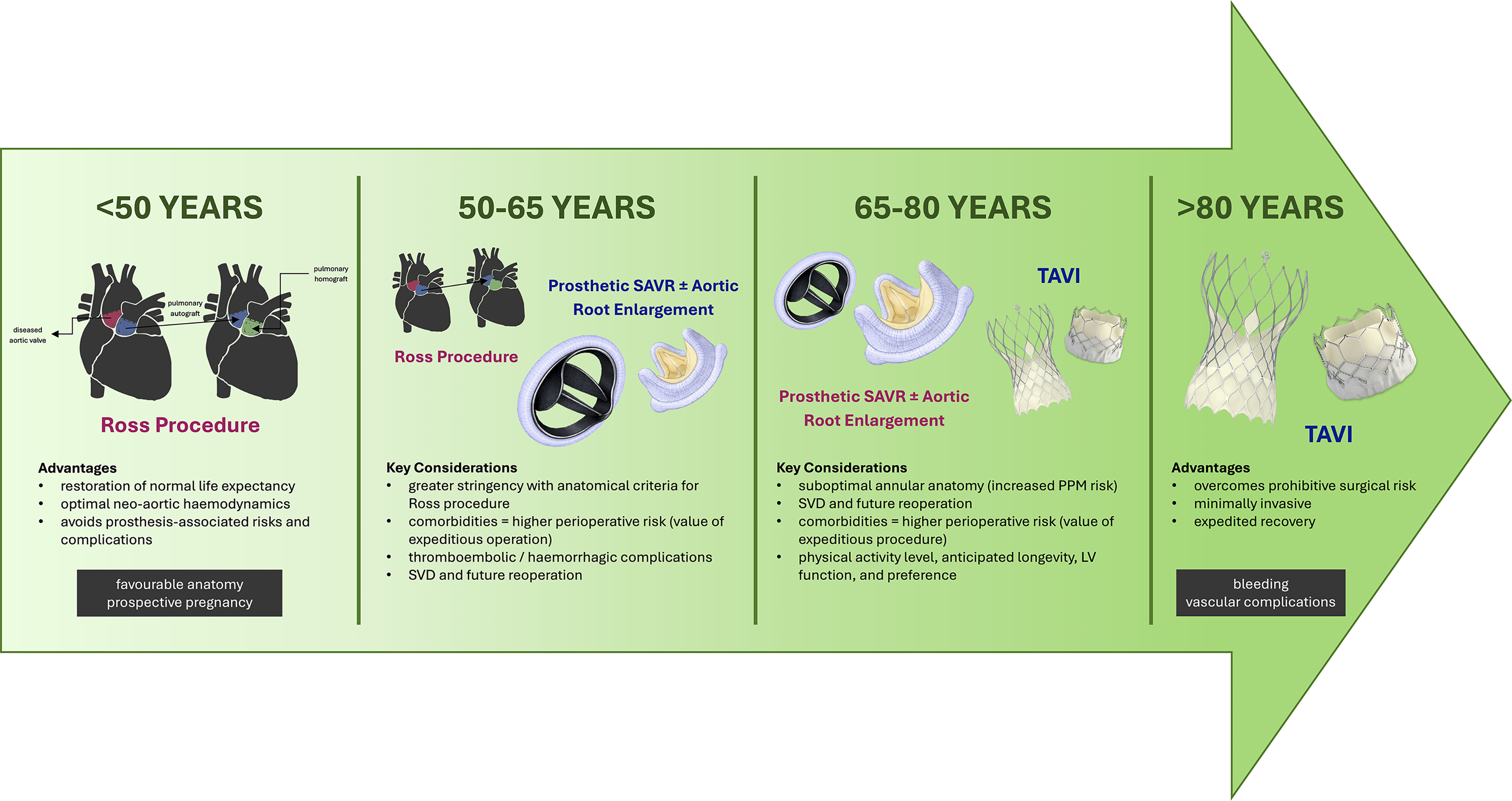
Age-stratified overview of the preferred primary interventions for severe aortic stenosis across the female adult lifespan. Various interventional strategies exist for the primary treatment of severe AS, each with associated advantages and disadvantages as well as important considerations unique to women. LV, left ventricular; PPM, prosthesis-patient mismatch; SAVR, surgical aortic valve replacement; SVD, structural valve deterioration; TAVI, transcatheter aortic valve implantation.
Ross Procedure
In otherwise healthy young and middle-aged women (i.e., <50 years of age), and in the setting of high-volume centres, the Ross procedure is the optimal aortic valve replacement strategy and should be prioritised in clinically and anatomically appropriate candidates.60,61 This operation is an autologous SAVR, whereby the diseased native aortic valve is excised and replaced with the patient's own pulmonary valve – the pulmonary autograft – while the right ventricular outflow tract is reconstructed with a conduit, which is ideally a pulmonary homograft. Although more technically complex than prosthetic SAVR, the advantages of the Ross procedure are multifold and include: restoration of life expectancy to that of the age-matched general population; use of a living graft that confers superior haemodynamics across the neo-aortic root; and reduction in the cumulative incidence of prosthesis-associated risks and complications, such as thromboembolism, haemorrhage, structural valve deterioration, and infective endocarditis.61–67 Independent predictors of autograft failure and reintervention include male sex, annuloaortic ectasia, and preoperative AR,62,68 meaning women – who have smaller annuli and a significantly lower prevalence of AR – are more likely to be anatomically favourable for a durable Ross procedure. Moreover, women in this age group may desire future pregnancy and consequently face additional nontrivial risks to prosthetic SAVR, including hypercoagulability and increased haemodynamic demand during pregnancy, 69 fetal susceptibility to the vitamin K antagonism required for mechanical prostheses,70,71 accelerated peripartum bioprosthesis degeneration, and endocarditis-associated bacteraemia during labour and delivery. 72 Collectively, these data indicate that women under the age of 50 years requiring aortic valve replacement ought strongly to be considered for a Ross procedure. In fact, based on their anatomy and biology, we believe they may garner a disproportionately greater benefit from this operation than men and should, therefore, be referred to high-volume Ross surgeons for consideration.
Prosthetic Surgical Aortic Valve Replacement
For women between the ages of 50 and 65 years, performance of a Ross procedure may also be reasonable, but we advocate for greater stringency in selection. While these women often also present with excellent anatomical substrates for a Ross procedure, they may have greater comorbidity and worse perioperative risk profiles, as explored in Clinical Presentation and Diagnosis.28,52,73 Indeed, women face significantly higher rates of in-hospital mortality, vascular complications, blood transfusions, and prolonged hospital stays after SAVR than men.73,74 As such, the benefits of an expeditious prosthetic SAVR ought to be balanced carefully against those of a Ross procedure, with the patient's lifestyle and priorities equally central to a shared decision-making discussion. If the patient is not deemed an optimal Ross procedure candidate, an important decision at this juncture is the type of SAVR prosthesis they receive – biological or mechanical.
A randomised control trial of 310 patients (ages 55 to 70 years) undergoing SAVR showed similar survival rates and linearised annual rates of thromboembolic or haemorrhagic complications at 13 years, regardless of choice of prosthesis. 75 In contrast, however, a recent analysis of the Society of Thoracic Surgeons’ Adult Cardiac Surgery Database – comprising 109,842 patients undergoing isolated SAVR – found that mechanical AVR was an independent predictor of survival in patients ≤60 years of age at 12 years of follow-up. 76 Similarly, a study of 5762 patients across 4 institutions found that the survival benefit of mechanical SAVR persists even longer, until an age of 70 years at 9 years of follow-up. 77 Nevertheless, patients are increasingly searching for a non-thrombogenic solution and the use of bioprosthetic valves in middle-aged patients is becoming increasingly common.78,79 Ultimately, lifetime disease management counseling in this age group ought to be comprehensive. In cases of primary bioprosthetic SAVR, anticipation of a need for future reintervention ought to be discussed given the inverse association between age at time of surgery and risk of valve failure; in cases of primary mechanical SAVR, emphasis ought to be placed on the importance of lifelong anticoagulation and regular monitoring of international normalised ratio to mitigate the risks of prosthesis- and warfarin-related adverse events.75,78,80–82
Unfortunately, however, the anatomical characteristics that make women excellent candidates for a Ross procedure at young ages pose as surgical challenges that increase their susceptibility to morbidity and mortality with prosthetic SAVR at more advanced ages. 52 Most women undergoing SAVR have small aortic annuli, and in turn, most patients with small aortic annuli are women.83,84 Accordingly, they develop prosthesis-patient mismatch (PPM; a haemodynamic phenomenon whereby the prosthetic valve's effective orifice area [EOA] is disproportionately small relative to the patient's body size) at significantly higher rates than men.85–87 As per the Valve Academic Research Consortium-3, sex-independent thresholds for defining PPM severity were set based on EOA indexed to body surface area (EOAi), with adjusted values for patients with body mass indices ≥30 kg/m2. 88 Importantly, however, a Québécois population-based study of 7319 patients who underwent SAVR between 2000 and 2021 revealed that refinement of these PPM definitions in a sex-stratified manner significantly improved their predictive performance, with men tolerating higher EOAi thresholds than women. 85 Consequences of inadequate cardiac output due to PPM include impaired regression of LV hypertrophy, persistent exercise intolerance and reduced functional capacity, increased incidence of structural valve deterioration, and increased mortality.85,87,89 In an effort to overcome these problematic sequelae, surgeons ought to consider concomitant aortic root enlargement if their patient's native anatomy cannot accommodate a large enough prosthesis, 90 and further investigation into standardised sex-specific EOAi thresholds is warranted to facilitate adoption into global clinical practice.
Transcatheter Aortic Valve Implantation
Significant PPM should be avoided in everyone, but its deleterious effects are particularly marked in patients with significant LV hypertrophy, systolic dysfunction, or pLFLG.87,91 Interestingly, in the first randomised control trials comparing outcomes between bioprosthetic SAVR and TAVI, the incidence of PPM was found to be significantly lower with TAVI, which employs prostheses with thinner stents and without sewing rings, thereby maximising patency of native outflow.86,92 This difference has been negated in the most recent trial of this series, possibly due to widespread adoption of surgical aortic root enlargement, which facilitates the implantation of a larger surgical prosthesis. 93 With the goal of comparing TAVI and SAVR outcomes in low-risk patients with severe AS and small aortic annuli, a recent randomised trial – comprising 151 patients with a median annular diameter of 21.1 mm, 93% of whom were women – demonstrated no significant differences in severe PPM incidence or either 2-year haemodynamic (i.e., transvalvular pressure gradients and AVA) or clinical (i.e., mortality, stroke, and cardiac rehospitalisation) outcomes. 94 Notably, among the SAVR patients, 7% underwent concomitant aortic root enlargement and 21.1% received sutureless prostheses, whereas among the TAVI patients, 59.2% received self-expandable supra-annular prostheses. 94
The advantages of TAVI in patients of high surgical risk – who are more frequently women – and the female-specific survival benefit of TAVI over SAVR have been cited extensively.95,96 Specifically, in a retrospective subanalysis of 699 high-risk patients with symptomatic severe AS, who were randomly assigned to TAVI or SAVR, women uniquely experienced a lower 2-year mortality if they underwent TAVI. 95 Similarly, a meta-analysis comprising 3758 randomly assigned patients showed a sex-specific 2-year survival advantage in TAVI recipients. 96 These favourable early clinical outcomes for women, specifically, were recently further supported by a randomised control trial of TAVI versus SAVR for severe AS in 420 women, which demonstrated a reduced 1-year composite incidence rate of death, stroke, or rehospitalisation in those who underwent TAVI. 97 This effect was driven primarily by the difference in rates of rehospitalisation. Haemodynamically at one year, however, mean transvalvular gradient was lower (11.7 mm Hg vs 14.3 mm Hg, P < .001), EOA was higher (1.9 cm2 vs 1.7 cm2, P = .003), and incidence of mild paravalvular regurgitation was lower (2.4% vs 15.5%, P < .001) in patients who underwent SAVR compared to those who underwent TAVI. 97
Nonetheless, women indeed constitute a greater proportion of patients undergoing TAVI and experience lower frequencies of paravalvular regurgitation and heart block requiring permanent pacemaker implantation than men.98,99 This may be due to their decreased likelihood of presenting with BAV and/or excess AVC, both of which predispose to the development of such complications. 100 Conversely, women do experience higher rates of major vascular complications (e.g., iliofemoral dissection or perforation) and life-threatening bleeding with TAVI than men.98,101 Taken together, these findings demonstrate superiority of TAVI in women of prohibitive surgical risk as well as non-inferiority of TAVI in women of low surgical risk.97,102,103 Of critical note, however, the majority of patients enrolled in these seminal studies were in their eighth or ninth decades of life, and their long-term outcomes have yet to be evaluated and reported. As such, these data must be interpreted and applied contextually, and their implications cannot be extrapolated to younger patients with longer life expectancies.104,105
Thus, in settings of severe AS meeting interventional indications, we advocate for the use of TAVI in anatomically eligible women over 80 years of age. The minimally invasive nature of the procedure and its resultant expedited recovery are particularly favourable for this age group, and significant valve deterioration is not likely to pose a major issue within these patients’ lifetimes. In fact, 10-year outcomes from the first randomised control trial of SAVR versus TAVI in 280 patients of low surgical risk revealed significantly lower rates of severe bioprosthetic structural and non-structural valve deterioration (as per Valve Academic Research Consortium-3 criteria) in those who underwent TAVI. 106 Between ages 65 and 80 years, however, due consideration must be paid to the patient's operative risk profile, physical activity level, anticipated longevity, LV function, anatomy (with special attention to aortic annular diameter and PPM risk), and preference. 94 A thorough conversation explaining the advantages and disadvantages of each approach is necessary, while a multidisciplinary heart team discussion about optimal strategies for immediate and lifetime management is strongly advised.
Conclusion
In an aging population with progressively increasing life expectancy, the burden of fibrocalcific AS is similarly expected to rise. Sex disparities in time to diagnosis, rates of referral to specialised care, intervention frequency, and mortality persist. As such, there is a critical need for enhanced disease understanding to improve lifelong management. The pathophysiology of and myocardial compensation for aortic stenosis in women demonstrate unique features that warrant deliberate sex-tailored investigations and haemodynamic stratifications for optimal diagnostic accuracy and interventional timing. With additional unique considerations, including childbearing potential, smaller aortic anatomy, and comorbid status at time of diagnosis, identifying an optimal treatment plan for women with AS is arguably much less straightforward. Fortunately, increased utilisation of the Ross procedure over the recent past has made it an increasingly available, excellent, durable, and safe solution to many of these challenges. Furthermore, the advent of TAVI, along with the continuous flow of promising device innovations it has sparked, has helped circumvent the obstacles of prohibitive surgical risk and PPM. Nevertheless, there is no perfect approach, and strides remain to be made in bridging the gap in care for women with AVD. We strongly believe that a collaborative, thorough, and personalised assessment of all relevant patient variables is the key to strategising the best possible plan.
Footnotes
Acknowledgments
Dr. Maral Ouzounian is partially supported by the Munk Chair in Advanced Therapeutics and the Antonio & Helga De Gasperis Chair in Clinical Trials and Outcomes Research at University Health Network.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
